Targeting Stem Cells in Therapy-Resistant TNBC
Stem Cell Targeting Natural Products for Therapy-Resistant Triple Negative Breast Cancer
Nitin T. Telang, Ph.D.1
- Cancer Prevention Research Program, Palindrome Liaisons Consultants, Montvale, NJ 07645-1559, USA
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Telang, NT., 2025. Stem Cell Targeting Natural Products for Therapy-Resistant Triple Negative Breast Cancer. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6584
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6584
ISSN 2375-1924
ABSTRACT
Background: The treatment options for breast cancer are dependent on the expression status of hormones and growth factor receptors. Hormone receptor positive Luminal A and Luminal B breast cancer subtypes exhibit favorable response to targeted endocrine therapy and/or human epidermal receptor-2 (HER-2) HER-2 therapy respectively. Hormone receptor negative, HER positive HER-2-enriched subtypes respond only to HER-2 targeted therapy. Hormone receptor negative and HER-2 negative triple negative (TNBC) subtypes respond only to conventional cytotoxic chemotherapy, and are notable for the presence of putative stem cell population. These subtype selective treatment options are universally associated with phenotypic therapy resistance, leading to emergence of chemo-resistant cancer initiating stem cell population that is responsible for initiation, progression and recurrence of therapy-resistant breast cancer. These limitations emphasize development of reliable drug-resistant cancer stem cell model for TNBC and identification of novel testable alternatives with stem cell selective efficacy.
Objectives: The present review article provides an overview of published evidence for i) Molecular characterization of TNBC subtype, ii) Limitations relevant to mainstream treatment options and advantages of natural products as testable therapeutic alternatives, and iii) Development and characterization of reliable drug-resistant TNBC stem cell model.
Conclusion: Published evidence discusses relevant information regarding molecular classification of TNBC subtype, significance of natural products as testable alternatives for TNBC subtype and applicability of drug-resistant stem cell model to investigate stem cell targeting efficacy of natural products.
Future Research: Collectively, discussed evidence defines a scientifically robust rationale to undertake future investigations on patient derived tumor explants and patient derived tumor organoids as a valuable strategy to reduce extrapolation of preclinical data for their clinical relevance and translatability for cancer stem cell targeting efficacy of natural products on breast cancer.
Keywords
Triple negative breast cancer, Therapy resistant stem cell, Natural products.
Introduction
The American Cancer Society has projected 316,950 newly diagnosed breast cancers and 42,170 cancer related deaths in 2026. Progression of advanced stage metastatic breast cancer represent a major cause of cancer related deaths. Selection of treatment options for breast cancer is dictated by the expression of hormone and growth factor receptors. Hormone receptor and growth factor receptor positive breast cancer subtypes exhibit favorable response to endocrine and growth factor targeted therapy. In contrast, breast cancer subtype that lacks hormone/growth factor receptor expression are classified as triple negative breast cancer (TNBC), and responds only to conventional cytotoxic chemotherapy.
Human breast carcinoma-derived cellular models, characterized by quantifiable mechanistic biomarkers, provide valuable experimental approaches to identify efficacious natural products as testable alternatives for conventional chemotherapy, endocrine therapy and pathway selective molecularly targeted therapy. Data generated from these approaches facilitate identification of mechanistic leads for cancer growth inhibitory efficacy of natural products and/or constitutive bioactive agents. Furthermore, experimental modulation of quantifiable mechanistic biomarkers identifies potential molecular targets.
The objective of present review is to provide a systematic discussion of published evidence relevant to unique aspects of TNBC. The discussion includes molecularly based classification, chemotherapy related limitations for treatment, advantages of natural products as testable alternatives, and applicability of drug-resistant Cancer Stem Cell Model for TNBC.
In view of therapy resistance commonly observed in TNBC investigations using the drug-resistant stem cell model provide an experimental approach to examine stem cell targeted inhibitory efficacy of natural products. Promising naturally-occurring bioactive agents may also represent potential new drug candidates for therapy resistant TNBC.
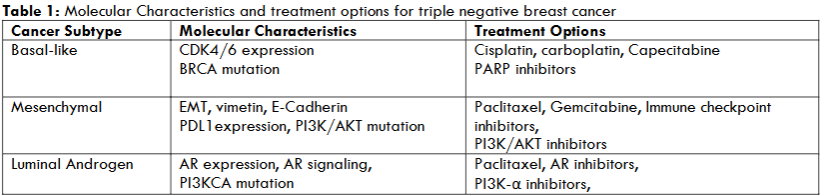
The TNBC subtypes are notable for cellular heterogeneity, cellular plasticity, high risk of therapy-resistant metastatic progression, and emergence of chemo-resistant cancer initiating stem cell population. Comparative lack of therapeutic options for TNBC emphasize molecular classification and identification of relevant molecularly targeted therapeutic strategies. Recent evidence has classified the TNBC in to basal-like, mesenchymal and Luminal androgen receptor positive subtypes. This classification is based on their molecular characteristics, and has documented subtype specific chemo-therapeutic options. It is notable that the expression of cyclin-dependent kinase, cyclin E and BRCA tumor suppressor gene mutation, epithelial-mesenchymal transition (EMT), expression of vimentin and E-cadherin, programmed death ligand1 (PDL1), mutation in PI3K/AKT, androgen receptor expression, receptor signaling and mutation in PI3K gene PI3KCA has been correlated in a subtype specific manner. Mainstream treatment options for individual subtypes include the use of cis-platin, carboplatin, capecitabine, gemcitabine and paclitaxel. These aspects are summarized in Table 1.
Cellular models for TNBC
Among the TNBC subtypes the mesenchymal subtype M-TNBC is notable for the presence of putative cancer stem cell population. Several human breast carcinoma derived cell lines such as MDA-MB-157, BT549, SUM 150PT and MDA-MB-231 have been extensively used as experimental models for clinical TNBC. The MDA-MB-231 model exhibits expression of mutant TP53, resulting in compromised tumor suppressive function of TP53. Overexpression and active EGFR signaling involves cyclin D1, cyclin E and MAPK pathways. Furthermore, EGFR signaling influences RAS, PI3K AKT and m TOR signaling pathways that confer growth advantage to parental TNBC as well as to chemo-resistant cancer stem cells. In this TNBC model active EGFR signaling represents a potential therapeutic target for small molecule tyrosine kinase inhibitors.
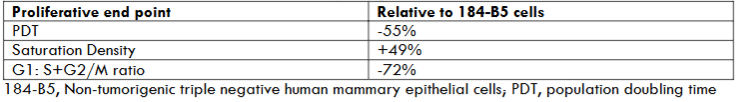
The MDA-MB-231 model has been stringently characterized for its growth pattern. Relative to the non-tumorigenic triple negative human breast epithelial 184-B5 cells, the carcinoma derived TNBC MDA-MB-231 cells exhibit substantial increase in cellular proliferation as evidenced by decreased population doubling time, increased saturation density and decreased quiescent: proliferative ratio in the cell cycle, collectively suggesting hyper-proliferation and accelerated cell cycle progression. These data are summarized in Table 2.
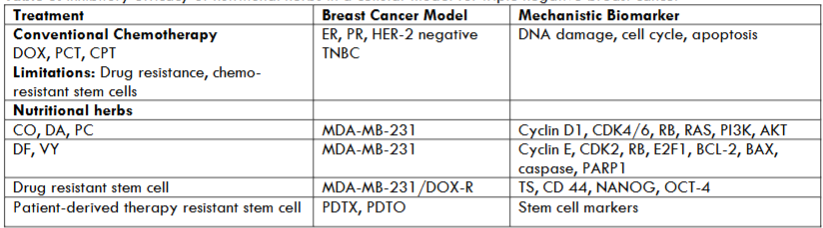
In contrast to chemo-endocrine therapy for breast cancer, natural products such as dietary phytochemicals and herbal formulations used in traditional Chinese medicine have documented low systemic toxicity, lack of phenotypic resistance preclinical efficacy, safe human consumption and stem cell efficacy. Preclinical efficacy of natural products for growth inhibition in cellular models for Luminal A, HER-2-enriched and TNBC breast cancer subtypes has been documented. These advantages emphasize the significance of natural products as alternatives for cancer stem cell targeting treatment of therapy resistant breast cancer. The data for front-line chemotherapy for TNBC, efficacy of nutritional herbs, drug-resistant stem cell models and experimental systems for future research are summarized in Table 3.
Cancer Stem Cell Model
The maintenance of normal stem cells is accomplished by Wnt/β-catenin, hedgehog and Notch signaling pathways that are responsible for preserving cellular homeostatic growth control essential for physiological function and regeneration of organs. In cancer stem cells these regulatory pathways are dysregulated and favor aberrant cell proliferation essential for tumor progression, metastasis and phenotypic resistance to cancer treatment. Additionally, drug-resistant cancer stem cells exhibit activation of cell survival pathways such as RAS/RAF. MEK/ERK, PI3K and AKT signaling that are responsible for growth advantage of stem cell population. TNBC cell lines MDA-MB-468, HCC38 and HCC 1806, in response to treatment with cytostatic concentrations of chemotherapeutics doxorubicin (DOX), paclitaxel (PCT), 5-fluoro-uracil (5-FU) and gemcitabine (GEM), favors the emergence of drug-resistant phenotypes. These resistant phenotypes represent potential stem cells population that facilitate identification of susceptible therapeutic targets and mechanistic biomarkers.
Putative drug-resistant stem cells have also been isolated using positive selection based on phenotypic resistance to selective estrogen receptor modulator tamoxifen (TAM), to growth factor receptor inhibitor lapatinib (LAP) and to chemotherapeutic DOX, respectively. The TAM-R, LAP-R and DOX-R stem cell models exhibit upregulated expression of stem cell specific phenotypic and molecular biomarkers such as tumor spheroid formation, cluster of differentiation 44 (CD44), DNA-binding nuclear transcription factors (NANOG) and octamer-binding protein-4 (OCT-4). These models provide reliable experimental systems for Luminal A, HER-2-enriched and TNBC subtypes, respectively.
Specific nuclear transcription factors such as OCT-4, Kruppel-like factor-4 (Klf-4), sex determining region Y-box-2 (SOX-2) and cellular Myc (c-MYC) are essential for reprogramming and maintenance of induced pluripotent cells derived from adult human somatic cells. These transcription factors are also upregulated in the cancer stem cell population and represent stem specific molecular markers.
Maintenance of MDA-MB-231 breast carcinoma-derived cell line under long-term selective pressure of the chemotherapeutic DOX promotes the growth of DOX-R phenotype. The putative DOX-R stem cells are characterized by monitoring the expression status of stem cell specific tumor spheroid (TS) formation and expression cell surface marker CD44, nuclear transcription factors NANOG and OCT-4. The expression of cell surface protein molecule CD44 and of the transcription factors is measured using a quantitative immuno-fluorescent assay that monitors cellular uptake of fluorescently labeled antibodies. The data are expressed as relative fluorescent units (RLU).
In comparison with the sensitive DOX-S phenotype the DOX-R phenotype exhibits a substantial upregulation of the stem cell markers. The evidence for upregulation of cellular and molecular stem cell specific markers are summarized in Table 4.
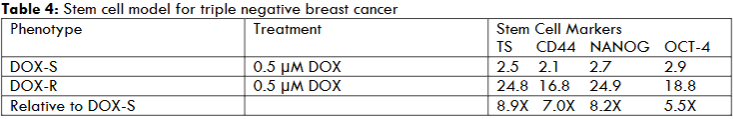
Preclinical Efficacy of Natural Products
Published evidence for therapeutic options, growth inhibitory efficacy of natural products, and drug resistant breast cancer stem cell models defines valuable experimental strategies for screening and prioritizing efficacious dietary phytochemicals and bioactive agents such as anthocyanins, prenylflavones, triterpenes, saponins and lignans present in nutritional herbal formulations used in traditional Chinese medicine. Naturally-occurring bioactive agents present in dietary phytochemicals and nutritional herbs are identified as potential drugs. Prior to evaluate their anti-cancer activity in the clinical setting these bioactive agents are subjected to preclinical in vitro assays for cell proliferation, cellular apoptosis and omics based assays to identify susceptible mechanistic pathways and potential molecular targets. Promising bioactive agents are subsequently subjected to in vivo assays for pharmacokinetics, pharmacodynamics absorption and, distribution, metabolism, excretion (ADME). Promising bioactive agents are evaluated for clinical safety and efficacy.
Stem cell targeting natural products
Dietary phytochemicals containing polyphenols, flavones and terpenes have documented cancer stem cell targeting efficacy. Nutritional herbs from traditional Chinese medicine have documented growth inhibitory efficacy in a cellular model for TNBC. Bioactive agents such as polyphenols, flavones, and terpenes present in dietary phytochemicals and androgopholide, baicalin, berberine present in herbal formulations used in traditional Chinese medicine have documented cancer stem cell specific efficacy for signaling pathways in breast cancer stem cell models. These regulatory pathways include Wnt/β-catenin, Notch, Hedgehog and TGF-β. Naturally-occurring bioactive agents also affect cancer stem cell survival pathways such as PI3K, AKT and m TOR that are involved in chemo-resistance.
In the lapatinib resistant stem cell model for HER-2-enriched subtype natural products such as vitamin A derivative all-trans retinoic acid and naturally-occurring terpene cornosol inhibit the expression of stem cell specific markers such as tumor spheroids, CD44, NANOG and OCT-4. These data provide mechanistic leads for stem cell targeting inhibitory efficacy for natural products, and thereby, validate an experimental approach for stem cell targeting efficacy of nutritional herbs and constitutive bioactive agents for TNBC stem cell population.
Conclusion
This mini-review discusses published evidence for preclinical efficacy of natural products on TNBC breast cancer subtype. Furthermore, systematic discussion regarding development of reliable drug-resistant stem cell model and mechanistic leads for stem cell targeted efficacy of dietary phytochemicals provides a scientifically robust rationale for future research.
Future research
Investigations focused to establish relevant stem cell models from patient derived tumor xeno-transplants and patient derived tumor organoid using clinical tumor samples from therapy-resistant breast cancer subtypes provide valuable experimental stem cell models. These models are likely to reduce data extrapolation for clinical relevance and translatability of stem cell targeted efficacy of novel therapeutic alternatives such as naturally occurring dietary phytochemicals and bioactive agents present in nutritional herbs.
Conflicts of interest statement: The author has no conflicts of interest to declare.
Funding Statement: The current research has not received extra-mural funding.
Acknowledgements: The author has conceived the theme, prepared the manuscript and has approved the final version of the submitted manuscript. The research program “Cellular models for molecular subtypes of clinical breast cancer: Molecular approaches for lead compound efficacy” has been supported in the past by extra-mural funding from the US National Cancer Institute FIRST Award CA 44741 and by the US Department of Defense Breast Cancer Research Program IDEA award DAMD-17-9-J-4208.
References:
- American Cancer Society Fact and Figures 2025, American Cancer Society, Atlanta GA, USA. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-and-figures/2025/2025-cancer-facts-and-figures.pdf. Accessed on February 25, 2025.
- Gardishar WJ, Moran MS, Abrahan J: NCCN Clinical Practice Guidelines in Oncology: Breast Cancer Version 4, National Comprehensive Network: Plymouth Meeting, PA, USA, 2022. Available online: https://ww.nccn.org Accessed on 25, February 2025.
- Lehmann BD, Colaprico A, Silva TC, et al: Multi-omics analysis identifies therapeutic vulnerabilities in triple negative breast cancer subtypes Nat. Commun. 2021, 12: 6276.
- Neve RM, Chin K, Fridlyand J, et al: A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 2006, 10: 515-527. Doi: 10.1016/j.ccr2006.10.008.
- Shapira L, Lee A, Vora R, Budman DR: P53 mutations in triple negative breast cancer upregulate endosomal recycling of epidermal growth factor receptor (EGFR) increasing its oncogenic potency. Crit. Rev. Oncology/Hematology. 2013, 88: 284-292. Doi: 10.1016/j.critrevonc.2013.05.003.
- Costa R, Shah AN, Santa-Maria CA, et al: Targeting epidermal growth factor receptor in triple negative breast cancer: New discoveries and practical insights for drug development. Cancer Treatment Review. 2017, 53: 111-119. Doi: 10.1016//j.ctrv.2016.12.010.
- Ye L, Jia Y, Ji KE, et al: Traditional Chinese medicine in prevention and treatment of breast cancer and metastasis. Oncol. Letts. 2015, 10: 1240-1250. Doi: 10.3892/ol.2015.3459.
- Hong M, Tan HY, Li S, et al: Cancer stem cells: The potential targets of Chinese medicines and their active compounds. Int. J. Mol. Sci. 2016, 17: 993.
- Naujokat C, Mc Kee DL: The big five phytochemicals targeting the cancer stem cells: Curcumin, EGCG, Sulforaphane, resveratrol, and genistein. Cur. Med. Chem. 2021, 28: 4321-4342.2021. Doi: 10.2174/0929867327666200228110738.
- Yang Z, Zhang Q, Yu L, et al: The signaling pathways and targets of traditional Chinese medicine and natural medicine in triple negative breast cancer. J. Ethno-pharmacol. 2021, 264: 113249. Doi: 10.1016/jep.2020.113249.
- Zhu M, Liu Y, Wen Z, et al: Exploration of Chinese medicine comprehensive treatment of triple negative breast cancer based on molecular pathology mechanisms. Breast Cancer Targets Ther. 2025, 17: 289-304. Doi: 10.2147/BCTT.S511059.
- Telang N: Natural phytochemicals as testable therapeutic alternatives for HER-2-enriched breast cancer. World Acad. Sci. J. 2020, 2: 19. Doi: 10.3892/wasj.2020.60
- Telang NT, Nair HB, Wong GYC: Growth inhibitory efficacy of Chinese herbs in a cellular model for triple negative breast cancer. Pharmaceuticals (Basel) 2021, 14: 1318. Doi: 10.3390/ph14121318.
- Telang N: Drug-resistant stem cell model for hormone responsive Luminal A breast cancer. Med. Res. Arch. 2023, 11: 3556. Doi: https://doi.org/10.18103/mrav11i2.3556.
- Lytle NK, Barber NG, Reya T: Stem cell fate in cancer growth, progression and therapy resistance. Nat. Rev. Cancer 2018, 18: 669-680. Doi: 10.1038/s41568-018-0056-x.
- Nunes T, Hamdan D, El Boutachtaoui MD, et al: Targeting cancer stem cells to overcome chemo-resistance. Int. J. Mol. Sci. 2018, 19: 4036.
- Yager R, Solit DB: overcoming adaptive resistance to RAS inhibitors through vertical pathway targeting. Clin. Cancer Res. 2020, 26: 1538-1540. Doi: 10.1158/1078-0432.CCR-19-4060.
- Takahashi K, Tanabe K, Ohnuki M, et al: Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131: 861-872. Doi: 10.1016/j.cell.2007.11.019.
- Park IH, Zhao R, West JA, et al: Reprogramming of human somatic cells to pluripotency with defined factors. Nature 2008, 451: 141-146. Doi: 10.1038/nature06534.
- Telang N, Li G, Katdare M, et al: Inhibitory effects of Chinese nutritional herbs in isogenic breast carcinoma cells with modulated estrogen receptor function. Oncol. Letts. 2016, 12: 3949-3957. Doi: 10.3892/ol.2016.5197.
- Telang N: Natural bioactive agents: Testable stem cell-targeting alternatives for therapy resistant breast cancer. Int. J. mol. Sci. 2025, 26: 2529. Doi: https://doi.org/10.3390/ijms26062529.
- Berman TA, Ben-Ayre E, Kienle GS, et al: Integrating botanicals in to oncology care: Consideration of FDA regulation of botanical products and botanical clinical trials. Clin. Cancer Res. 2025, 31: 1556-1572. Doi: 10.1158/1078-0432.CCR-24-3419.
- Jan A, Sofi S, Jan N, Mir MA: An update on cancer stem cell survival pathways involved in chemo-resistance in triple negative breast cancer. Future Oncol. 2025, 21: 715-735. Doi: 10.1080/14796694.2025.2461443.
- Manogaran P, Umapathy D, Karthikeyan M, et al: Dietary phytochemicals as a potential source for targeting cancer stem cells. Cancer Investig. 2021, 39: 349-368. Doi: 10.1080/07357907.2021.1894569.
- Mengitsu BA, Tsegaw T, Demessie Y, et al: Comparative review of drug-resistance in mammalian cancer stem cells: Implications for cancer therapy. Cancer Cell Int. 2024, 24: 406. Doi: 10.1186/s12935-024-03558-0.
- Elbaiomy MA, Tamer A, Atwan N, et al: Clinical impact of breast cancer stem cells in metastatic breast cancer patients. J. Oncol. 2020, 2561726. Doi: 10.1155/2020/2561726.eCollection2020.
- Telang N: Stem cell targeted therapeutic approaches for molecular subtypes of clinical breast cancer. World Acad. Sci. J 2019, 1: 20-24. Doi: 10.3892/wasj.2018.3.
- Blanchard Z, Brown EA, Ghazaryan A, et al: PDX models for functional precision oncology and discovery science. Nat. Rev. Cancer 2025, 25: 153-166. Doi: 10.1038/s41568-024-00779-3.
- Meng S, Cao Y, Lu LO, et al: Quercetin promotes the chemo-sensitivity in organoids derived from patients with breast cancer. Breast cancer Targets Ther. 2024, 16: 993-1004. Doi: 10.2147/BCTT.S494901.

