10-Year Study on PRESERFLO MicroShunt for Glaucoma
PRESERFLO™ MicroShunt Implantation in Patients with Primary Open-Angle Glaucoma: 10-Year Results from a Single-Center Nonrandomized Study
Juan F. Batlle, MD¹*, Rachel Albuquerque, MD¹ and Adalgisa Corona, MD¹
- Laser Center, Santo Domingo, Dominican Republic
OPEN ACCESS
PUBLISHED: 31 July 2025
CITATION Batlle, JF., Alburquerque, R., et al., 2025. PRESERFLOTM MicroShunt Implantation in Patients with Primary Open-Angle Glaucoma: 10-Year Results from a Single-Center Nonrandomized Study. Medical Research Archives, [online] 13(7). https://doi.org/10.18103/mra.v13i7.6631
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6631
ISSN 2375-1924
ABSTRACT
Intraocular implantation of the PRESERFLOTM MicroShunt has been shown to be effective in lowering intraocular pressure in patients with primary open-angle glaucoma. A single-site, open-label study in the Dominican Republic (NCT00772330) found that MicroShunt implantation resulted in sustained reductions in mean intraocular pressure and glaucoma medications after 1, 3 and 5 years, with no long-term sight-threatening adverse events and with low rates of post-operative interventions. The present study reports results in these patients after 10 years.
The original trial included patients aged 18-85 years with primary open-angle glaucoma inadequately controlled on maximum tolerated medical therapy, with medicated intraocular pressure ranging from 18-40 mmHg, who underwent monocular MicroShunt implantation. Patients who remained in this trial after 10 years, were included. The primary safety endpoint was the incidence of all device- and/or procedure-related adverse events. Secondary endpoints included the mean intraocular pressure and mean reduction in intraocular pressure from baseline, the mean number of glaucoma medications and changes in visual field.
Of the 23 patients initially enrolled in this trial, nine remained after 10 years. Mean medicated intraocular pressure in operated eyes was lower at 10 years than at baseline, with mean percent reductions in intraocular pressure being greater in operated than in non-operated eyes throughout the 10-year period. Glaucoma medication use returned to baseline after 10 years, with none of the nine patients being medication-free. Relative to endothelial cell counts at 5 years, endothelial cell counts at 10 years showed a greater decrease in non-operated than in operated eyes. The percentages of eyes with bullae began to decrease after 1 year, with 67% of eyes having bullae after 10 years. The mean deviation of change in visual field was stable for 36 months, but showed increasing variability thereafter.
The present study showed that the PRESERFLOTM MicroShunt is an effective and safe long-term option for the surgical management of primary open-angle glaucoma in this study population. Prospective clinical trials are needed to compare the long-term efficacy and safety of MicroShunt implantation with other treatments, such as trabeculectomy.
Keywords: PRESERFLO MicroShunt, primary open-angle glaucoma, intraocular pressure, long-term results, surgical management
Introduction
Glaucoma is a spectrum of diseases that frequently manifest as elevated intraocular pressure (IOP). Glaucoma is characterized by retinal ganglion cell death and cupping of the optic nerve head, resulting in visual field loss. Moreover, glaucoma is the leading cause of blindness worldwide, with a high proportion of these patients having primary open-angle glaucoma (POAG). IOP is an important modifiable risk factor in patients with glaucoma, with reduction of IOP targeted to prevent disease progression. Thus, the current management of glaucoma primarily consists of efforts to reduce IOP, using the fewest possible number of medications.
Although medical therapy, including topical and/or oral medications, is usually the first-line method for treating glaucoma, patient compliance is suboptimal. If medical therapy fails to adequately reduce IOP, laser or incisional surgery may be performed to reduce IOP. Trabeculectomy and tube shunt implantation are currently the most frequent types of incisional surgery, but these procedures have been associated with complications and the need for postoperative interventions.
Microinvasive glaucoma surgery is less invasive than traditional incisional surgery, but results in more modest IOP reductions. One type of device used in microinvasive glaucoma surgery is the PRESERFLO MicroShunt (Santen, Inc.), an 8.5-mm long microincisional filtration device of outer diameter 350 and a 70 lumen composed of poly(styrene block isobutylene block styrene [SIBS]), a polymer that is highly biocompatible and bioinert.
The MicroShunt is implanted subconjunctivally into eyes via an ab-externo approach. Several prospective clinical trials have evaluated the efficacy and safety of this device. For example, a large, 2-year multicenter randomized trial compared MicroShunt implantation (n=395) with trabeculectomy (n=132) in patients with POAG. Results have been reported at 1 year and 2 years, as have the preliminary results of a 5-year extension study (Panarelli et al, manuscript in preparation). In addition, a single-center prospective trial compared MicroShunt implantation (n=150) with trabeculectomy (n=150) in patients with moderate to advanced open-angle glaucoma (OAG), with results reported at 12 months. Another prospective trial compared MicroShunt implantation (n=26) with trabeculectomy (n=26) in patients with moderate to advance OAG, with these results reported at 6 months. All of these studies found that MicroShunt implantation reduced IOP and the number of glaucoma medications significantly, although the MicroShunt was generally less effective than trabeculectomy.
Other studies have also compared MicroShunt implantation with trabeculectomy. For example, a retrospective case-control study compared MicroShunt implantation (n=101) with trabeculectomy (historical controls; n=101) in patients at two London hospitals, with results reported at 18 months. A single-site, open label study evaluated 61 patients with POAG in France who underwent MicroShunt implantation with intraoperative administration of mitomycin C (MMC), and a multicenter study evaluated outcomes in 81 POAG patients in France, Spain, Switzerland and the Netherlands who underwent MicroShunt implantation with intraoperative administration of MMC. MicroShunt implantation, especially with intraoperative administration of MMC, was found to reduce IOP and the number of glaucoma medications over time.
The present study was a single-site, open label study of patients with POAG in the Dominican Republic who underwent MicroShunt implantation with intraoperative administration of MMC. Results in these patients have been reported after 3 years and 5 years, with these studies showing that MicroShunt implantation resulted in sustained reductions in mean IOP and glaucoma medications over the 5-year period. Long-term sight-threatening adverse events (AEs) were not observed, and there were low rates of post-operative interventions. The present study reports results in these patients after 10 years.
Patients and Methods
STUDY DESIGN
This single-center, nonrandomized, single-arm interventional study was performed at Centro Laser, Santo Domingo, Dominican Republic (NCT00772330). Although originally planned for 1-year, the observation period was extended to 3, 5, and 10 years. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of CONABIOS, the National Counsel of Bioethics and Health of the Dominican Republic.
PATIENTS
Eligible patients were aged 18-85 years with POAG inadequately controlled on maximum tolerated medical therapy, with medicated IOP ranging from 18-40 mmHg. All patients provided signed, written informed consent. Patient inclusion and exclusion criteria have been described in detail. Key exclusion criteria included previous ophthalmic surgery, excluding uncomplicated cataract surgery or corneal refractive surgery.
TREATMENTS AND ASSESSMENTS
The procedure for MicroShunt implantation has been described elsewhere in detail. Patients who also required cataract surgery underwent phacoemulsification prior to MicroShunt implantation. After making a 6-8 mm incision to form a fornix-based flap, three LASIK shields (Network Medical Ltd., UK) soaked with 0.4 mg/ml MMC were applied to the subconjunctival space for 3 minutes ± 15 seconds. The subconjunctival space was rinsed with >20 ml of balanced salt solution to flush out any remaining MMC.
The MicroShunt was subsequently implanted as described. Briefly, a 3-mm scleral marker and marker pen were used to mark the location for a scleral pocket 3 mm from the limbus. A 1-mm-side Mani knife was used to make a triangular pocket at this location, and a 25 to 27 G needle track was formed from the sclera into the anterior chamber. Using forceps, the proximal tip of the MicroShunt was inserted into the anterior chamber, with the 1.1-mm wingspan planar fins of the MicroShunt wedged into the 1-mm scleral pocket and positioned so that the fins were lying flat on the sclera, not protruding vertically. All implants were placed in the superotemporal quadrant between the superior and lateral rectus. The distal end of the MicroShunt was assessed for droplet formation before being tucked under the subconjunctival flap. The conjunctiva and flap were positioned over the MicroShunt to the limbus and sutured with 10-0 nylon sutures. Bleb leaks were monitored, with a light-pressure was placed on the eye.
The primary safety endpoint was the incidence of all device- and/or procedure-related AEs. Secondary endpoints included the mean IOP and mean reduction in IOP from baseline, the mean number of glaucoma medications and changes in visual field.
STATISTICAL METHODS
All statistical analyses were performed with SAS System, Version 9.1 or higher (Buckinghamshire, UK), as described. Quantitative endpoints were reported as mean ± standard deviation (SD) and qualitative endpoints as number (percentage). Descriptive summaries were based on observed patients, except for calculations of success rates and mean IOP. For the latter parameters, missing data were imputed using the last observed score. Data collected after reoperation (surgical failure) were excluded from the analyses.
Results
All 23 patients enrolled in this study underwent MicroShunt implantation in one eye, 14 as a standalone procedure and 9 in combination with cataract surgery. The demographics and baseline characteristics of these patients are summarized in Table 1.
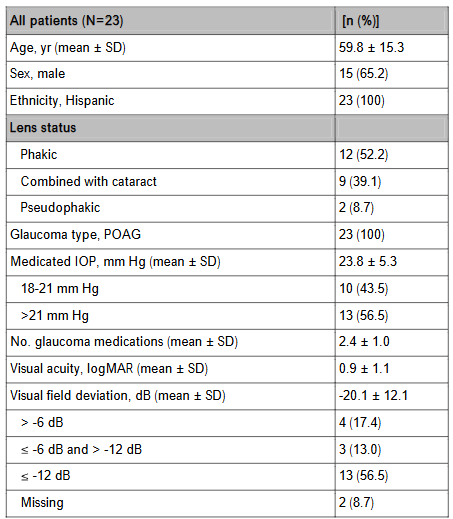
During the course of this study, 9 patients were discontinued or lost to follow-up, 5 patients experienced death not associated with the study device or glaucoma, and 9 patients remained after 10 years.
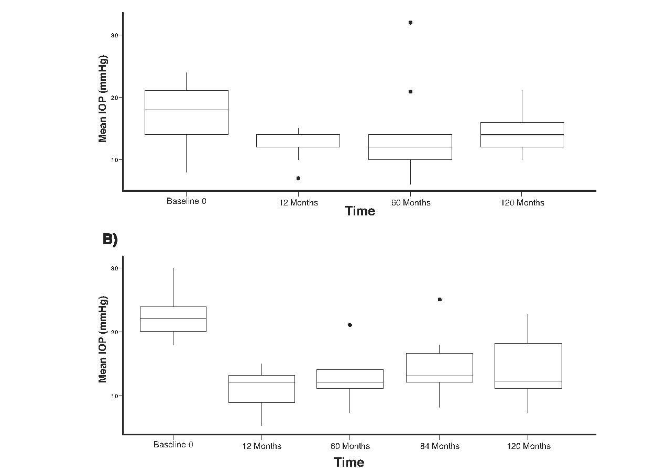
Mean medicated IOP in both the operated and non-operated eyes was lower at 1 year than preoperatively in the nine patients who completed the 10-year study. The reductions in mean medicated IOP in operated eyes were maintained through the 10-year follow-up period.
| Time | Mean ± SD | 95% CI |
|---|---|---|
| 1 day | 12.83 ± 5.46 | 17.03 to 8.64 |
| 7 days | 14.83 ± 3.55 | 17.56 to 12.10 |
| 3 weeks | 15.83 ± 4.29 | 19.13 to 12.54 |
| 6 weeks | 13.31 ± 4.51 | 17.08 to 9.54 |
| 3 months | 12.00 ± 3.95 | 15.04 to 8.96 |
| 6 months | 10.67 ± 4.13 | 13.84 to 7.49 |
| 9 months | 11.38 ± 4.45 | 15.10 to 7.65 |
| 12 months | 12.39 ± 4.72 | 16.01 to 8.76 |
| 24 months | 11.78 ± 3.34 | 14.34 to 9.21 |
| 36 months | 12.00 ± 4.04 | 15.38 to 8.62 |
| 48 months | 9.00 ± 4.78 | 16.60 to 1.40 |
| 60 months | 10.44 ± 5.82 | 14.92 to 5.97 |
| 120 months | 9.28 ± 4.79 | 12.96 to 5.60 |
The mean percentage reductions in IOP relative to baseline were greater in operated eyes than in non-operated eyes, with these reductions at 10 years being 39.36% (95% CI, 54.34% to 24.37%) in operated eyes and 8.67% (95% CI, 33.91% to +16.57%) in non-operated eyes.
| Operated group | Non-operated group | |||
|---|---|---|---|---|
| Time | Mean ± SD | 95% CI | Mean ± SD | 95% CI |
| 1 day | 56.13 ± 23.10 | 73.89 to 38.38 | +16.27 ± 36.91 | 12.10 to +44.64 |
| 7 days | 63.52 ± 13.25 | 73.71 to 53.34 | 18.31 ± 18.38 | 32.44 to 4.18 |
| 3 weeks | 67.12 ± 11.48 | 75.96 to 58.29 | 33.49 ± 20.24 | 49.05 to 17.93 |
| 6 weeks | 56.53 ± 17.87 | 71.47 to 41.59 | 16.93 ± 15.92 | 30.24 to 3.62 |
| 3 months | 51.19 ± 14.97 | 62.70 to 39.68 | 11.04 ± 29.03 | 33.36 to +11.27 |
| 6 months | 45.37 ± 16.27 | 57.88 to 32.86 | 10.93 ± 18.40 | 25.07 to +3.22 |
| 9 months | 46.93 ± 14.31 | 58.90 to 34.97 | 10.24 ± 23.16 | 29.60 to +9.12 |
| 12 months | 52.09 ± 15.54 | 64.03 to 40.15 | 23.64 ± 16.81 | 36.56 to 10.72 |
| 24 months | 50.62 ± 13.68 | 61.13 to 40.10 | 17.28 ± 16.07 | 29.63 to 4.92 |
| 36 months | 46.96 ± 16.30 | 63.58 to 36.33 | 32.54 ± 14.11 | 44.33 to 20.74 |
| 48 months | 36.73 ± 16.66 | 63.24 to 10.21 | 17.51 ± 36.39 | 75.41 to +40.39 |
| 60 months | 43.64 ± 21.22 | 59.95 to 24.37 | 17.63 ± 30.70 | 41.22 to +5.97 |
| 120 months | 39.36 ± 19.49 | 54.34 to 24.37 | 8.67 ± 32.84 | 33.91 to +16.57 |
Glaucoma medication use, which decreased in the first few years after MicroShunt implantation in the nine patients who completed the 10-year study, subsequently increased and was at baseline levels at 10 years. None of these patients was medication-free after 10 years.
| No. of medications | Preoperative (N=9) | 36 months (n=4) | 48 months (n=8) | 60 months (n=9) | 120 months (n=9) |
|---|---|---|---|---|---|
| 0 | 0.0% [0.0%, 33.6%] | 25.0% [0.63%, 80.6%] | 50.0% [15.7%, 84.3%] | 55.6% [21.2%, 86.3%] | 0.0% [0.0%, 33.6%] |
| 1 | 11.1% [0.28%, 48.2%] | 50.0% [6.76%, 93.2%] | 25.0% [3.19%, 65.1%] | 22.2% [2.81%, 60.0%] | 22.2% [2.81%, 60.0%] |
| 2 | 55.6% [21.2%, 86.3] | 25.0% [0.63%, 80.6%] | 0.0% [0.0%, 36.9%] | 11.1% [0.28%, 48.2%] | 0.0% [0.0%, 33.6%] |
| 3 | 22.2% [2.81%, 60.0%] | 0.0% [0.0%, 60.2%] | 25.0% [3.19%, 65.1%] | 0.0% [0.0%, 33.6%] | 66.7% [29.9%, 92.5%] |
| 4 | 11.1% [0.28%, 48.2%] | 0.0% [0.0%, 60.2%] | 0.0% [0.0%, 36.9%] | 11.1% [0.28%, 48.2%] | 11.1% [0.28%, 48.2%] |
Safety
Endothelial cell counts were measured at 5, 7, and 10 years in operated and non-operated eyes of the nine patients who completed the 10-year study. Relative to endothelial cell counts at 5 years, the decrease was greater in non-operated than in operated eyes.
| Operated eyes | 5 years post-op | 7 years post-op | 10 years post-op |
|---|---|---|---|
| Mean | 2249 | 2236 | 2267 |
| 95% CI | [1927; 2570] | [2083; 2388] | [1945; 2588] |
| % change | N/A | -0.6% | 0.8% |
| Time | No. of patients | Percentage with bullae, mean [95% CI] |
|---|---|---|
| 1 day | 9 | 88.9% [51.8%, 99.7%] |
| 7 days | 9 | 100% [66.4%, 100%] |
| 3 weeks | 9 | 100% [66.4%, 100%] |
| 6 weeks | 9 | 100% [66.4%, 100%] |
| 3 months | 9 | 100% [66.4%, 100%] |
| 6 months | 9 | 88.9% [51.8%, 99.7%] |
| 9 months | 9 | 100% [66.4%, 100%] |
| 12 months | 9 | 100% [66.4%, 100%] |
| 24 months | 9 | 88.9% [51.8%, 99.7%] |
| 36 months | 8 | 75.0% [34.9%, 96.8%] |
| 48 months | 7 | 85.7% [42.1%, 99.6%] |
| 60 months | 9 | 77.8% [40.0%, 97.2%] |
| 120 months | 9 | 66.7% [29.9%, 92.5%] |
Table 7 shows the mean deviation of change in visual field dB P <0.5% in the nine patients who completed the 10-year study. Mean deviation remained stable for 36 months, but showed increasing variability at later times. Percent changes were calculated to assess long-term efficacy, but the limited number of observations and increasing variability from 36 months made subsequent months unevaluable.
| Time | No. of patients | Mean deviation of change in VF [95% CI] | Percent change relative to baseline [95% CI] |
|---|---|---|---|
| Preoperation | 6 | -15.42 [-26.01, -4.83] | |
| 6 months | 6 | -15.27 [-25.51, -5.03] | -5.0% [-19%, 10%] |
| 12 months | 9 | -15.25 [-25.75, -4.75] | 1.0% [-23%, 25%] |
| 24 months | 6 | -15.52 [-24.40, -6.64] | -17.0% [-55%, 21%] |
| 36 months | 3 | -15.24 [-28.28, -2.19] | 3.0% [-47%, 54%] |
| 48 months | 8 | -8.26 [-28.44, 11.9] | -4.0% [-183%, 174%] |
| 60 months | 6 | -14.93 [-27.64, -2.22] | 5.0% [-46%, 55%] |
| 7 years | 8 | -22.67 [-34.52, -10.82] | -39.0% [-142%, 64%] |
| 10 years | 8 | -19.76 [-32.65, -6.87] | -119.0% [-442%, 205%] |
Discussion
The present study analyzed 10-year outcomes in nine patients who underwent MicroShunt implantation, with or without cataract surgery. These 10-year results were generally similar to the 3- and 5-year results reported in the same patient population.
Evaluation of efficacy outcomes showed that the MicroShunt was able to control IOP for up to 10 years, with mean IOP of 12-14 mmHg, beginning 1 year after surgery and remaining constant for up to 10 years. None of these nine patients remained medication-free after 10 years, with medication use after 10 years being similar to medication use at baseline.
The concentration and method of application of MMC may have contributed to the long-term success of the surgical procedure. This dose and method of application may have prevented blockage of the MicroShunt device by fibrosis, thereby contributing to long-term efficacy and safety. Serum factor clearance prior to MMC application is important, as these factors have been shown to interfere with the in vitro antifibrotic activity of MMC on fibroblast cells.
To date, no consensus has been reached about the optimal dose of MMC during glaucoma surgery. A recent analysis of three prospective, open label clinical trials comparing 0.2 and 0.4 mg/ml MMC found that both IOP and glaucoma medication use at year 2 were lower and complete success rate was higher in patients administered 0.4 than 0.2 mg/ml MMC. Although all patients in the present study were administered 0.4 mg/ml topical MMC for 3 minutes via three MMC-soaked LASIK shields, randomized clinical trials in larger numbers of patients are needed to determine the optimal dose, route of administration, and duration of application of MMC.
None of the MicroShunt devices showed signs of visibly apparent degradation 10 years after placement. This may have been due to their construction with SIBS, a polymer designed for long-term use in the body. SIBS has been extensively used as a coating for TAXUS drug-eluting coronary stents. Lack of MicroShunt degradation could not be evaluated directly, however, as these devices remained functional and there were no indications for removal. Nevertheless, the integrity of these devices when needled, as well as evaluation of the proximal tip through the cornea and lack of inflammation at the distal end, indicate a lack of biodegradation in the eye.
Evaluation of safety outcomes in this patient cohort showed that endothelial cell counts remained stable in the operated eyes through 10 years in the nine patients who completed the 10-year study. In contrast, endothelial cell counts in the non-operated eyes decreased 22.3% after 10 years. Leaking bullae formed in 100% of these nine operated eyes during the first year, decreasing to 66.7% after 10 years. Mean visual field deviation worsened slightly in these eyes, but this parameter varied widely.
Conclusion
In conclusion, the present study showed that the MicroShunt is an effective and safe long-term option for the surgical management of primary open-angle glaucoma in this study population. Although several clinical trials have compared MicroShunt implantation with trabeculectomy, additional prospective trials are required to compare their long-term efficacy and safety.
Conflict of Interest Statement:
Juan F. Batlle is a consultant for Santen, Inc. None of the other authors has any conflicts of interest to declare.
Funding Statement:
This trial was sponsored by Santen, Inc.
Acknowledgements: Editorial support was provided by BelMed Professional Resources, with funding by Santen.
References:
2. Jonas JB, Aung T, Bourne RR, Bron AM, Ritch R, Panda-Jonas S. Glaucoma. Lancet. 2017;390(1010 8):2183–2193. doi: 10.1016/S0140-6736(17)31469-1
3. Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006; 90(3):262–267. doi: 10.1136/ bjo.2005.081224
4. Flaxman SR, Bourne RRA, Resnikoff S, et al; Vision Loss Expert Group of the Global Burden of Disease Study. Global causes of blindness and distance vision impairment 1990-2020: a systematic review and meta-analysis. Lancet Glob Health. 2017;5(12):e1221–e1234. doi: 10.1016/S2 214-109X(17)30393-5
5. Mantravadi AV, Vadhar N. Glaucoma. Prim Care. 2015;42(3):437–449. doi: 10.1016/j.pop.2015.05.008
6. Gedde SJ, Vinod K, Wright MM, et al; American Academy of Ophthalmology Preferred Practice Pattern Glaucoma Panel. Primary open-angle glaucoma preferred practice pattern. Ophthalmology. 2021; 128(1):P71–P150. doi: 10.1016/j.ophtha.2020.10.022
7. European Glaucoma Society Terminology and Guidelines for Glaucoma, 4th Edition – Chapter 3: Treatment principles and options Supported by the EGS Foundation: Part 1: Foreword; Introduction; Glossary; Chapter 3 Treatment principles and options. Br J Ophthalmol. 2017;101(6):130–195. doi: 10.1136/bjophthalmol-2016-EGSguideline.003
8. Gedde SJ, Schiffman JC, Feuer WJ, Herndon LW, Brandt JD, Budenz DL; Tube versus Trabeculectomy Study Group. Treatment outcomes in the tube versus trabeculectomy (TVT) study after five years of follow-up. Am J Ophthalmol. 2012;153(5):789–803. doi: 10.1016/j. ajo.2011.10.026
9. Gedde SJ, Feuer WJ, Lim KS, et al; Primary Tube versus Trabeculectomy Study Group. Treatment outcomes in the primary tube versus trabeculectomy study after 5 years. Ophthalmology. 2022;129(12): 1344–1356. doi: 10.1016/j.ophtha.2022.07.003
10. Richter GM, Coleman AL. Minimally invasive glaucoma surgery: current status and future prospects. Clin Ophthalmol. 2016;10:189–206. doi: 10.2147/OPTH.S80490
11. Saheb H. Ahmed II. Micro-invasive glaucoma surgery: current perspectives and future directions. Curr Opin Ophthalmol. 2012;23(2):96–104. doi: 10.1097/ICU.0b013e32834ff1e7
12. Pinchuk L, Riss I, Batlle JF, et al. The development of a micro-shunt made from poly(styrene-block-isobutylene-block-styrene) to treat glaucoma. J Biomed Mater Res B Appl Biomater. 2017;105(1): 211–221. doi: 10.1002/jbm.b.33525
13. Baker ND, Barnebey HS, Moster MR, et al; INN005 Study Group. Ab-externo microshunt versus trabeculectomy in primary open-angle glaucoma: One-year results from a 2-year randomized, multicenter study. Ophthalmology. 2021;128(12): 1710–1721. doi: 10.1016/j.ophtha.2021.05.023
14. Panarelli JF, Moster MR, Garcia-Feijoo J, et al.; INN005 Study Group. Ab-externo microshunt versus trabeculectomy in primary open-angle glaucoma: Two-year results from a randomized, multicenter study. Ophthalmology. 2024;131(3):266–276. doi: 10.1016/j.ophtha.2023.09.023
15. Fili S, Kontopoulou K, Vastardis I, Perdikakis G, Kohlhaas M. PreserFlo™ MicroShunt versus trabeculectomy in patients with moderate to advanced open-angle glaucoma: 12-month follow-up of a single-center prospective study. Cureus. 2022;14(8):e28288. doi: 10.7759/cureus.28288
16. Pillunat KR, Herber R, Haase MA, Jamke M, Jasper CS, Pillunat LE. PRESERFLO™ MicroShunt versus trabeculectomy: first results on efficacy and safety. Acta Ophthalmol. 2022;100(3):e779-e790. doi: 10.1111/aos.14968
17. Fu MX, Normando EM, Luk SMH, et al. MicroShunt versus trabeculectomy for surgical management of glaucoma: a retrospective analysis. J Clin Med. 2022;11(18):5481. doi: 10.339 0/jcm11185481
18. Riss I. A 2-year, single-center study to assess the safety and effectiveness of the MicroShunt in primary open-angle glaucoma. Ophthalmic Res. 2023;66(1):206–217. doi: 10.1159/000526960
19. Beckers HJM, Aptel F, Webers CAB, et al. Safety and effectiveness of the PRESERFLO microshunt in primary open-angle glaucoma: Results from a 2-year multicenter study. Ophthalmol Glaucoma. 2022;5(2):195–209. doi: 10.1016/j.ogla.2021.07.008
20. Batlle JF, Fantes F, Riss I, et al. Three-year follow-up of a novel aqueous humor MicroShunt. J Glaucoma. 2016;25(2):e58–e65. doi: 10.1097/IJG. 0000000000000368
21. Batlle JF, Corona A, Albuquerque R. Long-term results of the PRESERFLO MicroShunt in patients with primary open-angle glaucoma from a single-center nonrandomized study. J Glaucoma. 2021;30 (3):281–286. doi: 10.1097/IJG.0000000000001734
22. Wilkins MR, Occleston NL, Kotecha A, Waters L, Khaw PT. Sponge delivery variables and tissue levels of 5-fluorouracil. Br J Ophthalmol. 2000;84 (1):92¬–97. doi: 10.1136/bjo.84.1.92
23. Dhingra S, Khaw PT. The moorfields safer surgery system. Middle East Afr J Ophthalmol. 2009;16(3): 112¬–115. doi: 10.4103/0974-9233.56220
24. Crowston JG, Wang XY, Khaw PT, Zoellner H, Healey PR. Human serum reduces mitomycin-C cytotoxicity in human Tenon’s fibroblasts. Invest Ophthalmol Vis Sci. 2006;47(3):946–952. doi: 10. 1167/iovs.05-1048
25. Al Habash A, Aljasim LA, Owaidhah O, Edward DP. A review of the efficacy of mitomycin C in glaucoma filtration surgery. Clin Ophthalmol. 2015;9:1945¬–1951. doi: 10.2147/OPTH.S80111
26. Garcia-Feijoo J, Batlle JF, Aptel F, et al. Pooled analysis of three MicroShunt studies in primary open-angle glaucoma evaluating different concentrations of applied mitomycin C.Ophthalmol Ther. 2025, in press. doi: 10.1007/s40123-025-01149-4
27. Pinchuk L. Development of SIBS and the PRESERFLO™ MicroShunt to treat advanced glaucoma: a review. Med Res Arch. 2025;13(2):61 69. doi: 10.18103/mra.v13i2.6169
28. Grube E, Silber S, Hauptmann KE, et al. TAXUS I: six- and twelve-month results from a randomized, double-blind trial on a slow-release paclitaxel-eluting stent for de novo coronary lesions. Circulation. 2003;107(1):38-42. doi: 10.1161/01.cir. 0000047700.58683.a1.




