Metadichol Enhances CD14 Expression in Stem Cells
Metadichol induces CD14 glycoprotein expression in human embryonic stem cells and fibroblasts
P.R. Raghavan1
- Nanorx Inc., PO Box 131, Chappaqua, NY 10514, USA
[email protected]
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION: Raghavan, P.R., 2025. Metadichol induces CD14 glycoprotein expression in human embryonic stem cells and fibroblasts. Medical Research Archives, [online] 13(5). https://doi.org/10.18103/mra.v1
3.5.6490
COPYRIGHT: © 2025 European Society of Medicine. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13.5.6490
ISSN 2375-1924
ABSTRACT
Cluster of differentiation 14 (CD14) is a glycoprotein essential to the immune system that is found primarily on monocytes, macrophages, and other immune cells. Despite its importance, there are no examples in the literature of small compounds that can substantially alter CD14 expression in human embryonic stem cells (hESCs) or fibroblasts. This study addresses this gap by exploring the potential of metadichol, a nanoemulsion of long-chain fatty alcohols, to induce CD14 expression in hESCs. Metadichol has been previously shown in our studies to express CD33 and CD34 as well as all the nuclear receptors and the family of sirtuins and Toll receptors. Using quantitative real-time PCR (qRT–PCR) and Western blotting, we showed that metadichol significantly upregulated CD14 expression in hESCs (by seventeen-fold) but downregulated it in fibroblasts. This novel finding indicates that metadichol can modulate CD14 expression in a cell type-specific manner, highlighting its potential to enhance stem cell-based therapeutics and advance our understanding of stem cell biology. The implications of these findings are substantial, suggesting new directions for research into the immunomodulatory functions of hESCs and their potential applications in regenerative medicine. Our work highlights the potential of metadichol as a powerful tool for modulating CD14 expression in stem and somatic cells, marking a significant step forward in the field of stem cell research and therapy development.
Keywords: CD14, hESC, fibroblasts, metadichol, VDR, SP1, PPAR gamma, retinoid X receptors.
Introduction
Cluster of differentiation (CD14) is a glycoprotein, and a coreceptor for toll-like receptors (TLRs), particularly TLR4, is essential in the innate immune response because it recognizes lipopolysaccharides (LPSs) from bacterial cell walls.¹ This interaction significantly enhances the immune response to bacterial infections, making CD14 a key component in pathogen recognition and immune activation. However, the expression and functional implications of CD14 in human embryonic stem cells (hESCs) remain underexplored, presenting a unique opportunity for novel therapeutic interventions.
Inducing the expression of CD14 in human embryonic stem cells (hESCs) and fibroblasts is challenging due to several factors related to the nature and regulation of CD14 expression. In terms of tissue distribution, CD14 is predominantly expressed in monocytes and macrophages, but it is also present in dendritic cells and, to a lesser extent, in neutrophils.² The expression of CD14 is tightly regulated by specific transcription factors and signaling molecules that are present in myeloid cells but may be absent or inactive in hESCs and fibroblasts. For example, stimuli such as LPS, dimethyl sulfoxide (DMSO), and 1,25-dihydroxyvitamin D3 can induce CD14 expression in promonocytic cell lines such as U937 and HL-60, but these factors may not have the same effect on hESCs or fibroblasts because of differences in receptor expression and intracellular signaling pathways.³
In some nonmyeloid cells, CD14 may be present in a soluble form (sCD14) that can participate in signaling pathways indirectly by interacting with other receptors, such as TLR4.⁴ However, the mechanisms and conditions under which sCD14 influences CD14 expression or function in hESCs and fibroblasts are poorly understood.
Overall, the difficulty in inducing CD14 expression in hESCs and fibroblasts is largely due to the specialized role of CD14 in immune cells and the lack of necessary transcriptional and signaling components in these nonmyeloid cell types. Metadichol, a nanoemulsion of long-chain alcohols, has shown promising effects on stem cells.⁵ Additionally, the antioxidant and anti-inflammatory properties of metadichol further support its beneficial effects on stem cells and their microenvironment, as well as their regenerative potential.⁶˒⁷
Although the literature does not provide specific evidence of small molecules that strongly increase CD14 expression in stem cells, previous work offers some insights into the expression and regulation of CD14 in different contexts.
Cluster of Differentiation (CD14) is not a marker typically associated with most stem cells, such as mesenchymal stem cells (MSCs), which are generally characterized by low or absent CD14 expression.⁸ However, CD14 expression has been observed in certain progenitor or stem-like cells, such as porcine spermatogonial stem cells (SSCs), where it is associated with stemness genes such as POU Class 5 Homeobox 1 (POU5F1), also known as Oct4, and Nanog homeobox (NANOG).⁹
Small molecules are used to influence stem cell characteristics and differentiation potential. For example, certain small molecules can affect gene expression patterns and enhance differentiation efficiency in amniotic fluid stem cells (AFSCs), although the studies that demonstrated this did not focus specifically on CD14 expression.¹⁰
In myeloid cells, small molecules that can either increase or decrease CD14 expression during differentiation processes have been identified. For example, one study showed that certain inhibitors can prevent CD14 upregulation in monocytes during macrophage differentiation.¹¹ However, this research focused more on immune cells than stem cells.
While small molecules can modulate gene expression and differentiation in stem cells, there is no direct evidence in the literature indicating that small molecules specifically increase CD14 expression in multiple types of stem cells. In this study, via qRT–PCR
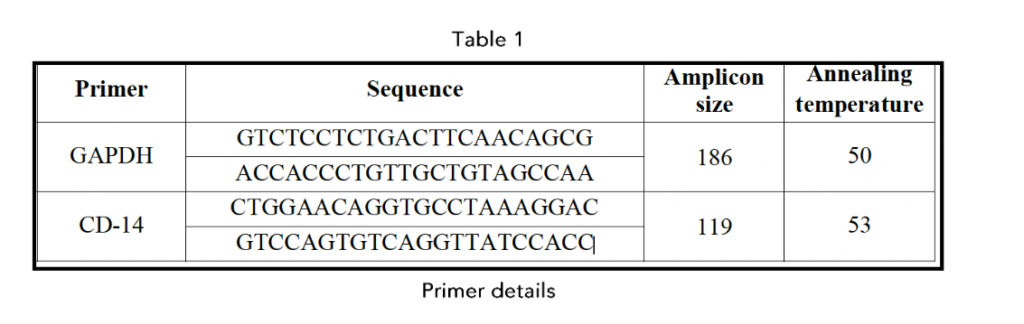
PCR and Western blotting, we demonstrated that in a recent preprint⁷, metadichol treatment significantly increased CD14 expression in hESCs. These findings suggest that metadichol modulates signaling pathways that regulate CD14 expression, potentially enhancing the immune modulatory functions and differentiation potential of hESCs.
Experimental
All work was outsourced commercially to Skanda Life Sciences Pvt. Ltd. (Bangalore, India). The primers used were obtained from EuroFin (Bangalore, India), and the antibodies used were from Elabscience® (Houston, Texas, USA). H-ESC BG01V and NHDF cells were obtained from ATCC®.
Western blotting
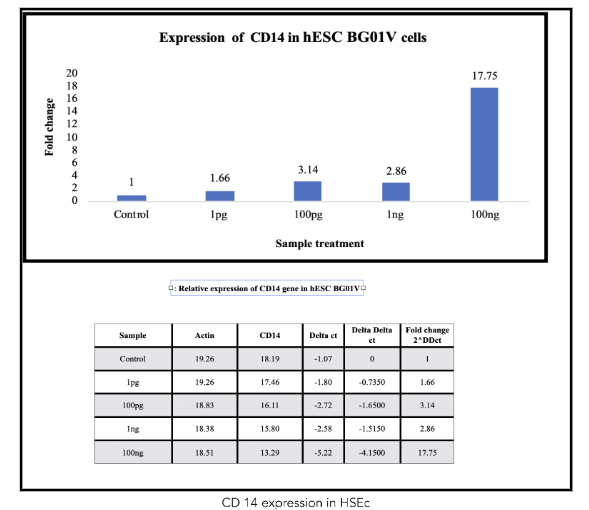
Protein was extracted using RIPA buffer and quantified using the BCA assay. Western blot analysis was performed using antibodies against GAPDH and CD14.
CLUSTER OF DIFFERENTIATION EXPRESSION (CD14) IN NORMAL HUMAN DERMAL FIBROBLASTS (NHDFs)
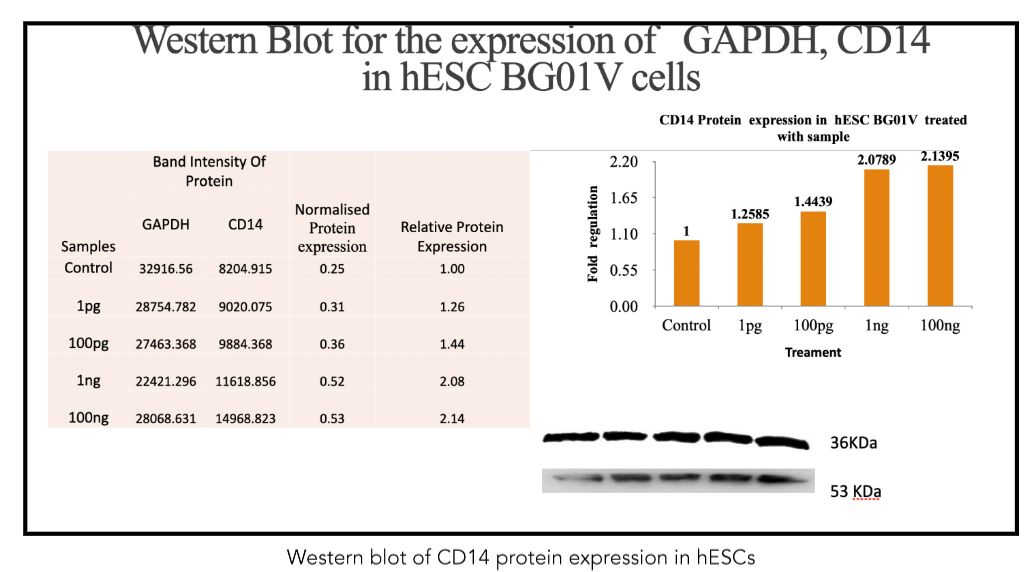
In stark contrast to the findings in hESCs, metadichol treatment resulted in a significant downregulation of CD14 expression in NHDFs. qRT-PCR analysis revealed a dose-dependent reduction in CD14 mRNA levels (Figure 3). At the highest tested concentration (100 ng/mL), CD14 mRNA expression was reduced to 10% of the control level (p < 0.01), effectively indicating near-complete suppression. Lower concentrations also suppressed CD14 expression, with 1 pg/mL, 100 pg/mL, and 1 ng/mL reducing mRNA levels to approximately 80%, 50%, and 20% of the control, respectively (p < 0.05 for all concentrations). These results indicate that metadichol exerts an inhibitory effect on CD14 transcription in fibroblasts, in direct opposition to its stimulatory effect in hESCs.
Discussion
While CD14 expression has been studied in various cell types,⁸ including mesenchymal stromal cells (MSCs) there is no information on its expression in hESCs. Previous studies have shown that CD14 expression can be induced in different cell types under various conditions.⁹
The effects of metadichol on CD14 expression in different cell types are multifaceted and can be understood in the context of its potential applications in regenerative medicine, cancer therapy, and cellular reprogramming.¹⁰˒¹¹˒¹²
The ability of metadichol to increase CD14 expression seventeen-fold in hESCs suggests a significant impact on the immunomodulatory functions of these cells. CD14 is a co-receptor for the detection of bacterial lipopolysaccharides and plays a role in the innate immune response. The increased expression of these
genes could increase the ability of stem cells to modulate immune responses, potentially improving their therapeutic efficacy in regenerative medicine and immune-related disorders.¹³
The complete downregulation of CD14 in fibroblasts by metadichol could imply a reduction in the inflammatory response typically mediated by these cells. Fibroblasts are involved in wound healing and tissue repair, and their role in inflammation is crucial. By downregulating CD14 in fibroblasts, metadichol might reduce chronic inflammation and fibrosis, which could be beneficial in treating fibrotic diseases and improving tissue.¹⁴˒¹⁵
Nuclear hormone receptors (NHRs) play a significant role in modulating CD14 expression, primarily through their ability to regulate immune and inflammatory responses. Several NHRs are involved in this regulatory process.
Peroxisome proliferator-activated receptors (PPARs), particularly PPAR gamma, are involved in modulating inflammatory responses and have been shown to influence the expression of CD14. PPARγ is associated with alternative (M2) macrophage polarization, which is linked to anti-inflammatory responses.¹⁶
Vitamin D receptor (VDR) is known to play a role in immune regulation. The activation of VDRs can influence the expression of CD14, as vitamin D is involved in modulating the immune response and inflammation.¹⁷
The literature indicates that the link between the vitamin D receptor (VDR) and CD14 involves at least one primary pathway, which is mediated through the interaction of the VDR with PI3-kinase signaling. The PI 3-kinase pathway is crucial for the D3-induced expression of CD14, even though the CD14 promoter does not contain a canonical vitamin D response element (VDRE).¹⁸
While direct binding of the VDR to the CD14 promoter is not evident, the VDR may influence CD14 expression through other signaling molecules or transcription factors that interact with the CD14 promoter.¹⁹˒²⁰
The expression of CD14 is tightly regulated at the transcriptional level, and specificity protein 1 (SP1) transcription factor is a critical transcription factor involved in this regulation. Specificity protein 1 (Sp1) binds to specific regions in the CD14 promoter, and its binding is essential for the tissue-specific expression of CD14 in monocytic cells.²¹˒²²˒²³ Specificity protein 1 (SP1) can also influence the expression of the VDR gene itself. The promoter region of the VDR gene contains Sp1 binding sites, which are essential for the transcriptional activation of the VDR gene. These findings suggest that Sp1 not only partners with the VDR to regulate other genes but also plays a role in modulating VDR expression levels. Metadichol is an inverse agonist, more likely a protean agonist, of the VDR.²⁴˒²⁵
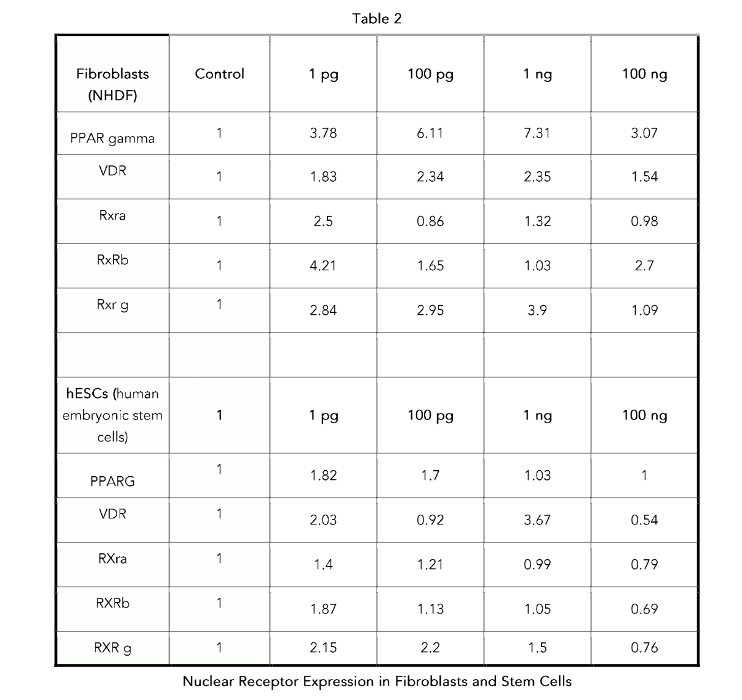
Vitamin D receptor (VDR) is known to regulate CD14 expression in immune cells, such as macrophages, where it is a target of vitamin D signaling. In these cells, vitamin D can upregulate CD14 expression, which is important for immune responses.²⁶ Retinoid X receptors (RXRs) form heterodimers with other NHRs, such as PPARs (peroxisome proliferator–activated receptors) and LXRs (liver X receptors), increasing their ability to regulate gene expression, including that of CD14.²⁷ These nuclear receptors interact with various signaling pathways to regulate immune responses, and their activation by specific ligands may lead to changes in CD14 expression. This modulation is crucial for maintaining immune homeostasis. Metadichol enhances the expression of nuclear receptors involved in CD14 expression. The results of our previous study²⁸ are shown in Table 2. Cluster of Differentiation (CD14) is likely expressed through the activation of multiple NHRs and the interaction of the VDR and SP1, which play a role in CD14 regulation.
Altering CD14 expression might increase the susceptibility of cancer cells to immune surveillance or therapeutic interventions, suggesting a novel approach for cancer treatment.²⁹ This is further aided and compounded by the fact that Metadichol is non-toxic.³⁰˒³¹˒³²
Conclusions
The findings from studies on the promotion of CD14 glycoprotein expression by Metadichol in human embryonic stem cells (hESCs) and fibroblast cells (NHDF) have several significant implications.
Advancements in regenerative medicine
The ability of metadichol to upregulate CD14 expression in hESCs suggests potential applications in regenerative medicine. By enhancing the immune-modulatory functions of stem cells, metadichol could improve the therapeutic efficacy of stem cell-based therapies, potentially leading to more effective treatments for various diseases and injuries.
Potential in Cancer Therapy
The modulation of CD14 expression in cancer cells by Metadichol could have implications for cancer therapy. CD14 is associated with inflammatory and proliferative tumor microenvironments. Altering CD14 expression might increase the susceptibility of cancer cells to immune surveillance or therapeutic
References
1. Funda DP, Tucková L, Farré MA, Iwase T, Moro I, Tlaskalová-Hogenová H. Human intestinal epithelial cells in vitro express and release soluble CD14: a reevaluation of LPS activation of epithelial cells. Infect Immun. 2001;69(6):3772-3781. doi:10.112 8/iai.69.6.3772-3781.2001
2. Zamani F, Zare Shahneh F, Aghebati-Maleki L, Baradaran B. Induction of CD14 expression and differentiation into monocytes or mature macrophages in promyelocytic cell lines: a novel strategy. Adv Pharm Bull. 2013;3(2):329-332. doi:10.5681/apb.2 013.053
3. Liu HZ, Gong JP, Wu CX, Peng Y, Li XH, You HB. The U937 cell line was induced to express CD14 protein by 1,25-dihydroxyvitamin D3 and became sensitive to endotoxin stimulation. Hepatobiliary Pancreat Dis Int. 2005;4(1):84-89.
4. Ichise Y, Saegusa J, Tanaka-Natsui S, et al. Soluble CD14 stimulates pro-inflammatory cytokine production in rheumatoid arthritis fibroblast-like synovial cells through toll-like receptor 4. Cells. 2020;9(7):1689. doi:10.3390/cells9071689
5. Raghavan PR, inventor; Nanorx Inc, assignee. Policosanol nanoparticles. US patents 8,722,093. May 13, 2014; 9,034,383. May 19, 2015; 9,006,292. April 14, 2015.
6. Raghavan PR. Metadichol serves as a natural ligand for the expression of Yamanaka reprogramming factors in human cardiac, fibroblast, and cancer cell lines. Med Res Arch. 2024;12(6). doi:10.18103/mra.v 12i6.5323
7. Raghavan PR. Metadichol stimulates CD14 glycoprotein expression in human embryonic stem cells and fibroblasts. Qeios. Published online 2024. Accessed April 17, 2025.
https://www.qeios.com/read/X9VA1H
8. Hirakawa MP, Tjahjono N, Light YK, et al. The upregulation of CD14 in mesenchymal stromal cells expedites the lipopolysaccharide-induced response and amplifies antibacterial capabilities. iScience. 2022;25(2):103759. doi:10.1016/j.isci.20 22.103759
9. Park HJ, Lee WY, Park C, Hong K, Song H. CD14 serves as a distinctive membrane marker for swine spermatogonial stem cells, governing their development. Sci Rep. 2019;9(1):9980. doi:10.103 8/s41598-019-46000-6
10. Zentelytė A, Žukauskaitė D, Jacerytė I, Borutinskaitė VV, Navakauskienė R. Small chemical therapies enhance the differentiation capacity of human amniotic fluid stem cells. Front Bioeng Biotechnol. 2021;9:623886. doi:10.3389/fbioe.202 1.623886
11. Na K, Oh BC, Jung Y. The complex function of CD14 in innate immunity and tissue equilibrium. Cytokine Growth Factor Rev. 2023;74:100-107. doi:10.1016/j.cytogfr.2023.08.008
12. Kapellos TS, Bonaguro L, Gemünd I, et al. Human monocyte subsets and phenotypes in major chronic inflammatory diseases. Front Immunol. 2019;10:2035. doi:10.3389/fimmu.2019.02035
13. van den Ancker W, Wijnands PG, Ruben JM, et al. Protocols for the proliferation of CD14(+) progenitors derived from acute myeloid leukemia cells to enhance dendritic cell-mediated immunotherapy. Immunotherapy. 2013;5(11):1183-1190. doi:10.2217/imt.13.125
14. Wu Z, Zhang Z, Lei Z, Lei P. CD14: its biological functions and involvement in disease etiology. Cytokine Growth Factor Rev. 2019;48:24-31. doi:10.1016/j.cytogfr.2019.06.003
15. Tsukamoto H, Takeuchi S, Kubota K, et al. Lipopolysaccharide (LPS)-binding protein enhances CD14-mediated internalization of toll-like receptor 4 and activates the LPS-induced TBK1-IKKϵ-IRF3 signaling pathway. J Biol Chem. 2018;293(26): 10186-10201. doi:10.1074/jbc.M117.796631
16. Di Gioia M, Zanoni I. CD14: not merely a chaperone, but a pivotal participant in inflammation. In: Rossetti C, Peri F, eds. The Role of Toll-Like Receptor 4 in Infectious and Non-Infectious Inflammation. Springer International Publishing; 2021:57-78.
17. Raghavan PR. Metadichol serves as a natural ligand for the expression of Yamanaka reprogramming factors in human cardiac, fibroblast, and cancer cell lines. Med Res Arch. 2024;12(6). doi:10.18103/mra.v 12i6.5323
18. Warwick T, Schulz MH, Günther S, et al. An investigation of the hierarchical regulatory network of the vitamin D-induced transcriptome uncovers novel regulators and demonstrates total VDR dependency in monocytes. Sci Rep. 2021;11(1): 6518. doi:10.1038/s41598-021-86032-5
19. Valdés-López JF, Velilla P, Urcuqui-Inchima S. Vitamin D regulates the expression of toll-like receptors and pro-inflammatory cytokines without influencing Chikungunya virus replication in monocytes and macrophages. Acta Trop. 2022;232:106497. doi:10.1016/j.actatropica.2022.106497
20. Hmama Z, Nandan D, Sly L, Knutson KL, Herrera-Velit P, Reiner NE. The differentiation of myeloid cells produced by 1α,25-dihydroxyvitamin D3 is governed by a signaling complex including the vitamin D receptor and phosphatidylinositol 3-kinase. J Exp Med. 1999;190(11):1583-1594.
21. Chakraborty B, Mondal P, Gajendra P, Mitra M, Das C, Sengupta S. Analyzing the genetic regulation of CD14 by SP1 through the analysis of the peripheral blood mononuclear transcriptome in malaria patients infected with P. falciparum and P. vivax. EBioMedicine. 2018;37:442-452. doi:10.101 6/j.ebiom.2018.09.049
22. Szpirer J, Szpirer C, Riviere M, et al. The SP1 transcription factor gene (SP1) and the 1,25-dihydroxyvitamin D3 receptor gene (VDR) are located together on human chromosomal arm 12q and rat chromosome 7. Genomics. 1991;11(1):168-173. doi:10.1016/0888-7543(91)90114
23. Zhang DE, Hetherington CJ, Tan S, et al. Sp1 is an essential element for the monocytic-specific expression of human CD14. J Biol Chem. 1994;269 (15):11425-11434. doi:10.1016/S0021-9258(19)78141-1
24. Kenakin T. Inverse, versatile, and ligand-specific agonism: issues of receptor conformation. FASEB J. 2001;15(7):598-611. doi:10.1096/fj.00-0438rev
25. Kenakin T. Innovative paradigms in pharmacological research: collateral efficacy and permissive antagonism. Nat Rev Drug Discov. 2005;4(11):919-927. doi:10.1038/nrd1875
26. Carlberg C, Seuter S, Heikkinen S. A comprehensive genome-wide analysis of vitamin D receptor localization and its molecular ramifications. Anticancer Res. 2012;32(1):271-282.
27. Joseph SB, Bradley MN, Castrillo A, et al. LXR-dependent gene expression is crucial for macrophage viability and the innate immune response. Cell. 2004;119(2):299-309.
28. Raghavan PR. Metadichol® is a nano lipid emulsion that activates all 49 nuclear receptors in stem and somatic cells. Arch Clin Biomed Res. 2023;7(4):524-536. doi:10.26502/acbr.50170370.
29. Cheah MT, Chen JY, Sahoo D, Contreras-Trujillo H, Volkmer AK, Scheeren FA, Volkmer JP, Weissman IL. CD14-expressing cancer cells establish the inflammatory and proliferative tumor microenvironment in bladder cancer. Proc Natl Acad Sci U S A. 2015 Apr 14;112(15):4725-30. doi: 10.1073/pnas.1424795112
30. Alemán CL, Más R, Hernández C, et al. A 12-month study of policosanol oral toxicity in Sprague Dawley rats. Toxicol Lett. 1994;70(1):77-87.
31. Alemán CL, Ferreiro RM, Puig MN, Guerra IR, Ortega CH, Capote A. Carcinogenicity of policosanol in Sprague Dawley rats: a 24 month study. Teratog Carcinog Mutagen. 1994;14(5):239-249.
32. Alemán CL, Puig MN, Elías EC, et al. Carcinogenicity of policosanol in mice: an 18-month study. Food Chem Toxicol. 1995;33(7):573-578.

