Cost-Effectiveness of Emicizumab in Severe Hemophilia A
Cost-effectiveness analysis of prophylaxis with emicizumab or bypassing agents in children with severe haemophilia A and high-response inhibitors after immunotolerance failure in Brazil
Luila Clicia Moura Henriques1, Francisco de Assis Acurcio1, Ricardo Mesquita Camelo1,2, Mariana Michel Barbosa1, Augusto Afonso Guerra Júnior1, Juliana Alvares-Teodoro1
- Faculty of Pharmacy, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil.
- Faculty of Medicine, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Emicizumab prophylaxis in children with severe haemophilia A and high-response inhibitor is more effective in preventing spontaneous bleeding when compared with bypassing agents prophylaxis. Brazil has approved emicizumab prophylaxis for high-response inhibitor who failed immune tolerance induction.
Methods: A cost-effectiveness analysis was performed to compare emicizumab prophylaxis with bypassing agents prophylaxis in high-response inhibitor during the first 5 years of treatment after immune tolerance induction failure, from the perspective of the Brazilian Ministry of Health (payer). The analysis included a 5-year-old male with high-response inhibitor (baseline weight: 20 kg). We obtained the regimens and costs (in US dollars, adjusted for Purchasing Power Parity) from the recommendations of the Brazilian Ministry of Health and the manufacturer’s package insert and purchasing information, respectively, for both emicizumab and bypassing agents prophylaxis, and the treatment of bleeding events with bypassing agents. We retrieved information on the frequency and treatment of bleeding during prophylaxis from the literature. The sensitivity analysis considered the regimen for prophylaxis and on-demand with bypassing agents and emicizumab, the probability of using a specific type of bypassing agents, and the costs associated with emicizumab and bypassing agents. Moreover, in the sensitivity analysis for emicizumab prophylaxis, we assumed settings with waste (amount of residual emicizumab discarded based on the weight regimen and vial availability) and without waste.
Results: The cost of emicizumab prophylaxis was US$1,012,021.4/ high-response inhibitor with 1.0 bleedings/ high-response inhibitor. Bypassing agents’ prophylaxis costs US$3,659,926.2/ high-response inhibitor with 16.5 bleedings/ high-response inhibitor. The incremental cost-effectiveness ratio was US$170,832.6 per bleed averted, favoring emicizumab prophylaxis. Emicizumab prophylaxis remained dominant after sensitivity analyses. The product regimen variables for bypassing agents prophylaxis and the likelihood of bypassing agents use had a high impact on incremental cost. Waste and on-demand bypassing agents doses had a minimal impact on incremental cost.
Conclusions: In high-response inhibitor, during the first 5 years following immune tolerance induction failure, emicizumab prophylaxis was a dominant option compared to bypassing agents’ prophylaxis, as per the Brazilian setting.
Keywords
haemophilia A; inhibitor; prophylaxis; emicizumab; bypass agent; cost-effectiveness analysis; Brazil.
Introduction
Haemophilia A (HA) is a rare hereditary coagulation disorder caused by pathogenic mutations in the F8 gene, which encodes coagulation factor VIII (FVIII). Due to the gene’s location on chromosome Xq28, haemophilia A predominantly affects males and is typically diagnosed in early childhood. The disease severity is classified based on residual FVIII plasma activity, with severe HA (FVIII <1%) associated with spontaneous haemorrhages, predominantly in joints and muscles. Treatment involves FVIII replacement therapy for both on-demand and prophylactic use. Prophylaxis involves the regular administration of clotting factors to maintain sufficiently high factor levels, regardless of bleeding episodes, with the aim of preventing them. Prophylaxis plays a crucial role in improving the quality of life of individuals with hemophilia, as it prevents bleeding episodes associated with this coagulopathy. Preventive treatment helps avoid joint and muscle damage, life-threatening hemorrhages, and long-term complications. Additionally, it enables individuals to engage more freely in physically demanding activities. However, approximately 30% of children with severe haemophilia A (CsHA) develop neutralizing alloantibodies (inhibitors), particularly high-response inhibitors, which substantially increase bleeding risk and morbidity. For these patients, FVIII replacement is ineffective, necessitating the use of bypassing agents such as activated prothrombin complex concentrate (aPCC) or recombinant activated factor VII (rFVIIa), which are associated with suboptimal haemostatic efficacy and higher treatment costs.
Immunotolerance induction (ITI) is the only recommended strategy for inhibitor eradication; however, 10–40% of severe haemophilia A with high-response inhibitors (CsHAhri) fail ITI and remain at an elevated risk of bleeding complications. Until recently, prophylaxis with bypassing agents (BpA) was the only therapeutic alternative for these patients. Emicizumab, a bispecific monoclonal antibody that mimics FVIII function by bridging activated factor IX (FIXa) and factor X (FX), has emerged as an effective prophylactic option. Clinical trials have demonstrated a significant reduction in annualized bleeding rate (ABR) with emicizumab in CsHAhri. Consequently, emicizumab prophylaxis is now recommended for this population. A systematic review and meta-analysis further confirmed superior efficacy of emicizumab compared to bypassing agents’ prophylaxis in reducing bleeding episodes.
In Brazil, 13,895 individuals with haemophilia A were registered in 2023, of whom 372 had active inhibitors. A national cohort study reported that 34.5% of CsHAhri failed ITI. In 2019, the Brazilian Ministry of Health incorporated emicizumab prophylaxis for CsHAhri who failed ITI as an alternative to bypassing agents prophylaxis. A previous economic evaluation conducted from the Brazilian Ministry of Health perspective demonstrated the dominance of emicizumab prophylaxis over bypassing agents prophylaxis in all assessed scenarios. When considering a maximum ITI duration of 37 months, the cost per CsHAhri undergoing ITI with emicizumab prophylaxis was estimated at US$546,358, compared to US$1,270,836 with bypassing agents prophylaxis, resulting in an incremental cost of US$724,478. The estimated bleeding rate during ITI was 0.7 for emicizumab prophylaxis versus 9.3 for bypassing agents’ prophylaxis, yielding an incremental cost-effectiveness ratio (ICER) of US$83,755 per bleeding episode avoided. However, this analysis did not account for drug waste associated with emicizumab. A subsequent budget impact analysis considered drug waste based on the Brazilian Ministry of Health protocol and manufacturer’s recommendations. The estimated expenditures ranged from US$2,917,877 to US$48,700,235, with waste representing 0.5% (US$17,420) to 38.2% (US$2,693,592) of the reconstituted product, depending on patient weight and dosing regimen.
Building on previous cost-effectiveness analyses, the present study evaluates the cost-effectiveness of emicizumab prophylaxis versus bypassing agents’ prophylaxis in severe haemophilia A with high-response inhibitors who failed immunotolerance induction over a five-year period from the perspective of the Brazilian Ministry of Health.
Methodology
MODEL DESIGN
This study builds upon the cost-effectiveness analysis conducted by Camelo et al. (2023). All analyses were performed using TreeAge Pro, LLC (Williamstown, MA) software. The target population consisted of male CsHAhri, aged five years at baseline, presenting a bleeding phenotype immediately following ITI failure. A five-year time horizon was adopted, and the analysis was conducted from the perspective of the Brazilian Ministry of Health. A decision tree model was developed to compare the costs and effectiveness (measured by the number of bleeding episodes) of prophylaxis with emicizumab (considering drug wastage) versus prophylaxis with bypassing agents, specifically activated prothrombin complex concentrate (aPCC) or recombinant activated factor VII (rFVIIa).
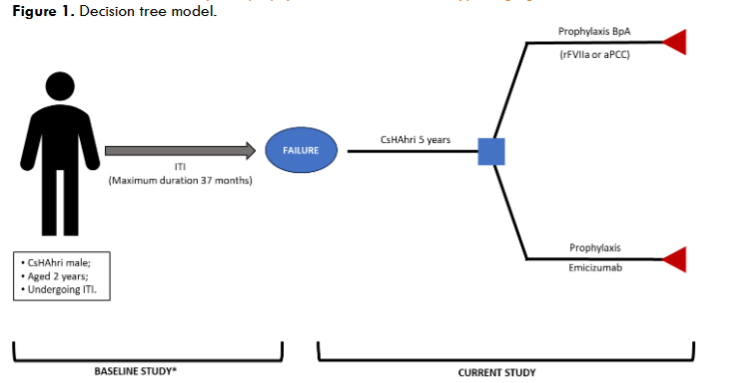
CsHAhri was defined as a child with residual plasma factor VIII (FVIII) activity <1% of normal and an inhibitor titre >5 BU/mL in at least one lifetime measurement. Given the high risk of spontaneous bleeding associated with this population, prophylaxis was considered the standard of care for ethical reasons, and on-demand treatment was excluded from the analysis. The hypothetical cohort assumed that individuals started prophylaxis with minimal lesions and remained stable over five years without complications. The initial age of five years was chosen based on previous studies in which ITI failure was reported at approximately 4.7 years. Treatment regimens remained unchanged throughout the five-year follow-up. Analyses were conducted at annual intervals (52 weeks), and median body weights for each interval were derived from Brazilian Institute of Geography and Statistics (IBGE) data, with a variability range of ±20%. Within each annual interval, weight was assumed to be constant but adjusted between intervals. Mortality and orthopaedic surgical procedures were not considered due to the limited time horizon.
Table 1. Parameters used in the economic model for children with severe haemophilia A and high-response inhibitors who failed to induce immunotolerance.
| Parameters | Data Sources |
|---|---|
| Patient characteristics | |
| Age, in years | 5 Assumption |
| Mean weight, in kg | IBGE |
| 5.0-5.9 years | 20 |
| 6.0-6.9 years | 22 |
| 7.0-7.9 years | 25 |
| 8.0-8.9 years | 28 |
| 9.0-9.9 years | 32 |
| Weight variability | ±20% Assumption |
| Male gender | 100% Assumption |
| Haemophilia A characteristics | |
| Severe | 100% Assumption |
| High-response inhibitor | 100% Assumption |
| Failure of immunotolerance | 100% Assumption |
| Hemorrhagic phenotype | 100% Assumption |
| Prophylaxis | 100% Assumption |
| Prophylaxis characteristics | |
| Product | Assumption |
| Emicizumab | 100% |
| aPCC (variation) | 50% (0-100%) |
| rFVIIa (variation) | 50% (0-100%) |
| Regimen, in mg/kg/week | 3.0 |
| Duration, in weeks | 4 |
| Maintenance | |
| Regimen, in mg/kg/month | 6.0 |
| Duration, in weeks | 1st year 48; 2nd to 5th years 52 – per year |
| Bypass agents | Brazilian Guidance of Haemophilia Treatment, Astermark et al, Brazilian ITI protocol, López-Fernández et al |
| aPCC, in U/kg/week (interval) | 243.8 (162.5-700.0) |
| rFVIIa, in µg/kg/week (interval) | 630.0 (292.5-1890.0) |
| On-demand treatment characteristics | |
| Bleedings (annualized rate) | Muniz et al |
| Emicizumab, in prophylaxis (interval) | 0.2 (0.0-1.0) |
| aPCC, in prophylaxis (interval) | 3.3 (0.0-6.1) |
| rFVIIa, in prophylaxis (interval) | 3.3 (0.0-6.1) |
| On-demand treatment regimen (product used for prophylaxis) | |
| rFVIIa (emicizumab), in µg/kg/bleeding (interval) | 180.0 (90.0-240.0) |
| aPCC (aPCC), in U/kg/bleeding (interval) | 87.5 (50.0-100.0) |
| rFVIIa (rFVIIa), in µg/kg/bleeding (interval) | 180.0 (90.0-270.0) |
| Waste | |
| Emicizumab (interval) | 0% (0%-16.4%) |
| CCPa | 0% Assumption |
| rFVIIa | 0% Assumption |
The prophylactic regimen for emicizumab included a loading dose of 3.0 mg/kg weekly for four weeks, followed by a maintenance dose of 6.0 mg/kg/month, irrespective of the administration frequency (1.5 mg/kg weekly, 3.0 mg/kg biweekly, or 6.0 mg/kg every four weeks). The total emicizumab requirement for the first year included both the loading and maintenance doses, while subsequent years considered only the maintenance regimen. The base-case analysis did not account for drug wastage. Bypassing agents prophylaxis consisted of either aPCC at 75.0 U/kg on alternate days (3.25 times per week; 243.8 U/kg/week) or rFVIIa at 90.0 µg/kg daily (630.0 µg/kg/week). Drug wastage was not included, and rounding to full vial use, as commonly practiced in clinical settings, was not considered. Annualized bleeding rates for CsHAhri receiving emicizumab or bypassing agents prophylaxis were derived from a previously published meta-analysis. The rates reported in forest plots were weighted according to study size, and additional studies excluded from the meta-analysis were reconsidered. The mean annualized bleeding rate (ABR) was calculated, with a range defined by the lowest and highest mean values. Bleeding episodes associated with surgical procedures were excluded. All bleeding episodes were managed with bypassing agents. For patients on emicizumab prophylaxis, bleeding was treated with rFVIIa, while those on bypassing agents prophylaxis received the same agent as their prophylactic regimen. Recommended doses for the treatment of bleeding episodes in CsHAhri included aPCC at 50 to 100 U/kg/week and rFVIIa at 90 to 270 µg/kg/week in the emicizumab cohort or 90 to 240 µg/kg/week in the bypassing agents cohort.
COSTS
Factor concentrate acquisition costs constitute approximately 99.5% of total haemophilia treatment expenditures; thus, only the direct drug acquisition costs of emicizumab and bypassing agents were considered. Cost data were obtained from the Brazilian Ministry of Health, which oversees the procurement and distribution of these agents within Brazil’s Unified Health System (Sistema Único de Saúde, SUS). Public procurement prices were extracted from the Health Price Database and adjusted per unit of measurement.
Table 2. Costs of emicizumab and bypassing agents.
| Products | Price, in PPP – US$ – public purchase, 2022* | Minimum, in PPP – US$ – as per CONITEC recommendation report, 2021 | Maximum in PPP – US$ – according to the values suggested by CMED, 2022† |
|---|---|---|---|
| Emicizumab | 100.47/mg | 80.47/mg | 187.94/mg |
| aPCC | 0.35/U | 0.28/U | 1.63/U |
| rFVIIa | 1.03/µg | 0.82/µg | 1.56/µg |
Minimum prices were calculated by applying a ±20% variation from baseline values. Maximum prices were adapted following Drug Market Regulation Chamber (CMED) guidelines, incorporating an 18% value-added tax (VAT) rate for 2024. Costs were reported in Brazilian Reals (R$) and converted to US dollars (US$) using purchasing power parity (PPP) for 2023 (PPP-US$1.00 = R$2.44). Due to the short time horizon, discounting was not applied.
SENSITIVITY ANALYSIS
Deterministic and probabilistic sensitivity analyses were conducted to assess model uncertainty. Deterministic sensitivity analysis examined variations in coagulation factor and emicizumab costs, emicizumab wastage, medication regimens, bleeding frequency, and associated treatment costs. Drug wastage was evaluated between 0% (complete utilization) and a maximum of 16.4%. Minimum and maximum values for all variables are detailed in Table 1. A tornado diagram was generated to illustrate the incremental cost variation from baseline, with the center representing the base-case estimate. Incremental costs were depicted as blue (low estimate) and red (high estimate) bars for each parameter. Probabilistic sensitivity analysis was performed via Monte Carlo simulation with 10,000 iterations. Triangular distributions were applied to dosing regimens and bleeding frequency parameters, while gamma distributions were used for cost variables.
Results
In the cost-effectiveness analysis, for the 5 years evaluated, prophylaxis with emicizumab and bypassing agents cost PPP-US$1,012,021.4 and PPP-US$3,659,926.2, respectively, resulting in an incremental cost of PPP-US$2,647,904.8 for bypassing agents prophylaxis per treatment of CsHAhri. Emicizumab prophylaxis resulted in a mean of 1.0 CsHAhri bleedings, while bypassing agents prophylaxis resulted in a mean of 16.5 bleedings/CsHAhri, giving rise to an additional ABR of 15.5 bleedings per CsHAhri treatment. Thus, bypassing agents prophylaxis resulted in higher costs and ABR than emicizumab prophylaxis. The incremental cost-effectiveness ratio (ICER) was PPP-US$170,832.6 per bleeding averted, with emicizumab prophylaxis dominant over bypassing agents prophylaxis.
Table 3. Results of the baseline case cost-effectiveness analysis.
| Treatment | Cost, in PPP-US$ | Incremental cost, in PPP-US$ | Effectiveness in number of bleedings | Incremental effectiveness, in number of bleedings | ICER in PPP-US$ |
|---|---|---|---|---|---|
| Emicizumab | 1,012,021.4 | NA | 1.0 | NA | NA |
| Bypassing agents | 3,659,926.2 | 2,647,904.8 | 16.5 | -15.5 | -170,832.6 |
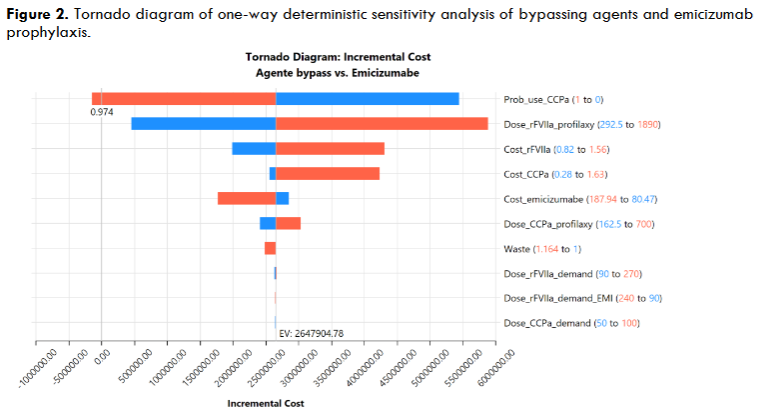
Deterministic sensitivity analysis was performed to identify variables influencing the incremental cost when comparing emicizumab and bypassing agents prophylaxis. The baseline value of the tornado diagram was US$170,832.6. The results were ranked in descending order, considering the lower and upper estimates of the 10 most influential parameters in the analysis. The dose of prophylaxis with rFVIIa was the variable with the most significant power to change the incremental cost, representing 70.7% of the total risk. Subsequently, the dose regimen of prophylaxis with aPCC represented 12.5% of the total risk of change in the incremental cost. The increase in the use of bypassing agents in prophylaxis had a power of 12.1% to change bypassing agent’s incremental cost. Other variables influencing the incremental cost were costs associated with emicizumab and bypassing agents. Waste had an impact of only 0.06% on the incremental cost, showing that it is a low-impact variable compared to the others. On-demand doses for treating bleeding events with bypassing agents also had a minimal impact on the incremental cost, and emicizumab prophylaxis remained dominant in all settings.
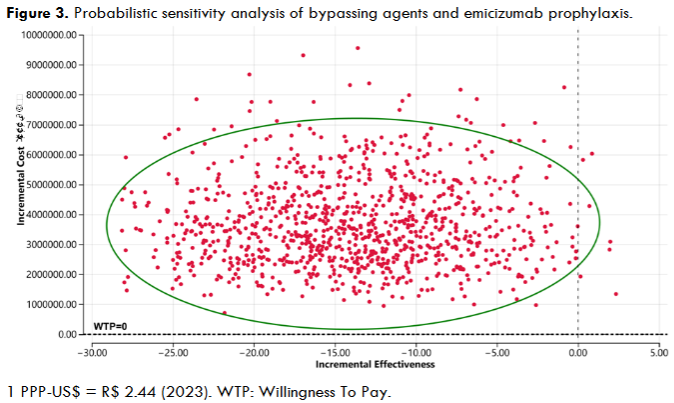
The probabilistic analysis corroborates the findings of the baseline case analysis, showing that the simulated mean cost reduction and increasing incremental effectiveness agree with the primary results. Although some situations favor bypassing agents’ prophylaxis, most favor prophylaxis with emicizumab.
Discussion
This study analyzed the cost-effectiveness of emicizumab prophylaxis compared to bypassing agents prophylaxis for CsHAhri who failed ITI, from the perspective of the Brazilian Ministry of Health, using a decision tree model with a 5-year time horizon. Emicizumab prophylaxis had a significantly lower total cost and ABR per treatment of CsHAhri than BpA prophylaxis in the evaluated setting. The incremental cost-effectiveness ratio (ICER) was PPP-US$170,832.6 per bleeding avoided, showing that emicizumab prophylaxis dominates over bypassing agents prophylaxis. Probabilistic and deterministic sensitivity analyses confirmed the dominance of emicizumab prophylaxis in most of the evaluated settings.
The deterministic sensitivity analysis identified the 10 most influential parameters on the incremental cost. The dose of prophylaxis with rFVIIa and aPCC stands out, accounting for 83.2% of the total risk of incremental cost change. The bypassing agents regimens are variable to maintain a reduced ABR for CsHAhri. This variability in regimens explains the significant influence of bypassing agents prophylaxis doses on modifying the incremental cost. The probability of bypassing agents use was the third parameter with the highest impact on incremental cost, corroborating the results of the effect of bypassing agents regimens and variability presented previously when relating the probability of rFVIIa and aPCC use. Given its regimen’s variability, the impact on incremental cost is higher for using rFVIIa. Regardless of this variability, prophylaxis with emicizumab remained at a lower cost than prophylaxis with bypassing agents. Regarding the unit costs of the products, the impact was more significant for the unit cost of emicizumab and lower for the unit cost of bypassing agents. However, the unit costs remained with a lower impact than the regimens adopted. Thus, although emicizumab has a higher unit cost, the regimens adopted for bypassing agents had a more significant impact on the incremental cost.
Emicizumab prophylaxis is a potential cost-reducing treatment for CsHAhri against bypassing agents prophylaxis in different settings. From the perspective of the Italian National Health Service, for severe haemophilia A with inhibitors and who failed immunotolerance induction at 1 year, emicizumab prophylaxis showed a gain of 0.94 quality-adjusted life years and lowered the cost of treatment by €25,272,190.0 (PPP-US$70,418,430.2) and €19,984,465.0 (PPP-US$55,684,713.3) against rFVIIa and aPCC prophylaxis, respectively. A retrospective observational study conducted in Taiwan investigated and compared the efficacy, safety, and cost before and after prophylaxis with emicizumab in individuals with HA and inhibitors. The median age of participants was 23 years. The study evidenced an ABR drop from 24 to 0 bleedings, a 71% reduction in treatment costs, and no reports of serious adverse events after treatment with emicizumab for 1 year. Pharmacoeconomic models indicate prophylaxis with emicizumab as cost-effective and associated with higher quality-adjusted life-year gains than the use of different procedures.
Emicizumab offers advantages by reducing costs, having subcutaneous administration with the potential for longer intervals between doses, and requiring less stringent storage conditions compared to other products used in the treatment of haemophilia A. Despite the high cost of emicizumab prophylaxis, it is more cost-effective than prophylaxis with bypass agents. The World Federation of Haemophilia supports the use of lower doses for bypass agents and exogenous factor VIII in resource-limited countries, although these are less effective. However, the Federation does not yet endorse lower-dose regimens for emicizumab. Strategies aimed at reducing treatment costs with emicizumab are under investigation. Adjusting therapeutic regimens to utilize lower doses than conventional and approved emicizumab dosing regimens appears promising in reducing the costs of haemophilia A treatment. Countries like India and Thailand have shown favorable results with a reduced dosing schedule, ensuring the full use of the vial in emicizumab prophylaxis. Additionally, adapting regimens to proportional administration based on pharmacokinetics is a strategy that ensures emicizumab’s effectiveness in controlling bleeds. Thus, reducing conventional dosing regimens within acceptable pharmacokinetic and pharmacodynamic parameters, as approved for the treatment of haemophilia A, may impact a reduction in ICER and overall costs.
In this context, reducing emicizumab waste may complement cost-saving strategies in haemophilia A treatment. Waste can account for 0.5% to 38.2% of the reconstituted product, representing considerable values in treatment. The reality and conditions must be assessed to choose the most appropriate action for implementation. Implementing strategies to prevent or reduce emicizumab waste could allow budget reallocation to fund treatment for more individuals with haemophilia A. Examples of waste-reduction alternatives include changes in product presentations and leftovers, as well as vial sharing within healthcare institutions while maintaining product sterility. New technologies may alter existing protocols for haemophilia A treatment, raising discussions about the cost-effectiveness of emicizumab. New non-factor agents are in clinical trial phases and show efficacy in treating haemophilia A and B, with or without inhibitors. Concizumab, a monoclonal antibody targeting the tissue factor pathway inhibitor, has been approved for use in haemophilia B patients with inhibitors. Similarly, marstacimab, with the same mechanism of action as concizumab, is in clinical trials. Fitusiran, an antithrombin inhibitory ribonucleic acid, has shown efficacy and safety results in a phase 3 clinical trial. These products may soon enter discussions regarding the effectiveness and costs of haemophilia A treatment, potentially making bypass agent prophylaxis obsolete.
Finally, individuals with hemophilia generally tend to report a good quality of life; however, this can be adversely affected by low income, limited access to healthcare services, chronic pain, and restricted employment opportunities. Hemophilia is a condition with significant biopsychosocial implications such as the need to promote the social inclusion of individuals living with hemophilia by encouraging access to education, sports, and healthcare services, as well as facilitating their integration into the labor market. It is essential to develop targeted treatment programs based on a multidisciplinary approach, ensure the home delivery of lyophilized clotting factor concentrates, and implement preventive strategies to reduce the incidence of bleeding episodes—all aimed at enhancing the quality of life for people with hemophilia.
Limitations
This study did not consider the association between the wastage rate and the CsHAhri weight variation. Furthermore, since the calculations were based on realities similar to those in Brazil, they may not apply to the reality of haemophilia A treatment in other countries. It is worth noting that the evaluation considered a 5-year follow-up due to the lack of individual long-term data on prophylaxis with emicizumab. Due to the rarity of occurrence, the introduction of haemorrhagic complications, the risk of developing adverse events, including thrombosis, and the development of anti-drug antibodies were not considered. Mortality was not considered due to the low age range for the occurrence of risk of death, given the implementation of prophylaxis.
Although this is not a limitation, we should underscore that the study performed, by definition, estimates values and results of outcomes within the settings considered. A real-world analysis can be conducted to estimate these differences. Real-world studies are underway in the country. The National Registry of People with Haemophilia A Using Emicizumab in Brazil (Emicizumab Cases, EMCase) is a retrospective and prospective cohort study that aims to create a national registry for formal monitoring of patients using emicizumab as prophylaxis against bleeding in haemophilia A (ReBec-UTN: U1111-1249-1876). The results are expected in 2025 and can be used to more reliably estimate the costs and outcomes of emicizumab prophylaxis in Brazil against previous treatments.
Conclusion
The pharmacoeconomic model adopted showed that emicizumab prophylaxis for severe haemophilia A CsHA with high-response inhibitors who failed immunotolerance induction was dominant (reduced costs and bleeding events in any setting), with an incremental cost of PPP-US$2,647,904.8 against haemophilia A prophylaxis from the perspective of the Brazilian Ministry of Health.
Conflicts of Interest Statement
RMC received speaker fees from Bayer, NovoNordisk, Hoffman-La Roche, and Takeda; consultancy fees from Hoffman-La Roche and Takeda, and scientific event grants from Bayer, NovoNordisk, Hoffman-La Roche, and Takeda. MMB, LCMH, AAGJ, FAA, and JAT declare no competing conflict of interest.
Funding Statement
This research did not receive any specific grant from funding agencies in the public, commercial, and not-for-profit sectors.
References
- Srivastava A, Santagostino E, Dougall A et al. WFH Guidelines for the Management of Haemophilia, 3rd edition. Haemophilia.2020; 26(S6):1–158.
- Federação Brasileira de Hemofilia. 2023. Caxias do Sul. FBH. Relatório Anual 2023. Disponível em: https://www.hemofiliabrasil.org.br
- Ljung R, Auerswald G, Benson G et al. Inhibitors in haemophilia A and B: Management of bleeds, inhibitor eradication and strategies for difficult-to-treat patients. Eur J Haematol.2019;102(2):111–122.
- Earnshaw SR, Graham CN, McDade CL, Spears JB, Kessler CM. Factor VIII alloantibody inhibitors: cost analysis of immune tolerance induction vs. prophylaxis and on-demand with bypass treatment. Haemophilia.2015; 21(3):310–319.
- Brasil. Imunotolerância: protocolo de uso de indução de imunotolerância para pacientes com hemofilia A e inibidor. 2015. Ministério da Saúde, Secretaria de atenção à saúde, Departamento de Atenção Especializada e temática.
- Oldenburg J, Young G, Santagostino E, Escuriola Ettingshausen C. The importance of inhibitor eradication in clinically complicated haemophilia A patients. Expert Rev Hematol. 2018;11(11):857–862.
- Oldenburg J, Mahlangu JN, Kim B et al. Emicizumab Prophylaxis in Haemophilia A with Inhibitors. N Engl J Med. 2017; 377(9):809–818.
- Mahlangu J, Oldenburg J, Paz-Priel I et al. Emicizumab Prophylaxis in Patients Who Have Haemophilia A without Inhibitors. N Engl J Med. 2018;379(9):811–822.
- Young G, Liesner R, Chang T et al. A multicenter, open-label phase 3 study of emicizumab prophylaxis in children with haemophilia A with inhibitors. Blood. 2019;134(24):2127–2138.
- Pipe SW, Shima M, Lehle M et al. Efficacy, safety, and pharmacokinetics of emicizumab prophylaxis given every 4 weeks in people with haemophilia A (HAVEN 4): a multicentre, open-label, non-randomised phase 3 study. Lancet Haematol. 2019;6(6):e295–e305.
- Callaghan MU, Negrier C, Paz-Priel I et al. Long-term outcomes with emicizumab prophylaxis for haemophilia A with or without FVIII inhibitors from the HAVEN 1-4 studies. Blood. 2021; 137(16):2231–2242.
- Muniz RL, Camelo RM, Araújo MS et al. Efficacy/effectiveness and safety of emicizumab prophylaxis of people with haemophilia A: a systematic review and meta-analysis. Expert Rev Hematol. 2023;16(12):1087–1097.
- World Federation of Haemophilia. 2024. Report on the Annual Global Survey 2023, https://www1.wfh.org/publications/files/pdf-2525.pdf (Accessed on 14 April 2025)
- Camelo RM, Dias MM, Caram-Deelder C et al. Time between inhibitor detection and start of immune tolerance induction: Association with outcome in the BrazIT study. J Thromb Haemost. 2022;20(11):2526–2537.
- Brasil. CONITEC (Comissão Nacional de Incorporação de Tecnologia). Emicizumabe para tratamento de indivíduos com hemofilia A e inibidores do fator VIII refratários ao tratamento de imunotolerância. 2019. Brasília, Brasil. Ministério da Saúde.
- Camelo RM, Barbosa MM, Araújo MS et al. Economic Evaluation of Immune Tolerance Induction in Children With Severe Haemophilia A and High-Responding Inhibitors: A Cost-Effectiveness Analysis of Prophylaxis With Emicizumab. Value Health Reg Issues. 2023; 34:31–39.
- Camelo RM, Barbosa MM, Henriques LCM et al. Emicizumab prophylaxis for people with haemophilia A: Waste estimation and the Brazilian perspective. Saudi Pharm J. 2023;31(12):101867.
- Tabela 2645: Estimativas populacionais das medianas de altura e peso de crianças, adolescentes e adultos, por sexo, situação do domicílio e idade – Brasil e Grandes Regiões. https://sidra.ibge.gov.br/tabela/2645.
- Hoffmann-La Roche. HEMLIBRA ® (emicizumab injection) HEMLIBRA ® emicizumab injection. 2018.
- Brasil. Ministério da Saúde. Secretaria de Atenção a Saúde. Departamento de Atenção Especializada e Temática. Manual de Hemofilia. 2015. https://bvsms.saude.gov.br/bvs/publicacoes/manual_hemofilia_2 ed.pdf.
- Astermark J, Donfield SM, DiMichele DM et al. A randomized comparison of bypassing agents in haemophilia complicated by an inhibitor: the FEIBA NovoSeven Comparative (FENOC) Study. Blood. 2007;109(2):546–551.
- López-Fernández MF, Roca CA, Álvarez-Román MT et al. Spanish Consensus Guidelines on prophylaxis with bypassing agents in patients with haemophilia and inhibitors. Thromb Haemost. 2016;115(05):872–895.
- Linari S, Castaman G. Concomitant Use of rFVIIa and Emicizumab in People with Haemophilia A with Inhibitors: Current Perspectives and Emerging Clinical Evidence. Ther Clin Risk Manag. 2020;16:461–469.
- Oldenburg J, Shima M, Kruse-Jarres R et al. Outcomes in children with haemophilia A with inhibitors: Results from a noninterventional study. Pediatr Blood Cancer. 2020;67(10).
- Ferreira AA, Brum IV, Souza JV de L, Leite ICG. Cost analysis of haemophilia treatment in a Brazilian public blood center. Cad Saude Colet. 2020;28(4):556–566.
- BPS – Banco de Preços em Saúde. https://bps.saude.gov.br/login.jsf.
- Listas de preços de medicamentos — Agência Nacional de Vigilância Sanitária – Anvisa. https://www.gov.br/anvisa/pt-br/assuntos/medicamentos/cmed/precos.
- OECD (2024), Purchasing power parities (PPP) (indicator). doi: 10.1787/1290ee5a-en (Accessed on 10 April 2024).
- Cortesi PA, Castaman G, Trifirò G et al. Cost-Effectiveness and Budget Impact of Emicizumab Prophylaxis in Haemophilia A Patients with Inhibitors. Thromb Haemost. 2020;120(02):216–228.
- Shen M, Chou S, Chiou S et al. Efficacy, safety and cost of emicizumab prophylaxis in haemophilia A patients with inhibitors: A nationwide observational study in Taiwan. Haemophilia. 2023;29(6):1499–1508.
- Bukkems LH, Fischer K, Kremer-Hovinga I et al. Emicizumab Dosing in Children and Adults with Haemophilia A: Simulating a User-Friendly and Cost-Efficient Regimen. Thromb Haemost. 2022;122(02):208–215.
- Mannucci PM, Hermans C. Low-dose emicizumab for more equitable access to prophylaxis in resource limited countries. Haemophilia. 2024;30(3):575–576.
- Chuansumrit A, Sirachainan N, Jaovisidha S et al. Effectiveness of monthly low dose emicizumab prophylaxis without 4-week loading doses among patients with haemophilia A with and without inhibitors: A case series report. Haemophilia. 2023;29(1):382–385.
- Bansal S, Donners AAMT, Fischer K et al. Low dose emicizumab prophylaxis in haemophilia A patients: A pilot study from India. Haemophilia. 2023;29(3):931–934.
- Srivastava A, Iorio A. Lower-dose emicizumab prophylaxis: can less be more? J Thromb Haemost. 2024;22(4):922–925.
- Liu G, Huang K, Li G et al. Real-world experience of emicizumab prophylaxis in young children with haemophilia A: retrospective data from China. Front Pediatr. 2022 Oct 19;10:992267. doi: 10.3389/fped.2022.992267.
- Donners AAMT, van der Zwet K, Rademaker CMA, Egberts TCG, Schutgens REG, Fischer K. The efficacy of the entire-vial dosing of emicizumab: Real-world evidence on plasma concentrations, bleeds, and drug waste. Res Pract Thromb Haemost. 2023;7(2):100074.
- Hatswell AJ, Porter JK. Reducing Drug Wastage in Pharmaceuticals Dosed by Weight or Body Surface Areas by Optimising Vial Sizes. Appl Health Econ Health Policy. 2019;17(3):391–397.
- Damuzzo V, Russi A, Chiumente M et al. Optimization of resources by drug management: A multicentred web-administered study on the use of ipilimumab in Italy. J Oncol Pharm Pract. 2019;25(4):787–792.
- Ripoll Gallardo A, Meneghetti G, Ragazzoni L et al. Multiple withdrawals from single-use vials: A study on sterility. Int J Pharm. 2015;485(1–2):160–163.
- Matsushita T, Shapiro A, Abraham A et al. Phase 3 Trial of Concizumab in Haemophilia with Inhibitors. N Engl J Med. 2023;389(9):783–794.
- Mahlangu J, Luis Lamas J, Cristobal Morales J et al. Long-term safety and efficacy of the anti-tissue factor pathway inhibitor marstacimab in participants with severe haemophilia: Phase II study results. Br J Haematol. 2023;200(2):240–248.
- Young G, Srivastava A, Kavakli K et al. Efficacy and safety of fitusiran prophylaxis in people with haemophilia A or haemophilia B with inhibitors (ATLAS-INH): a multicentre, open-label, randomised phase 3 trial. The Lancet. 2023;401(10386):1427–1437.
- Nunes AA, Rodrigues BSC, Soares EM, Soares S, Miranzi SSC. Qualidade de vida de pacientes hemofílicos acompanhados em ambulatório de hematologia. Rev. Bras. Hematol. Hemoter. 2009;31 (6). https://doi.org/10.1590/S1516-84842009005000085 (Accessed on 14 April 2025).
- Jiménez-Yuste V, Rodríguez-Merchán EC, Matsushita T, Holme PA. Concomitant use of bypassing agents with emicizumab for people with haemophilia A and inhibitors undergoing surgery. Haemophilia. 2021;27(4):519–530.

