Triple Therapy for Ethambutol-Induced Optic Neuropathy
Therapeutic role of triple drug regimen of pentoxifylline, cilostazol, and methylcobalamin for visual recovery in ethambutol induced neuropathy
Dr. Perwez Khan, Dr. Shweta Singh, Dr. Alok Ranjan, Dr. Lubna Khan*, Dr. Indu Yadav
*Professor, Department of Ophthalmology, GSVM Medical College, Kanpur, UP, India.
Senior resident, Department of Ophthalmology, GSVM Medical College, Kanpur, UP, India.
Assistant Professor, Department of Ophthalmology, Government Medical College Kanpur, UP, India.
Professor, Department of Pathology, GSVM Medical College Kanpur, UP, India.
Junior Resident, Department of Ophthalmology, GSVM Medical College Kanpur, UP, India.
OPEN ACCESS
PUBLISHED 31 May 2025
CITATION Khan, P., Singh, S., et al., 2025. Therapeutic role of triple drug regimen of pentoxifylline, cilostazol, and methylcobalamin for visual recovery in ethambutol induced neuropathy. Medical Research Archives, [online] 13(5). https://doi.org/10.18103/mra.v13i5.6588
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i5.6588
ISSN 2375-1924
ABSTRACT
Purpose: To evaluate the efficacy of a novel oral combination therapy—pentoxifylline, cilostazol, and methylcobalamin—in improving visual and electrophysiological outcomes in patients with ethambutol-induced optic neuropathy (EON).
Methods: A prospective interventional study was conducted on 34 patients (68 eyes) diagnosed with EON. Patients were randomly divided into two groups. The intervention group (n = 17) received oral pentoxifylline (16–18 mg/kg/day in two divided doses), cilostazol (50 mg/day), and methylcobalamin (1500 mcg/day) for 6 months. The control group (n = 17) received methylcobalamin alone for the same duration. Patients were evaluated at baseline and followed up at 1.5 months intervals for 6 months. Assessments included best-corrected visual acuity (BCVA) using early treatment diabetes retinopathy study (ETDRS) chart, contrast sensitivity via Pelli-Robson chart, color vision using Ishihara plates and fundus examination. Visual evoked potentials (VEP) with reverse pattern stimulation were performed at baseline and 6 months.
Results: The intervention group demonstrated significantly greater improvement in BCVA (mean gain: 0.71 ± 0.21; p = 0.001) and contrast sensitivity (0.697 ± 0.02; p = 0.002) compared to controls. VEP revealed a significant reduction in P100 latency in the intervention group (4.18 ± 1.23 ms; p = 0.003), indicating better optic nerve recovery, whereas latency worsened in the control group. Color vision improvement was not statistically significant (p > 0.05) in both groups.
Conclusion: The combination of pentoxifylline, cilostazol, and methylcobalamin significantly improved visual and electrophysiological parameters in EON patients. This multi-targeted neuro-regenerative therapy may represent a promising adjunct in the management of drug-induced optic neuropathies.
Keywords: Ethambutol-induced optic neuropathy, Pentoxifylline, Cilostazol, Methylcobalamin, Visual Evoked Potential, Best-Corrected Visual Acuity.
INTRODUCTION
Ethambutol is a cornerstone of first-line anti-tubercular regimens owing to its bacteriostatic efficacy and role in preventing resistance. However, its use is limited by ethambutol-induced optic neuropathy (EON), which if severe and prolonged can lead to permanent vision loss. EON typically presents as bilateral, painless, progressive visual loss, with accompanying signs such as central or cecocentral scotomas, dyschromatopsia (particularly red-green), and decreased contrast sensitivity. Visual evoked potentials (VEP) commonly reveal prolonged P100 latency, indicating visual pathway dysfunction. The reported incidence of EON ranges from 1% to 5%, depending on treatment duration, cumulative dose, renal function, and individual susceptibility.
Mechanism of optic neuritis due to ethambutol toxicity is unknown, however various studies suggest that ethambutol and its metabolites can chelate zinc, causing a disturbance in retinal homeostasis. Additionally, ethambutol interrupts oxidative phosphorylation by acting on iron-containing complex I and copper-containing complex IV, leading to reactive oxygen species production which may cause injury to the retinal ganglion cells. Additional factors such as deficiencies in trace elements (zinc, copper) and neurotrophic vitamins (e.g., B1, B12, E, folate) may exacerbate neural vulnerability.
Currently, the cornerstone of EON management involves early cessation of ethambutol and supportive therapy with neurotrophic agents like methylcobalamin, which is widely used. However, clinical outcomes are often suboptimal, with many patients experiencing incomplete or delayed visual recovery. This limitation has prompted interest in adjunctive therapies that target additional pathogenic mechanisms such as ischemia, inflammation, and oxidative damage.
Pentoxifylline, a non-selective phosphodiesterase inhibitor and methylxanthine derivative, has demonstrated neuroprotective and microcirculatory benefits in ischemic optic neuropathies. It improves erythrocyte deformability, reduces blood viscosity, inhibits platelet aggregation, and reduces oxidative stress. It also has immunomodulatory properties which results in vascular endothelial stabilization and autoimmune inhibition. It decreases peripheral nerve injury by preventing free radical production by neutrophil inhibition and decreasing the levels of cytokines, malondialdehyde and myeloperoxidase. Previous studies have shown functional improvement in neuropathies by oral pentoxifylline.
Cilostazol, a selective phosphodiesterase-III (PDE3) inhibitor, exerts vasodilatory and anti-platelet effects by increasing intracellular cAMP levels, thereby enhancing microvascular perfusion and endothelial function.
Zinc and copper deficiencies, caused by the metal-chelating effects of ethambutol, are believed to contribute to the development of neuropathy. Similarly, deficiencies in vitamins such as E, B1, B9, and B12 may worsen optic neuritis, in order to mitigate this risk, supplementing these minerals and multivitamins has been suggested. Methylcobalamin supports remyelination and axonal regeneration, and its deficiency has been implicated in various optic neuropathies. Together, these agents may augment optic nerve recovery when combined with methylcobalamin, due to its role in promoting remyelination and axonal regeneration.
To the best of our knowledge, no previous study has evaluated the combined therapeutic efficacy of pentoxifylline, cilostazol, and methylcobalamin. This study aimed to evaluate the efficacy of this novel oral combination therapy compared with methylcobalamin monotherapy to improve visual function and VEP parameters in patients with ethambutol-induced optic neuropathy.
MATERIAL AND METHODS
An institutional prospective, interventional study was conducted in the tertiary care center located in north India. 34 patients (n = 68 eyes) diagnosed with ethambutol-induced optic neuritis (EON) were enrolled after obtaining informed written consent. The diagnosis of EON was based on clinical features, history of ethambutol use, and supportive electrophysiological findings. The study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Ethics Committee.
Inclusion criteria: Patients of ethambutol optic neuropathy with minimum baseline visual acuity of hand movements or better were included in the study. Patients diagnosed within 6 months of cessation of ethambutol therapy were included.
Exclusion criteria: Patients with other causes of optic neuropathy (e.g., nutritional, hereditary, ischemic), systemic illnesses affecting visual function (e.g., diabetes mellitus with retinopathy), or those on concurrent neuroprotective therapies were excluded.
Patients were randomly divided into two equal groups (n = 17 per group, 34 eyes each) using a simple randomization technique. The intervention group received oral pentoxifylline (16–18 mg/kg/day) in divided doses, cilostazol (50 mg per day single dose), along with methylcobalamin (1500 mcg single dose) daily. The control group received only methylcobalamin 1500 mcg once daily. All treatments were administered orally and were continued for a duration of six months. Patients were followed at baseline, 1.5 months, 3 months, 4.5 months, and 6 months. At each visit, visual and ocular parameters were assessed, including BCVA measured by using the ETDRS chart, contrast sensitivity using the Pelli-Robson chart, and color vision using the Ishihara pseudoisochromatic plate test. Fundus examination was conducted by indirect ophthalmoscopy using a 20-diopter lens. These assessments were performed bilaterally and documented at each visit to monitor progression or improvement.
Visual evoked potential (VEP) testing was performed using the reverse pattern stimulation method to evaluate optic nerve conduction. VEP was recorded only at baseline and at the 6-month follow-up.
Data were compiled and analysed using IBM SPSS Statistics software version 24.0 (IBM Corp., Chicago, USA). Descriptive statistics were applied for baseline demographic and clinical characteristics. The independent samples t-test was employed to compare mean values between the intervention and control groups for BCVA, contrast sensitivity, and VEP latency. Levene’s test was used to assess the equality of variances. A p-value less than 0.05 was considered statistically significant.
The primary outcome measures included change in BCVA and VEP P100 latency from baseline to six months. Secondary outcomes included changes in contrast sensitivity and color vision. Adverse effects, medication tolerance, and compliance were monitored throughout the study duration. This methodology allowed for a comprehensive evaluation of visual function, integrating both subjective (acuity, color vision, contrast sensitivity) and objective (VEP) assessments, while comparing the efficacy of pentoxifylline and cilostazol adjunct therapy with standard neurotrophic treatment in patients affected by ethambutol-induced optic neuropathy.
RESULTS
A total of 34 patients (68 eyes) were enrolled in the study. Female preponderance (n=21) was noted with M:F ratio being 1.6:1. Majority of patients (n=31) were in the age group of 50-65 years with remaining 3 patients in age group of 25-35 years. The intervention group consisted of 17 patients (34 eyes), and the control group also included 17 patients (34 eyes).
Visual Improvement: The mean improvement in BCVA in the intervention group was 0.71 ± 0.21, which showed a significant improvement (p = 0.001). In contrast, the control group (0.21 ± 0.07) exhibited no significant change, with a p-value of 0.09.
| Group | Mean±SD | P-value |
|---|---|---|
| Intervention group | 0.71 ±0.21 | 0.001 |
| Control group | 0.21± 0.07 | 0.09 |
On applying Levene’s test for equality of variance for visual acuity across subsequent visits, the p-values were 0.94, 0.03, 0.02, and 0.001, respectively, indicating a progressively significant improvement in the intervention group over time.
| Visit | Intervention group (Mean ± SD) | Control group (Mean ± SD) | P-value |
|---|---|---|---|
| 1st | 1.522±0.6 | 1.51±0.5 | 0.94 |
| 2nd | 1.2±0.5 | 1.47±0.49 | 0.03 |
| 3rd | 0.99±0.4 | 1.3±0.5 | 0.02 |
| 4th | 0.81±0.4 | 1.3±0.5 | 0.001 |
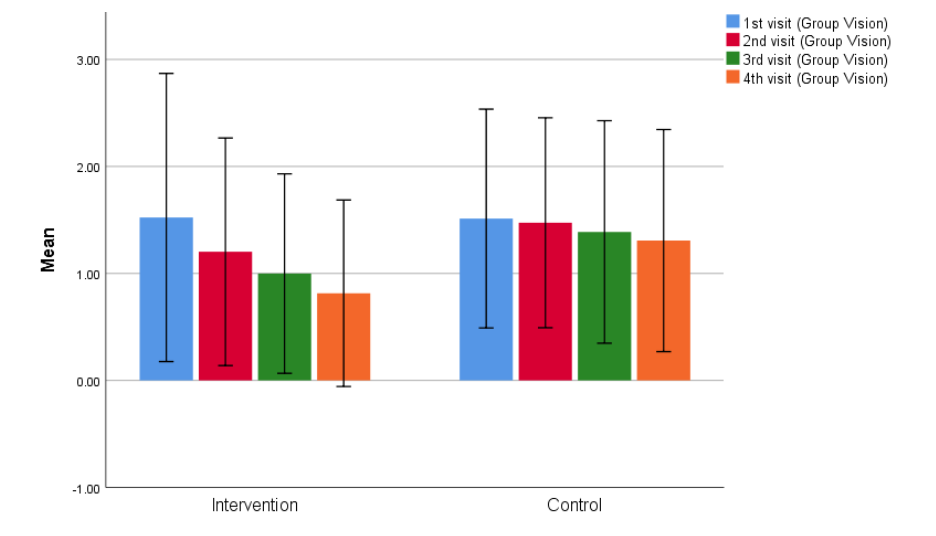
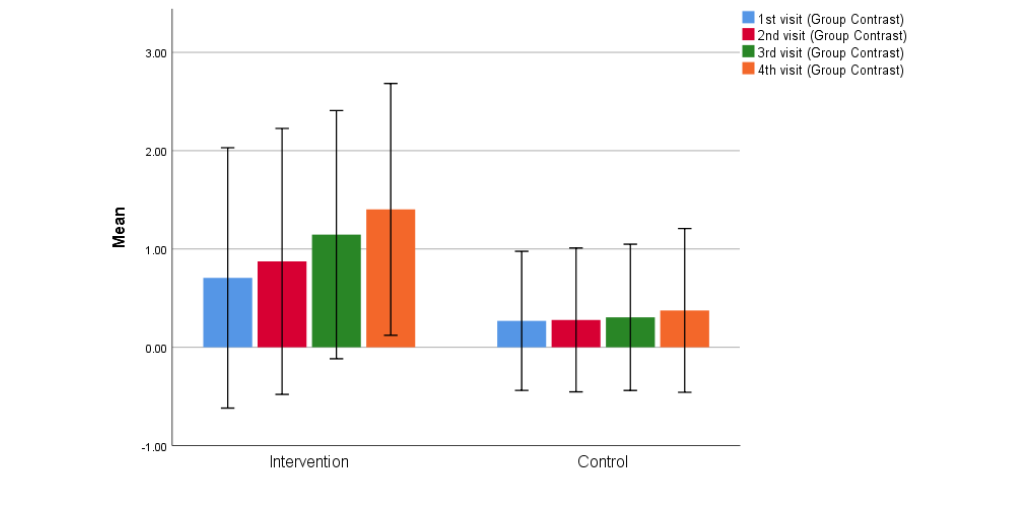
Contrast Sensitivity: The mean improvement in contrast sensitivity in the intervention group was 0.697 ± 0.02 (p = 0.002), while the control group showed only a slight improvement of 0.013 ± 0.06 (p = 0.08), which was not statistically significant.
| Group | Mean±SD | P-value |
|---|---|---|
| Intervention | 0.697± 0.02 | 0.002 |
| Control | 0.106 ±0.06 | 0.08 |
| Visit | Intervention group (Mean ± SD) | Control group (Mean ± SD) | P-value |
|---|---|---|---|
| 1st | 0.7±0.6 | 0.2±0.3 | 0.05 |
| 2nd | 0.8±0.6 | 0.2±0.3 | 0.02 |
| 3rd | 1.1±0.5 | 0.3±0.3 | 0.01 |
| 4th | 1.4±0.5 | 0.3±0.4 | 0.001 |
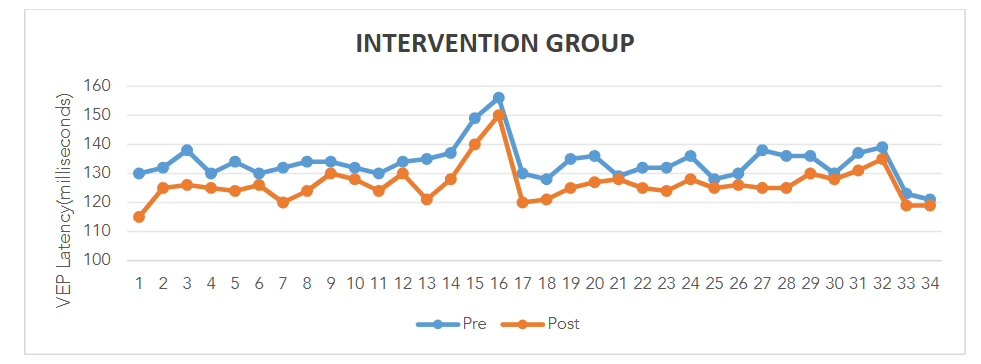
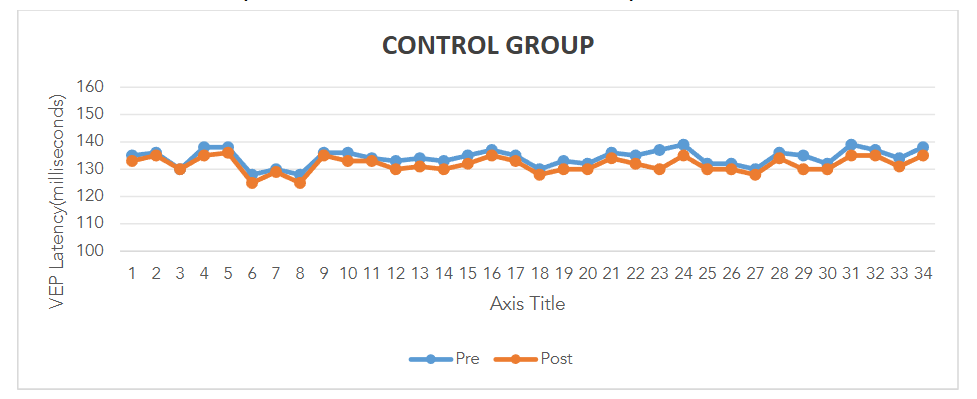
Visual Evoked Potential (VEP): The mean reduction in P100 latency in the intervention group was 4.18 ± 1.23 millisecond (p = 0.003), indicating significant improvement in optic nerve function. In contrast, the control group showed a mean increase in P100 latency of 4.65 ± 0.52 ms (p = 0.004), suggesting worsening of optic nerve conduction.
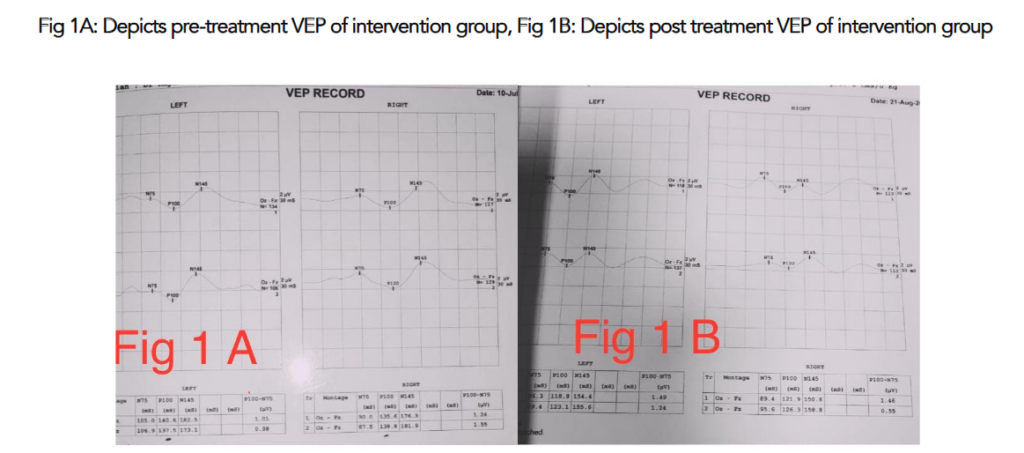
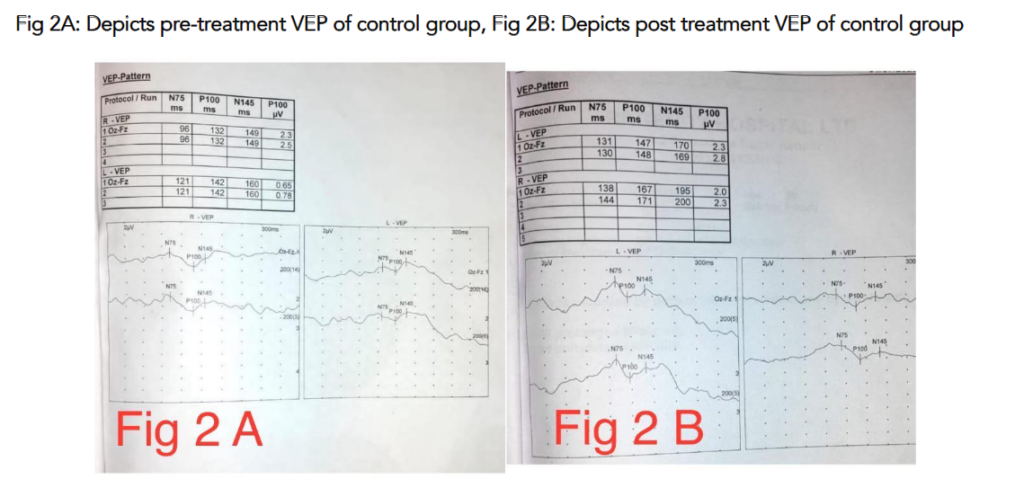
Color Vision: Statistical analysis using the Z-test for proportions revealed no significant difference between the two groups (Z = 0.551, p = 1.164), indicating that color vision did not improve in either group.
Statistical Analysis and Observations: This study demonstrated that pentoxifylline, when used in combination with cilostazol and methylcobalamin, significantly improved BCVA and contrast sensitivity, as well as reduced P100 latency in patients with ethambutol-induced optic neuropathy. These findings suggest that combination therapy by above agents may offer neuroprotective benefits, enhancing visual and electrophysiological outcomes, although its effect on color vision was not statistically significant.
DISCUSSION
Natural course of recovery of EON despite timely discontinuation of ethambutol suggests persistent visual impairment and irreversible damage to optic nerve, especially in older age patients (individuals over 60 years of age). The present study was aimed to evaluate the therapeutic efficacy of a neuro-regenerative combination – pentoxifylline, cilostazol, and methylcobalamin in managing ethambutol-induced optic neuropathy, a condition with limited established treatment options. The treatment strategy was designed to target both vascular and neuronal pathways implicated in optic nerve damage, thereby offering a multidimensional approach to neuroprotection.
Methylcobalamin was administered to the control arm orally to assess the role of combination therapy in comparison to monotherapy of methylcobalamin as it is widely used for treatment of various neurological disorders. It has also showed significant improvement in cases of crush injury of optic nerve, however its role in treatment of EON has been inconsistent. Available literature show comparison of methylcobalamin monotherapy with buqihuoxue formula combined with methylcobalamin which is consistent with the present methodology.
The combination therapy exhibited marked visual and electrophysiological improvement in the intervention group compared to controls. Visual acuity improvement emerged as the most clinically significant outcome, revealing the potential of a neuro-regenerative cocktail composed of pentoxifylline, cilostazol, and methylcobalamin in reversing functional visual loss in patients with ethambutol-induced optic neuritis (EON). Supporting evidence for the efficacy of pentoxifylline in optic neuropathies exists but is limited. However; this study focused solely on visual acuity and lacked data on contrast sensitivity, VEP latency, or structured combination therapy, leaving an important therapeutic gap. In contrast, the current study adds to the field by demonstrating that the combination of pentoxifylline with cilostazol and methylcobalamin confers enhanced visual recovery, potentially due to synergistic effects. Therapeutic dose of cilostazol for peripheral vascular disease is 100mg twice a day. However; no dosage has been defined for the treatment of EON, hence, subtherapeutic dose of cilostazol (50mg per day in single dose) was administered in order to limit adverse drug reactions and to achieve better compliance. Headache, palpitation, dizziness, diarrhoea, peripheral edema and tachycardia are some of the frequent side effects of cilostazol which may lead to poor compliance.
Contrast sensitivity is a sensitive indicator of optic nerve function, often affected in EON. Contrast sensitivity testing may detect subclinical optic neuropathy even in the presence of normal visual acuity and color perception. In our study, the intervention group exhibited a significant improvement in contrast sensitivity. Our study is distinguished by the use of combination therapy, which not only halts further deterioration but also enhances contrast sensitivity, indicating a potential role in reversing Ethambutol-induced visual impairment.
Visual evoked potential (VEP) provide objective measure of optic nerve function and serve as a sensitive electrophysiological tool for detecting optic nerve dysfunction in patients receiving ethambutol, with prolongation of P100 latency and reduction in amplitude. Some studies have reported cases where VEP changes were not evident despite clinical signs of optic neuropathy, suggesting that VEP may not always detect early or subclinical changes, indicating that no single parameter is sufficient to make diagnosis of EON. Our study found a significant reduction in VEP P100 latency in the intervention group following combination therapy, indicating improved optic nerve conduction. Our study suggests that VEPs, beyond their established role in early diagnosis, may also serve as a biomarker for monitoring therapeutic response in patients with ethambutol-induced optic neuropathy.
Previous studies have emphasized the importance of color vision testing in the early detection of ethambutol-induced optic neuropathy, with one reporting abnormal color perception among affected patients and another noting that 71.8% of eyes exhibited color vision defects in cases of toxic optic neuropathy secondary to ethambutol. Our study also supports existing evidence that color vision is frequently affected in EON, however, our novel combination therapy did not result in a statistically significant improvement in color vision, suggesting that this parameter may be less responsive to pharmacologic intervention compared to other visual and electrophysiological measures or in our study’s use of Ishihara plates may have limited sensitivity in detecting subtle color vision changes, especially in patients with severe visual acuity loss.
The unique aspect of our study lies in its pioneering assessment of a neuro-regenerative combination therapy-pentoxifylline, cilostazol, and methylcobalamin-in managing EON, an area previously unexplored in clinical literature. Unlike earlier studies that primarily focused on visual acuity or isolated electrophysiological changes, our research provides a comprehensive evaluation of multiple visual parameters, including contrast sensitivity, VEP P100 latency, and color vision. Additionally, this is the first interventional study to demonstrate that visual parameters and VEP abnormalities in EON are not only detectable but potentially reversible with targeted pharmacologic intervention. These findings underscore the significance of our study in advancing both the diagnostic refinement and therapeutic approach for EON.
LIMITATIONS:
Despite the promising results, this study has several limitations. The relatively small sample size (34 patients) limits the generalizability of the findings. The study was conducted at a single center, which may introduce selection bias and limit external validity. Furthermore, the follow-up period of six months, though adequate for observing short-term outcomes, may not fully capture the long-term efficacy or relapse following cessation of therapy. Lastly, serum levels of ethambutol or oxidative stress markers were not evaluated, which could have provided mechanistic insights and further validated the therapeutic effects of the intervention.
CONCLUSION:
This prospective interventional study demonstrates that a novel combination of pentoxifylline, cilostazol, and methylcobalamin significantly improves visual acuity, contrast sensitivity, and VEP latency in patients with EON, suggesting both functional and electrophysiological recovery of the optic nerve. These results highlight the therapeutic potential of a multi-targeted neuro-regenerative approach in managing drug-induced optic neuropathies. However, the absence of significant improvement in color vision suggests differential responsiveness among visual pathways, indicating the need for further exploration into selective neuroprotection. Larger, multicentric studies with longer follow-up and objective biochemical markers are needed to validate the efficacy and durability of this therapeutic regimen. Clinically, our results support the early use of targeted neuro-regenerative therapy in EON to enhance visual recovery and mitigate permanent visual disability.
Conflict of Interest: None.
Funding Statement: None.
Acknowledgements: None.
REFERENCES:
- Chamberlain PD, Sadaka A, Berry S, Lee AG. Ethambutol optic neuropathy. Curr Opin Ophthalmol. 2017;28:545-551. doi:10.1097/ICU.0000000000000416. PMID: 28759559.
- Koul PA. Ocular toxicity with ethambutol therapy: timely precaution. Lung India. 2015;32:1-3. doi:10.4103/0970-2113.148395. PMID: 25624586; PMCID: PMC4298909.
- Garg P, Garg R, Prasad R, Mishra AK. A prospective study of ocular toxicity in patients receiving ethambutol as a part of directly observed treatment strategy therapy. Lung India. 2015;32:16-19. doi:10.4103/0970-2113.148428. PMID: 25624590; PMCID: PMC4298911.
- Citron KM, Thomas GO. Ocular toxicity from ethambutol. Thorax. 1986;41:737-739. doi:10.1136/thx.41.10.737. PMID: 3787505; PMCID: PMC460464.
- Menon V, Jain D, Saxena R, Sood R. Prospective evaluation of visual function for early detection of ethambutol toxicity. Br J Ophthalmol. 2009;93:1251-1254. doi:10.1136/bjo.2008.148502. Epub 2009 Jun 11. PMID: 19525243.
- Kim J, Kwon HY, Ahn SJ. Nationwide usage of ethambutol and incidence and screening practices of optic neuropathy. Front Pharmacol. 2024;15:1461111. doi:10.3389/fphar.2024.1461111. PMID: 39474613; PMCID: PMC11518835.
- Islam IC, Umar BT, Minhajat R, Bukhari A, Sari DK, Kaelan C. Ethambutol effect on retinal and optic nerve structural changes in toxic optic neuropathy: a literature review. Ophthalmol Res Int J. 2022;16:26-36.
- Lee EJ, Kim SJ, Choung HK, Kim JH, Yu YS. Incidence and clinical features of ethambutol-induced optic neuropathy in Korea. J Neuroophthalmol. 2008;28:269-277. doi:10.1097/WNO.0b013e31818e3c6b. PMID: 19145123.
- Sadun AA. Metabolic optic neuropathies. Semin Ophthalmol. 2002;17:29-32. doi:10.1076/soph.17.1.29.10290. PMID: 15513453.
- Grzybowski A, Zülsdorff M, Wilhelm H, Tonagel F. Toxic optic neuropathies: an updated review. Acta Ophthalmol. 2015;93:402-410. doi:10.1111/aos.12515. Epub 2014 Aug 27. PMID: 25159832.
- Rasool M, Malik A, Manan A, Aziz K, Mahmood A, Zaheer S, et al. Determination of potential role of antioxidative status and circulating biochemical markers in the pathogenesis of ethambutol-induced toxic optic neuropathy among diabetic and non-diabetic patients. Saudi J Biol Sci. 2015;22:739-743. doi:10.1016/j.sjbs.2014.09.019. Epub 2014 Oct 12. PMID: 26587002; PMCID: PMC4625422.
- Ciuffetti G, Mercuri M, Ott C, Lombardini R, Paltriccia R, Lupattelli G, et al. Use of pentoxifylline as an inhibitor of free radical generation in peripheral vascular disease: results of a double-blind placebo-controlled study. Eur J Clin Pharmacol. 1991;41:511-515. doi:10.1007/BF00314976. PMID: 1667754.
- Dawson DL, Cutler BS, Hiatt WR, Hobson RW 2nd, Martin JD, Bortey EB, et al. A comparison of cilostazol and pentoxifylline for treating intermittent claudication. Am J Med. 2000;109:523-530. doi:10.1016/s0002-9343(00)00569-6. PMID: 11063952.
- Ward A, Clissold SP. Pentoxifylline: a review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic efficacy. Drugs. 1987;34:50-97. doi:10.2165/00003495-198734010-00003. PMID: 3308412.
- Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34:10-16. doi:10.1097/SHK.0b013e3181cdc3e2. PMID: 19997047.
- Savaş C, Aras T, Cakmak M, Bilgehan A, Ataoğlu O, Türközkan N, Ozgüner F, Yücesan S, Dindar H. Pentoxifylline inhibits overflow and reduces intestinal reperfusion injury. J Pediatr Surg. 1997;32:905-910. doi:10.1016/s0022-3468(97)90648-5. PMID: 9200098.
- Khan P, Khan L, Awasthi U, Khan A, Siddique Z, Gupta RC. Pentoxifylline: a therapeutic remedy for optic neuropathies. J Evol Med Dent Sci. 2013;2:3516-3522.
- Goto S. Cilostazol: potential mechanism of action for antithrombotic effects accompanied by a low rate of bleeding. Atheroscler Suppl. 2005;6:3-11. doi:10.1016/j.atherosclerosissup.2005.09.002. Epub 2005 Nov 4. PMID: 16275169.
- Rondina MT, Weyrich AS. Targeting phosphodiesterases in anti-platelet therapy. Handb Exp Pharmacol. 2012;(210):225-238. doi:10.1007/978-3-642-29423-5_9. PMID: 22918733; PMCID: PMC3682780.
- Guerra R, Casu L. Hydroxycobalamin for ethambutol-induced optic neuropathy. Lancet. 1981;2:1176. doi:10.1016/s0140-6736(81)90631-0. PMID: 6118620.
- Tsai RK, Lee YH. Reversibility of ethambutol optic neuropathy. J Ocul Pharmacol Ther. 1997;13:473-477. doi:10.1089/jop.1997.13.473. PMID: 9326729.
- Kumar A, Sandramouli S, Verma L, Tewari HK, Khosla PK. Ocular ethambutol toxicity: is it reversible? J Clin Neuroophthalmol. 1993;13:15-17.
- Sivakumaran P, Harrison AC, Marschner J, Martin P. Ocular toxicity from ethambutol: a review of four cases and recommended precautions. N Z Med J. 1998;111:428-430.
- DeVita EG, Miao M, Sadun AA. Optic neuropathy in ethambutol-treated renal tuberculosis. J Clin Neuroophthalmol. 1987;7:77-86.
- Melamud A, Kosmorsky GS, Lee MS. Ocular ethambutol toxicity. Mayo Clin Proc. 2003;78:1409-1411. doi:10.1016/S0025-6196(11)62071-9.
- Sharma V, Biswas D. Cobalamin deficiency presenting as obsessive-compulsive disorder: case report. Gen Hosp Psychiatry. 2012;34:578.e7-e8.
- Kong X, Sun X, Zhang J. The protective role of Mecobalamin following optic nerve crush in adult rats. Yan Ke Xue Bao. 2004;20:171-177.
- Matsumoto T, Kusabiraki R, Arisawa A, Fujiki T, Noda A, Tanaka A, Yamamoto N, Aihara K, Yamaoka S, Mishima M. Drastically progressive ethambutol-induced optic neuropathy after withdrawal of ethambutol: a case report and literature review. Intern Med. 2021;60:1785-1788. doi:10.2169/internalmedicine.6178-20.
- Xu J, Dai Q, Wu S, Sheng W, Zhao S, Zhong L. Treatment of ethambutol-induced optic neuropathy by Buqihuoxue formula combined with methycobal. Int J Clin Exp Med. 2017;10:7011-7015.
- Balinski AM, Preuss CV. Cilostazol. Updated March 27, 2023. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; January 2025-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK544363/.
- Salmon JF, Carmichael TR, Welsh NH. Use of contrast sensitivity measurement in the detection of subclinical ethambutol toxic optic neuropathy. Br J Ophthalmol. 1987;71:192-196. doi:10.1136/bjo.71.3.192. PMID: 3828274; PMCID: PMC1041117.
- Yiannikas C, Walsh JC, McLeod JG. Visual evoked potentials in the detection of subclinical optic toxic effects secondary to ethambutol. Arch Neurol. 1983;40:645-648. doi:10.1001/archneur.1983.04050090081014. PMID: 6615272.
- Woung LC, Jou JR, Liaw SL. Visual function in recovered ethambutol optic neuropathy. J Ocul Pharmacol Ther. 1995;11:411-419. doi:10.1089/jop.1995.11.411. PMID: 8590273.
- Choi SY, Hwang JM. Optic neuropathy associated with ethambutol in Koreans. Korean J Ophthalmol. 1997;11:106-110. doi:10.3341/kjo.1997.11.2.106. PMID: 9510653.
- Somers LP, Bosten JM. Screening for mild anomalous trichromacy using the Ishihara plates test. J Opt Soc Am A Opt Image Sci Vis. 2023;40:A208-A219. doi:10.1364/JOSAA.476536. PMID: 37133039.




