Diabetic Foot Microangiopathy: Risks and Management
Microangiopathic disease in the diabetic neuro-ischemic feet: A threatening, often understated contributor, for tissue and limb loss
Vlad Adrian Alexandrescu MD, PhD¹, Arnaud Kerzmann MD¹, Evelyne Boesmans MD¹, Vincent Tchana-Sato MD, PhD¹.*
- Cardiovascular and Thoracic Surgery Department, CHU Sart-Tilman University Hospital Center, Liège, Belgium.
OPEN ACCESS
PUBLISHED 31 July 2025
CITATION Alexandrescu, VA., Kerzmann, A., et al., 2025. Microangiopathic disease in the diabetic neuro-ischemic feet: A threatening, often understated contributor, for tissue and limb loss. Medical Research Archives, [online] 13(7). https://doi.org/10.18103/mra.v13i7.6730
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6730
ISSN 2375-1924
ABSTRACT
Conventionally, vessels exhibiting diameters between 100 μm and 5 μm are related to the ubiquitarian microcirculatory realm that gathers the arterioles, the capillaries and the venules. This important network represents a fundamental anatomical and functional unit involved in all vital processes of human tissues. The microcirculatory system secures and regulates the microvascular flow and intraluminal pressure within specific organs (including the lower extremities). These purposes are achieved by harmonising the luminal diameter of regional microvessels to the changing metabolic needs, oxygen demand, neurologic signals, and eventual pathological surrounding conditions. Particularly in patients with diabetes mellitus, hyperglycaemia can generate characteristic microcirculatory damage through molecular, biochemical, structural, and functional alterations, which add reactional, self-unfolding, hypoxic and pro-coagulant local tissue conditions. Among various localisations of diabetic microvascular disease (retinopathy, nephropathy, peripheral neuropathy, etc.), diabetic foot microangiopathy essentially produces aberrant arteriolar wall remodelling, coupled with progressive thickening and stiffness of the concerned microvessels, which can lead to increased regional blood flow occlusions and increasing local tissue ischemia. Progressive structural and functional impairment of endothelial cells in arterioles and capillaries is well known in diabetes mellitus and mirrors the gradual loss of microvascular repair ability. Recent video-capillaroscopy research specifically conducted in patients with diabetes documented that the duration of diabetes and the severity of chronic hyperglycaemic levels, significantly influenced the spread and gravity of peripheral diabetic microangiopathy. This latest combined with peripheral macrovascular arterial disease manifested a 12-fold higher risk for major amputation, while suboptimal glycaemic control during and post-revascularization showed a significantly higher propensity for major adverse limb events in patients with chronic arterial disease and diabetic microangiopathy. This review equally avails a succinct overview of current diagnostic and treatment methods applied to diabetic microangiopathy concomitantly to prompt macrovascular revascularization, owing systematic surveillance by a multidisciplinary diabetic foot team. Significant clinical implications of inferior limb microangiopathy isolated, versus associated to diabetic arteriopathy and neuropathy, or to other parallel systemic microvascular manifestations are developed in distinct paragraphs, following referred recommendations, for better clinical knowledge and applicability.
Keywords
chronic limb-threatening ischemia, hyperglycaemia, diabetic microangiopathy, microcirculation, diabetic peripheral neuropathy, diabetic foot syndrome, arterial calcification, capillary disease, peripheral angioplasty, inferior limb salvage.
Introduction
Persistent hyperglycaemia is an important triggering factor for macrovascular (cardiac, cerebral, and peripheral) complications as well as microvascular (retinopathy, nephropathy, and peripheral neuropathy) hindrances in type 1 and 2 diabetes mellitus. Despite increasing dedicated clinical research, particularly for inferior limbs suffering from chronic ischaemia, high glycaemic levels are reputed to initiate or aggravate diabetic macroangiopathy and microangiopathy at the local and systemic levels. This may lead to increased cardiovascular morbidity, lower limb amputations of all types, and high mortality (up to 50% at five years) in these patients. Contemporary reports show that approximately 15% of diabetic patients develop foot ulcers during their lifetime and that, unfortunately, 14%–43% of them will end with inferior limb amputations. The presence of lower limb microvascular disease appears to significantly increases the risk of amputation in patients with evidence of peripheral artery disease (PAD) and chronic limb-threatening ischaemia (CLTI). This latest, represents the most severe form of PAD and an important risk factor for high-level vascular morbimortality and socioeconomic burden, even after patent macrovascular revascularization. The microcirculatory system can be pictured as a paired structure to any organ or tissue region in the body, with which is linked through oxygen and nutrient supply, tissue regeneration activity, metabolic, hormonal, genetic and epigenetic signalling. Among all these systemic interactions, the threatened diabetic, neuro-ischemic inferior limb is also included. The complex and independently unfolding diabetic peripheral microvascular hurdle shows equal importance in CLTI prevention and treatment as the macrovascular occlusive damage, generally more deeply analysed in the literature, for tissue and inferior limb preservation. Diabetic foot microangiopathy gained an increasing interest for limb salvage in the last decades (parallel to diabetic retinopathy, nephropathy, cardiomyopathy, or neuropathy knowledge), that continues to grow. This autonomous piece of the wider metabolic syndrome puzzle embodies nowadays a characteristic and undeniable threat for major amputation in patients with, or without associated macrovascular occlusive CLTI.
The aim of this review was to update novel research focusing on diabetic peripheral microangiopathy in CLTI neuro-ischemic limbs, by integrating current aetiologic, structural, pathophysiologic, diagnostic, and therapeutic observations. This paper also endeavours to highlight the importance of correct multidisciplinary assessment, adapted treatment and follow-up, for every concomitant macro- and microcirculatory presentation in CLTI diabetic patients; we believe that this scope may prove useful for every knowledgeable clinician and interventionist in their current practice.
Material and Methods
PUBLICATION SCREENING
A parallel MEDLINE database analysis associated with unrestricted online exploration was conducted for all publications related to diabetic foot microangiopathy (Figure 1). A preferential data collection from the last decade publications was employed, that involved 16 keywords, including ‘hyperglycaemia’, ‘chronic limb-threatening ischemia’, ‘macro- and microcirculation’, ‘diabetic neuro-ischemic foot’, ‘microangiopathy’, ‘arterial calcification’, ‘chronic limb-threatening limb ischemia’, among others, without restriction on paper language.
DATA ASSEMBLING AND SELECTION
Marked inhomogeneity in paper designs, research protocols, and pathological process analysis was observed. The article identification, matching criteria, and selection process are illustrated in Figure 1.

Structural and physiological microcirculatory aspects
The human body vasculature gathers a wide variety diameter perfusion structures, ranging from 2 cm to 5 μm. Conventionally, vessels with > 100 μm diameter are referred to the macrocirculation, while vessels exhibiting diameters between 5 μm and 100 μm are related to the ubiquitarian microcirculatory realm that gathers the arterioles, the capillaries and venules. From a structural perspective, one contemporary publication underscoring on this topic, proposes a simplified “six-level” scale arrangement concerning the inferior limb arterial segmentation (Figure 2), mainly based on characteristic branching levels and on structural histologic criteria. This model differentiates the arterial macrocirculatory levels (1–4) starting from the iliac arteries to the foot arches and their collaterals (all featuring a reproducible five-layer wall structure), followed by the microcirculatory levels (5 and 6) represented by the proximal and distal arterioles and capillaries. These tiny microcirculatory structures afford a specific wall configuration that is fully adapted to each organ or tissue region, owing to five or fewer wall structural layers (with variable thickness).
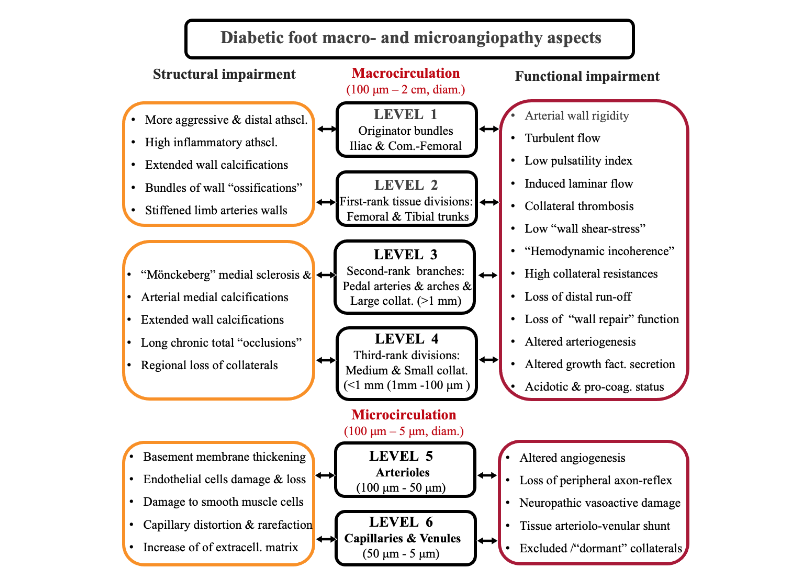
The capillaries wall structure is reinforced by a basement membrane that longitudinally sustains a kilometre pavement of endothelial cells surrounded by pericytes. The arterioles exhibit a thin layer of smooth muscle cells (adapted to each host organ) and are connected to smaller-diameter meta-arterioles which dispose of the precapillary sphincters. These act as important peripheral flow resistance-coordinating conduits that harmonise regional blood flow towards specific tissue territories. Hence, any specific organ microperfusion pathology (including the foot) can be assessed as a unique ‘anatomo-functional unit’ that associates countless of microvascular pathways to various target tissue-flow distribution.
From a functional perspective, the integrity of the capillary wall ensures a bidirectional passive and active permeability function, which influences the macrocirculatory haemodynamic parameters. All of these bilateral interchanges need to be conceived in an indissoluble circle of interactions. This bilateral unceasing interplay between the macro- and microcirculation is known as ‘hemodynamic coherence’. Muscular and skin microcirculatory flow regulation of the normal foot is governed by both a central neural-mediated flow balance (vasoconstriction vs. vasodilatation) and the veno-arteriolar axon reflex, a local neural-mediated reflex. These functions are essential for controlling harmful variations in foot capillary pressure. Displaying grim consequences since abolished by peripheral neuropathy and by diabetic foot microangiopathy.
Etiological and pathophysiological pathways for diabetic foot microangiopathy
ACUTE AND CHRONIC PRESENTATIONS
Microcirculatory manifestations can be classified into acute and chronic clinical presentations. Among the acute forms, various types of abrupt microvascular occlusions can be identified, including those associated with acute limb ischemia, cardiogenic or septic shock, massive distal limb microembolism, vasospastic diseases, critical vasoconstrictive medications, and local septic thrombosis of collateral vessels in the context of diabetic foot syndrome.
The chronic forms of microangiopathy are often associated with systemic pathologies, such as hypertension, diabetes mellitus, vasculitis, chronic kidney disease, various medications and drug abuse, and peripheral arterial chronic occlusion at the macro- and microcirculatory levels. The severity of the diabetic foot microvascular disease appears to be significantly related to the concomitant presence and gravity of other microangiopathic manifestations, such as retinopathy, nephropathy, and neuropathy, as linked markers of diabetic complications in same individuals.
CAPILLARY DAMAGE
Persistent hyperglycaemia induces both direct systemic cellular toxicity and indirect targeted organ hypoxia by damaging the ubiquitin-dependent capillary network and endothelial cells. Progressive structural and functional impairment of endothelial cells is well known in diabetes mellitus and mirrors the gradual loss of microvascular repair ability. More specifically, hyperglycaemia affects the mitochondria of the endothelial cells that consequently generate excessive amounts of ‘reactive oxygen species’ with recognised tissue toxicity. Parallel and deleterious metabolic pathways are involved, such as the polyol, hexosamine, and protein kinase pathways. A specific increase in the glycation process leads to an excessive load of irreversible advanced glycation end products (AGE), resulting in a parallel increase in their receptors for AGE. Thus, further activation of macrophages and microvascular smooth muscle cells, together with stressed endothelial capillary cells, is observed. A primordial participation of AGE interactions in the genesis of the complex diabetic peripheral microvascular disease was also postulated. AGEs can also directly alter the neuro-ischemic limb’s regional arteriolar and capillary endothelial cells and their extracellular matrix proteins including type IV collagen and laminin. Through this detrimental effect, AGEs simultaneously impair the structural integrity and function of the normal microvessel walls by promoting sclerotic tissue accumulation, while concurrently enhancing muscular and connective tissue fibrosis in the diabetic neuroischemic limb.
In a recent study using nailfold videocapillaroscopy by Kintrup et al., the authors performed direct capillary quantification in patients with a critical haemodynamic status (20% of diabetic limbs). The authors observed significant changes in capillary morphology including severe rarefaction, distortions, ‘brushy and small loops formation’, that inflict a brake-down of the normal capillary shape. Parallel functional changes lead to 1) microvascular uncontrolled vasoconstriction (by nitric oxide and prostacyclin decline, following any endothelial injury), 2) to increased local coagulation, and to 3) fluid extravasation and supplementary wall thickening. The abovementioned structural functional damage inflicts a significant increase in microvascular resistance, which decreases the normal blood flow distribution in specific foot territories.
Similar capillaroscopy research by Neubauer-Geryk et al. specifically conducted in patients with diabetes suggested that the duration of diabetes significantly influences the spread and severity of peripheral diabetic microangiopathy. The authors described characteristic hyperglycaemic capillary irregularities, which first included a specific basement membrane thickening that may vary in different types of microvascular beds. For example, the retina, myocardium, peripheral nerves, skin, and muscles may exhibit different types of capillary distortions. Additional pathological changes in capillaries include glycocalyx degeneration, progressive loss of endothelial cells and pericytes, subsequent decline and migration of smooth muscle cells, and dysregulation of vasodilation and vasoconstriction driven by persistent oxidative stress and newly circulating ‘vasoactive agents’. Moreover, the loss of pericytes induces disorganised local angiogenesis represented by acellular capillaries which inflict complementary impaired perfusion in tissues. The presence of peripheral neuropathy appears to be directly correlated to a reduction in toe nailfold capillaries perfusion (as microangiopathic issue), and with higher morbidity and risk for limb loss following revascularization, in diabetic microangiopathic feet. Hyperglycaemia appears to independently inhibit the growth of vascular progenitor cells, making capillary wall repair impossible.
ARTERIOLAR IMPAIRMENT
At rest, approximately 25% of the arteriolar network is actively perfused. Unlike capillaries, arterioles feature an extremely thin vascular smooth muscle tunica media that triggers specific pathological transformations. Madonna et al. synthetized the main microcirculatory (arteriolar and capillary) structural and functional damages, inflicted by hyperglycaemia as: 1) biochemical changes (by increasing AGE and subsequent oxidative stress); 2) structural changes (extracellular, medial layer, excessive storage of matrix proteins, collagen, and fibronectin), and 3) functional changes (impaired endothelial cell regeneration, abnormal growth and migration of vascular smooth muscle cells, reduced angiogenesis, neuropathic affliction and loss of local vascular reactivity, ultimately resulting in ‘cutaneous shunt’ phenomenon). These changes promote an independent, chronic, procoagulant, and ischemic state within the microcirculation.
Previous publications documented a thickening process that develops at the level of capillaries and arterioles initially designed as a progressive ‘basement membrane condensing’ phenomenon. The resulting lumen narrowing was identified as a key structural change responsible for reduced blood flow and diminished delivery of oxygenated haemoglobin to the target foot tissues, leading to impaired tissue healing and an increased risk of septic complications. Diabetic foot microangiopathy enhances aberrant arteriolar wall remodelling. This process involves initial wall oedema, damage to smooth muscle cells, and their subsequent replacement through the accumulation of extracellular connective matrix and calcifications, including lack of regenerative stem cell activation. The resulting medial layer extracellular matrix (altered by chronic hyperglycaemia) may transform into specific local sclerosis, increased wall stiffness, and initiation of medial layer focal calcification hubs. These characteristic infragenicular arterial and arteriolar wall calcifications were previously described as ‘medial layer degenerative sclerosis’ that can associate the ‘Mönckeberg calcifications’, particularly encountered in diabetic neuro-ischaemic limbs, also in renal patients. Regarding functional arteriolar changes, beyond the above-mentioned ‘cutaneous shunting phenomenon’, a consecutive increase in the peripheral vascular resistance leads to a decline in the mean diastolic arterial pressure.
REVERSIBLE AND NON-REVERSIBLE MICROVASCULAR EFFECT
Microangiopathic manifestations of diabetic foot can harbour both reversible and, more frequently, non-reversible forms of occlusive disease. For the former, transient hyper-pressure in specific foot regions can give rise to non-circulating (yet non-occluded) ‘dormant’ small collaterals or temporarily shunted arteriolo-capillary territories, which may play a critical role following macrovascular revascularization. The irreversible modifications assemble all structural and functional wall alterations in collaterals, arterioles, and capillaries inflicted by hyperglycaemia. This state was synthetized by O’Neal et al. as follows: ‘In diabetic limbs, areas ranging from a few millimetres of skin to the entire foot may rely on a single end-territory vessel, in accordance with the ‘end-artery occlusive disease’ (EAOD) theory.
Diabetic foot microangiopathy and major adverse limb events
Patients with diabetes exhibit a more aggressive and distally situated below-the-knee and below-the-ankle atherosclerotic occlusive disease, expressed by peripheral arterial disease (PAD) and by CLTI, as an extreme form of inferior limb chronic flow deprivation. It has been estimated that patients with diabetes with PAD have a 10-fold higher risk of major amputation than patients who are non-diabetic. Seemingly, in a vast cohort of 933,597 individuals in a study by Kris et al., they found that diabetic microvascular disease combined with PAD manifested a 12-fold higher risk for major amputation, throughout a seven-year study period. The severity of PAD or CLTI (stratified by macro- and microvascular analyses) was significantly influenced by the duration of diabetes and levels of uncontrolled hyperglycaemia. In another large cohort of patients, Behroozian et al. assessed the reciprocal relationship between predominant microvascular diseases (retinopathy, neuropathy, and/or nephropathy) and peripheral limb microangiopathy, either in isolation or in association with PAD or CLTI. The authors found an increase in the amputation risk of 3.7-fold, in the presence of lower-limb microvascular disease only; this risk increased to 22.7-fold with the additional presence of PAD. One in six below-the-knee amputations occurred in the background of microangiopathy only.
Similarly, previous research by our team revealed that diabetic peripheral neuropathy is an independent and influential factor for poor clinical outcomes of below-the-knee endovascular revascularization. According to a recent multicentre retrospective study by Cha et al. on diabetic limb revascularization, perioperative suboptimal glycaemic control showed a significantly higher propensity for major adverse limb events (through macro- and microvascular diagnostics) and proved to be an independent predictor for re-interventions after endovascular therapy in patients with diabetes with PAD. Parallel research based on a 979-patient prospective cohort study by Chen et al. recently demonstrated that the presence of medial arterial (Mönckeberg) calcifications in the foot arteries was significantly associated with higher amputation and mortality rates in patients with diabetic foot syndrome. Interestingly, in this cohort, the same type of medial calcifications was analysed, which added a characteristically unfavourable predictive value to limb preservation other than PAD. In a corresponding publication by Ferraresi et al., the authors scored the medial arterial calcification and the small artery disease in a retrospective, 259-inferior limb CLTI analysis. The authors found that these two pathological entities represent manifestations of the same underlying occlusive disease and have powerful prognostic value for major adverse limb events in patients with CLTI. Our group’s related research on the same topic developed an original four-grade (A–D) severity scale for occlusive infra-malleolar macrovascular and microangiopathic disease in CLTI-affected feet. In this study, microcirculation was specifically analysed using TcPO2 and SPECT scan imaging. The results confirmed a significant difference in the limb salvage rates observed in crescendo severity grades A, B, C, and D, which were distinctively considered for patients with diabetes and no diabetes.
All the aforementioned studies appear to match other parallel contemporary publications on microcirculatory disease implications, including consensual recommendations for CLTI treatment.
Practical point:
Patients with diabetes and particularly those with PAD need to be timely evaluated for eventual microangiopathy manifestations, such as retinopathy, neuropathy, and nephropathy. Concomitant microvascular disease should be suspected in the threatened diabetic foot, as it can increase the risk of amputation by up to 23-fold if coordinated management by a diabetic foot team is not implemented.
Diagnostic methods and stratification
Regarding diabetic foot microangiopathy, no uniform consensus is currently available regarding the best diagnostic methodology. Common clinical findings associate a worm limb (by arteriolo-venular cutaneous shunting), to chronically inflammatory oedema, and variable degrees of pain, or sensory deficit (by associated neuropathy or denervation). Distal pulses may be present (although more difficult to assess in the presence of oedema), while the capillary filling appears significantly hindered. Variable degrees of tissue loss and local sepsis may be present. Beyond morphological stratification by cutaneous video capillaroscopy (discussed above), other methods are currently in use (owing to their advantages and inherent drawbacks), such as the following:
- Pedal acceleration time and arterial pulsatility index, both based on ultrasound technology, can provide functional foot vascular resistance evaluation and parallel foot collateral reserve estimation.
- Transcutaneous pressure oxygen monitoring (TcPO2) measures skin oxygen levels (through indirect microcirculatory blood flow appraisal).
- Near-infrared spectroscopy (NIRS) provides continuous tissue oxygen distribution (StO2) values in specific skin and muscle regions.
- Hyperspectral imaging can monitor the tissue microcirculatory oxygen saturation in targeted territories (by indirect flow measurements).
- Laser Doppler flowmetry and laser speckle imaging provide non-invasive, direct, and real-time assessments of territorial capillary flow.
- Indocyanine green angiography provides a detailed anatomical and hemodynamic imaging of regional foot perfusion enabling targeted revascularization.
- Contrast-enhanced magnetic resonance is associated with arterial spin labelling (ASL) and to blood oxygenation level-dependent (BOLD) techniques; both methods enable precise, non-invasive tissue micro-vessel flow measurement and synchronous blood and tissue oxygenation appraisal.
- Spatial frequency domain imaging associates structural skin papillary haemoglobin, to tissue oxygen saturation (StO2) measurements.
- SPECT and PET scan technologies that provide direct intracellular and mitochondrial hypoxic tissue information. This aspect may be particularly important for microcirculatory diagnostics, as angiographic visualization alone of very small collaterals (<500 μm) can be challenging or scarce, owing to the limitations of human eye acuity.
Practical point:
The purpose of precise microcirculatory assessment is to detect specific foot regions with surviving small-vessel blood supply (run-off) for suitable revascularization, prompt wound healing, and reduced amputations.
Current treatment approaches
During the last few decades, the established paradigm for PAD and CLTI treatment has required macrovascular valuation and revascularization that remains the gold standard in today’s practice. However, specific methods for treating microvascular occlusive disease in diabetic neuroischemic feet are scarce despite their equal importance in limb salvage and currently involve systemic control of hyperglycaemic harm. Macrovascular flow restoration (by surgery or endovascular techniques), even by providing successful intentional direct wound revascularization, cannot constantly and utterly predict flawless tissue regeneration and limb preservation in all patients, which is particularly true in limbs exhibiting poor collateral and microvascular foot run-off. Diabetic microangiopathy should be associated with the systematic implications of a multidisciplinary diabetic foot team. Parallel therapies (without standardized application) directed to microcirculation improvement were also proposed, such as the following: 1) controlled aerobic exercise; 2) use of antiplatelet agents and lipid-lowering and glucose-lowering medications; 3) use of ‘phosphodiesterase-3 inhibitors’ that add antiplatelet and vasodilatory properties; 4) specific “Betain” supplementation in patient’s daily diet (owing to decreased hypothesized diminution of ‘reactive oxygen species’); 5) use of pro-angiogenetic factors, among others. Unlike macrovascular diseases, diabetic microangiopathy does not provide homogeneous recommendations for treatment to date. All the above-mentioned therapeutic observations are important, yet remain under clinical investigation owing to a lack of uniform evidence presently.
One specific limitation of this review is related to the vastness of diabetic microangiopathic disease (not entirely acknowledged), and to inherent difficulty to detail most of interrelated topics that it covers. Future publications, specifically focusing on preclinical, or clinical aspects, may afford much extensive introspection for selected subjects in this extensive research ground.
Practical point:
The diabetic microangiopathy requires a prompt macro- and microvascular diagnostic followed by punctual macrovascular revascularization. Although with variable accessibility in daily practice, the microcirculatory diagnostic should integrate any CLTI basic assessment. A systematic surveillance by a regular diabetic foot multidisciplinary team appears mandatory.
Perspectives
Recent research documented that genes associated with type 2 diabetes are also linked to retinopathy and neuropathy as microvascular-related pathologies, which require future targets for clinical investigation. Until novel, or improved diagnostic technology (e.g. ‘laser speckle’, ‘implantable micro-oxygen sensors’, or ‘peripheral fractional flow reserve’), adding specific biomarkers, and standardised methods for micro-flow restoration become available, stringent preventive surveillance of all feet at risk of microcirculatory compromise by a multidisciplinary team appears essential to avoid amputation.
Conclusion
The presence of diabetic lower limb microangiopathy requires clinicians’ awareness and understanding that microvascular disease always represents a multiorgan disorder with a myriad systemic presentations. Its association with peripheral arterial disease and neuro-ischemic diabetic foot syndrome indicates a potentially high cardiovascular morbidity, mortality, and amputation risk, which necessitates close surveillance of patients with prompt referral to specialised centres for expeditious revascularization and multidisciplinary monitoring.
Conflict of Interest Statement:
The authors have no conflicts of interest to declare.
Funding Statement: None.
Acknowledgements: The corresponding author acknowledges the Cardio-Vascular and Thoracic Surgery Department academic team, for their courtly support in preparing and refining this paper.
ORCID ID: Alexandrescu Vlad Adrian: https://orcid.org/0000-0002-9181-7993, Kerzmann Arnaud: https://orcid.org/0000-0002-9434-3076, Vincent Tchana-Sato: https://orcid.org/0000-0001-8838-814X
References:
- Mauricio D, Gratacos M, Franch-Nadal J. Diabetic microvascular disease in non-classical beds: the hidden impact beyond the retina, the kidney, and the peripheral nerves. Cardiovasc Diabetol. 2023; 22: 314. doi: 10.1186/s12933-023-02056-3.
- Mansour A, Mousa M, Abdelmannan D, Tay G, Hassoun A, Alsafar A. Microvascular and macrovascular complications of type 2 diabetes mellitus: exome wide association analyses. Front. Endocrinol. 14:1143067. doi: 10.3389/fendo.2023.1143067
- Hinchliffe RJ, Andros G, Apelqvist J, et al. A systematic review of the effectiveness of revascularization of the ulcerated foot in patients with diabetes and peripheral disease. Diabetes Metab Res Rev. 2012;28(suppl 1):179–217.
- Alexandrescu VA, Van Overmeire L, Makrygiannis G, et al. Clinical implications of diabetic peripheral neuropathy in primary infrapopliteal angioplasty approach for neuro-ischemic foot wounds. J Endovasc Ther. 2022;1-11. DOI: 10.1177/15266028221106312
- Smith AD, Hawkins AT, Schaumeier MJ, et al. Predictors of major amputation despite patent bypass grafts. J Vasc Surg. 2016; 63: 1279-88. http://dx.doi.org/10.1016/j.jvs.2015.10.101
- Alexandrescu VA, Pottier M, Balthazar S, Azdad K. The foot angiosomes as integrated level of lower limb arterial perfusion: amendments for chronic limb threatening ischemia presentations. J Vasc Endovasc Ther. 2019;4(1):1-7. http://vascular-endovascular-surgery.imedpub.com/
- Behroozian A, Beckman JA. Microvascular disease increases amputation in patients with peripheral artery disease. Arterioscler Thromb Vasc Biol. 2020;40:534-540. www.ahajournals.org/journal/atvb. DOI: 10.1161/ATVBAHA.119.312859
- Ince C. Hemodynamic coherence and the rationale for monitoring the microcirculation. Crit Care. 2015;19(3):S8.
- Sharma S, Schaper N, Rayman G. Microangiopathy: is it relevant to wound healing in diabetic foot disease? Diabetes Metab Res Rev. 2020;36(S1):e3244 https://doi.org/10.1002/dmrr.3244
- Hingorani A, LaMuraglia GM, Henke P, et al. The management of the diabetic foot: a clinical practice guideline by the Society for Vascular Surgery in collaboration with the American Podiatric Medical Association and the Society for Vascular Medicine. J Vasc Surg. 2016;63(2 Suppl):3S–21S. doi: 10.1016/j.jvs.2015.10.003.
- Jett S, Thompson MR, Awasthi S, et al. Stratification of microvascular disease severity in the foot using spatial frequency domain imaging. J Diabetes Science Technology. 2023;17(1):25-34.
- Alexandrescu VA, Kerzmann A, Tchana-Sato V. Diabetic neuro-ischaemic foot syndrome and the key underlying interactions between chronic arterial disease and peripheral neuropathy. Medical Research Archives, 2025;13(2). ISSN 2375-1924
- Yang P, Feng J, Peng Q, Liu X, Fan Zhongcai. Advanced glycation end products: potential mechanism and therapeutic target in cardiovascular complications under diabetes. Hindawi, Oxidative Medicine and Cellular Longevity Volume 2019, ID: 9570616, 12 pages https://doi.org/10.1155/2019/9570616
- Kintrup S, Listkiewicz L, Arnemann PH, Wagner NM. Nailfold videocapillaroscopy – a novel method for the assessment of hemodynamic incoherence on the ICU. Critical care. 2024;28:400. https://doi.org/10.1186/s13054-024-05194-6
- Neubauer-Geryk J, Wielicka M, Hoffmann M, Mysliwiec M, Bieniaszewski. The impact of disease duration on microcirculatory dysfunction in young patients with uncomplicated type 1 diabetes. Biomedicines. 2024, 12, 1020. https://doi.org/10.3390/biomedicines12051020
- Madonna R, Balistreri CR, Geng YJ, De Caterina R. Diabetic microangiopathy: pathogenetic insights and novel therapeutic approaches. Vascul Pharmacol. 2017;90:1-7. doi: 10.1016/j.vph.2017.01.004.
- Guven, G.; Hilty, M.P.; Ince, C. Microcirculation: physiology, pathophysiology, and clinical application. Blood Purif. 2020, 49,143–150.
- Siperstein M, Unger RH, Madison L. Studies of muscle capillary basement membranes in normal subjects, diabetic, and prediabetic patients. J Clin Investig.1968;47(9):1973-1999.
- Jaalouk DE, Lammerding J. Mechano-transduction gone awry. Nat Rev Mol Cell Biol. 2009;10:63–73.
- Lanzer P, Hannan FM, Lanzer D, et al. Medial arterial calcification JACC state-of-the art review. JACC.2021;78(11): 1146-1165. https://doi.org/10.1016/j.jacc.2021.06.049
- Ngai D, Lino M, Rothenberg KE, Simmons CA, Fernandez-Gonzalez R, Bendeck MP. DDR1 (discoidin domain receptor-1)-RhoA (Ras homolog family member A) axis senses matrix stiffness to promote vascular calcification. Arterioscler Thromb Vasc Biol. 2020;40:1763–1776.
- Edmonds ME, Morrison N, Laws JW, et al. Medial arterial calcification and diabetic neuropathy. Br Med J. 1982;284(6320):928–930. doi:10.1136/bmj.284.6320.928.
- Duque A, Mediano MF, De Lorenzo A, et al. Cardiovascular autonomic neuropathy in diabetes: pathophysiology, clinical assessment and implications. World J Diabetes.2021;12(6):855–867. doi:10.4239/wjd.v12.i6.855.
- Mustapha JA, Diaz-Sandoval LJ, Saab F. Infra-popliteal calcification patterns in critical limb ischemia: diagnostic, pathologic and therapeutic implications in the search for the endovascular holy grail. J Cardiovasc Surg (Torino). 2017;58: 383–401.
- Alexandrescu VA, Kerzmann A, Boesmans E, et al. Singularities of the pedal circulation in CLTI: time for novel merging guidelines. J Crit Limb Ischem. 2023;3:E1-E8.
- O’Neal LW. Surgical pathology of the foot and clinicopathologic correlations. The end-artery occlusive disease conception. In: Bowker JH, Pfeifer MA. Levin and O’Neal’s The Diabetic Foot. 7th Ed. Mosby Elsevier, Philadelphia. 2007:367-401.
- Conte MS, Bradbury AW, Kolh P, et al. Global Vascular Guidelines on the Management of Chronic Limb-Threatening Ischemia. Eur. J. Vasc. Endovasc. Surg.2019, 58, S1-S109.e133.
- Thiruvoipati T, Kielhorn CE Armstrong EJ. Peripheral artery disease in patients with diabetes: epidemiology, mechanisms, and outcomes. World J. Diabetes. 2015; 6: 961–969.
- Jude EB, Oyibo SO, Chalmers N, Boulton AJ. Peripheral arterial disease in diabetic and nondiabetic patients: a comparison of severity and outcome. Diabetes Care. 2001; 24: 1433–1437.
- Kris K, Olesen W, Anand S, Thim T, Gyldenkerne C, Maeng M. Microvascular disease increases the risk of lower limb amputation – a western danish cohort study. Eur J Clin Invest. 2022; 52(10): e13812 doi: 10.1111/eci.13812.
- Arya S, Binney ZO, Khakharia A, et al. High haemoglobin A. J. Vasc. Surg. 2018; 67: 217–228.e211.
- Cha JJ, Kim H, Ko YG, et al. Influence of preprocedural glycemic control on clinical outcomes of endovascular therapy in diabetic patients with lower extremity artery disease: an analysis from a Korean multicenter retrospective registry cohort. Cardiovasc. Diabetol. 2020; 19: 1–10.
- Chen L, Chen D, Gong H et al. Pedal medial arterial calcification in diabetic foot ulcers: a significant risk factor of amputation and mortality. Journal of Diabetes. 2024;16:e13527. https://doi.org/10.1111/1753-0407.13527
- Ferraresi R, Ucci A, Pizzuto A, et al. A novel scoring system for small artery disease and medial arterial calcification is strongly associated with major adverse Limb events in patients with chronic limb-threatening ischemia. J Endovasc Ther. 2021;28(2):194-207.
- Alexandrescu VA, Brochier S, Schoenen S, et al. Grades of below-the-ankle arterial occlusive disease following the angiosome perfusion: a new morphological assessment and correlations with the inframalleolar GVG stratification in CLTI patients. Ann Vasc Surg. 2022.1;1-20. https://doi.org/10.1016/j.avsg.2021.09.031
- Biscetti F, Nardella E, Rando MM, et al. Outcomes of lower extremity endovascular revascularization: Potential predictors and prevention strategies. Int. J. Mol. Sci. 2021; 22: 2002. https://doi.org/10.3390/ijms22042002
- Lowry D, Saeed M, Narendran P, Tiwari A. The difference between the healing and the nonhealing diabetic foot ulcer: a review of the role of the microcirculation. Journal of Diabetes Science and Technology. 2017;11(5): 914–923
- LoGerfo FW, Coffman JD. Vascular and microvascular disease of the foot in diabetes. Implications for foot care. N Engl J Med. 1984; 311:1615–1619.
- Song K, Chambers AR. Diabetic foot care. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. 2025, PMID: 31971750.
- Farber A. Chronic limb-threatening ischemia. N Engl J Med. 2018;379:171–180. doi: 10.1056/NEJMcp1709326
- Teso D, Sommerset J, Dally M, et al. Pedal acceleration time (PAT): A novel predictor of limb salvage. Ann Vasc Surg 2021; 75: 189–193. https://doi.org/10.1016/j.avsg.2021.02.038
- Li FM, Liang HL, Wu TH. Pulsatility-index adjusted doppler flow measurement of pedal arteries in peripheral artery disease patients. J. Clin. Med. 2021, 10, x. https://doi.org/10.3390/xxxxx

