Variations in Hemostasis and Blood Loss in ECLS
Individual variations in hemostasis, thrombosis and blood loss during extracorporeal life support
Wanda L. Chandler MD1
- Department of Laboratory Medicine, University of Washington, Seattle, WA
OPEN ACCESS
PUBLISHED:30 June 2025
CITATION: Chandler WL, 2025. Individual variations in hemostasis, thrombosis and blood loss during extracorporeal life support. Medical Research Archives, Volume 13 Issue 6. https://doi.org/10.18103/mra.v13i6.4261
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i6.6619
ISSN 2375-1924
Abstract
Multiple factors affect how a patient responds to extracorporeal life support (ECLS) including the patient’s age, size, underlying problem, clinical condition, genetic differences and clinical progress. To better understand this highly variable response, we must separate what is happening in the patient versus the ECLS circuit. Pre-ECLS risk factors, including disseminated intravascular coagulation and recent surgery, increase the risk of platelet loss and bleeding during ECLS. Exposure of blood to artificial surfaces in the circuit activates the contact system which in turn activates coagulation. High shear in the cannula, tubing and pumps, along with coagulation activation, damages and activates platelets, red cells, endothelial cells and leukocytes. Platelets and red cells are lost during ECLS through bleeding, phlebotomy, activation, hemolysis, sub-lethal injury induced extravascular clearance, and other causes. Activation of coagulation and platelets leads to circuit thrombus formation. Clinically important circuit thrombi include arterial tubing thrombi associated with arterial embolism, oxygenator surface membrane thrombosis resulting in oxygenator failure/circuit replacement and venous pump emboli leading to severe hemolysis. The variability in response means some patients require almost continuous transfusion support while others receive almost no transfusions. This review summarizes what is known and what research is needed to improve ECLS.
Keywords
extracorporeal life support, hemostasis, thrombosis, blood loss, platelet activation, red blood cells
Introduction
While extracorporeal life support (ECLS) can be beneficial for patients suffering from severe cardiopulmonary distress, it is associated with an increased risk of blood loss, due to the hemostatic variations and thrombotic formation in the ECLS circuit. Current studies report that patients on ECLS show a continuing loss of red cells, platelets and coagulation factors. Pediatric patients on ECLS lose blood cells faster than adults. This patient blood loss leads to recurrent, at times daily, transfusion support including red cell, platelet and plasma transfusions. Increased blood loss and transfusion requirements are associated with worse outcomes in ECLS. Anticoagulant medications used during ECLS expose patients to an increased risk of bleeding and thrombosis.
While we have a good understanding of the overall blood loss, transfusion and thrombotic issues in ECLS, there is still much to learn about the individual variations that lead to these outcomes. This study aims to reduce these discrepancies by helping to understand the individual variations in blood loss and thrombotic events during ECLS.
Patient clinical condition
The first factor affecting the patient’s response to ECLS is their genetic makeup and size, 2) underlying problem leading to ECLS and 3) clinical condition. As blood flows through the ECLS circuit, cells and other humoral factors in the blood may be activated and damaged. As blood returns from the circuit to the patient these activation and damage products must be cleared by the patient’s liver, spleen and kidneys. In a patient with a larger blood volume, the 500-600 mL of blood from the ECLS circuit is diluted 5 to 10-fold in the 3 to 6 L of blood in the patient. In contrast, in a neonate with a 250 mL blood volume, the same volume of blood from the ECLS circuit is concentrated and can lead to increased activation of the coagulation cascade.
Contact system factor XII binds to circuit surfaces where it auto-activates to factor XIIa, which in turn activates coagulation factor XI. Activated coagulation factor XI initiates thrombin generation, coagulation activation and thrombus formation on circuit surfaces. Activation of the contact system peaks during the first 24 hours of ECLS. Initial contact activation is followed by progressive increases over the following days in thrombin generation and circuit fibrin deposition.
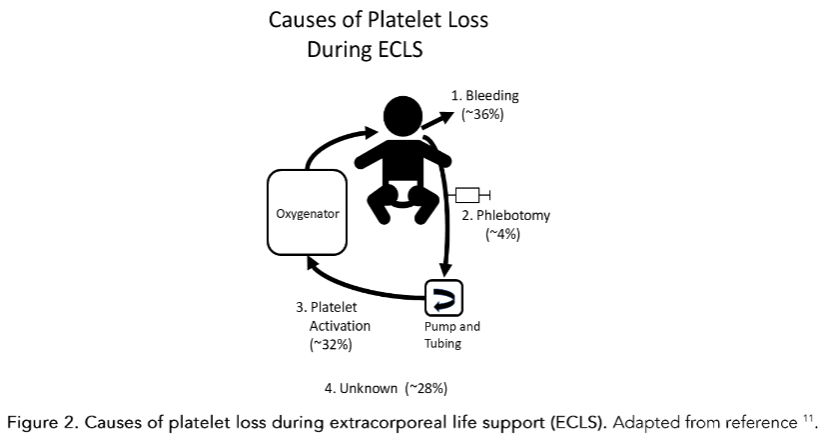
Causes of Platelet Loss During ECLS
Some patients show little to no platelet activation, limited platelet loss and require no platelet transfusions while others with a similar clinical history and circuit dynamics show rapid platelet activation and loss. This suggests there may be an intrinsic contribution to a patient’s response to the circuit. Genetic variation in platelet activation by the circuit surfaces and by humoral systems like coagulation and complement may play a role in how patients respond to ECLS.
The platelet activation marker PEV1 has been shown to measure phosphatidylserine on the surface of activated platelets, a phospholipid that promotes coagulation activation (termed platelet extracellular vesicle type 1 or PEV1). Levels of PEV1 in blood can be used to estimate the number of platelets that have been activated and in turn lost due to activation. The patient’s pre-existing condition also affects platelet activation and loss. When patients with a history of pre-existing DIC are placed on ECLS, the combination of platelet activation in the circuit and the platelet activation in the patient due to their DIC results in a vicious cycle that produces up to 100-fold increases in platelet activation that can lead to significant platelet loss.
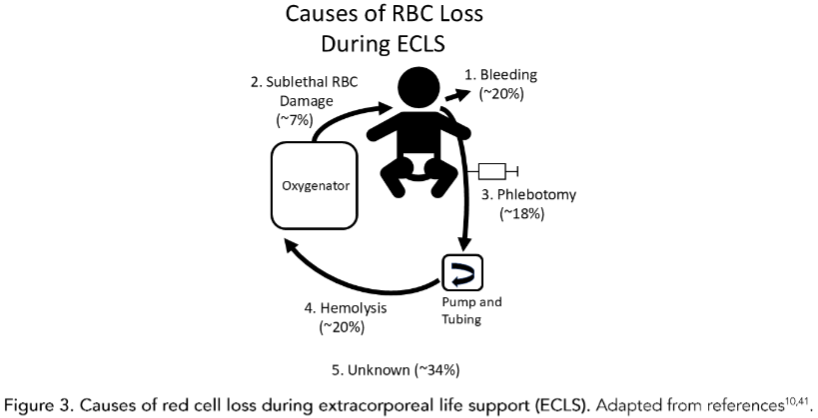
Causes of RBC Loss During ECLS
In addition to hemolysis, red cells suffer repeated sublethal damage as they flow through the ECLS circuit as indicated by decreased red cell deformability, increased exposure of phosphatidylserine on their surface and red cell extracellular vesicle release. Sublethal red cell injury does not lyse red cells, but marks them for clearance by the liver and spleen resulting in extravascular hemolysis and post-ECLS transfusion requirements. Pediatric ECLS patients with hemolysis, thrombosis, platelet dysfunction and bleeding are often the most challenging to manage.
Endothelium
Vascular endothelial cells in normal blood vessels release small numbers of endothelial extracellular vesicles (EEV) that are detected in blood. When endothelium are activated or damaged they release increased numbers of EEV. High levels of EEV in blood are an indication of vascular injury in a variety of cardiovascular disorders. On average EEV are increased about 2 to 3-fold during ECLS indicating some level of endothelial activation and vascular injury. Some patients show massive release of red cell and endothelial extracellular vesicles, which can lead to increased red cell loss and leukocyte deposition on the circuit surfaces.
Leukocytes
Thrombi are most common in tubing and connectors, affecting ~40% of circuits. Fibrin is initially deposited on the tubing by the leukocytes and platelets, red blood cells, white blood cells and von Willebrand factor. Thrombi in the arterial side of the circuit increase the risk of arterial thromboembolism in the patient.
Another clinically important circuit problem is the development of oxygenator membrane surface thrombus which can occlude the oxygenator, resulting in rapid increases in circuit delta pressures, oxygenator failure and emergent circuit replacement. Very few thrombi were apparent visually when the circuit was running, but they were detected by the circuit pressure alarms. Thrombi that are not apparent can still lead to significant blood loss and transfusion requirements.
Red Cell Transfusion Rates
Red cell transfusions on ECLS typically average about 15 to 30 mL/kg/day with some centers reporting as high as 50 mL/kg/day. The median platelet loss index in children on ECLS is 2.8×107/L, equivalent to a 67,000/L decrease in platelet count per day.
Conflict of interest statement
The authors have no conflicts of interest to declare.
References
- Thomas J, Kotsuvos V, Teruya J, Bledsoe M et al. Factors Associated with Bleeding and Thrombosis in Children Receiving Extracorporeal Membrane Oxygenation. Am J Respir Crit Care Med. 2017; 196:762-771.
- Musszynski JA, Reeder RW, Hall MW, et al. RBC Transfusion Practice in Pediatric Extracorporeal Membrane Oxygenation Support. Crit Care Med. 2018;46:e552-e559.
- Abudoh, Cheng AC, Pilcher D, et al. Factors associated with bleeding and thrombosis during extracorporeal membrane oxygenation support: a 5-year cohort study. Syst Rev. 2018;7:1-12.
- Chandler WL. Coagulation activation during extracorporeal membrane oxygenation. Thromb Res. 2022;211:154-160.
- Walsh-Sukys MC, Cornell DL, Stork EK. The incidence of late-onset hyperbilirubinemia associated with extracorporeal membrane oxygenation. J Surg Res. 1998;78:1-6.
- Smith A, Hardison D, Bridges B, Pritchard J. Red blood cell transfusion volume and mortality among patients receiving extracorporeal membrane oxygenation. Perfusion. 2013;28:54-60.
- Cashen K, Patton H, Reeder RW, et al. Platelet Transfusion Practice and Related Outcomes in Pediatric Extracorporeal Membrane Oxygenation. Pediatr Crit Care Med. 2020;21:112-118.
- Yuan S, Ferrell C, Chandler WL. Comparing the prothrombin time during extracorporeal membrane oxygenation. Thromb Res. 2007;120:29-37.

