Spirochetal Pathway Linking Alzheimer’s Disease and Biofilms
THE 3-D Pictorial Spirochetal Pathway to Alzheimer’s Disease
Herbert B. Allen1, MD Judith Miklossy, MD2, PhD
- Herbert B. Allen, MD Department of Dermatology, Drexel University College of Medicine, Philadelphia, PA, USA Dermatology, Eastern Virginia Medical School, Norfolk, VA, USA Rowan School of Osteopathic Medicine Geriatrics and Gerontology, Stratford, NJ, USA
- Judith Miklossy, MD, PhD International Alzheimer Research Centre, Prevention Alzheimer International Foundation Martigny-Croix, Switzerland
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION:ALLEN, Herbert B.; MIKLOSSY, Judith. THE 3-D Pictorial Spirochetal Pathway to Alzheimer’s Disease. Medical Research Archives, [S.l.], v. 13, n. 6, june 2025. ISSN 2375-1924. Available at: <https://esmed.org/MRA/mra/article/view/6568>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i6.6568
ISSN 2375-1924
Abstract
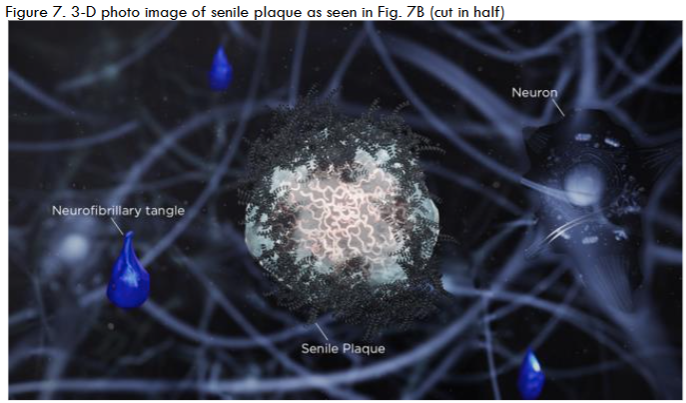
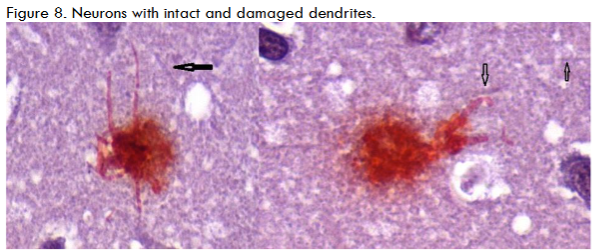
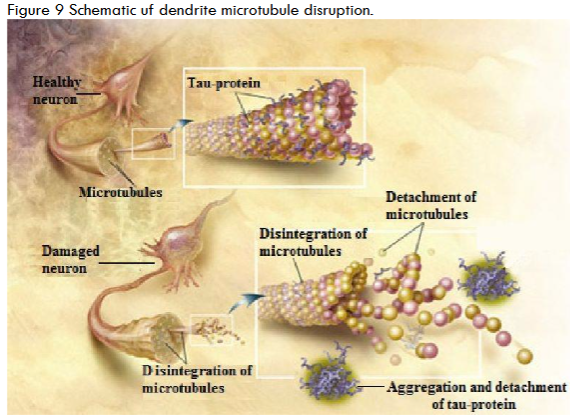
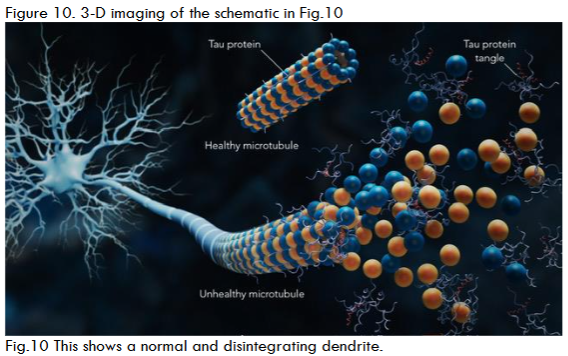
This perspective article features a visual depiction supporting a potential link between spirochetal infection and Alzheimer’s disease (AD). Two-dimensional (2D) images, alongside innovative three-dimensional (3D) imaging derived from 2D photomicrographs, provide the basis for a pathway through which spirochetes travel to the brain, with the hippocampus identified as their initial target. Upon arrival at this specific region, spirochetes can be found both extracellularly and intracellularly. Despite their slow growth, spirochetes eventually create a biofilm. Biofilm formation requires 10 cells in every direction, which can take two or more years for a significant biofilm to develop. To establish the biofilm, the spirochetes produce a slime coating that shields them from antibiotics, the immune system, and harmful irritants. Simultaneously with biofilm formation, spirochetes may induce beta-amyloid precursor protein (AβPP) and beta-amyloid (Aβ) production. In the extracellular space, biofilms can be covered with Aβ to be part of senile plaques. Intracellularly, during biofilm formation, spirochetes also generate amyloid precursors. It can also be hypothesized that spirochetes may induce tau hyperphosphorylation (p-tau), perhaps through a mechanism involving phosphatase inhibition. Accumulation of p-tau prevents the stabilization of microtubules within dendrites, leading to the disintegration of tubules and dendrites. Overall, neurofibrillary tangles are formed that, together with amyloid plaques, contribute to neuronal death. As more dendrites and neurons are destroyed, impulse transmission is impaired, resulting in cognitive deficits.Keywords
Alzheimer’s Disease, spirochetes, biofilms, beta-amyloid, tau hyperphosphorylationIntroduction
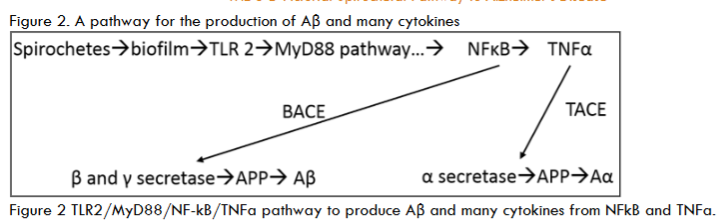
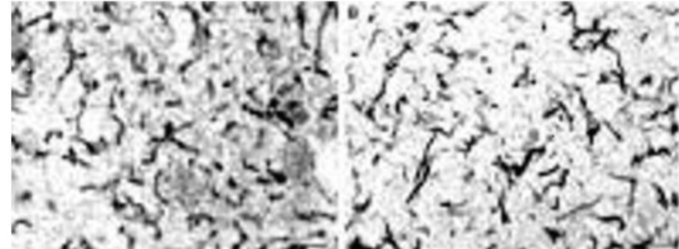
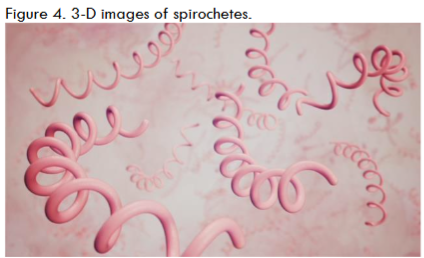
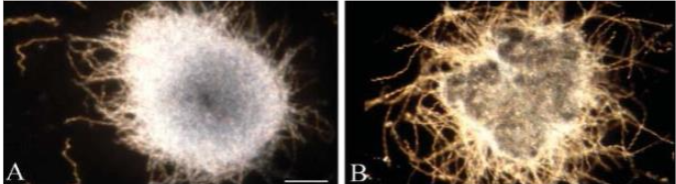
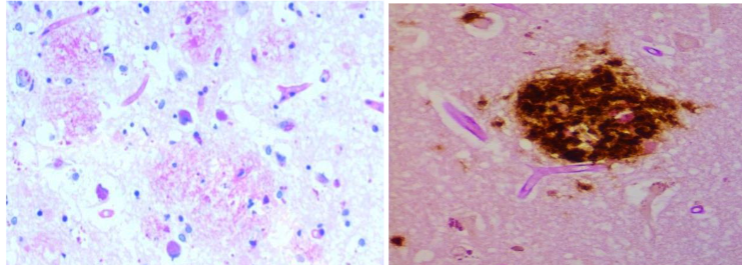
The scope and purpose of this is perspective article is to present the many different precepts present. The most important is the presentation of 3-D vs 2-D images of the documented pathology, immunopathology and microbiology photomicrographs, Bright field microscopy enlarges the images 1000X vs 100,000X in 3D images. Also, the spirochetes are presented as in a darkfield examination. Another precept is the pathology of AD and tertiary syphilis with dementia are similar: the same senile plaques, same tau tangles, spirochetes, etc in each. The background information includes those pathology changes and a discussion of the microbes involved. The methods in the original article included observation and categorization of the changes noted. There are three different spirochetes documented to cause dementia: Treponema pallidum (TP) that causes syphilis and general paresis (GP) or syphilitic dementia; Borrelia burgdorferi (BB) that causes Lyme disease, neuroborreliosis, and Alzheimer’s disease (AD); and oral spirochetes, represented by Treponema denticola (TD), that cause dental plaque and AD. The evidence supporting BB and TD in AD is strong: they have been cultivated from AD brains, BB directly and TD as Spirochaetales. Further, both BB and TD have not only been cultured from AD brains but also have been characterized by immunopathology, fluorescent in situ hybridization (FISH) analysis, atomic force microscopy, and electron microscopy. BB has been further characterized by 16s rRNA and taxonomical analysis by electron microscopy. TP was the first to be noted because it was seen decades ago on silver pathology stains of the brain and was recognized by the number of coils (up to 14) it contained. TD, in its 4 multiple iterations (T. socranskii, T. pectinovorum, T. medium T. amylovorum, T. maltophilum, and T. denticola) was the last to be noted, but it was found on species-specific polymerase chain reaction (PCR). GP forms up to 40% of tertiary neurosyphilis and neurosyphilis forms up to 30% of tertiary syphilis. Thus, syphilitic dementia is seen in a maximum of 12% of patients with syphilis. The same is likely to be true for BB and neuroborreliosis as well. BB has the fewest spirals (as few as three), TD is next with 6-10 and TP has 6-14. Both TP and TD have three characteristic movements: undulation, rotation, and angulation. TP is further distinguishable by sharp angulation as opposed to TD that bends and by TP’s ability to “snap back” from an angulation. The mouth, incidentally, has been a difficult location to determine whether a lesion is a syphilitic chancre because of the close similarity of TP and TD. The three different spirochetes have been fully discussed in the commentary section of this article. The spirochetes in the mouth have the shortest distance to travel: it is but a few centimeters from the mouth to the hippocampus, the first site involved in AD. From there, they can travel to other portions of the brain. BB would have the longest distance to travel, if the tick bite that injected the organism, into the skin was on the lower extremity. The next key occurrence is the formation of biofilms because it is very likely there is no AD without biofilms. Biofilms form when organisms divide and form a quorum which is ten organisms in every direction. To obtain a quorum of spirochetes takes up to 2 years because they divide so slowly. These fits with the clinical appearance of the disease at advanced age. Fischer who studied 12 cases of dementia to Alzheimer’s one, thought the disease was infectious because the senile plaques resembled the sulfur granules of actinomycosis. It is interesting that both the senile plaques and sulfur granules are biofilm structures. One obvious difference is the sulfur granules are surrounded by inflammatory cells of the adaptive immune system while the senile plaques are not (those cells, neutrophils and lymphocytes, are too large to fit through the blood brain barrier). The innate immune system molecule (TLR2) is present, and it creates numerous cytokines such as TLR2/MyD88/TNFa/NF-kB.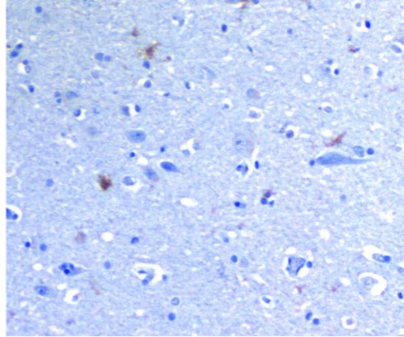
Approval
All work in this paper was done under the approval of the Drexel College of Medicine institutional Review Board and nearly all the text was peer reviewed in J Alz Dis 2021; 84: 61-67.References:
- Miklossy J. Historic evidence to support a causal relationship between spirochetal infections and Alzheimer’s disease. Front Aging neuroscience, 2015;7: 46
- MacDonald AB. Borrelia in the Brains of Patients Dying with Dementia. JAMA. 1986; 256: 2195-2196.
- Riviere GR, Riviere GH, Smith KS. Molecular and immunological evidence of oral treponemes in the human brain and their association with Alzheimer’s disease. Oral Microbiol Immunol. 2002; 17: 113-118.
- Senejani AG, Maghsoudlou J, El-Zohiry D, Gaur G, Wawrzeniak K, Caravaglia C, Khatri VA, MacDonald A, and Sapi E. Borrelia burgdorferi Co-Localizing with Amyloid Markers in Alzheimer’s Disease Brain Tissues. J Alzheimer’s Dis 2022; 85(2); 889-903.
- Miklossy J. Alzheimer’s Disease-A Neurospirochetosis Analysis of the Evidence following Koch’s and Hill’s Criteria. J Neuroinflammation. 2011; 8: 90.
- Miklossy, J., Khalili, K., Gern,L, Ericson R, Darekar, P, Bolle, L., … & Paster, B. J. (2004). Borrelia burgdorferi persists in the brain in chronic lyme neuroborreliosis and may be associated with Alzheimer disease. Journal of Alzheimer’s disease, 6(6), 639-649.
- Clark EG, Danbolt N. The Oslo Study of the natural history of untreated syphilis:An epidemiologic investigation based on a study of Bruusgaard material a reviewed appraisal. J Chronic Diseases 1955;2(3): 311-344.
- Landry T, Smyczek P, Cooper R, Gratix J, Bartholet L, Read R, Romanowski B, and Singh AE. Retrospective review of tertiary and neurosyphilis cases in Alberta, 1973- 2017. BMJOpen.2019; 9(6)e025995 doi: 10.1136/bmjopen-2018-025995.
- Allen HB. Alzheimer’s disease: Assessing the Role of Spirochetes, Biofilms, the Immune System, and Beta Amyloid with regard to potential Treatment and Prevention. J Alz Dis. 2016; 53: 1271-1276.
- Fischer O and Alzheimer A as reported in Miklossy. Historic evidence to support a causal relationship between spirochetal infections and Alzheimer’s disease. Front Aging Neurosci. 2015; 7: 46.
- Allen HB, Allawh R, Touati A, et al. Alzheimers Disease: The Novel Finding of Intracellular Biofilms. J Neuroinfect Dis. 2017; 8: 247.
- Miklossy J. Bacterial Amyloid and DNA are Important Constituents of Senile Plaques:Further Evidence of the Spirochetal and Biofilm Nature of Senile Plaques. J Alz Dis. 2016; 53: 1479-1473.
- Iqbal K, Alonso AC, Chen S, et al. Tau pathology in Alzheimer disease and other tauopathies. Biochim Biophys Acta. 2005; 1739: 198-210.
- Zempel H, Thies E, Mandelkow E, et al. Aβ Oligomers Cause Localized Ca2+ Elevation, Missorting of Endogenous Tau into Dendrites, Tau Phosphorylation, and Destruction of Microtubules and Spines. J Neurosci. 2010; 30: 11938-11950.
- Imahori K and Uchida T. Physiology and pathology of tau protein kinases in relation to Alzheimer’s disease. J Biochem. 1997; 121(2):179-188.
- Wang J-Z, Grundke-Iqbal I. and Iqbal K. Kinases and phosphatases and tau sites involved in Alzheimer neurofibrillary degeneration. Eur J Neurosci. 2007; 25(1): 59-68.
- Allen HB, Vin H, Warner C, Joshi S. Lyme Disease: Beyond Erythema Migrans. 2016; J Clin Exp Dermatol Res 7: 330. doi: 10.4172/2155-9554.1000330
- Ebady R, Niddam AF, Boczula AE, Simmons CA, Skare JT, and Moriarty TJ. Biomechanics of Borrelia bugdorferi vascular interactions. 2016: Cell Reports16: 2593-2064.
- Singhrao SK, Harding A, Poole S, Kesavalu L, CreanS (2015) Porphyromonas gingivalis periodontal infection and its putative links with Alzheimer’s disease. Mediators Inflamm 137
- Itzhaki RF,Wozniak MA (2008) Herpes simplex virus type in Alzheimer’s diseas. The enemy within. J Alzheimers Dis 13, 393–405.
- Balin BJ, Little CS, Hammond, CJ, Appelt DM, Whittum- Hudson JA, G´erard HC, Hudson AP (2008) Chlamydophila pneumoniae and the etiology of late-onset Alzheimer’s disease., J Alzheimers Dis 13, 371–380.
- Tukel C, Wilson RP, Nishimori M, Pezeshki M, Chromy BA, Baumier AG (2009) Responses to amyloids of microbial and host origin are mediated through toll-like receptor 2. Cell Host Microbe 6, 45-53.
- Sapi E, Gupta K, Wawrzeniak K, Gaur G, Torres J, Filush K, Melillo A, Zelger B (2019) Borrelia and Chlamydia can form mixed biofilms in infected human skin tissues. Eur J Microbiol Immunol 9, 46–55.
- Maheshwari P, Eslick GD. Bacterial infection and Alzheimer’s disease: a metaanalysis. J Alzheimers Dis. 2015;43(3):957-66. doi: 10.3233/JAD-140621.
- Allen HB. A Novel Approach to the Treatment and Prevention of Alzheimer’s Disease Based on the Pathology and Microbiology. Journal of Alzheimer’s Dis. 2021; 84: 61–67.
- Allen HB. Alzheimer’s Disease: A Chronic Infection (2020), e book DOI: 10.9734/bpi/mono/978-81-947979-7-5.
- Allen HB, Allen RA, Kannan K, Fransko L. The Presence and Impact of Bacteria and Biofilms in Chronic Skin and Systemic Diseases. (2023) doi.org/10.18103/mra.v11i7.2.4160.
- Shrag in Piller C (2025) “Doctored: The Devastating Legacy of Lies in Alzheimer’s Disease.” doi: .org/11177/13872877251315762, p 93.
Interested in publishing your own research?
ESMED members can publish their research for free in our peer-reviewed journal.
Learn About Membership

