Genotoxic Assessment via Conjunctival Mucosa in Workers
Suitability of Conjunctival Mucosa Cytological Samples for Genotoxic Assessment: Micronucleus Assay Applied to Workers Exposed to Ionizing Radiation
Cátia Martins Leite Padilha 1,2; Laura Maria Campello Martins 1,4; Lucas Gomes Padilha Filho 4; Anderson Schutz dos Santos 3,4; Nadja de Abreu Carvalho 2; Sérgio Augusto Lopes de Souza 4
OPEN ACCESS
PUBLISHED 31 August 2025
CITATION Padilha, CML., Martins, LMC., et al., 2025. Suitability of Conjunctival Mucosa Cytological Samples for Genotoxic Assessment: Micronucleus Assay Applied to Workers Exposed to Ionizing Radiation. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6819
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6819
ISSN 2375-1924
Abstract
This study investigated an alternative, less invasive approach for implementing genetic biomonitoring in healthcare professionals occupationally exposed to ionizing radiation. The main objectives were to evaluate the suitability of conjunctival mucosa as a cellular sample for the micronucleus assay in occupationally exposed individuals and to contribute to the development of less invasive strategies for occupational genetic monitoring. Using sterile brushes for ocular mucosa sampling, 20 volunteers were recruited from hospital and academic institutions, following strict ethical and clinical criteria. Samples were processed using a cytocentrifuge and stained with Giemsa, allowing morphological analysis under light microscopy. Despite challenges related to low cellularity, structural preservation was sufficient for the identification of micronuclei and other relevant nuclear abnormalities. The results indicate that the methodology is well tolerated, low-cost, technically simple, and feasible for large-scale application, particularly in clinical settings with limited infrastructure. Therefore, conjunctival mucosa may represent a promising matrix for occupational genotoxic screening, offering significant contributions to public health surveillance strategies and the prevention of cumulative genetic effects in exposed professionals.
Keywords:
- Occupational Genotoxicity
- Micronucleus Assay
- Conjunctival Mucosa
- Ionizing Radiation
Introduction
Occupational exposure to ionizing radiation poses a recognized risk to the genomic integrity of healthcare professionals, particularly those involved in diagnostic and therapeutic radiological procedures. Although exposure levels are generally maintained within regulatory limits, studies have shown that chronic low-dose exposures can lead to cumulative genetic damage, with potential long-term health consequences. The understanding of the biological effects of ionizing radiation has been built upon theoretical and experimental models, considering variables such as intensity, frequency, and duration of exposure. The energy transferred by radiation can induce structural alterations in cellular molecules, especially DNA, thereby affecting vital functions and compromising genomic stability.
Despite compliance with regulatory dose limits, increasing evidence suggests that chronic low-dose exposures may induce cumulative genetic alterations with implications for long-term health. Studies involving workers occupationally exposed to ionizing radiation are essential to deepen the understanding of these cumulative effects on genetic material.
Occupationally Exposed Individuals (OEI) in healthcare, particularly those working in diagnostic and interventional radiology, represent a particularly vulnerable group due to their routine handling of radiation sources. While international guidelines establish dose limits and recommend the use of personal protective equipment, the scientific literature highlights critical gaps between recorded doses and the actual extent of cellular damage accumulated over time.
In this context, the use of cytogenetic biomarkers has emerged as an effective strategy for biomonitoring exposed populations. The micronucleus assay (MNA), widely validated in various cell types, enables the detection of genetic damage caused by chromosomal breaks or abnormal mitotic segregation and is considered a sensitive in vivo method for genotoxic assessment.
Traditionally, this assay has been applied to lymphocytes (peripheral blood), as well as cells from oral, nasal, and vaginal mucosa. However, the search for less invasive and more specific alternatives has prompted experimental applications in other mucosal tissues exposed to harmful agents, including ionizing radiation.
The superficial anatomical position of the conjunctival mucosa grants this epithelial tissue unique characteristics of direct exposure to physical agents, including ionizing radiation. Unlike deeper mucosae or hematologic cells, which reflect systemic exposure, conjunctival cells offer a localized assessment of genotoxic damage, especially in areas not fully shielded by protective barriers.
In this regard, the conjunctival mucosa has emerged as a promising matrix. It is a highly regenerative epithelial tissue, easily accessible, and potentially capable of reflecting early cellular damage induced by radiation—particularly in the ocular region, which may still be significantly exposed despite the use of personal protective equipment.
The biological viability of conjunctival mucosa samples remains underexplored, and few studies have evaluated their suitability for micronucleus assay application in occupational settings. This gap underscores the importance of investigating its potential as a tool for genotoxic surveillance, particularly among workers in radiological services, who are subject to continuous and cumulative exposure risks.
Given that the effects of occupational exposure to ionizing radiation do not always manifest clinically in the short term, the implementation of strategies for early detection of cellular damage becomes crucial to safeguarding workers’ long-term health. Accordingly, the objective of this study was to assess the suitability of conjunctival mucosa as a cellular sample for the micronucleus assay in OEI and to contribute to the development of less invasive strategies for occupational genetic biomonitoring.
Methodology
This is a qualitative and descriptive study with an exploratory approach, approved by the Research Ethics Committee involving Human Subjects (Approval Number: 7.190.292).
Twenty volunteers of both sexes (female and male), from the general population, were recruited from higher education institutions and public and private hospitals. All participants were informed about the study objectives and signed the Informed Consent Form prior to inclusion. Eligible participants were aged 18 years or older, presented no signs of ocular or systemic infection at the time of sampling, and had not used any eye or facial cosmetics (such as eyeshadow, eyeliner, mascara, eyebrow products, or foundation) within the 24 hours preceding collection.
The selection of participants did not take prior occupational exposure to ionizing radiation into account at this initial stage, since the primary objective was to verify the technical feasibility of using conjunctival mucosa as a biological matrix for the micronucleus assay. Thus, the inclusion of volunteers from the general population allowed for the assessment of the morphological adequacy and stability of the collected samples, as well as the reproducibility of the proposed technique, regardless of the individuals’ occupational history.
As an additional criterion, participants were instructed to wash their faces with running water prior to the procedure. When necessary, a sterile 0.9% sodium chloride (NaCl) solution was applied directly to the eyes to optimize conjunctival hydration before sampling.
BIOLOGICAL SAMPLE COLLECTION
Sample collection was carried out between February and March 2025, in higher education institutions and hospital units located in the city of Rio de Janeiro, Brazil. All stages of the study—including recruitment, collection, and processing of the samples—were conducted in a controlled environment, with technical support from a qualified professional from the Brazilian National Cancer Institute, in accordance with the operational conditions described in this protocol.
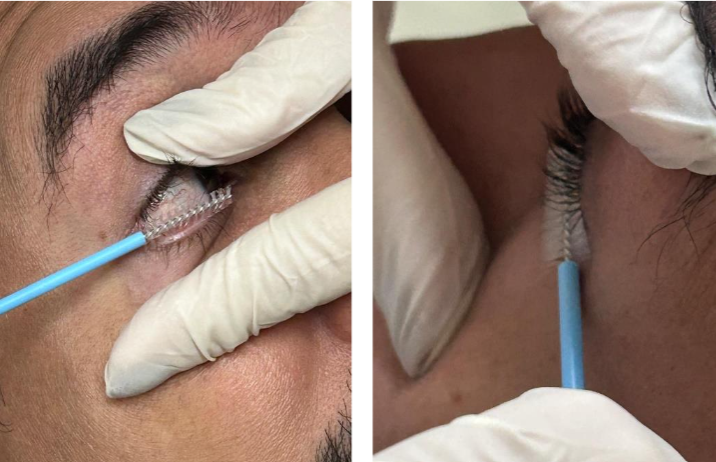
Sample collection was performed using a sterile cervico-vaginal cytobrush (Citobrush®) under adequate illumination. Volunteers were seated with their heads slightly tilted back. Using the dominant hand, a qualified and experienced professional gently rotated the brush 360° over the inferior palpebral conjunctiva, sweeping from the medial to the lateral canthus without causing abrasion.
Samples were collected from both eyes, starting with the right eye, followed by the left. The procedure was standardized to minimize discomfort and maximize the retrieval of viable epithelial cells. Each brush (right and left eye) was immediately immersed in a sterile Falcon tube containing 5 ml of isotonic saline solution (0.9% NaCl) and gently agitated for 30 seconds to facilitate cell detachment.
When same-day processing was not feasible, samples were stored under controlled refrigeration (2–8 °C) for a maximum of 48 hours, in accordance with literature recommendations to preserve cellular morphological integrity.
CYTOLOGICAL PROCESSING
The collected material was homogenized and subjected to cytocentrifugation using glass slides and medium-porosity filter paper at 800 rpm for 5 minutes, to concentrate the cellular content at the center of the slide. After air drying, the smears were fixed in absolute methanol for 10 minutes. Staining was performed with 5% Giemsa solution in phosphate buffer (pH 6.8) for 10 minutes, followed by rinsing in distilled water and air drying at room temperature. The slides were mounted with Canada balsam and coverslips (24 × 50 mm) and allowed to rest for 24 hours prior to analysis.
MICROSCOPIC EVALUATION
Analysis was performed using a binocular light microscope with 10x and 40x objectives. Sample suitability for the micronucleus assay was assessed based on the following parameters:
- Minimum presence of 1,000 well-preserved epithelial cells (intact nucleus and cytoplasm, without overlap or lysis).
- Proportion of epithelial cells relative to contaminants and/or artifacts (e.g., leukocytes, mucus, cellular debris).
- Homogeneous cellular distribution on the slide.
- Adequate staining quality, absence of precipitates, and preserved morphological integrity after staining.
Results
The conjunctival brushing technique employed in this study proved to be operationally feasible, with no refusal reported among participants.
Regarding cellularity, it was necessary to consider the total number of cells obtained from both eyes (left and right) to ensure quantitatively relevant analysis. Although the main challenge was obtaining samples with sufficient cellularity to meet the minimum criteria for morphological evaluation, as described in the literature, most cases showed well-preserved epithelial cells, with intact nuclei and cytoplasm, as well as homogeneous distribution across the slide.
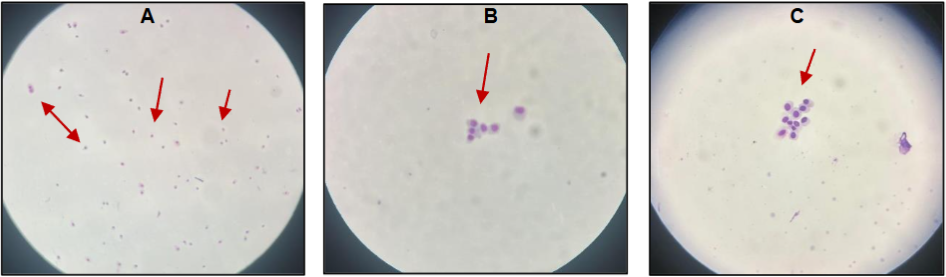
The laboratory protocol adopted in this study enabled the preparation of slides with well-defined cytoplasmic and nuclear features. The absence of staining artifacts, precipitates, or cellular overlap contributed to the accuracy in identifying nuclear alterations such as micronuclei, nuclear budding, and cytotoxicity markers (karyolysis, karyorrhexis, and pyknosis). These findings support the suitability of the methodology for detecting early genotoxic damage, even in samples obtained through less invasive techniques.
The approach used in this study demonstrated substantial advantages over the conjunctival impression technique described in the literature, which requires the use of topical anesthetics and high-cost materials. The simplicity of the sampling procedure, combined with the high acceptance rate among volunteers, highlights its potential for large-scale application, particularly in occupationally exposed populations. Moreover, its feasibility in clinical settings with limited resources underscores its value as a screening tool in genomic surveillance programs.
Discussion
Chronic occupational exposure to ionizing radiation, even at levels deemed low and within regulatory thresholds, has been associated with subtle but cumulative genetic effects that do not always manifest clinically in the short term. Recent studies demonstrate that ionizing radiation can induce DNA double-strand breaks, chromosomal rearrangements, and progressive genomic instability, particularly in rapidly renewing tissues such as epithelial surfaces.
These findings underscore the importance of continuous cytogenetic monitoring, even among workers who do not present with acute clinical signs of exposure. The conjunctival mucosa, in turn, has gained increasing attention in experimental and clinical studies as a suitable epithelial matrix for genotoxic surveillance. Its superficial anatomical position, high cellular turnover rate, and direct interface with the external environment favor the early detection of nuclear alterations such as micronuclei and nuclear budding.
Several studies employing the micronucleus assay in epithelial cells adhere to morphological criteria that include, in addition to the identification of micronuclei per se, the observation of other nuclear alterations associated with genotoxicity (e.g., nuclear budding) and cytotoxicity (e.g., karyorrhexis, karyolysis, and pyknosis). When these analyses are supported by rigorous inter-observer quality control strategies, they provide robustness and reliability to data interpretation—even in cases where the total number of cells analyzed does not meet the thresholds recommended by classical protocols.
In this sense, the careful application of these markers can partially compensate for the limited cellularity observed in certain conjunctival samples, enabling effective qualitative analysis with sufficient sensitivity to detect early nuclear damage.
The relevance of detecting cytogenetic damage in professionals chronically exposed to ionizing radiation justifies a judicious flexibility of certain quantitative standards, especially when the analyzed sample directly reflects the anatomical route of exposure as is the case with conjunctival epithelium in radiology workers. Therefore, the growing need for comparative studies lies in the ability to detect morphological alterations even with lower cellularity. Combined with the lower invasiveness of the collection method and its potential for large-scale application, these features support the use of conjunctival mucosa as a viable and scientifically sound matrix for occupational health and genomic surveillance.
Findings by Leonardi et al. (2020), who used buccal mucosa cells to assess early genetic damage in construction workers, reinforce the feasibility of using exfoliated epithelia as biological material for the micronucleus assay. Similar to buccal mucosa, conjunctival mucosa displays favorable characteristics such as a high renewal rate, accessibility, and direct interface with environmental agents, qualifying it as a promising material for genotoxic biomonitoring in occupational contexts.
In a study evaluating the bulbar conjunctival cytology of 33 healthy individuals and 16 patients with neoplastic or inflammatory diseases, the sample collection was performed using the impression cytology technique on the ocular surface. Although this method provided adequate cellularity for micronucleus analysis, it required the use of topical anesthetic and additional handling precautions—factors that may pose practical limitations, particularly in large-scale or occupational studies.
Typically, protocols for ocular genotoxicity studies involve the use of nitrocellulose membrane fragments, polycarbonate filters, sterilized cellulose paper, or specialized slides such as Biopore™ or Millipore™, with pore sizes ranging from 0.2 to 0.45 µm. These procedures also require sterile forceps and topical ocular anesthetics (e.g., 0.5% proxymetacaine hydrochloride eye drops) to minimize discomfort and blinking reflexes.
In contrast, the technique adopted in the present study proved to be less invasive, simpler to perform, and better accepted by participants, establishing itself as a feasible and more cooperative alternative for genotoxic biomonitoring in populations exposed to ionizing radiation.
Conclusion
The findings of this study demonstrate that conjunctival mucosa can be considered a viable alternative for the application of the micronucleus assay in workers chronically exposed to ionizing radiation, as well as to other deleterious agents. Although the cellularity obtained was lower compared to other commonly used epithelia, such as buccal or vaginal mucosa—mainly due to the fragility of ocular tissue and the need for less aggressive sampling techniques—it was possible to obtain samples with sufficient morphological preservation for reliable cellular analysis.
The methodology employed, based on conjunctival brushing using a sterile instrument without the use of topical anesthetics, proved to be technically simple, well tolerated by participants, and operationally suitable for clinical environments with limited infrastructure. These attributes support its applicability in large-scale genotoxic screening strategies.
In addition to enabling early detection of cytogenetic damage, the approach adopted in this study has the potential to contribute valuable insights for the improvement of occupational protection measures and the development of public policies aimed at preserving the genomic health of radiology professionals. Continued longitudinal investigations may further elucidate the relationship between micronucleus frequency, cumulative exposure dose, and the effectiveness of radiological protection practices.
In conclusion, the application of the micronucleus assay to conjunctival mucosa represents a promising and accessible strategy for the biological monitoring of occupational risks associated with ionizing radiation, with significant potential impact in preventing cumulative genetic effects in exposed populations.
Acknowledgments
The authors acknowledge the institutional support provided by Dr. Verônica Goulart Moreira, Pathologist and Head of the Division of Pathology (DIPAT) at the Brazilian National Cancer Institute (INCA), which enabled the technical collaboration necessary for the development of this study. Special recognition is extended to Eduardo Pereira Ribeiro Lima and Fabíola Alves Soares, members of the DIPAT/INCA technical team, whose expertise and dedicated contribution to the processing and standardization of the cytological samples were essential to the methodological rigor and analytical reliability of this research.
Bibliography
- Batista VMD, Bernardo MO, Morgado F, Almeida FA de. Radiological protection in the perspective of health professionals exposed to radiation. Rev Bras Enferm. 2019 Feb;72:9–16.
- Kuchi Bhotla H, Balasubramanian B, Rengasamy KRR, Arumugam VA, Alagamuthu KK, Chithravel V, Chaudhary A, Alanazi AM, Pappuswamy M, Meyyazhagan A. Genotoxic repercussion of high-intensity radiation (x-rays) on hospital radiographers. Environ Mol Mutagen. 2023 Feb;64(2):123–131. PMID: 36541415
- Okuno E. Efeitos biológicos das radiações ionizantes: acidente radiológico de Goiânia. Estud Av. Instituto de Estudos Avançados da Universidade de São Paulo; 2013;27:185–200.
- Okuno E. Radiação: Efeitos, Riscos e Benefícios. 2018; Available from: https://s3-sa-east-1.amazonaws.com/ofitexto.arquivos/Radiacao-Efeitos-Riscos-e-Benefícios-DEG.pdf
- Çobanoğlu H, Çayır A. Occupational exposure to radiation among health workers: Genome integrity and predictors of exposure. Mutat Res Toxicol Environ Mutagen. 2024 Jan;893:503726.
- Antonio EL, Nascimento AJ do, Lima AAS de, Leonart MSS, Fernandes Â. GENOTOXICIDADE E CITOTOXICIDADE DOS RAIOS X EM CRIANÇAS SUBMETIDAS À RADIOGRAFIA PANORÂMICA. Rev Paul Pediatr. Sociedade de Pediatria de São Paulo; 2017 Jul 20;35:296–301.
- Gallo AM, Lima FAC de, Reis LM dos, Cremer E. Exposição Ocupacional À Radiações Ionizantes Sob A Ótica De Profissionais De Enfermagem Em Hemodinâmica. Rev Rene. 2013;14(1):109–119.
- Navarro MVT, Leite HJD, Alexandrino J da C, Costa EA. Controle de riscos à saúde em radiodiagnóstico: uma perspectiva histórica. História Ciênc Saúde-Manguinhos. 2008 Dec;15(4):1039–1047.
- Higgins A. Measurement of occupational doses of ionising radiation to the lens of the eyes of interventional radiologists. J Radiol Prot. 2016 Mar;36(1):74–92.
- Leyton F, Canevaro L, Dourado A, Castello H, Bacelar A, Navarro MT, Vañó E, Nogueira M do S, Batista WO, Furquim TAC, Lykawka R, Melo CS, Borges F, Rodrigues B. Riscos da Radiação X e a Importância da Proteção Radiológica na Cardiologia Intervencionista: Uma Revisão Sistemática. Rev Bras Cardiol Invasiva. Sociedade Brasileira de Hemodinâmica e Cardiologia Intervencionista – SBHCI; 2014 Mar;22:87–98.
- Vaiserman A, Koliada A, Zabuga O, Socol Y. Health Impacts of Low-Dose Ionizing Radiation: Current Scientific Debates and Regulatory Issues. Dose-Response. 2018 Jul 1;16(3):1559325818796331.
- Padilha CML, Filho LGP, Ferreira R da P, Souza SAL de. Cytopathology and the Micronucleus Test in Monitoring Post-Radiotherapy Patients for Cervical Cancer. Med Res Arch [Internet]. 2023 Oct 25 [cited 2024 Aug 8];11(10). Available from: https://esmed.org/MRA/mra/article/view/4555
- Leonardi S, Poma AM, Colafarina S, D’Aloisio F, Scatigna M, Zarivi O, Mastrantonio R, Tobia L, Fabiani L. Early genotoxic damage through micronucleus test in exfoliated buccal cells and occupational dust exposure in construction workers: a cross-sectional study in L’Aquila, Italy. Ecotoxicol Environ Saf. 2020 Oct 15;203:110989. PMID: 32888600
- Thomas P, Holland N, Bolognesi C, Kirsch-Volders M, Bonassi S, Zeiger E, Knasmueller S, Fenech M. Buccal micronucleus cytome assay. Nat Protoc. 2009 Jun;4(6):825–837.
- Padilha C. Avaliação Cito-Oncótica de Pacientes com Câncer de Colo Uterino Submetidas a Radioterapia. [Departamento de Radiologia]: UFRJ. Faculdade de Medicina; 2021.
- Jirsova K, Juklova K, Alfakih A, Filipec M. Presence of snake-like chromatin in epithelial cells of keratoconjunctivitis sicca followed by a large number of micronuclei. Acta Cytol. 2007;541–6.
- Silva RCG, Figueirêdo RDPV, Silva ACO, Lima CEQ, Oliveira SR, Peres AL. Cytopathologic follow-up of women with cervical cancer post-radiotherapy: case series. J Bras Patol E Med Lab [Internet]. 2018 [cited 2023 Aug 11];54(2). Available from: http://www.gnresearch.org/doi/10.5935/1676-2444.20180018
- Barros JDN, Almeida SRAD, Lowen MS, Cunha MCD, Gomes JÁP. Impression cytology in the evaluation of ocular surface tumors: review article. Arq Bras Oftalmol. 2015;78(2):126–132.
- Drummond RL, Rhoden CR, Lubianca Neto JF, Fleck A da S, Padoin RCPK, Amantéa SL. Micronucleus count in nasal epithelial cells from patients with chronic rhinosinusitis and polyps. Braz J Otorhinolaryngol. Associação Brasileira de Otorrinolaringologia e Cirurgia Cérvico-Facial.; 2020 Dec 11;86:743–747.
- Bannier-Hélaouët M, Korving J, Ma Z, Begthel H, Giladi A, Lamers MM, Van De Wetering WJ, Yawata N, Yawata M, LaPointe VLS, Dickman MM, Kalmann R, Imhoff SM, Van Es JH, López-Iglesias C, Peters PJ, Haagmans BL, Wu W, Clevers H. Human conjunctiva organoids to study ocular surface homeostasis and disease. Cell Stem Cell. Elsevier BV; 2024 Feb;31(2):227-243.e12.
- Branisteanu D, Stoleriu G, Branisteanu D, Boda D, Branisteanu C, Maranduca M, Moraru A, Stanca H, Zemba M, Balta F. Ocular cicatricial pemphigoid (Review). Exp Ther Med [Internet]. Spandidos Publications; 2020 Jul 7 [cited 2025 Jul 9]; Available from: http://www.spandidos-publications.com/10.3892/etm.2020.8972
- Blakely EA, Kleiman NJ, Neriishi K, Chodick G, Chylack LT, Cucinotta FA, Minamoto A, Nakashima E, Kumagami T, Kitaoka T, Kiuchi Y, Chang P, Fujii N, Shore RE. Radiation Cataractogenesis: Epidemiology and Biology. Radiat Res. 2010 May;173(5):709–717.
- Silva RR, De Carli JP, Collares K, Vanini J, Presotto JS, Vargas JE, Loguercio AD, Benetti P. Reply to the opinion paper: is micronucleus test a suitable method for monitoring oral mucosa exposed to dental bleachings in smokers? Arch Toxicol. 2023 Jul;97(7):2031–2033.
- Wojcik A, Kowalska M, Bouzyk E, Buraczewska I, Kobialko G, Jarocewicz N, Szumiel I. Validation of the micronucleus-centromere assay for biological dosimetry. Genet Mol Biol. 2000 Dec;23(4):1083–1085.
- Valente D, Costa-Amaral IC, Carvalho LVBD, Santos MVCD, Castro VSD, Rodrigues DDRF, Falco AD, Silva CB, Nogueira SM, Gonçalves ES, Moreira JC, André LC, Teixeira LR, Sarcinelli PDN, Sisenando HA, Oliveira MSD, Perini JA, Mattos RDCODC, Larentis AL. Utilização de biomarcadores de genotoxicidade e expressão gênica na avaliação de trabalhadores de postos de combustíveis expostos a vapores de gasolina. Rev Bras Saúde Ocupacional [Internet]. 2017 [cited 2024 May 5];42(suppl 1). Available from: http://www.scielo.br/scielo.php?script=sci_arttext&id=S0303-76572017001000402&lng=pt&tlng=pt
- Brand CI, Fontana RT, Santos AV dos. A saúde do trabalhador em radiologia: algumas considerações. Texto Contexto – Enferm. 2011 Mar;20(1):68–75.
- Tolbert PE, Shy CM, Allen JW. Micronuclei and other nuclear anomalies in buccal smears: methods development. Mutat Res Mutagen Relat Subj. 1992 Feb;271(1):69–77.
- Méndez NP, Azevedo MG, Cargnin LS, Seibel MP, Silva AFD, Franceschini MEM, Rocha RS, Pigatto JAT. Morfologia das células endoteliais corneanas bovinas obtidas com alizarina vermelha e microscopia óptica. Ciênc Anim Bras [Internet]. FapUNIFESP (SciELO); 2024 [cited 2025 Jul 9];25. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&id=S1809-68912024000100214&tlng=pt
- Willegaignon J, Fernandes SCP, Pelissoni RA, Coura-Filho GB, Sapienza MT, Buchpiguel CA. Radiation safety measures in diagnostic nuclear medicine, based on the potential radiation dose emitted by radioactive patients. Radiol Bras. Publicação do Colégio Brasileiro de Radiologia e Diagnóstico por Imagem; 2023 Mar 13;56:13–20.
- Ganesan N, Phansalkar M, Ambroise M, Varghese R. Validating micronucleus score in effusion fluids. J Cytol. 2017;34(4):193.
- Bhattathiri VN, Bindu L, Remani P, Chandralekha B, Davis CA, Nair MK. Serial cytological assay of micronucleus induction: a new tool to predict human cancer radiosensitivity. Radiother Oncol. 1996 Nov 1;41(2):139–142.
- Ferreira MDCM, Nogueira MC, Ferreira LDCM, Bustamante-Teixeira MT. Early detection and prevention of cervical cancer: knowledge, attitudes and practices of FHS professionals. Ciênc Saúde Coletiva. 2022 Jun;27(6):2291–2302.
- Tariq F. Allergic Conjunctivitis: Review of Current Types, Treatments, and Trends. Life. MDPI AG; 2024 May 21;14(6):650.

