Biomarker Testing Inequities in Lung Cancer in Ghana
Inequities in Biomarker Testing for Lung Cancer in a Low Resource Setting
Jane Sandra Afriyie-Mensah1,2, Mary-Ann Dadzie3, Andrew Yaw Nyantakyi3,4, Charles Domotey5
- Jane Sandra Afriyie-Mensah Department of Medicine and Therapeutics, Korle Bu Teaching Hospital, Accra, Ghana; Department of Internal Medicine, University of Ghana, Legon, Accra, Ghana
- Mary-Ann Dadzie National Centre for Radiotherapy, Oncology and Nuclear Medicine, Korle Bu Teaching Hospital, Accra, Ghana http://orcid.org/0000-0003-4800-4724
- Andrew Yaw NyantakyiNational Centre for Radiotherapy, Oncology and Nuclear Medicine, Korle Bu Teaching Hospital, Accra, Ghana; Department of Oncology, Cape Coast Teaching Hospital, Cape Coast, Ghanahttp://orcid.org/00000003-0742-6007
- Charles Domotey Korle Bu Teaching Hospital, Accra Ghana
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION: AFRIYIE-MENSAH, Jane Sandra et al. Inequities in Biomarker Testing for Lung Cancer in a Low Resource Setting. Medical Research Archives, [S.l.], v. 13, n. 3, mar. 2025. Available at: <https://esmed.org/MRA/mra/article/view/6300>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i3.6300
ISSN 2375-1924
Abstract
Introduction: Despite the novel advances in molecular biology which has revolutionized treatment of Non-Small Cell Lung cancers, biomarker testing remains limited in low resource settings including Ghana making the adoption of personalized treatment pathways difficult.
Methodology: This was a retrospective review of histology-confirmed lung cancer cases from January 2019 to December 2023. Data extracted from patients’ medical records were analyzed using descriptive statistics.
Aim: To describe the proportion and pattern of biomarker testing requested by clinicians involved in diagnosing and managing lung cancer patients.
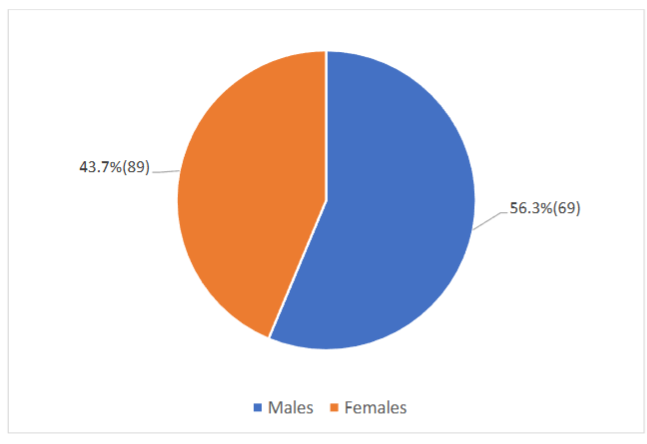
Results: A total of 158 medical records cases were retrieved of which male and female proportions were 89(56.3%) and 69(43.7%) respectively. Non-small cell lung cancer constituted 127 (80.4%) all cases. Out of the total number of non-small cell lung cancer cases, 117(92.1%) had adenocarcinoma, 9(7.1%) had squamous cell carcinoma and 1(0.8%) had Adenosquamous carcinoma. Only 22 (17%) of the non-small cell lung cancer cases had biomarker analysis. A total of 49 biomarker reports were retrieved from the 22 patients and the variety as well as the proportions of the assays were as follows; Programmed Death-Ligand-1(PDL1) were 13/49(26.5%), Epidermal Growth Factor Receptor (EGFR) were 12/49(24.5%), Anaplastic Lymphoma Kinase (ALK) were 12/49(24.5%), Kirsten rat sarcoma viral oncogene homolog (KRAS) and ROS proto-oncogene 1 (ROS1) were 6 each representing 1.2% of all cases. The results show that 11/22(50%) of these patients had two different biomarker reports whiles only 1 patient had all 5 biomarker test reports.
Discussion: The findings of this study reflect a low level of biomarker profiling of Non-Small Cell Lung cancer cases among our cohort of patients. This certainly has significant implications on lung cancer management and prognosis in our environment.
Conclusion: There is an urgent need to address the gaps in biomarker profiling of non-small cell lung cancers diagnosed in our setting. This may be achieved through continuous medical education of treating clinicians, improved molecular testing capacity and advocating for governmental support policies.
Keywords
Lung cancer, Non-Small Cell Lung cancers, biomarker analysis
Introduction
Lung cancer is the most common cause of cancer-related deaths worldwide, with approximately 1.8 million deaths recorded in the year 2020. The prognosis of lung cancer is significantly influenced by the stage of the disease at the time of diagnosis. Since the majority of patients are diagnosed in advanced stages and effective treatment options often limited, lung cancer remains the disease with the poorest prognosis worldwide, with a 5-year survival rate below 20% in most countries.
Lung cancers are traditionally classified into two main histologic groups; Small Cell Lung Cancer [SCLC] and Non-Small Cell Lung Cancer [NSCLC] based on the differences in morphological appearance. NSCLC constitutes approximately 85% of all lung cancers, and its most common subtypes are adenocarcinoma, squamous cell carcinoma and large cell carcinoma. Research has shown that these subtypes are not only morphologically different but also have different molecular characteristics which are possible therapeutic targets. In approximately 60% and 20% of adenocarcinomas and squamous cell carcinomas respectively, these defined genetic changes has paved way for the successful development of treatments that targets these molecules appropriately. In NSCLC, key molecular alterations include mutations such as Epidermal Growth Factor Receptor (EGFR), B-Raf proto-oncogene, serine/threonine kinase, valine-to-glutamic acid mutation at residue 600 (BRAFV600E), Kirsten Rat Sarcoma viral oncogene homolog (KRASG12C) and Erb-B2 Receptor Tyrosine Kinase 2 (ERBB2 (HER2)), fusions such as Anaplastic Lymphoma Kinase (ALK), ROS proto-oncogene 1 (ROS1), Neurotrophic Tyrosine Receptor Kinase genes 1/2/3 (NTRK 1/2/3) and “Rearranged during Transfection” (RET) as well as Mesenchymal Epithelial Transition (MET) exon-14 skipping mutations. Molecular understanding is continually revolving and has further advanced the paradigm of “personalized cancer treatment” with novel agents that selectively target a specific tumoral molecular alteration.
There is overwhelming evidence that these agents with better safety profiles compared to conventional chemotherapy have impacted the quality of life and overall survival of lung cancer patients across all stages. In 2021, the American Lung association documented an increase in 5-year survival from 14.5% to 23.7% for lung cancer patients in the United States due to incorporation of novel targeted therapies. Thus, in-depth molecular profiling in NSCLC is not only considered a standard practice at initial diagnosis per international guidelines, but also fully explored at the point of disease progression to identify potential resistant genes or actionable mutations for treatment adjustments.
While mandatory molecular profiling of these cancers is being practiced on a large scale in the western world, the situation is different in most parts of Africa where there is significant underdiagnoses, inadequate management and limited data on use of targeted therapies.
It has been shown that key biomarkers do not typically overlap and most patients have only one actionable mutation. With continuous discovery of new biomarkers, it is worth noting that there are variations in the prevalence of these markers across geographical locations, hence the need for molecular profiling of tumors in various jurisdictions. In Ghana, although there is lack of national epidemiological data, a recent review in a tertiary facility documented a modest increase in lung cancer cases with over 80% being adenocarcinomas and majority reporting with Stage IV disease. There is also lack of data on the prevalence of molecular markers among NSCLC patients in Ghana. The aim of this study was to determine the proportion of NSCLC patients with biomarker testing and the pattern of tests requested by treating clinicians at a tertiary facility in Ghana.
Methodology
This was a retrospective study conducted at the Korle-Bu Teaching Hospital in Accra, Ghana between the periods of January 2019 to December 2023. Medical records of patients with histologically confirmed primary lung cancer were retrieved from the database of the respiratory department, cardiothoracic center, and the pathology lab within the stipulated period. Demographic and disease related variables which included histological type and subtype of lung cancer as well as biomarker test reports were retrieved and transferred unto an Excel spreadsheet. Data obtained was cleaned and analyzed using the Statistical Package for Social Sciences (SPSS) version 22. Descriptive statistics, including frequency distributions, percentages, pie charts and tables were used to present categorical data using Excel 2024 version. Medical records of suspected cases without histological confirmation were excluded from the study. Ethical approval was obtained from the Ethical and Protocol Review Committee of the College of Health Sciences, University of Ghana.
Table 1: Age distribution of participants
| Age (years) | Frequency | Percentage (%) |
|---|---|---|
| 20-29 | 6 | 3.8 |
| 30-39 | 15 | 9.5 |
| 40-49 | 24 | 15.2 |
| 50-59 | 33 | 20.9 |
| 60-69 | 40 | 25.3 |
| 70-79 | 35 | 22.2 |
| 80 and above | 5 | 3.2 |
| Total | 158 | 100 |
Results
A total of 158 cases were included in the analysis. The mean age was 57.3 years (SD 14.9) with a peak age group of 60 to 69 years. Approximately 20(13%) of the cases were below the age of 40 years.
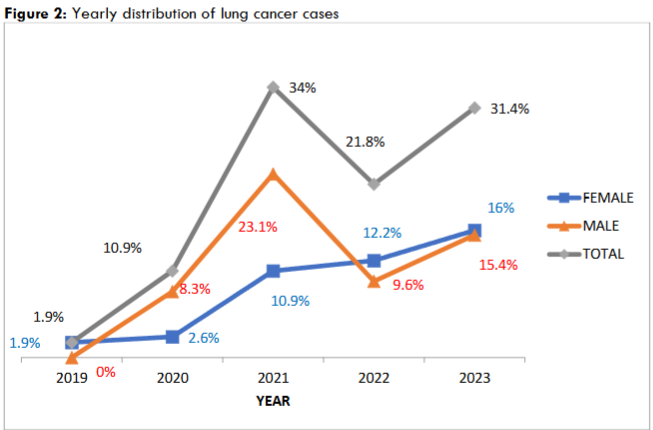
Figure 2 shows the yearly distribution of lung cancer cases diagnosed. The number of cases diagnosed per year (grey line) as well as the proportion of cases that are males (orange) and female (blue) is depicted in this figure. Generally, there was a steady rise in the number of lung cancer cases seen yearly with the highest peak noted in the year 2021. Although, there was a dip in the number of male cases diagnosed in 2022 that of the females rather showed a steady rise, exceeding the number males diagnosed.
Table 2: Types and subtypes of lung cancer diagnosed among study participants.
| HISTOLOGY | FREQUENCY | PERCENTAGE (%) |
|---|---|---|
| NSCLC | ||
| Adenocarcinoma | 127 | 80.4% |
| Squamous cell carcinoma | 9 | |
| Adenosquamous carcinoma | 1 | |
| Bronchial carcinoid cancer | 20 | 12.7 |
| Undifferentiated neuroendocrine cancer | 3 | 1.9 |
| Primary Pulmonary Lymphoma | 2 | 1.3 |
| High Grade Bronchial Dysplasia | 2 | 1.3 |
| Primary Pulmonary Plasmacytoma | 1 | 0.6 |
| Primary Pulmonary Choriocarcinoma | 1 | 0.6 |
| Solitary Fibrous Tumour | 1 | 0.6 |
| Malignant Fibromyxoid Neoplasm | 1 | 0.6 |
| Total | 158 | 100 |
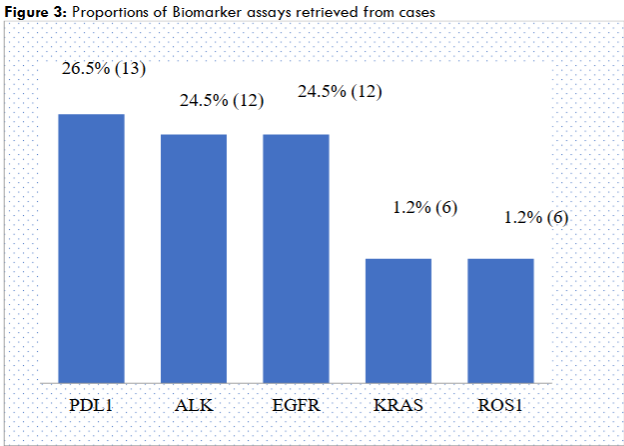
Figure 3 displays the variety of biomarker analysis performed and respective proportions. A total of 49 test reports were obtained from the 22 cases who had biomarker assays done. The five specific tests reported on were PDL1, EGFR, ALK, KRAS and ROS1. The most predominant biomarker reported was PDL1 13(26.5%) followed by 12 (24.5%) reports each for EGFR and ALK mutations.
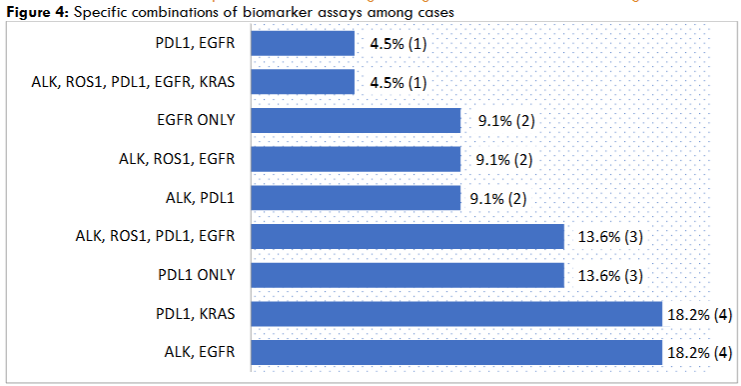
Table 4 shows the combinations of biomarker tests requested per individual patient. The result indicates that majority 11(50%) of the cases performed two biomarker tests. These included 4(18.2%) combining EGFR and ALK mutation assays, 4(18.2%) with PDL1 and KRAS assays, 2(9.1%) with PDL1 and ALK and finally 1(4.5%) with PDL1 and EGFR. Three (13.6%) patients had only PDL1 test and 2 (9.1%) had only EGFR mutation assays. A combination of ALK, ROS and EGFR was reported in 2(9.1%) cases, ALK, ROS1, PDL1 and EGFR in 3(13.6%) cases and only one patient had all 5 biomarker assays done.
Discussion
Lung cancer statistics in low to middle-income countries are believed to be underestimated due to significant diagnostic challenges and misdiagnosis as Pulmonary Tuberculosis which tends to be endemic in these regions. In spite of these challenges, especially in sub-Saharan Africa, recent reports on lung cancer cases could indirectly signal increasing prevalence as well as growing awareness of the disease. In Ghana, GLOBOCAN statistics in 2020 ranked lung cancer as the 8th most prevalent cancer, being 2.2% of all new cancer cases and among the top 10 causes of cancer mortality. As previously reported by Afriyie-Mensah et al, the current study similarly shows a steady rise in the diagnosis of lung cancer in both men and women from the year 2019 with a significant peak in the year 2021. The observed peak could be attributed to the COVID-19 pandemic which increased the rate of chest imaging among patients with respiratory symptoms, ultimately leading to increased detection of incidental lung masses. A total of 158 cases were diagnosed in the tertiary facility over the study period and this was higher than the number reported in other institutional reviews in Ethiopia and Ivory Coast over similar periods. In the current study, males were in the majority as described in literature, but it is worth noting the steady rise of lung cancer diagnoses among the females, surpassing their male counterparts in the years 2022 and 2023. The observation similarly reflects a worrying global trend of increasing lung cancer diagnosis in women, notably among female never-smokers. The typical age of lung cancer diagnosis has been shown to be 65 years and above, with an average age of 70 years. However, in our study, we found that just about half of the patients (50.6%) were at least 60 years implying a relatively younger age of diagnosis. In contrast to data from the National Cancer Institute in the US in which lung cancer diagnosis below age 45 was about 5.2%, we observed that 20% of our cohort were below age 45 years. This could however be due to the significant proportion of cases diagnosed with Bronchial carcinoids in our cohort. The usual age range of onset of bronchial carcinoids in adults is between 40 – 50 years with a mean of 42 years reported in a study by Martini et al.
Traditionally, NSCLCs account for about 80% of all lung cancers, and this was similarly observed in the current review. Adenocarcinoma is known to be the commonest histological subtype of NSCLCs constituting about 40 – 50% but our proportion of adenocarcinomas was significantly higher, accounting for 74% of all the NSCLCs diagnosed. Interestingly, there was no record of SCLC among our cases which aligns with the low smoking rates among lung cancer cases diagnosed in Ghana. This mirrors the trend of declining estimates of SCLC attributed to decreases in smoking habits worldwide.
Management practices in low resource setting are largely lagging behind international standards for surgical and systemic (specifically targeted) therapies for both resectable and advanced lung cancer. Although lung cancer has the poorest cancer prognosis, the gloomy picture is gradually fading due to the rapid development of novel molecular therapies targeting specific tumor mutations and fusions as well as immunotherapeutic agents that promote tumor cell destruction by activation of the immune system. The effectiveness of these novel therapies with regards to improved progression free survival and better adverse effect profile has led to their recommendation in numerous lung cancer management guidelines. Employing such therapies depends on the histological detection of these specific mutations and proteins in the obtained biopsy specimen.
The landscape of lung cancer management in Ghana is yet to shift appreciably toward personalized care although the local lung multi-disciplinary team (MDT) recommends biomarker profiling for all patients with NSCLC. The study results however show a very low level of molecular testing with only 17% of the NSCLC population having tested for tumor biomarkers. We observed that about half of these patients had reports for two different biomarker assays with predominant combinations being both EGFR and ALK and PDL1 and KRAS. It is however unclear what informed the choices of these combinations. This observation is similar to reports in other African studies where molecular profiling is either performed in only a small fraction of patients or not done at all. These studies provide proof of the low uptake of current biomarker profiling in the sub-region compared to high resource settings. The reasons for this trend in Ghana are multiple and quite challenging. Testing for these biomarkers is not available in public health facilities and only available in a limited number of privately-run pathology laboratories. Currently, the cost of assay for all 5 molecular markers noted in the study (ALK, EGFR, ROS 1, PD1 and KRAS) stands at about $500 equivalent and this is not affordable to the ordinary Ghanaian, many of whom pay out of pocket for their health needs. Immunotherapies and targeted medications available in Ghana include Atezolizumab and Pembrolizumab for PDL-1, Elortinib and Osimertinib for EGFR mutations, Alectinib for ALK mutations and Crizotinib for ALK and ROS 1 fusion. None of these expensive medications is reimbursed by the National health Insurance Scheme in its current form. Thus, some clinicians find it futile and unethical to request for expensive biomarker profiling when it is obvious to them patients may likely not be able to afford the medications. Crizotinib has been made accessible in Ghana through the MAX FOUNDATION to patients at no cost and Elortinib is available in the generic brand at a price of approximately $200 equivalent for a month’s supply. It is therefore not surprising that EGFR and ALK biomarkers were among the frequent biomarker reports retrieved in this review although few. It is clear from these findings that improved financial accessibility to medications could be a catalyst to boost biomarker testing rates among patients.
We observed with interest the predominance of PDL1 testing among the cohort of this study although the medication is exorbitant and not immediately available in-country. Since most of our patients have advanced disease, this could represent increasing knowledge on novel immunotherapies in advanced lung cancers among treating physicians. Since a number of the study patients were able to afford one or two tests, could requesting for a biomarkers with ready access to medications been more beneficial to patient rather than a PDL1 test? In recent years, there has been a surge in the volume of scientific publications on the integration of immune therapy in the treatment of lung cancer, highlighting its significant survival benefits. Anti PDL1 monotherapy or in combination with other treatments has become the standard treatment regimen for advanced lung cancer. With the current prohibitive cost of biomarker testing as well as the expanding molecular panel, it is important to know the prevalence of specific tumor mutations in our population of NSCLC to direct relevant biomarker testing and minimize cost effects on patients.
Conclusion
Lung cancer management needs to gradually shift towards personalized medicine even in low resourced setting such as ours. The cost of biomarker testing is a major hindrance to the implementation of recommended treatment guidelines. Immediate efforts must be made through research to determine the molecular profile of NSCLC in Ghana as knowledge of this will help narrow the scope of biomarkers assays to those that are common in our environment thereby reducing cost of testing. Deliberations with relevant stakeholders such as pharma industry and governmental health agencies will be a necessary action to equip testing capacity of public pathology laboratories.
Limitations
The study was retrospective and relied on manually archived medical records. We, therefore, encountered challenges such as missing patient information. Secondly, due to the low numbers with biomarker assays, the study cannot define the most common genetic mutation among the NSCLC cases.
Disclosure
The authors declare no conflict of interest.
References
- Cao W, Chen HD, Yu YW, Li N, Chen WQ. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Chin Med J 2021; 7:783–791.
- Allemani C, Matsuda T, Di Carlo V, et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37,513,025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018; 391(10125): 1023-1075. doi:10.1016/s0140-6736(17)33326-3.
- Soerjomataram I, Cabasag C, Bardot A, et al. Cancer survival in Africa, Central and South America, and Asia (SURVCAN-3): a population-based benchmarking study in 32 countries. Lancet Oncol. 2023;24(1):22-32. doi:10.1016/s1470-2045(22)00704-5.
- Forde PM, Ettinger DS. Targeted therapy for non-small-cell lung cancer: Past, present and future. Expert Rev Anticancer Ther 2013;13:745-58.
- Van Christ Manirakiza, A., Rubagumya, F., Rugengamanzi, E, et al. Trends of Molecular Testing for Lung Cancer at the King Faisal Hospital, Kigali: Therapeutic and Survival Implications. JTO clinical and research reports, 3(4),100304. https://doi.org/10.1016/j.jtocrr.2022.100304.
- Thunnissen E, van der Oord K, den Bakker M. Prognostic and predictive biomarkers in lung cancer. A review. Virchows Arch 2014;464:347 58.
- Mina SA, Shanshal M, Leventakos K, Parikh K. Emerging Targeted Therapies in Non-Small-Cell Lung Cancer (NSCLC). Cancers (Basel). 2025 Jan 22;17(3):353. doi: 10.3390/cancers17030353. PMID: 39941723.
- Shen FF, Guo W, Tian RF, et al. Long-term survival with targeted therapy in an advanced non-small cell lung cancer patient based on genetic profiling. Transl Lung Cancer Res. 2020 Apr;9(2):373-378. doi: 10.21037/tlcr.2020.01.21. PMID: 32420078; PMCID: PMC7225131.
- Musika W, Kamsa-Ard S, Jirapornkul C, et al. Lung Cancer Survival with Current Therapies and New Targeted Treatments: Comprehensive Update from the Srinagarind Hospital-Based Cancer Registry from (2013 to 2017). Asian Pac J Cancer Prev. 2021 Aug 1;22(8):2501-2507.
- American Lung Association; State of Lung cancer 2021 Report: https://www.lung.org/getmedia/ba972351-ddc5-46b2-8e0d-028002d16c72/solc-2021-print-report-final.pdf.
- The European Society for Medical Oncology (ESMO). (2021). Early and locally advanced non-small-cell lung cancer: an update of the ESMO Clinical Practice Guidelines focusing on diagnosis, staging and systemic and local therapy. https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guidelines-lung-and-chest-tumours/early-stage-and-locally-advanced-non-metastatic-non-small-cell-lung-cancer-esmo-clinical-practice-guidelines.
- The National Comprehensive Cancer Network (NCCN). (2024). Non-Small Cell Lung Cancer (Version 7). https://www.nccn.org/login?ReturnURL=https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf.
- American Society of Clinical Oncology (ASCO). (2022). Adjuvant Chemotherapy and Adjuvant Radiation Therapy for Stages I-IIIA Resectable Non-Small Cell Lung Cancer. https://society.asco.org/practice-patients/guidelines/thoracic-cancer#/10226.
- American Society of Clinical Oncology (ASCO). (2024). Management of Stage III Non-Small Cell Lung Cancer. https://society.asco.org/practice-patients/guidelines/thoracic-cancer#/168762.
- Lubuzo B, Ginindza T, Hlongwana K. The barriers to initiating lung cancer care in low- and middle-income countries. Pam Afr Med J. 2020; 35:38.
- Said, N S, Degu, A. Assessment of Survival outcomes among lung cancer patients at the National and Referral Hospital in Kenya. Cancer Med. 2023 Jan 27;12(8):9194-9201.
- Van Christ Manirakiza A, Rubagumya F, Rugengamanzi E, et al. Trends of Molecular Testing for Lung Cancer at the King Faisal Hospital, Kigali: Therapeutic and Survival Implications. JTO Clin Ress Rep. 2022 Mar 1;3(4)100304.
- Slavik T, Asselah F, Fakhruddin N, et al. Diagnosis and Predictive Molecular Analysis of Non-Small-Cell Lung Cancer in the Africa-Middle-East Region: Challenges and Strategies for Improvement. Clinical Lung Cancer. 15(6):398-404.
- Mosele F, Remon J, Mateo J, et al. Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: a report from the ESMO Precision Medicine Working Group. Ann Oncol. 2020;31(11):1491–505.
- Batra U, Prabhash K, Noronha V, et al. Prevalence of EGFR Mutations in Patients with Resected Stage I to III Non-Small Cell Lung Cancer: Results of India Cohort. JCO Glob Oncol. 2025 Jan; 11:e2400353.
- Zhang Y-L, Yuan J-Q, Wang K-F, et al: The prevalence of EGFR mutation in patients with non-small cell lung cancer: A systematic review and meta-analysis. Oncotarget 7:78985-78993,2016.
- Liao BC, Chiang NJ, Chang GC, et al. Registry of Genetic Alterations of Taiwan Non-Small Cell Lung Cancer by Comprehensive Next-Generation Sequencing: A Real-World Cohort Study-Taiwan Cooperative Oncology Group T1521. JCO Glob Oncol. 2024 Sep;10:e2400125.
- Afriyie-Mensah, J. S., Kwarteng, E., Tetteh, J., et al. A three-year review of lung cancer patient characteristics in a tertiary hospital. Ghana medical journal, 57(3), 167–174. https://doi.org/10.4314/gmj.v57i3.2.
- Okonta KE, Echieh CP, Abubakar U, et al. Management of lung cancer in Africa: Underdiagnosis and poor access to treatment – A close look at Nigeria and West African Sub-region. J Pan Afr Thorac Soc 2021;2:122-9.
- Mbeje NP, Ginindza T, Jafta, N. Epidemiological Study of Risk factors for Lung Cancer in KwaZulu-Nata, South Africa. Int. J. Environ. Res. Public Health 2022, 19(11), 6752.
- Gebremariam TH, Haisch DA, Fernandes H, et al. Clinical Characteristics and Molecular Profiles of Lung Cancer in Ethiopia. JTO Clin Res Rep 2021 May 31; 2(7):100196.
- Moctar T, Kouassi KKY, Narcisse SE, et al. Non-Small Cell Lung Cancer: Therapeutic and Evolutionary Aspects in Cote d’Ivoire. Advances in Lung Cancer. 7(1):1-8.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021 Feb 4. doi: 10.3322/caac.21660. Epub ahead of print. PMID: 33538338.
- Mederos N, Friedlaender A, Peters S, Addeo A. Gender-specific aspects of epidemiology, molecular genetics and outcome: lung cancer. ESMO open 2020; 5: e000796.
- Lung Cancer Statistics. How Common is Lung Cancer? Available at https://www.cancer.org/cancer/lung-cancer/about/key-statistics.html. Accessed 17th February, 2025.
- National cancer Institute. Age and cancer risk. Available at https://www.cancer.gov/about-cancer/causes-prevention/risk/age. Accessed: 17th February, 2025.
- Martini N, Zaman MB, Bains MS, et al. Treatment and prognosis in bronchial carcinoids involving regional lymph nodes. The Journal of Thoracic and Cardiovascular Surgery, 107(1):1-7.
- Walker S. Updates in non-small cell lung cancer. Clin J Oncol Nurs. 2008; 12:587-96.
- Govindan R, Page N, Morgenszten D et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol. 2006 Oct 1; 24(28):4539-44.
- Mamdani H, Mastosevic S, Khalid AB, et al. Immunotherapy in Lung cancer: Current Landscape and Future Directions. Front Immunol. 2022 Feb 9;13:823618.
- Olatunji E, Patel S, Graef K, et al. Utilization of cancer immunotherapy in sub-Saharan Africa. Front. Oncol. 13:1266514. doi: 10.3389/fonc.2023.1266514.
- Spigel DR, Faivre-Finn C, Gray JE, et al. Five-Year Survival Outcomes with Durvalumab After Chemoradiotherapy in Unresectable Stage III NSCLC: An Update from the PACIFIC Trial. J Clin Oncol (2021) 39:(15_suppl):8511.
- Felip E, Altorki N, Zhou C, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB–IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. The Lancet. 2021 Oct 9;398(10308):1344-57.
- Reck M, Rodríguez-Abreu D, Robinson AG, et al. Five-year outcomes with pembrolizumab versus chemotherapy for metastatic non–small-cell lung cancer with PD-L1 tumor proportion score≥ 50%. Journal of Clinical Oncology. 2021 Jul 20;39(21):2339-49.
- Borghaei H, Gettinger S, Vokes EE, et al. Five-year outcomes from the randomized, phase III trials checkmate 017 and 057: nivolumab versus docetaxel in previously treated non–small-cell lung cancer. Journal of Clinical Oncology. 2021 Mar 1:39(7):723-33.




