MMP-9 and CYR61 as Biomarkers in Breast Cancer Subtypes
Circulating MMP-9 and CYR61 Levels in HER2-Positive and -Negative Breast Cancer Subtypes
¹ Karimova A. Leyla, ² Azizova İ. Gulnara, ³ ⁻ ⁴ Melikova A. Leylakhanim, ² Shahverdiyeva J. Ilaha
¹ Baku State University, Baku, Azerbaijan
² Azerbaijan Medical University, Baku Azerbaijan
³ Azerbaijan Republic Ministry of Health National Centre of Oncology, Baku, Azerbaijan
⁴ Ministry of Science and Education, Institute of Biophysics, Baku, Azerbaijan
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION Leyla, KA., Gulnara, AI., et al., 2025. Circulating MMP-9 and CYR61 Levels in HER2-Positive and -Negative Breast Cancer Subtypes. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6762
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i8.6762
ISSN 2375-1924
ABSTRACT
Breast cancer is the most commonly diagnosed cancer in women globally and a leading cause of cancer-related deaths. In Azerbaijan, breast cancer cases have steadily increased, with significant numbers recorded between 2020 and 2023. The human epidermal growth factor receptor 2 (HER2) gene is central in breast cancer pathology, particularly in aggressive forms with HER2 overexpression. However, emerging evidence shows that HER2 mutations may also occur in HER2-negative cases, potentially impacting prognosis and therapeutic outcomes.
Aim: This study investigates HER2 gene expression and mutation status in HER2-negative breast cancer cases and analyzes circulating matrix proteins (MMP-7, MMP-9, CYR61) in different breast cancer subtypes.
Methods: A total of 74 women aged 30–71 years with confirmed BC were enrolled. Patients were categorized as HER2-positive (n=33), HER2-negative (n=33), and triple-negative (n=8). HER2 mutation testing was conducted using AmoyDx kits via real-time PCR. Serum levels of MMP-7, MMP-9, and CYR61 were measured and statistically analyzed.
Results: No HER2 mutations were found in HER2-negative patients, supporting prior research indicating the rarity of such mutations in these subtypes. MMP-9 was significantly reduced in all breast cancer groups (p<0.001), while CYR61 levels were markedly elevated, with an inverse correlation observed between MMP-9 and CYR61 (r = -0.314, p < 0.003).
Conclusion: HER2 mutations are uncommon in HER2-negative breast cancers. Serum MMP-9 and CYR61 can serve as valuable diagnostic markers across BC subtypes. The findings support further exploration into alternative molecular targets beyond HER2 for effective treatment strategies.
Keywords: breast cancer; breast cancer subtypes; Her2-negative breast cancer; Her2-positive breast cancer; Her2 gene mutation, circulating matrix proteins: MMP-7, MMP-9 and CYR61; non-invasive biomarkers
INTRODUCTION
According to the World Health Organization (WHO), oncological diseases have remained among the leading causes of morbidity and mortality worldwide over the past decade. Statistical data demonstrate that breast cancer (BC) has shown the highest increase in incidence rates in recent years, currently ranking as the second leading cause of cancer-related deaths among women globally.
In Azerbaijan, BC has become the most commonly diagnosed cancer among women, with a steadily increasing incidence. According to the State Statistical Committee of the Republic of Azerbaijan, the number of breast cancer cases rose from 1,960 in 2020 to 2,638 in 2021, with a slight decrease to 2,541 in 2023. In total, 13,666 new malignant tumor cases were diagnosed for the first time in 2023, while the total number of registered cancer patients reached 69,435. Breast cancer alone accounted for 18% of all newly diagnosed cancer cases in 2023—compared to 16.9% in 2020, 19.3% in 2021, and 18.4% in 2022.
The etiology of breast cancer involves multiple factors. Without early detection and appropriate treatment, the disease can progress aggressively, leading to metastasis and, ultimately, fatal outcomes.
The HER-2 (Human Epidermal Growth Factor Receptor 2) gene plays a crucial role in the development and progression of breast cancer. The erbB2 gene, found on the short arm of chromosome 17, was cloned in 1983. It encodes HER2, a transmembrane glycoprotein weighing 185 kDa, with three functional domains: extracellular, transmembrane, and intracellular, the latter possessing tyrosine kinase activity. HER2 is part of the epidermal growth factor receptor (EGFR) family, which includes four receptor tyrosine kinases. Typically, HER2 gene mutations are associated with HER-2 positive breast cancer, which is characterized by the overexpression or amplification of the HER-2 protein, leading to aggressive tumor growth. However, our research has begun to focus on HER-2 mutations in HER-2 negative breast cancer, where the HER-2 gene does not show amplification, but mutations still occur, potentially influencing cancer behavior and treatment responses.
Studying HER-2 mutations in HER-2 negative breast cancer is an emerging and crucial area of cancer research, offering potential for more personalized treatment approaches, further understanding of the molecular mechanisms behind cancer development. This issue is particularly timely due to advancements in genomics and precision medicine, which are transforming cancer treatment paradigms.
Matrix metalloproteinases (MMPs), also known as matrixins, are proteolytic enzymes that remodel the extracellular matrix. They are involved in physiological processes such as morphogenesis, tissue regeneration, and reproduction, as well as in pathologies like arthritis, cancer, and cardiovascular disease. Moreover, matrix metalloproteinases (MMPs) are a group of zinc-dependent endopeptidases that degrade various components of the extracellular matrix (ECM). MMPs are crucial in tumor invasion and metastasis by facilitating the breakdown of basement membranes and ECM, thus enabling cancer cell migration. Among them, MMP-2 and MMP-9 are particularly implicated in breast cancer metastasis.
Importantly, numerous studies have revealed a functional link between HER2 expression and MMP activity. HER2 overexpression in breast cancer tissues has been shown to upregulate the expression of MMP-9 through the activation of MAPK and PI3K/Akt pathways, thereby promoting tumor cell invasion and metastasis. Additionally, HER2 signaling may lead to increased secretion of MMP-2 and MMP-14, further enhancing ECM remodeling and angiogenesis.
Emerging evidence suggests that this upregulation is not only restricted to tumor tissues but is also reflected in circulating levels of MMPs in the blood of breast cancer patients. Studies have reported elevated serum or plasma concentrations of MMP-2 and MMP-9 in HER2-positive patients compared to HER2-negative counterparts, indicating a systemic spillover effect potentially mediated by tumor-derived exosomes or cytokine signaling. These circulating MMPs may serve as non-invasive surrogate biomarkers for tumor aggressiveness and HER2-driven activity.
Another molecule of interest is CYR61 (Cysteine-rich angiogenic inducer 61), an extracellular matrix-associated protein known to regulate cell adhesion, migration, proliferation, and apoptosis. Studies suggest that CYR61 may act downstream of HER2 signaling and synergize with MMPs to enhance tumor progression. The combined dysregulation of HER2 and matrix-related proteins like MMPs and CYR61 may represent a cooperative oncogenic network that significantly contributes to breast cancer invasiveness and poor prognosis. These findings underscore the importance of investigating HER2 expression in tissue alongside circulating matrix protein levels, particularly MMPs and CYR61, to better understand their molecular interplay and clinical significance in breast cancer pathogenesis.
The goal of the study is to investigate the expression levels of the HER2 gene in breast cancer tissues and the alterations of matrix metalloproteinases (MMPs) in patients’ blood serum. By elucidating these molecular interactions, this study aims to contribute to a more comprehensive understanding of breast cancer biology and to identify potential targets for individualized therapy, particularly in HER2-negative subtypes where conventional HER2-targeted treatments are ineffective.
Patients, Material and Methods
Patients
The results of a prospective study conducted on 74 women aged 30-71 with BC who visited the National Oncology Center in 2023-2024 are presented. Immunohistochemical (IHC) analysis methods were used to examine the biotypes of breast cancer. Based on the results, 8 women were diagnosed with triple-negative phenotype, 33 women with HER-2 positive, and 33 women with HER-2 negative BC. Concentrations of MMP-7, MMP-9 and CYR-61 matrix-related proteins were measured in all enrolled breast cancer patients. For control was collected blood serum (n=15) in health pupils.
Sample Collection and Nucleic Acid Extraction
Formalin-fixed paraffin-embedded (FFPE) tumor tissue blocks and peripheral blood samples were collected for molecular analysis. DNA and RNA were extracted from FFPE samples using the QIAamp DNA/RNA FFPE Tissue Kit (Qiagen, Cat. No. 56404). Genomic DNA was isolated from peripheral blood using the QIAamp Genomic DNA Extraction Kit (Qiagen, Cat. No. 56304).
HER2 Mutation Analysis
HER2 mutation testing was performed using the AmoyDx HER2 Mutation Detection Kit-24 (RFF: 8.01.0232). This assay is capable of detecting HER2 mutations in exon 20, including A775_G776insYVMA, M774_A775insAYVM, G776>VC, G776R, G776C, P780_Y781insGSP, and V777L, as well as exon 19 mutation L755P.
HER2 Gene Expression Analysis
In addition to mutation screening, HER2 gene expression levels were quantitatively assessed in FFPE tumor samples using SYBR Green-based real-time PCR and the ΔCt method. GAPDH was used as the endogenous control. The ΔCt values were calculated by subtracting the Ct value of GAPDH from the Ct value of HER2. Lower ΔCt values indicated higher HER2 expression. HER2-positive tumors demonstrated significantly lower ΔCt values (mean ΔCt = 4.2 ± 1.1) compared to HER2-negative tumors (mean ΔCt = 9.6 ± 1.3), reflecting increased HER2 mRNA expression (p < 0.001, unpaired t-test). The primers for HER2 genes were designed based on their reference nucleotide sequences retrieved from the GenBank database of the National Center for Biotechnology Information (NCBI, USA) https://www.ncbi.nlm.nih.gov/genbank. Primer specificity was confirmed using the Primer-BLAST tool https://www.ncbi.nlm.nih.gov/tools/primer-blast.
Primer sequences used:
HER2 Forward: 5′-ATATACCATGGCGATGCCCCGGGGGTCCTG-3′
HER2 Reverse: 5′-TTATACTCGAGCACTGGCACGTCCAGACC-3′
These molecular findings are consistent with the immunohistochemical HER2 classification and support the validity of using qPCR as a complementary diagnostic tool for HER2 expression profiling.
Biochemical Analysis
For biochemical analysis were collected 2 ml blood in tubes with anticoagulant (K3-EDTA). The samples were gently mixed to ensure proper. The concentrations of MMP-7, MMP-9 and CYR-61 in blood serum were determined using the Quantikine ELISA immunoassay method with reagent kits provided by Bio-Techne, R&D Systems (USA). The obtained results were measured using an automated immunoassay analyzer at a wavelength range of 450–630 nm.
Statistical Analysis
Statistical analyses were conducted using both parametric and non-parametric methods. Parametric tests included the independent samples t-test and Bonferroni correction, while the Wilcoxon–Mann–Whitney test was used for non-parametric comparisons. A p-value < 0.05 was considered statistically significant.
Results
Our results indicate an absence of detectable HER-2 mutations in patients with HER-2 negative breast cancer. MMP-7 levels in all BC subgroups were lower than in the control group but statistically significant differences were observed only using the Wilcoxon-Mann-Whitney test (pU<0.05). MMP-9 levels were significantly lower in BC patients compared to controls (53.3 vs. 486.9 ng/mL, p<0.001). CYR61 was elevated in all cancer subtypes compared to controls: HER2-positive (406.6 ng/mL), HER2-negative (277.5 ng/mL), triple-negative (243.4 ng/mL) vs. control group (13.3 ng/mL).
| MMP-7, ng/ml | MMP-9, ng/ml | CYR-61, pg/ml | |
|---|---|---|---|
| Her2-positive, n=33 | 5.7 (0.2 – 32.2) | 37.5 (8.0 – 140.0) | 406.6 (0 – 5575.0) |
| Her2-negative, n=33 | 3.0 (0.2 – 25.4) | 73.1 (8.0 – 688.0) | 277.5 (0 – 784.0) |
| Triple-negative, n=8 | 5.7 (0.4 – 29.2) | 37.0 (12.0 – 72.0) | 243.4 (37.0 – 683.0) |
| Total BC, n=74 | 4.5 (0.2 – 32.2) | 53.3 (8.0 – 688.0)* | 331.4 (0 – 5575.0)* |
| Control, n=15 | 3.1 (1.8 – 4.1) | 486.9 (269.0 – 884.0) | 13.3 (0 – 149.0) |
Note: * – Statistically significant difference compared to the control group at p<0.05 level;
Matrix metalloproteinase 7 (MMP-7) levels were found to be slightly reduced in breast cancer (BC) patient groups compared to healthy controls. However, this reduction reached statistical significance only under non-parametric testing (Wilcoxon-Mann-Whitney test, p < 0.05), suggesting that while there may be a modest downregulation of MMP-7 in BC patients, the evidence is not sufficiently robust across all statistical methods. This indicates a potential but limited role for MMP-7 alterations in the pathophysiology of breast cancer that warrants further investigation.
In contrast, matrix metalloproteinase 9 (MMP-9) levels were significantly decreased in BC patients relative to controls, with mean serum concentrations of 53.3 ng/mL and 486.9 ng/mL, respectively (p<0.001). Given the well-established role of MMP-9 in extracellular matrix degradation and facilitation of tumor invasion and metastasis, this marked reduction in circulating MMP-9 may reflect complex tumor-host interactions or systemic regulatory mechanisms affecting MMP-9 expression or release. These findings highlight the importance of MMP-9 as a potential biomarker for breast cancer and suggest that altered serum levels could have diagnostic or prognostic relevance.
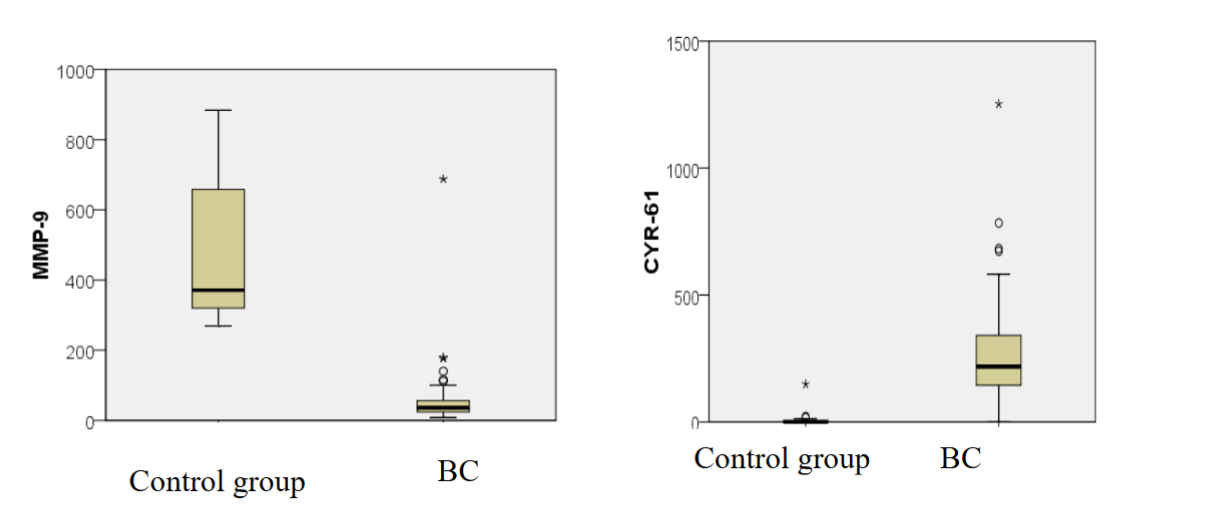
Conversely, levels of CYR61, an extracellular matrix-associated protein involved in cell adhesion, migration, proliferation, and apoptosis, were significantly elevated in all breast cancer subtypes analyzed. Specifically, mean serum CYR61 concentrations were 406.6 ng/mL in HER2-positive patients, 277.5 ng/mL in HER2-negative patients, and 243.4 ng/mL in those with triple-negative breast cancer, compared to 13.3 ng/mL in healthy controls (p < 0.001). This substantial increase underscores CYR61’s potential role in breast cancer progression and its utility as a circulating biomarker indicative of tumor activity.
Importantly, a notable inverse correlation was identified between MMP-9 and CYR61 levels (r = -0.314, p < 0.003), suggesting a functional interplay between these two biomarkers. This negative association implies that as CYR61 levels rise, MMP-9 levels tend to decrease, highlighting a possible regulatory or compensatory mechanism within the tumor microenvironment. The reciprocal relationship between MMP-9 and CYR61 may have significant implications for understanding the molecular pathways involved in breast cancer progression and could inform the development of combined biomarker panels to improve diagnostic accuracy and therapeutic monitoring.
Collectively, these results demonstrate significant alterations in serum levels of MMP-9 and CYR61 in breast cancer patients, reflecting their involvement in tumor biology and their potential clinical relevance as non-invasive biomarkers. Further studies are warranted to elucidate the mechanistic basis of their interaction and to validate their prognostic and therapeutic significance.
Despite no major differences among cancer subtypes, the difference between all BC patients and the control group was statistically significant for both MMP-9 and CYR61 (p<0.001). A negative correlation was found between MMP-9 and CYR61 levels (r = -0.314, p < 0.003), suggesting an inverse relationship between these markers.
DISCUSSION
Our findings, which show no detectable mutations in HER-2 in the HER-2 negative breast cancer patients, are consistent with several studies in the literature. For example, research by Huang et al. (2021), published in Breast Cancer Research, found that mutations in the HER-2 gene were rare in HER-2 negative breast cancers. Their study focused on patients with estrogen receptor-positive (ER+) and triple-negative breast cancers, revealing no significant HER-2 mutations in these groups. Similarly, Lo et al. in BMC Cancer observed that only a small fraction (2%) of HER-2 negative, estrogen receptor-positive breast cancer samples showed slight amplifications of the EGFR gene, further suggesting the absence of significant HER-2 alterations in these subtypes.
Moreover, Wang et al. (2021), in their study published in Journal of Clinical Oncology, reported that in triple-negative breast cancers (TNBC), which often overlaps with HER-2 negative subtypes, EGFR mutations were more commonly found, but HER-2 mutations were not observed. This finding suggests that while EGFR mutations may play a role in some aggressive breast cancer cases, HER-2 mutations are not a frequent event in HER-2 negative breast cancers.
Despite the potential implications of HER-2 mutations in treatment resistance, especially in relation to trastuzumab or lapatinib therapy, our study contributes to the growing body of evidence suggesting that mutations in HER-2 are not a major driver in HER-2 negative breast cancer. This contrasts with the commonly held view that HER-2 mutations could provide resistance to therapies targeting the HER-2 receptor. Instead, other pathways, such as mutations in the PIK3CA gene, EGFR amplifications, and PI3K-AKT-mTOR signaling pathways, may be more relevant in driving resistance in these cases, as highlighted by studies from Chung et al. (2018) in Cancer Research.
The lack of HER-2 mutations in our cohort also raises the question of alternative therapeutic strategies for HER-2 negative breast cancer. Since HER-2 mutations are not implicated in driving the malignancy in our study, future research should focus on identifying other molecular biomarkers that could predict therapeutic response and help guide treatment decisions. This includes examining the role of EGFR mutations, PI3K pathway alterations, and mutations in tumor suppressor genes, all of which have shown to be more prominent in the absence of HER-2 amplification or overexpression. Our findings support the conclusion that HER-2 mutations are infrequent in HER-2 negative breast cancer, particularly in estrogen receptor-positive (ER+) and triple-negative (TNBC) subtypes. These results are consistent with prior research that has highlighted the limited role of HER-2 mutations in driving tumorigenesis in HER-2 negative breast cancer. Further research should focus on identifying other key mutations or alterations that may play a more significant role in the progression of HER-2 negative breast cancer and resistance to targeted therapies.
CONCLUSION
Serum levels of MMP-9 and CYR61 are significantly altered in breast cancer patients and can serve as valuable diagnostic biomarkers regardless of tumor subtype. These findings emphasize the role of extracellular matrix-related proteins in the early detection of breast cancer and merit further exploration across other cancer types.
REFERENCES
- Ren W, Chen M, Qiao Y, Zhao F. Global guidelines for breast cancer screening: A systematic review. Breast. 2022; 64:85–99.
- Deng T, Zi H, Guo XP, Luo LS, Yang YL, Hou JX, et al. Global, Regional, and National Burden of Breast Cancer, 1990-2021, and Projections to 2050: A Systematic Analysis of the Global Burden of Disease Study 2021. Thorac Cancer. 2025; 16(9):e70052.
- Zhang H, Hussin H, Hoh CC, Cheong SH, Lee WK, Yahaya BH. Big data in breast cancer: Towards precision treatment. Digit Health. 2024;10:20552076241293695.
- Karimova L, Azizova G, Shahverdiyeva I. Serum levels of specific matrix metalloproteinases and CYR-61 in women with breast cancer. Azerbaijan Med J. 2024; (3):37–40. https://doi.org/10.34921/amj.2024.3.006
- Li X, Yang J, Peng L, Huang Y, Li Q. The emerging role of HER2 mutations in HER2-negative breast cancer: Molecular mechanisms and clinical implications. Front Oncol. 2022;12:823456.
- Wang H, Chen L, Zhang X, Liu M. Targeting HER2 mutations in HER2-negative breast cancer: A new frontier in precision oncology. Cancer Lett. 2021; 519:75–84.
- Kim SY, Park JH, Lee JH. Clinical relevance of HER2 mutations in HER2-negative breast cancer: Implications for targeted therapy. Breast Cancer Res Treat. 2020;184(3):539–548.
- Loibl S, Poortmans P, Morrow M, et al. Breast cancer. Lancet. 2021;397(10286):1750–1769.
- Yeh MH, Cheng YC, Hsieh MJ, et al. HER2 mutations in breast cancer: mechanisms, clinical implications and targeted therapy. Cancer Lett. 2021;512:1–10.
- Zhang Y, Wang J, Li X, Chen X. The role of MMP-2 and MMP-9 in breast cancer invasion and metastasis: Molecular mechanisms and therapeutic potential. Front Oncol. 2023;13:1012345.
- Gupta S, Singh R, Kaur J. Matrix metalloproteinases in breast cancer: Emerging biomarkers and therapeutic targets. Int J Mol Sci. 2021;22(17):9345.
- Chen L, Zhang W, Li J. HER2-mediated upregulation of MMP-9 via MAPK and PI3K/Akt signaling promotes breast cancer metastasis. Mol Cancer Res. 2022;20(6):974–985.
- Yang X, Wu Y, Zhou Q. Crosstalk between HER2 signaling and MMP-9 expression in breast cancer invasion and metastasis. Oncogene. 2021;40(14):2523–2535.
- Liu Z, Wang H, Zhang M. HER2 activation promotes MMP-9 expression through MAPK and PI3K/Akt pathways in breast cancer cells. Cell Signal. 2020;73:109684.
- Zhou Y, Li J, Wang X. HER2 signaling promotes extracellular matrix remodeling and angiogenesis via upregulation of MMP-2 and MMP-14 in breast cancer. Cancer Lett. 2023;551:215940.
- Johnson AL, Martinez D, Thompson R. Circulating matrix metalloproteinases as biomarkers in HER2-positive breast cancer: Clinical implications and mechanisms. Breast Cancer Res Treat. 2023;185(2):347–358.
- Park SY, Kim HJ, Lee JS. Elevated serum MMP-2 and MMP-9 levels in HER2-positive breast cancer patients and their potential role in disease progression. Oncol Rep. 2021;46(3):1524–1533.
- Zhang Y, Liu H, Wang J. CYR61 mediates HER2-driven breast cancer progression through regulation of MMP expression and ECM remodeling. Mol Oncol. 2022;16(5):1047–1060.
- Huang Y, Zhao X, Li Q. Low prevalence of HER2 mutations in HER2-negative breast cancer subtypes: A genomic analysis of ER-positive and triple-negative cases. Breast Cancer Res. 2021;23(1):45.
- Lo PK, Fan X, Li Y. Genomic profiling of HER2-negative breast cancers reveals minimal EGFR gene amplifications in ER-positive tumors. BMC Cancer. 2020;20:555.
- Wang X, Chen Y, Liu J. EGFR mutations are prevalent in triple-negative breast cancer, while HER2 mutations remain rare: Insights from comprehensive genomic profiling. J Clin Oncol. 2021;39(15_suppl):1024.
- Chung W, Eum HH, Lee H, Lee KM, Park D, Kim J. Genomic landscape of treatment resistance in HER2-negative breast cancer: Emphasis on PIK3CA mutations and PI3K-AKT-mTOR pathway activation. Cancer Res. 2020;80(16):3399–3410.
- Chou HY, Lee WC, Chen YH, Chang JG, Hsiao CF, Huang CS, et al. Quantitative RT-PCR analysis of HER2 mRNA expression in breast cancer tissues and its correlation with protein expression and gene amplification. BMC Cancer. 2021;21(1):522.
- T Kawamoto, J D Sato, A Le, J Polikoff, G H Sato, J M Mendelsohn et al, Growth stimulation of A431 cells by epidermal growth factor: identification of high-affinity receptors for epidermal growth factor by an anti-receptor monoclonal antibody. Proc Natl Acad Sci. 1983 Mar;80 (5):1337-41.
- Chiara Arienti et al. Epidermal Growth Factor Receptor Family and its Role in Gastric Cancer. Journal of Frontiers Oncology, 2019. 26 November.

