Pharmacokinetics of Topical Phosphosulindac in Mice
Pharmacokinetics and biodistribution of topical phosphosulindac and its metabolites in mice and a mouse model for chemotherapy-induced peripheral neuropathy
Ishan Amin Khwaja 1, Jonathan Souferian 1, Fatima B Khwaja 1, Andrew Loiacono 1, Cameron Takmil 1, Vivian Zhu 1, Owen Dong 1, Jennifer Zeng 1, Sean Hatzidakis 1, Atahan Yetimoglu 1, Ishan Deen Khwaja 1, Emanuel Mamakas 2, Ernest Natke 3, Robert Honkanen 3, Liqun Huang 1,2 and Basil Rigas 1,2,*
- Department of Preventive Medicine, Renaissance School of Medicine, Stony Brook University, Stony Brook, NY 11794, USA.
- Medicon Pharmaceuticals, Inc, Setauket, NY 11733, USA.
- Department of Ophthalmology, Renaissance School of Medicine, Stony Brook University, Stony Brook, NY 11794, USA.
Email: [email protected]
OPEN ACCESS
PUBLISHED 31 July 2025
CITATION Khwaja, IA., Souferian, J., et al., 2025. Pharmacokinetics and biodistribution of topical phosphosulindac and its metabolites in mice and a mouse model for chemotherapy-induced peripheral neuropathy. Medical Research Archives, [online] 13(7). https://doi.org/10.18103/mra.v13i7.6792
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6792
ISSN 2375-1924
ABSTRACT
Background: Our purpose was to determine the pharmacokinetics and biodistribution of phosphosulindac and its metabolites when applied to mouse skin. Phosphosulindac differs from sulindac by adding a diethyl-phospho-butane moiety, which enhances efficacy and safety. In preclinical studies, phosphosulindac has anti-cancer and anti-inflammatory properties and prevents and reverses chemotherapy-induced peripheral neuropathy.
Methods: Phosphosulindac gel was applied topically to the hind paws of normal mice and those with chemotherapy-induced peripheral neuropathy. Samples from the paw skin, paw muscle, leg muscle, sciatic nerve, dorsal root ganglia and blood, obtained at various time points, were assayed using HPLC for phosphosulindac, and its metabolites (sulindac, sulindac sulfide, sulindac sulfone).
Results: Topically applied phosphosulindac was detected in paw skin, paw and adjacent leg muscles where it reached its Tmax in 0.5 h. Smaller amounts of phosphosulindac were detected in sciatic nerve (Tmax = 3 h) and dorsal root ganglia (at 24 h). Phosphosulindac was not found in blood. Absorption of phosphosulindac was concentration-dependent and pH-sensitive. Its metabolites were detected in paw skin, vicinal muscles, sciatic nerve, and blood but not in dorsal root ganglia. Topically applied equimolar sulindac was detected in paw skin and muscle and in smaller amounts in leg muscle and sciatic nerve.
Conclusion: Phosphosulindac is absorbed through the paw skin and transported from paw and leg muscle to the sciatic nerve and dorsal root ganglia, its target tissues in normal and chemotherapy-induced peripheral neuropathy mice. In contrast, sulindac is transported through the same tissues as well as by the circulation.
Keywords: Topical application, Preclinical pharmacokinetics, Phosphosulindac, OXT-328, Sulindac, Modified NSAIDs, CIPN
INTRODUCTION
Phosphosulindac (PS, OXT-328) is a novel agent belonging to the broad class of modified nonsteroidal anti-inflammatory drugs (NSAIDs). Structurally, PS can be considered a derivative of the conventional NSAID sulindac, which has been covalently bound through a carboxylic ester to a diethyl-phospho-butane moiety. This modification has been considered crucial for its safety, as it removes the carboxylic moiety of sulindac, which is responsible for sulindac’s gastroduodenal toxicity. Compared to sulindac, PS displayed enhanced pharmacological efficacy in various systems, in some cases as high as 10-fold.
In preclinical studies, PS has shown robust anticancer and anti-inflammatory properties, altering pathways central to cell growth and inflammation. Recently, we reported that PS has a strong preventive and therapeutic effect on chemotherapy-induced peripheral neuropathy (CIPN). This side effect of chemotherapeutic agents can be dose-limiting, negatively affecting the patient’s treatment regimen and their quality of life. CIPN affects peripheral nerves, as its clinical manifestations are prominent in hands and feet characterized as a “glove-and-stocking” distribution. In our study, PS was applied topically to the hind paws of mice with CIPN, a site of marked allodynia. PS was dose-dependently efficacious on CIPN induced by three different classes of commonly prescribed chemotherapeutic agents. In each case, PS improved and often normalized mechanical and cold allodynia scores as measured on the hind paws of mice. The mechanism by which PS relieves the neuropathy of CIPN appeared to be the suppression of neuroinflammation through its effect on cytokine expression and macrophage activation. The effect of PS on CIPN did not interfere with the anticancer effect of these chemotherapeutic agents. Interestingly, sulindac, its parent NSAID, was devoid of any effect on CIPN, indicating that the intact PS molecule was required for its anti-CIPN activity, in agreement with similar findings in other systems.
The metabolism of PS, as well as its pharmacokinetics (PK) and biodistribution following its oral administration to mice, have been reported elsewhere. PS undergoes reduction and oxidation, yielding PS sulfide and PS sulfone; it is also hydrolyzed, releasing sulindac, which generates sulindac sulfide and sulindac sulfone, all of which are glucuronidated. In mice, oral PS is rapidly absorbed, metabolized, and found in blood and other tissues. The central metabolic transformation of PS is the rapid hydrolysis of its carboxylic ester catalyzed by carboxylesterases, an event that determines its efficacy. In both mouse blood and tissues, the levels of PS were low or undetectable, with the dominant species being its metabolites; of those, sulindac had the highest Cmax.
Given PS’s efficacy in CIPN, we undertook a detailed PK and biodistribution study to map where and when detectable levels of PS and its metabolites appear in various tissues of the mouse after its topical application. Our assumption was that to relieve allodynia, the hallmark of CIPN, PS must reach the affected nerves. Tissues analyzed included the paw skin and its vicinal leg muscles associated with the site of application; the sciatic nerve and the dorsal root ganglia (DRG), its presumed targets of action; and blood, a potential conduit to nerves.
MATERIALS AND METHODS
MATERIALS
Phosphosulindac was a gift of Medicon Pharmaceuticals, Inc, Setauket, NY. Sulindac and other reagents used in these studies including the excipients ethyl alcohol (derived from grain), triethanolamine, hydroxypropyl methyl cellulose, xanthan gum, sodium citrate, benzalkonium chloride, citric acid and glycerol, were purchased from Sigma-Aldrich (St. Louis, MO). PS was formulated in a gel as described; hydroxypropyl-methylcellulose was the gelling agent and xanthan gum the phase stabilizer. The pH of each formulation was adjusted to the desired value using citric acid.
ANIMALS
Adult male C57BL/6J mice, eight weeks of age at the beginning of the experiments and weighing 20–30 g (The Jackson Laboratory, Bar Harbor, ME) were housed in an AAALAC-accredited facility in groups of four. Food and water were available ad libitum. Experiments were performed during the light cycle (7:00 am to 7:00 pm) and animals were euthanized with CO2 asphyxiation. Studies were approved by the Institutional Animal Care and Use Committees of Stony Brook University and followed the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. Animal studies are reported in compliance with the ARRIVE guidelines.
PHARMACOKINETICS/BIODISTRIBUTION IN MICE
Phosphosulindac or sulindac gel, each at various concentrations, was applied topically to each hind paw (50 µl per paw) with gentle rubbing. At the time points 0.5h, 1h, 3h, 6h, 12h, and 24h, mice (n = 5-6 mice/time point) were euthanized with CO2 inhalation. Blood was drawn immediately after death by cardiac puncture. Tissues, including the sciatic nerve and lumbar DRG bilaterally, were dissected quickly, immediately frozen in liquid nitrogen and stored at -80°C until analyzed. As previously described, each plasma sample was mixed with double volume of acetonitrile and centrifuged at 13,200 rpm for 15 min. Tissue samples were weighed, ddH2O (100-300 µL, depending on tissue weight) was added and they were homogenized. Following addition of acetonitrile (twice the volume of the homogenate), the mixture was sonicated for 10 min, centrifuged at 13,200 rpm for 15 min, and analyzed by HPLC, as reported. The limit of quantitation is 0.1 μM for PS and 0.05 μM for sulindac, sulindac sulfone, and sulindac sulfide.
INDUCTION OF CHEMOTHERAPY-INDUCED PERIPHERAL NEUROPATHY
Chemotherapy-induced peripheral neuropathy was induced in male C57BL/6 mice with paclitaxel using established protocols. Dosing regiments for paclitaxel as follows: Four i.p. injections of 8 mg/kg paclitaxel every other day, resulting in a cumulative dose of 32 mg/kg. Mechanical allodynia thresholds (paw withdraw thresholds, PWT) were determined using von Frey filaments as described.
STATISTICAL ANALYSIS
Pharmacokinetic parameters were calculated by Microsoft Excel and PKSolver. We employed non-compartmental analyses.
RESULTS
PHARMACOKINETICS/BIODISTRIBUTION OF PHOSPHOSULINDAC 8% TOPICALLY APPLIED TO THE SKIN OF MICE
We studied the kinetics and biodistribution of PS when it was applied topically as an 8% pH 9.4 gel to the hind paws of normal mice. To validate our methods, we aimed to measure the amount of PS in the tissues proximal to the site of application as well as tissues more distal with particular attention to the sciatic nerve (SN) and the dorsal root ganglia (DRG) that are altered in CIPN.
As shown in Figure 1A-1B and Table I, PS was detected in paw skin, the site of its application, the muscles underneath the skin (paw muscle), vicinal leg muscles, and the sciatic nerve after it was applied topically to the hind paws of the mice. The concentration of PS progressively decreased from the skin (site of application) to more distal tissues like the SN, as evidenced by the respective values of both Cmax (from 126.3 ± 4.8 µM to 2.0 ± 0.7 µM) and AUC0-24h (from 918.4 µM.h to 17.0 µM.h). PS was detected in DRG only after 24h at a low concentration of 0.9 µM. However, no PS was detected in the systemic circulation. The Tmax of PS was the same in all tissues (0.5 h) except for SN, which showed a prolonged Tmax (3 h). Another interesting feature is the difference in T1/2 of the skin, the muscles, and SN, which varied within a wide range (9.6 – 63.7 h); the T1/2 of DRG was more prolonged but could not be determined with reasonable accuracy.
| Tissue | Cmax, µM mean ±SEM | Tmax, h | T1/2, h | AUC0-24h, µM.h |
|---|---|---|---|---|
| Paw skin | 126.3 ± 4.8 | 0.5 | 63.7 | 918.4 |
| Paw muscle | 76.6 ± 4.3 | 0.5 | 9.6 | 461.6 |
| Leg muscle | 19.2 ± 3.0 | 0.5 | 81.8 | 114.9 |
| SN | 2.0 ± 0.7 | 3 | 11.3 | 17.0 |
| DRG | – | – | – | – |
| Blood | – | – | – | – |
We detected three metabolites of PS: sulindac, sulindac sulfone, and sulindac sulfide (Figure 2A-D and Table II) in the tissues and the circulation. However, no glucuronidated metabolites were detected and all three appeared in circulation. Sulindac was the dominant metabolite, with sulindac sulfide and sulindac sulfone levels being <20% of those of sulindac. However, sulindac sulfide was only detected in the circulation. When comparing Cmax data from Tables I and II, sulindac levels were about 1/3 of those of PS in all tissues except the sciatic nerve (where it was greater than 50%). No sulindac was detected in DRG.
| Tissue | Cmax, µM mean ±SEM | Tmax, h | T1/2, h | AUC0-24h, µM.h |
|---|---|---|---|---|
| Paw Skin | 36.3 ± 3.6 | 0.5 | 9.8 | 486.9 |
| Paw muscle | 28.8 ± 3.4 | 0.5 | 5.9 | 352.5 |
| Leg muscle | 6.5 ± 1.9 | 6 | – | 97.3 |
| SN | 1.2 ± 0.2 | 3 | 14.0 | 11.1 |
| DRG | – | – | – | – |
| Blood | 4.0 ± 1.0 | 0.5 | 13.7 | 14.0 |
Having detected PS and sulindac in SN tissue, we compared the levels of PS and its metabolites in the proximal and distal half of the sciatic nerve of mice 30 min after its application to the hind paw (Table III). The two values were strikingly different, with those of the distal half being 18.9-fold higher than those of its proximal half (17 ± 5.1 µM vs. 0.9 ± 0.3 µM; Table 3). Also shown in Table III, the concentration of the three metabolites of PS (sulindac, sulindac sulfide, and sulindac sulfone) was also higher in the distal half compared to the proximal half (4.5-8.5-fold higher).
| Distal | Proximal* | Fold difference | |
|---|---|---|---|
| PS | 17.0 ± 5.1 | 0.9 ± 0.3 | 18.9 |
| Sulindac | 3.4 ± 1.1 | 0.4 ± 0.03 | 8.5 |
| Sulindac sulfide | 3.5 ± 1.0 | 0 | – |
| Sulindac sulfone | 0.9 ± 0.2 | 0.2 ± 0.08 | 4.5 |
aAll differences between proximal and distal values are statistically significant (p<0.001). N=8 mice/group
EFFECT OF PS DOSE ON PHARMACOKINETICS/BIO-DISTRIBUTION
Given the dose dependence of the effect of PS in reversing mechanical allodynia in CIPN, we investigated the effect of increasing doses of PS on the PK/biodistribution of PS in areas such as the site of its application, contiguous (paw muscle) or vicinal (leg muscle) tissues as well as the sciatic nerve and blood. Mice were treated with 1.2%, 5%, and 8% PS pH 7.5 gel, in which PS was administered topically to their hind paws, and their tissues were harvested at the indicated time points. As shown in Figure 3A-D and Table IV, PS was detected in paw skin, its application site, the muscles underneath the skin (paw muscle), leg muscles, and the sciatic nerve in a dose-dependent manner. Minimal PS levels were detected in DRG (data not shown) and no PS was detected in the circulation.
| Tissue | Cmax, µM mean ± SEM | Tmax, h | T1/2, h | AUC0-24h, µM.h |
|---|---|---|---|---|
| Paw skin | 1.2% 9.8 ± 2.3 | 0.5 | 21.9 | 117.9 |
| Paw muscle | 1.2% 6.6 ± 1.9 | 0.5 | 15.8 | 79.0 |
| Leg Muscle | 1.2% 62.5 ± 4.1 | 0.5 | 8.5 | 49.2 |
| SN | 1.2% 0.6 ± 0.2 | 3 | 27.7 | 9.9 |
a, cannot be determined. N =5-8 mice/time point
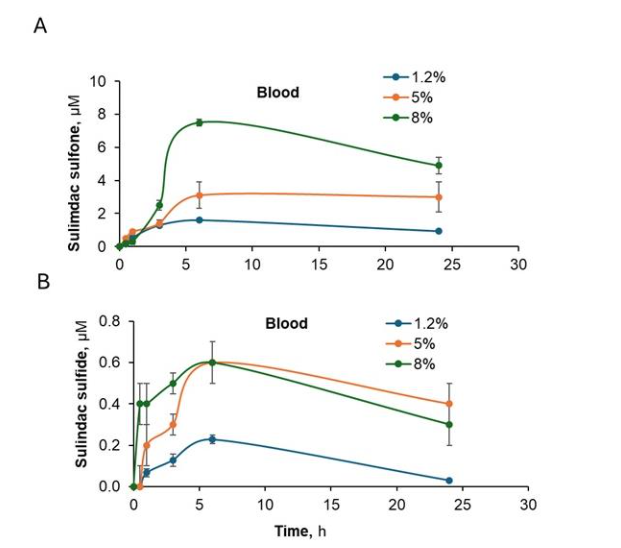
We also detected the three metabolites (sulindac, sulindac sulfone, and sulindac sulfide) for all PS doses administered. As expected, sulindac was the most abundant metabolite and it was present in all tissues including blood with higher concentrations detected in mice treated with 5% and 8% PS gels as compared to 1.2% PS. However, there were similar sulindac levels in all tissues treated with 5% and 8% PS gels. The other two metabolites, sulindac sulfone and sulindac sulfide, were only detected in blood in a dose dependent manner and showed minimal levels in other tissue.
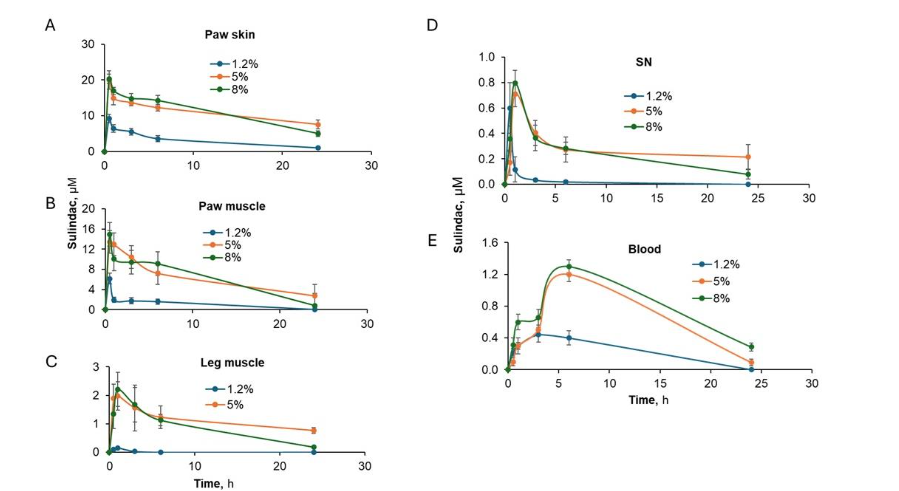
| Tissue | Cmax, µM mean ±SEM | Tmax, h | T1/2, h | AUC0-24h, µM.h |
|---|---|---|---|---|
| Paw skin | 1.2% 9.1 ± 2.2 | 0.5 | 8.7 | 73.5 |
| Paw muscle | 1.2% 6.2 ± 1.8 | 0.5 | 3.9 | 27.2 |
| Leg Muscle | 1.2% 14.9 ± 2.7 | 0.5 | 5.5 | 146.8 |
| SN | 1.2% 0.6 ± 0.1 | 0.5 | 2.0 | 0.6 |
| Blood | 1.2% 0.4 ± 0.08 | 3 | – | 2.1 |
a, cannot be determined. N =5-8 mice/time point
We also detected the three metabolites of PS at both pH levels: sulindac, sulindac sulfone, and sulindac sulfide. As shown in figures 7A-E, higher concentrations of sulindac, the metabolite of PS, were detected in all tissues (including blood) in mice treated with the higher pH gel.
pH EFFECT ON PK/BIODISTRIBUTION OF PS AND ITS EFFICACY ON MECHANICAL ALLODYNIA IN PACLITAXEL INDUCED CIPN
To explore the effect of pH on the efficacy of PS in mechanical allodynia, we compared the efficacy of a 3% gel of PS at pH 7.5 and pH 9.4. Higher doses may mask the effect of pH since PS was proved effective from 1.2% to 8%. We have reported that PS at both pH levels significantly improved the mechanical allodynia as measured by paw withdrawal threshold from D0=0.71g, to D15, being 1.11g for pH 7.5 and 1.50 g for pH 9.4; p values were <0.03 and <0.005, respectively. However, PS pH 9.4 showed greater efficacy at D15 in reversing mechanical allodynia in CIPN (pH 9.4 vs. pH 7.5, p<0.007).
To explore this, we tested the effect of pH on the biodistribution of PS in CIPN mice. We administered a pH 7.5 and pH 9.4 gel of 8% PS to the hind paws of the two different groups of CIPN mice. Tissues were harvested at the indicated time points as shown in Figure 6B-E. PS was detected in all tissues, including paw skin, the site of its application, the muscles underneath the skin (paw muscle), leg muscles, and the sciatic nerve but not in the circulation (data not shown). As shown in Figures 6B-E, a higher pH showed a mild increase in drug uptake in all tissues.
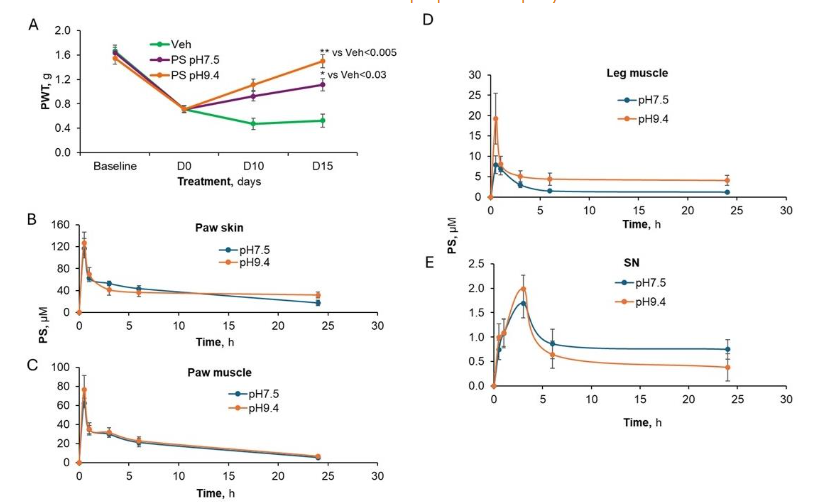
Figure 6. An elevated pH increases the effect of PS on CIPN and its biodistribution. (A) CIPN was induced by repeated injections of paclitaxel which decreased the PWT of all groups. Mice were treated with topical applications of 8% PS at two different pH’s or vehicle on D0. The higher pH of 9.4 was more efficacious than the pH 7.5 gel at restoring PWT back to baseline by D15. The p values were ()<0.005 and (*)<0.03 when compared to vehicle. The effect of pH on PS uptake was measured in (B) paw skin, (C) paw muscle, (D) vicinal leg muscle and (E) sciatic nerve and appears to be only a mild improvement.**
We also detected the three metabolites of PS at both pH levels: sulindac, sulindac sulfone, and sulindac sulfide. As shown in figures 7A-E, higher concentrations of sulindac, the metabolite of PS, were detected in all tissues (including blood) in mice treated with the higher pH gel. As shown in figure 8 A, B, respectively, sulindac sulfone and sulindac sulfide were only detected in blood and higher levels were associated with the administration of higher pH 9.4 PS gel.
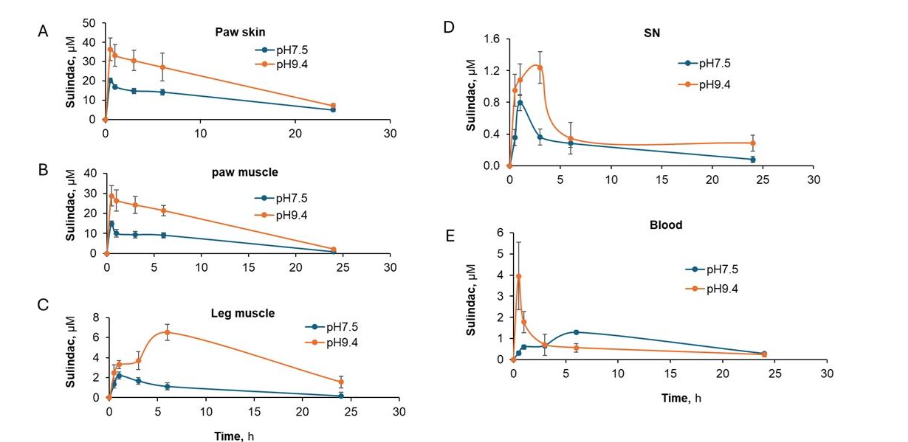 Figure 7. Kinetic and biodistribution of sulindac, the main metabolite of PS, is pH dependent. When tissues from CIPN mice were treated with topically applied 8% PS at two different pH’s 7.5 and 9.4, it main metabolite sulindac was measured in (A) paw skin, (B) paw muscle, (C) leg muscle, (D) sciatic nerve and (E) blood. In each tissue, a high pH was associated with a large amount of sulindac.
Figure 7. Kinetic and biodistribution of sulindac, the main metabolite of PS, is pH dependent. When tissues from CIPN mice were treated with topically applied 8% PS at two different pH’s 7.5 and 9.4, it main metabolite sulindac was measured in (A) paw skin, (B) paw muscle, (C) leg muscle, (D) sciatic nerve and (E) blood. In each tissue, a high pH was associated with a large amount of sulindac.
Figure 8. Kinetic profiles of sulindac sulfone and sulindac sulfide in blood are pH sensitive. The PS metabolites of (A) sulindac sulfone and (B) sulindac sulfide are only found in the blood of CIPN mice treated topically with 8% PS gels at pH 7.5 and 9.4. A higher pH was associated with a more rapid appearance of the PS metabolites in blood.
COMPARISON OF PHARMACOKINETICS/BIODISTRIBUTION OF EQUIMOLAR SULINDAC AND PS GELS
As mentioned earlier, sulindac has no efficacy on CIPN, in contrast to PS, when both were applied topically to the hind paws at equimolar concentrations (sulindac 0.7% and PS 1.2% gels). We performed a PK/biodistribution study of sulindac in which a 0.7% gel was applied to the hind paws of mice. This concentration of sulindac is equimolar to a 1.2% gel of PS and is the highest achievable concentration of sulindac in this formulation. Below, we summarize our findings.
As shown in Table VI, when 0.7% sulindac gel is topically administered to the hind paws of mice, higher levels of sulindac were detected in the paw skin, paw muscle and blood when compared to the amount of PS found in these same tissues of mice treated with equimolar 1.2% PS gel. However, lower levels of sulindac were detected in leg muscle and sciatic nerve of mice treated with equimolar sulindac when compared to PS levels in the same tissues from mice treated with equimolar PS (compared to data in Table IV). Intact sulindac is also found in blood, in contrast to PS which is quickly hydrolyzed by carboxylesterases 1 and 2 and the two metabolites of sulindac, sulindac sulfone and sulindac sulfide, were detected in the blood of mice treated with topically applied equimolar sulindac as shown in Table VI.
| Tissue | Cmax, µM mean ±SEM | Tmax, h | T1/2, h | AUC0-24h, µM.h |
|---|---|---|---|---|
| Sulindac | Paw skin 35.1 ± 3.6 | 0.5 | 13.7 | 431.0 |
| Paw muscle | 11.0 ± 2.4 | 0.5 | 46.8 | 92.1 |
| Leg muscle | 0.2 ± 0.05 | 1 | – | 0.4 |
| SN | 0.2 ± 0.04 | 0.5 | 2.8 | 0.6 |
| Blood | 13.6 ± 2.6 | 1 | – | 5.9 |
| Sulindac sulfone | Blood 3.6 ± 1.2 | 1 | 35.7 | 70.7 |
| Sulindac sulfide | Blood 5.2 ± 1.6 | 1 | 5.5 | 49.1 |
a, cannot be determined. N =5 mice/time point.
DISCUSSION
Global cancer incidence is estimated at 19.3 million new cases and 10 million deaths annually. Advances in cancer diagnosis and treatment are expected to increase the number of survivors to 26.1 million by 2040. Many patients will receive chemotherapy agents such as platinum compounds (cisplatin, carboplatin, oxaliplatin), taxanes (paclitaxel, docetaxel), vinca alkaloids (vincristine, vinblastine), and bortezomib. A common off-target effect of these therapies is CIPN, with an incidence ranging from 19% to 85%, depending on the drug and dose. Though symptoms vary slightly between agents, CIPN often causes severe, debilitating pain, significantly reducing quality of life and sometimes necessitating dose reduction, treatment delays, or discontinuation of life-prolonging therapy.
Currently, no agents are approved for CIPN prevention, and only duloxetine is recommended for treatment, with ASCO guidelines assigning it moderate benefit and low harm based on intermediate-quality evidence. The need for effective therapies remains urgent.
CIPN is associated with the loss of sensory nerve fibers in the skin, including those responding to touch, pain, and temperature. Given the peripheral nature of the damage, topical therapies may offer a targeted, safer alternative. Prior trials using lidocaine or capsaicin patches yielded limited success. An effective topical therapy could relieve pain with good tolerability, minimal systemic exposure, and a favorable safety profile. In a preclinical model, we found that topical phosphosulindac both prevented and reversed mechanical and cold allodynia in CIPN without compromising the anticancer efficacy of paclitaxel.
The current study provides the first pharmacokinetic and biodistribution profile of PS when it is applied topically to the paw skin of mice. The findings provide valuable information about its potential therapeutic uses and safety profile. Applying 8% PS gel to the hind paws of mice revealed a noticeable variation in the concentration of the drug, with higher levels observed closer to the application site and lower levels in more distant tissues. Notably, PS was discovered in substantial amounts in the skin and muscles of the paw, with gradually decreasing levels observed in the sciatic nerve and dorsal root ganglia. The Tmax and T1/2 values exhibited substantial variations among tissues, suggesting distinct rates of absorption and metabolism. The prolonged maximum concentration of the drug in the dorsal root ganglia indicates a distinct mechanism of drug transportation to neural tissues, which is likely to involve direct diffusion or uptake through the tissues rather than being distributed through the bloodstream, as indicated by the lack of presence of PS in the blood plasma.
The rapid hydrolysis of PS and its subsequent metabolism are highlighted by the detection of PS metabolites, specifically sulindac, sulindac sulfone, and sulindac sulfide, in the circulation. At the same time, the parent compound, PS, is not detected. Of the PS metabolites analyzed, higher concentrations of sulindac (quantitatively the primary metabolite of PS) were found in the various tissues. This is consistent with previous studies on the metabolic transformation of PS.
To estimate the relative distribution of PS after an administered dose, we examined the AUC0-24h as shown in Table 1 and 2 which represents the amount of phosphosulindac and sulindac measured in paw skin, paw muscle, leg muscle and sciatic nerve. If we assume that the sum of the two substances is equivalent to that of the administered dose of 8% PS, then over half of the administered dose remains on the paw skin as PS or sulindac its main metabolite. Only about a third is transported to the paw muscle and about 9% is measured in the leg muscle. Our estimate from this calculation is that only about 1% of an administered dose is transported to the sciatic nerve. This may imply that the continuous presence on paw skin of PS acts as a depot for drug to target sensory nerve endings damaged by chemotherapeutic agents and associated with peripheral neuropathy. Also important is the observation that only small amounts of PS reach the sciatic nerve and the dorsal root ganglia. If these tissues are a target for PS in CIPN, then they must be exquisitely sensitive to PS’s effect.
These data also indicate that further improvements in drug efficacy may be achieved by optimizing its formulation and thus enhancing its delivery to the target neural tissues.
We have also shown that the higher the concentration of PS (5 and 8% verses 1.2%) the higher the drug levels in the sciatic nerve as accompanied by a proportional increase in AUC0-24h. This correlation between dosage and biodistribution is consistent with the therapeutic benefits observed in previous studies on CIPN. Remarkably, the pH of the PS formulation had a significant impact on its biodistribution. Although there was no significant difference in the maximum concentration of PS in paw skin and muscle between pH 7.5 and 9.4 solutions, the concentration in leg muscle was considerably higher at pH 9.4. We also found higher concentrations of PS metabolites in mice treated with 8% PS pH 9.4 indicating that the formulation’s pH can influence the substance’s ability to enter tissues, be metabolized and appear in the bloodstream. The practical significance of these observations must be tempered by the pH-dependent vulnerability of the PS molecule. Specifically, its carboxylic ester linkage between the sulindac molecule and its diethyl-phospho-butane moiety is more susceptible to being hydrolyzed at high pH, thus inactivating the PS molecule. Thus, the dual effect of high pH on biodistribution and metabolism of PS probably merits further scrutiny in future studies.
When sulindac, the main derivative of PS, was applied directly to the skin, it showed distinct pharmacokinetic and biodistribution characteristics. Although sulindac shares a similar structure with PS, it did not reach similar levels in neural tissues. The presence of the intact compound in the circulation revealed the distinct metabolic pathways and biodistribution profiles of the two compounds. Thus, in contrast to PS, the ineffectiveness of equimolar sulindac in CIPN models may among other factors be a consequence of low delivery to neural tissues in contrast to its higher levels in skin and vicinal muscle tissues and highlights the distinct difference in its therapeutic potential as a treatment for CIPN.
Our results can be summarized as follows. Phosphosulindac is found in all tissues analyzed including paw skin its site of application, vicinal muscles, sciatic nerve and dorsal root ganglia but not the circulation. Like other phospho-NSAIDs, PS is metabolized through the P450 system, whose expression can differ between tissues. Its principal metabolite, sulindac is found in all the tissues analyzed including blood but not in dorsal root ganglia. The other metabolite, sulindac sulfone, is found in paw skin, paw muscle and blood. Sulindac sulfide, the active metabolic of sulindac, is only found in blood.
The results of this study support the effectiveness of applying PS to the skin as a specific treatment for conditions such as CIPN, where delivering the medication directly to the peripheral nerves and their superficial nerve endings has a beneficial therapeutic effect. Phosphosulindac’s ability to reach therapeutic levels in neural tissues in spite of the near total absence in the circulation makes it a promising candidate for further clinical evaluation as a topically applied treatment/prevention of CIPN. Future research should focus on optimizing the pH, drug concentration and perhaps the formulation to improve therapeutic outcomes while reducing systemic exposure and toxicity.
This extensive study on the pharmacokinetics and biodistribution of dermal applied PS gel in a preclinical model provides a foundation for its potential therapeutic application in human patients, specifically for conditions characterized by peripheral neuroinflammation and pain.
CONCLUSION
This study presents the first pharmacokinetic and biodistribution analysis of PS, a novel therapeutic candidate for CIPN. Our findings demonstrate that a significant fraction of PS remains at the site of its dermal application and reaches directly contiguous nerves, where it presumably exerts its pharmacological actions in CIPN. While the amounts found in sciatic nerve and dorsal root ganglia are much smaller, they may still be indicative of another possible site of action for PS in CIPN, demonstrating targeted biodistribution to key sites of CIPN pathology. The detection of PS in the various tissues at or near its site of application (4-52 pmol/mg tissue) and more distal neural tissues such as sciatic nerve and dorsal root ganglia provide important mechanistic support for its previously reported in vivo efficacy. It also provides data for dose selection and the rationale for its topical dermal application for treating CIPN in mice. Importantly, our results support the continued development of topical PS as a treatment for human CIPN.
Conflict of Interest Statement:
BR has an equity position in Medicon Pharmaceuticals, Inc. and LH was employed at Medicon Pharmaceuticals during the conduct of this study. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Statement:
This work was supported in part by funds provided by Stony Brook University and Medicon Pharmaceuticals, Inc. (Setauket, NY).
Acknowledgments:
Phosphosulindac was a gift of Medicon Pharmaceuticals, Inc. (Setauket, NY).
Data Availability Statement:
Research data used in preparation of this manuscript is available on reasonable request to the corresponding author.
References:
- Mackenzie GG, Sun Y, Huang L, et al. Phospho-sulindac (OXT-328), a novel sulindac derivative, is safe and effective in colon cancer prevention in mice. Gastroenterology. 2010;139(4):1320-32. doi: 10.1053/j.gastro.2010.06.044.
- Murray OT, Wong CC, Vrankova K, Rigas B. Phospho-sulindac inhibits pancreatic cancer growth: NFATc1 as a drug resistance candidate. Int J Oncol. 2014;44(2):521-9. doi: 10.3892/ijo.2013.2190.
- Cheng KW, Wong CC, Alston N, et al. Aerosol administration of phospho-sulindac inhibits lung tumorigenesis. Mol Cancer Ther. 2013;12(8):1417-28. doi: 10.1158/1535-7163.MCT-13-0006-T.
- Zhu R, Cheng KW, Mackenzie G, et al. Phospho-sulindac (OXT-328) inhibits the growth of human lung cancer xenografts in mice: enhanced efficacy and mitochondria targeting by its formulation in solid lipid nanoparticles. Pharm Res. 2012;29(11):3090-101. doi: 10.1007/s11095-012-0801-x.
- Cheng KW, Mattheolabakis G, Wong CC, et al. Topical phospho-sulindac (OXT-328) is effective in the treatment of non-melanoma skin cancer. Int J Oncol. 2012;41(4):1199-203. doi: 10.3892/ijo.2012.1577.
- Mattheolabakis G, Mackenzie GG, Huang L, Ouyang N, Cheng KW, Rigas B. Topically applied phospho-sulindac hydrogel is efficacious and safe in the treatment of experimental arthritis in rats. Pharm Res. 2013;30(6):1471-82. doi: 10.1007/s11095-012-0953-8.
- Basu A, Yang JY, Tsirukis VE, et al. Phosphosulindac (OXT-328) prevents and reverses chemotherapy induced peripheral neuropathy in mice. Front. Neurosci. 2024;17 doi: 10.3389/fnins.2023.1240372.
- Colvin LA. Chemotherapy-induced peripheral neuropathy: where are we now? Pain. 2019;160 Suppl 1:S1-S10. doi: 10.1097/j.pain.0000000000001540.
- Wong CC, Cheng KW, Papayannis I, et al. Phospho-NSAIDs have enhanced efficacy in mice lacking plasma carboxylesterase: implications for their clinical pharmacology. Pharm Res. 2015;32(5):1663-75. doi: 10.1007/s11095-014-1565-2.
- Wong CC, Cheng KW, Xie G, et al. Carboxylesterases 1 and 2 hydrolyze phospho-nonsteroidal anti-inflammatory drugs: relevance to their pharmacological activity. J Pharm Exp Ther. 2012;340(2):422-32. doi: 10.1124/jpet.111.188508.
- Xie G, Nie T, Mackenzie GG, et al. The metabolism and pharmacokinetics of phospho-sulindac (OXT-328) and the effect of difluoromethylornithine. Br J Pharmacol. 2012;165(7):2152-66. doi: 10.1111/j.1476-5381.2011.01705.x.
- Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8(6):e1000412. doi: 10.1371/journal.pbio.1000412.
- Wen Z, Muratomi N, Huang W, et al. The ocular pharmacokinetics and biodistribution of phospho-sulindac (OXT-328) formulated in nanoparticles: Enhanced and targeted tissue drug delivery. Int J Pharm. 2019;557:273-9. doi: 10.1016/j.ijpharm.2018.12.057.
- Currie GL, Angel-Scott HN, Colvin L, et al. Animal models of chemotherapy-induced peripheral neuropathy: A machine-assisted systematic review and meta-analysis. PLoS Biol. 2019;17(5):e3000243. doi: 10.1371/journal.pbio.3000243.
- Eldridge S, Guo L, Hamre J III. A comparative review of chemotherapy-induced peripheral neuropathy in in vivo and in vitro models. Toxicol Pathol. 2020;48(1):190-201. doi: 10.1177/0192623319861937.
- Carozzi VA, Canta A, Oggioni N, et al. Neurophysiological and neuropathological characterization of new murine models of chemotherapy-induced chronic peripheral neuropathies. Exp Neurol. 2010;226(2):301-9. doi: 10.1016/j.expneurol.2010.09.004.
- Toma W, Kyte SL, Bagdas D, et al. Effects of paclitaxel on the development of neuropathy and affective behaviors in the mouse. Neuropharmacol. 2017;117:305-15. doi: 10.1016/j.neuropharm.2017.02.020.
- Hab Y, Smith MT. Pathobiology of cancer chemotherapy-induced peripheral neuropathy (CIPN). Front Pharmacol. 2013;4:1-16. doi: 10.3389/fphar.2013.00156.
- Ferly J, Colombet M, Soerjomataram I, et al. Cancer statistics for the year 2020: An overview. Int J Cancer. 2021;149:778-789. doi: 10.1002/ijc.33588.
- Shapiro CL. Cancer survivorship. N Engl J Med. 2018;379(25):2438-2450. doi: 10.1056/NEJMra1712502.
- Flatters SJL, Dougherty PM, Colvin LA. Clinical and preclinical perspectives on chemotherapy-induced peripheral neuropathy (CIPN): a narrative review. BJA. 2017;119(4):737-49. doi: 10.1093/bja/aex229.
- Chen X, Gan Y, Au NPB, Ma CHE. Current understanding of the molecular mechanisms of chemotherapy-induced peripheral neuropathy. Front Mol Neurosci. 2024;17:1345811. doi: 10.3389/fmol.2024.1345811.
- Loprinzi CL, Lacchetti C, Bleeker J, et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: ASCO guideline update. J Clin Oncol. 2020;38(28):3325-3348. doi: 10.1200/JCO.20.01399.
- Maihofner C, Diel I, Tesch H, Quandel T, Baron R. Chemotherapy-induced peripheral neuropathy (CIPN): current therapies and topical treatment option with high-concentration capsaicin. Support Care Cancer. 2021;29:4223-4238. doi: 10.1007/s00520-021-06042-x.
- Desforges AD, Hebert CM, Spence A, et al. Treatment and diagnosis of chemotherapy-induced peripheral neuropathy: An update. Biomed Pharmacother. 2022;147:112671. doi: 10.1016/j.biopha.2022.112671.
- Xie G, Wong CC, Cheng KW, Huang L, Constantinides PP, Rigas B. Regioselective oxidation of phospho-NSAIDs by human cytochrome P450 and flavin monooxygenase isoforms: implications for their pharmacokinetic properties and safety. Br J Pharmacol. 2012;167(1):222-32. doi: 10.1111/j.1476-5381.2012.01982.x.
- Zanger UM, Schwab M. Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther. 2013;138(1):103-41. doi:10.1016/j.pharmthera.2012.12.007.