Innovations in Blood Systems for Low-Middle Income Countries
Global Challenges and Innovations in Low-and Middle-Income Country Blood Systems
Cees Th. Smit Sibinga, MD, PhD, FRCP Edin, FRCPath
IQM Consulting Zuidhorn, The Netherlands, emProfessor International Development of Transfusion Medicine, University of Groningen, Groningen, The Netherlands
OPEN ACCESS
PUBLISHED: 31 July 2025
CITATION Smit, Sibinga, CT., 2025. Global Challenges and Innovations in Low-and Middle-Income Country Blood Systems. Medical Research Archives, [online] 13(7). https://doi.org/10.18103/mra.v13i7.6807
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DO:I https://doi.org/10.18103/mra.v13i7.6807
ISSN 2375-1924
ABSTRACT
Blood transfusion has come a long way. Since the discovery of Landsteiner late 19th century clinical use has become possible. However, it took decades before an effective quality blood supply system (managerial and operational, well-governed) was developed serving different hospitals. The World Health Assembly resolution 28.72 adopted in 1975 has been the take-off point of innovation. Together and supported by United Nations Development Program the world was classified in three and later four groups of countries based on three essential domains: longevity and health, education, income; Low-, Medium- and High-Human Development Index. World Health Organization, analyzing these Human Development Index groups for their health care systems and blood systems, recognized Human Development Index groups and the high considerable gaps between the Low- and Medium-Human Development Index groups and the High-Human Development Index group of countries seemingly caused by a weak governance structure and Infrastructure and education, almost absent quality culture and poor to mediocre economies. The International League of Red Cross and Red Crescent Societies, World Health Organization and several transfusion societies and associations attempted to fill the gap, but so far made insufficient progress. Recent innovative initiatives of World Health Organization and United Nations Development Plan are promising.
Keywords: UNP, HDI, WHO, Blood systems, Quality, Management.
Introduction
Since ancient times blood has been given mythical and healing strength. The Egyptian Faros made their wounded warriors bath in oxblood assuming they would regain their health and strength1. In 1492 Pope Innocentius VIII was treated for a lethal renal disease with the blood from three healthy young boys2. At the court of the French King Louis IV in Paris Jean Baptiste Denis and Paul Emmerez treated several patients with an exchange transfusion of calf blood3. In 1818 the gynecologist and obstetrician James Blundell treated women for obstetrical bleeding with human blood transfusion. Blundell and his colleague Henry Leacock from Barbados discovered the principle that transfusion should always be done species specific4.
In 1900 the Viennese scientist Karl Landsteiner discovered the existence of red cell antigens, the ABO blood group system and the principle of cross matching donor blood with blood from the potential recipient for compatibility before transfusion5. With World War I the first blood transfusions were practiced at the battlefield lazarettes in France, for which preservation of the blood was needed. The bottles closed with a cotton stopper were flown over from England. The Belgian scientist Albert Hustin developed in 1913 and used in 1914 as an anticoagulant citrate as a divalent cation (Ca2+) scavenger, originally in a 1:1 ratio in the blood collected6. A British scientist group (Rous and Turner) developed a preservation solution containing 0.9% physiological saline, isocitrate and glucose7.
Blood transfusion became a feasible clinical practice. However, the practice was individual and was not the outcome of a system and structure on a larger scale, recognizing that the practice should be made possible for all patients in need of blood.
Organization, governance and leadership
A blood bank was established during WWI in France by Oswald Hope Robertson8, followed by similar organizations in Moscow (cadaver blood), Cook County hospital in Chicago (Bernard Fantus 1937), Deaconess hospital in Rotterdam (Henri van Dijk, 1930) and Barcelona (Spanish civil war). However, most of the facilities that mushroomed before World War II were small and accommodating just the need of one hospital hospital based blood banks, active in collection and procurement of blood as well as supporting patient care through compatibility testing and immunohematology testing. None of the officials and practitioners realized that they had created a legal conflict of interest. Procuring blood and blood products holds opponents cannot be united under one final responsibility and need clear separation in their organization, operations and responsibilities9,10.
A variety of organizational and governance structures were created at the National, regional and local level, public and private, ministerial and Non-Governmental Organization (NGO), fragmented and concentrated at regional or national level and each serving their own kingdom. It was not until the early years post-World War II that France endorsed in 1956 a specific legal framework to regulate blood transfusion in the country, followed in 1961 by the Netherlands. However, these frameworks were not complete and did allow mediocre governance and practices. The Federation of Red Cross and Red Crescent Societies in Geneva decided that individual societies were allowed to organize a blood transfusion system in their country e.g., Finland, Germany, Switzerland Thailand, Japan, Indonesia, Surinam, and many more.
The first breakthrough came with the outbreak of World War II when the American war industry introduced for its technical standardization of equipment and vehicles in the battlefield the concept of Good Manufacturing Practice (GMP) which became the first comprehensive quality system in manufacturing of products. The quality Guru, electrotechnical engineer and statistician William Edward Deming developed in the 1950s while in Japan the principle of the cycle of improvement consisting of four key actions Plan, Do or implement, Check or control, and Act or improve11. GMP was adopted as the basic quality system for the blood supply. However, also that system needed management. To standardize the management of quality it was set up as a system (Quality Management System or QMS) based on five key elements12:
- Organization, (infra-)structure, governance and leadership, which include policies and planning, both managerial and operational;
- Standards and references, regularly revised and updated;
- Documentation, archiving and traceability;
- Education: teaching and training, and competency;
- Assessment through monitoring and evaluation, including hemovigilance.
The first three key elements are the fundaments, what is absolutely necessary. Where the last two key elements are needed for the implementation and the quality management. For instance, what you monitor is the work and immediate result, but what you evaluate is the outcome and impact, both against the standards as a Golden reference. These systems are dynamic and (should) change over time.
The second and major breakthrough came with the HIV/AIDS epidemic early 1980s which created a tsunami of liability issues all over the world and disclosed the paucities and weaknesses in organization, leadership, legal structure, governance, management, and quality awareness and culture. People recognized that a system like QMS needs also management in a structured way – a Quality System Management (QSM) to make sure that the five key elements are properly managed as a flow12.
Any organization is in principle managed and operated through a series of processes, starting at the top with the steering processes, followed by the supportive or secondary processes that allow a smooth and harmonized functioning of the primary or operational processes. These fundamental to which Transfusion Medicine bridges to allow optimization of the governance, management and operations of the vein-to-vein blood transfusion chain. Management Science is the broad interdisciplinary study of problem solving and decision making in human organizations, with strong links to management, economics, business, engineering, management consulting, and other sciences. It uses various scientific research-based principles, strategies, and analytical methods including mathematical modelling, statistics and numerical algorithms to improve an organization’s ability to enact rational and accurate management decisions by arriving at optimal or near optimal solutions to complex decision problems. In short, management sciences help businesses to achieve goals using various scientific methods e.g., SPC, Six Sigma, ISO, and lean Six Sigma supported by artificial intelligence (AI). This science field was initially an outgrowth of applied mathematics, where early challenges were problems relating to the optimization of systems which could be modelled linearly, i.e., determining the optima (maximum value of outcome, procurement performance, quality of products and services, bandwidth and economy of scale, etc. or minimum of loss, risk, costs, etc.) of an objective function. Today, management science encompasses any organizational activity for which the problem can be structured as a functional governance system so as to obtain a solution set with identifiable characteristics. To be able to understand and practice this supportive science in the governance of a blood establishment competent leadership is needed at top (steering processes) and middle management (supportive processes) level organization.
Human Development Indices
United Nations Development Program (UNDP) developed three developmental dimensions each with a separate dimension, together the Human Development Index (HDI): Long and Healthy Life, Knowledge, A Decent Standard of Living, with the indices Live Expectancy, Education and Gross National Income13. Originally countries were divided in three and later in four HDI groups; Low-, Medium-, High- and Very High-, or L-HDI, M-HDI, H-HDI and VH-HDI. Together the data of the Indices provide per country the overall HDI. The UNDP Human Development Report 202514 presents of 193 in 2023 responding countries (Table 1):
| HDI | Number | Index range |
|---|---|---|
| VH-HDI | 74 countries | 0.972 – 0.804 |
| H-HDI | 50 countries | 0.799 – 0.703 |
| M-HDI | 43 countries | 0.698 – 0.550 |
| L-HDI | 26 countries | 0.544 – 0.226 |
Looking at the table, 37.75% (n=69) of the countries belongs to the Low- and Medium-HDI group home to over 80% of the global population. The 2023 World Health Organization (WHO) Key Facts for Blood Safety and Availability 202515 show that of the close to 200 million annual blood donations collected globally, 40% are collected in high-income countries, home to only 16% of the global population, while in Low- and Middle-income countries (LMICs), up to 54 % of blood transfusions are given to children under 5 years of age; whereas in high-income countries, the most frequently transfused patient group is over 60 years of age, accounting for up to 76% of all transfusions. The clinical indications for and quality of the transfusion practices in that part of the world are far from ideal.
In the more advanced countries the blood supply has been captured away from fragmentation in a national system based on a governmental policy and anchored in a framework legislative and regulatory system. The daily operations vein-to-vein were in many situations delegated to the national Red Cross or Red Crescent Societies. This happened e.g., in Switzerland (Hässig), Finland (Nevanlina), Germany (Seidl), The Netherlands (van Loghem), Japan and Australia (Beal), but to an extend also in the US (Barker).
World Health Organization responsibility
In 1975 the World Health Assembly (WHA) in Geneva endorsed a World Health Organization proposed resolution WHA28/7216 on Utilization and Supply of Human Blood and Blood Products in an attempt to stop the commercialization of the production of blood and plasma derived source material, largely paid blood donors and for sale. The resolution is often referred to as the defining WHA resolution on a universal access to blood and blood products including plasma and plasma derived medicinal products (PDMP) like albumin, gamma globulins and coagulation factors. During the early days of the dramatic HIV/AIDS pandemic WHO, in 1988, initiated the Global Blood Safety Initiative (GBSI) to which the League of Red Cross and Red Crescent Societies (LRCRCS), the International Society of Blood Transfusion (ISBT) and the World Federation of Hemophilia (WFH) were invited to participate17. GBSI mapped the world for the existence, safety and effectivity of the blood supply and believed that the challenge of poor and inadequate education in large parts of the world and particularly in the developing world was by far the most important factor and resulted in the design of a comprehensive series of aspects of the vein-to-vein transfusion chain but also a Distance Learning Program with modular distance learning material published in five global languages. In 2000 a Quality Management Training (QMT) course was designed and implemented, followed by a series of other WHA resolutions of which the 2010 resolution WHA63.1218 on universal access to safe, effective and quality-assured blood products 2020-2023: Strategic Objectives each with a number of high-level outcomes to be achieved and a series of 8 guidance documents to support the developing world implementing the strategic objectives and achieving the high-level outcomes19,20. A number of Low- and Medium-HDI countries reported having difficulties implementing the strategic objectives and achieving the high-level outcomes listed in the Action framework. WHO decided to support these countries more intensively in their efforts of implementation designing a special educational and practical services using the blood system self-assessment Group (BCG) and financed by the then United States Agency for International Development (USAID) the tool was developed as a web annex to the guidance document21,22.
Existing Blood Supply systems challenges and innovations
Based on the UNDP HDI groups of countries and the World Bank sectioning: Low- Middle-, High-Middle and High-Income countries, WHO has recognized three classes of development of the healthcare and blood systems: Low-HDI, Medium HDI and High-, Very-High-HDI countries. Preferably there should be a National Blood System19 with regional blood establishments serving uniformly the hospitals in the region. Such system should be based on a national blood policy, translated in a specific legislative framework and regulatory oversight system, a national blood system (governance, education, standards, documentation and quality) and at the operational level licensed regional blood establishments (Figure. 1)
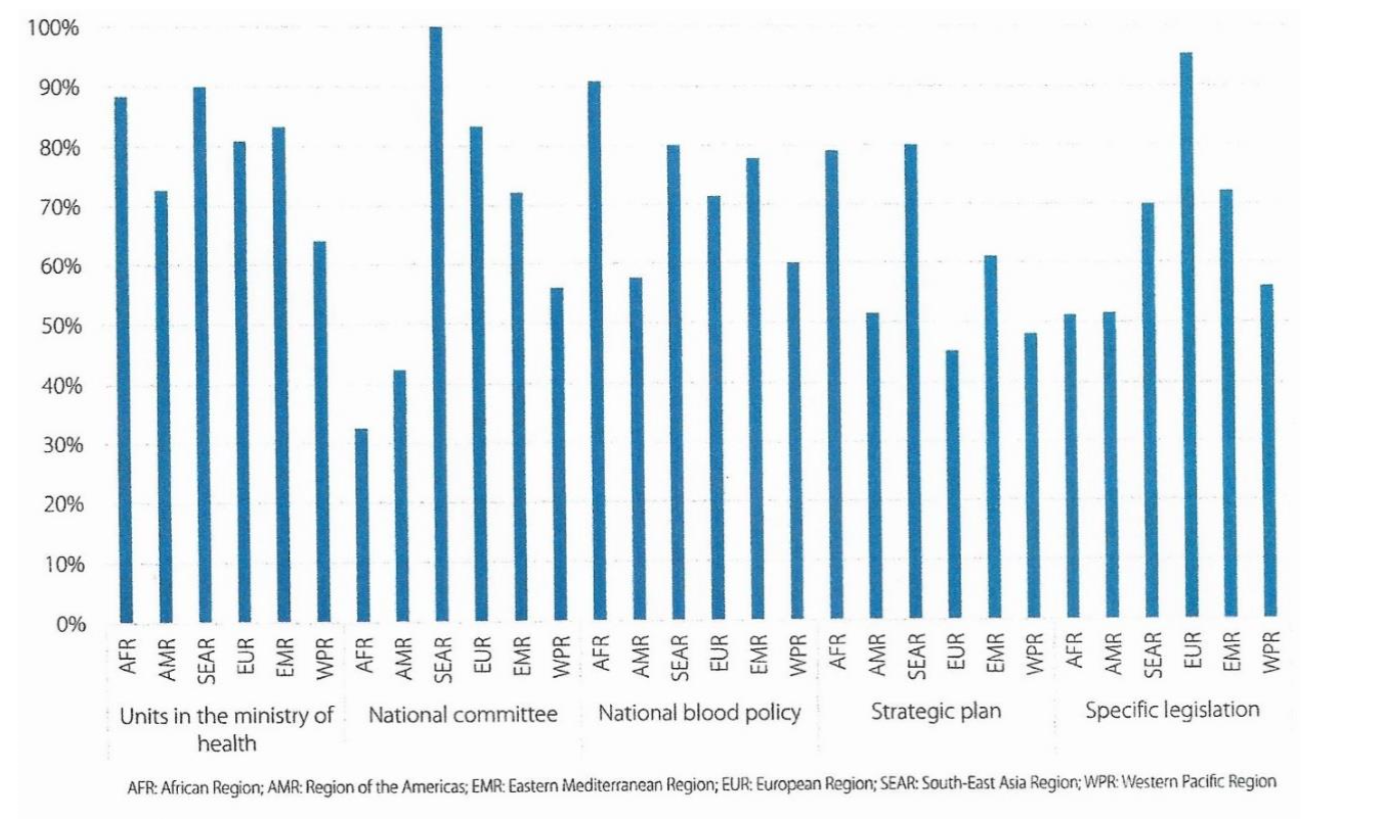
The hospital should have a hospital blood bank responsible for the patient care oriented logistic functions: an appropriate blood stock, immunohematology and cross matching, the distribution of the cross matched units to the wards, operation rooms (ORs), and other locations in the hospital, adverse transfusion events analysis, documentation and hemovigilance.
Not all these mechanisms have been implemented in practice! The only WHO Region where these mechanisms have been implemented successfully is Europe and in particular in the European Union (EU) countries.
In most of the Low-HDI type LMICs such organizational and structural setup is unfortunately still missing or weak due to a failing governance at high level, continuing misunderstandings and interpretations, poor to absent documentation and quality. The major and protracting challenges observed23 are:
- deficiencies in national policy, governance and financing; insufficient supply of safe, effective and quality-assured blood products for transfusion;
- deficiencies in blood product safety, effectiveness and quality;
- insufficient availability of plasma derived medicinal products (PDMPs);
- suboptimal clinical practices in transfusion of blood components;
- insufficient access to blood during emergency situations;
Extremely prominent is the challenge of knowledge:
- poor and inadequate education.
The UNDP in its 2025 Report on Human Development14 the introduction of AI as an innovative pathway to accelerate development and strengthen more effectively organizations and operations of underdeveloped Health Care and Blood Systems in the LMIC part of the globe, using the slogan Focused attention on organization and structure.
Over the past decades projects, workshops and training courses had predominantly focused on bench work and related technologies, the primary functions. Most of these were organized in the advanced world and privileged a limited number of selected employees. There has been very little attention for the managerial aspects, organization and structure, legislation and regulations, leadership, quality principles and governance, hygiene and cleanliness, systematic documentation, education and continuous learning programs, biohazardous waste, technical service and maintenance. A quality culture24 24/7 is virtually absent and comprehensive stewardship is hardly noticeable. Laboratory personnel, when lucky, may be invited to one of these technical bench workshops far from their daily environment in a foreign and sophisticated laboratory and conducted by strangers who are usually not well familiar with the environments and possibilities of their routine work. Different kitchens, cooks and recipes.
Foreign aid projects and their impact. What could have been done better? Development programs and projects or consultations are usually far too time-limited to accomplish sustainable progress. Progress depends for a small part on available money and predominantly on the sharing of knowledge and expertise. Change takes time and a willingness or positive attitude to the proposed change, and accompanied by an adequate and effective change management.
One of the major foreign aid blood safety initiatives was the 2003-2004 launched Emergency Plan for AIDS Relief (PEPFAR)24 besides other initiatives such as the HIV/AIDS specific safe blood investments by the Global Fund to Fight Aids, Tuberculosis and Malaria (GFATM), the World Bank (WB), the Asian Development Bank (AB), the European Union Development Bank (EUDB) and others. These funding initiatives made dramatic and historically unprecedented changes in the amount of funding available for blood safety projects, specially in Sub-Saharan Africa countries with high HIV/AIDS burdens25. While much has been done, mostly not well coordinated, intended to strengthen blood transfusion services in developing countries, the impact of externally funded blood safety projects has not been well documented and evaluated. Follow-up was scarcely and over a short period given, concluding that the intended blood safety aid and strengthening of blood supply systems de facto has been poor or at the best limited. The existing development gap has not really been narrowed or bridged!
The role of sharing, education and discipline.
In a world in which we try to come to equality in any respect, as documented in the 1948 proclaimed and adopted by the United Nations General Assembly Universal Declaration of Human Rights26, the principle of sharing and respecting which is most important (living circumstances, food, health care, knowledge and skills, justice and protection, education etc.) has still not 100% materialized nor been implemented. The international community has a duty to uphold and defend these universal human rights. It is therefore important to ensure those people and communities who most need their rights protected are made aware that this 1948 Declaration exists also for them. That counts in particular for the more advanced part of the world (the haves) to actively and above self show the discipline to share and educate these principles in transfusion medicine, an integral part of the health care, with the developing world (the have nots) as a national system and operational. These principles are a living reality for every individual (man, woman and child) irrespective of gender wherever on this globe. It raises the burning question why, after more than three quarters of a century, these blood supply and transfusion gaps still exist?
Conclusion
There is still a remarkable variety of blood supply systems existing in the world despite the large number of efforts, projects, guidance initiatives and recommendations that were launched and funded over the past century. The data collection done by UNDP and WHO over the last decades show a number of still existing serious challenges caused by weaknesses in education, sharing of knowledge and experience between the advanced and the underdeveloped world. Weaknesses in government and governance, legislation, organization and structure, leadership and stewardship, quality culture and operations, cleanliness and waste management, hygiene and asepsis, documentation and ownership still prevail and are not given the attention needed to sustainably overcome the challenges. The UNDP changes emphasis and advocates the implementation and use of artificial intelligence (AI) more widely in societal life as a matter of choice of people and structures in the age of AI. That includes health care and the supportive fields like pharmacy, engineering, waste management and the blood supply.
Conflict of Interest Statement: The author declare to have no conflicts of interest.
Funding Statement: None.
Acknowledgements: None.
Orcid ID: http://orcid.org/0000-0003-0156-5620
References:
- Maluf, N. S. R. History of Blood Transfusion. J Histf Med Allied Sci, 1954;9:59-107. DOI: https://doi.org/10.1093/jhmas/ix.1.59
- Lindeboom, G. A. The Story of a Blood Transfusion to a Pope. J Hist Med Allied Sci, 1954;9:455-459. DOI: https://doi.org/10.1093/jhmas/ix.4.455
- Denis, J. Concerning a New Way of Curing Sundry Diseases by Transfusion of Blood. Philosoph Transactions Royal Soc, 1667;3: 489-504.
- Young J.H. James Blundell (1790-1878). Experimental physiologist And obstetrician. J Histf Med Allied Sci,1964;8:t59.
- Landsteiner K. Uber Agglutinatioserscheinungen normalem menschlichen Blutes. Wiener Klein Wochenschr 1901;14:1132-113.
- Hustin A. Note sur une nouvelle; méthode de transfusion. Communication préliminaire. Ann Bull Soc Royale Sci Méd 1917;87:141-142.
- Rous P., Turner J.W. The preservation of living red cells in vitro. J Exp Med 1916;23:219-237.
- Robertson O.H. Transfusion with preserved red cells. Br J Med 1918;1:691-695.
- Directive 2002/98/EC of the European Parliament and the Council of 27 January 2003 setting standards of quality and Safety for the collection, testing, processing, storage and distribution of human blood and blood components and amending Commission Directive 2004/33/EC of 22 March 2004 implementing Directive 2002/98/EC European Parliament and the Council as regards certain technical requirements for blood and blood components. Official J Europ Union 2004;L91:preamble.
- Smit Sibinga CTh. Recommended Legislative Framework for a National Blood Policy and Blood System. In: Sawadogo RW ed. New Visions in Medicine and Medical Science, Vol.1. BP International, India, UK. 2024: Chapter 2:09-23 DOI: http://doi.org/10.9734/bpi/nvmms/v1/8728A
- Deming W.E. The new economics for industry, government and education Massachusetts Institute of Technology Center for Advanced Engineering Study. Cambridge, MA. 1993.
- Smit Sibinga C.Th., Seidl C., Nunes E., Aubuchon J.P. Quality and quality management systems application in transfusion medicine. In: Smit Sibinga C.Th. ed. Quality Management in Transfusion Medicine. Nova Sci Publ 2013; chapter III:55-130.
- UNDP HDI [Available at https://hdr.undp.org/data-center/human-development-index#/indicies/HDI]
- UNDP Human Development Report 2025, Overview. UNDP Human Development Report 2023-2024. A matter of choice. People and possibilities in the age of AI.. United Nation Development Programme. 2025 New York, NY.
- WHO Blood Safety and availability.Key Facts. 2025 [Available at https://www.who.int/news-room/fact-sheets/detail/blood-safety-and-availability]
- WHA Resolution 28.72. Utilization and supply of human blood and blood products. Geneva, World Health Assembly 1975. [Available at https://iris.who.int/handle/10665/93010]
- Harris D. Global Blood Safety Initiative (GBSI) 1989. [Available at https://aps.who.int/iris/handle/10665/49318]
- WHA Resolution 63.12. Availability, safety and quality of blood and blood products. Geneva, World Health Assembly 2010 [Available at https://www.who.int/publications/i/item/WHA63.12]
- WHO. Action framework to advance universal access to safe, effective and quality-assured blood and blood products 2020-2023. Geneva, World Health Organization 2020. Licence: CC By-NC-SA 3.0 IGO.
- Abdella YE., Maryuningsih S., Smit Sibinga CTh. Historical Background and current Global Efforts. In: Eichbaum QG et al. eds. Global Perspectives and Practices in Transfusion Medicine. AABB Press, Bethesda MA. 2023;chapter 1:1-26.
- WHO. Guidance to identify barriers in blood services using the blood system self-assessment (BSS) tool. Geneva, World Health Organization 2023. Licence: CC By-NC-SA 3.0 IGO.
- Web Annex: Blood System self-assessment. In: Guidance to identify barriers in blood services using the blood system self-assessment (BSS) tool. Geneva, World Health Organization 2023. Licence: CC BY-NC-SA 3.0 IGO.
- WHO. Global Status Report on Blood Safety and Availability 2021. Geneva: World Health Organization 2022. Licence: CC BY-NC-SA 3.0 IGO.
- Centers for Disease Control and Prevention. Progress toward strengthening blood transfusion services 14 countries. 2003-2007. MMWR Morb Mortal Wkly Rep 2008; 57:1237.
- Pitman J.P. The influence and impact of the US President’s Emergency Plan for AIDS Relief (PEPFAR) on blood transfusion services in Africa. Case studies from Namibia. Academic Dissertation University of Groningen, NL 2015 Grafimedia RUG.
- UN. Universal Declaration of Human Rights. United Nations New York, NY. 2015.




