Coronary Calcification and Cognitive Decline in ELSA-Brasil
CORONARY AND AORTIC CALCIFICATION AND COGNITIVE PERFORMANCE CHANGE IN ELSA-BRASIL: A LONGITUDINAL STUDY
Igor Vasconcelos Barros Cronemberg1, Jesiane Ferreira Pedrosa1, Daniel Bos2, Luana Giatti Gonçalves3, Sandro Maria Barreto1
- Postgraduate program in Public Health, Universidade Federal de Minas Gerais, Brazil
- Medical School and Clinical Hospital/ EBSERH, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil
- Department of Epidemiology, Erasmus MC, Rotterdam, The Netherlands
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION:Cronemberger, IVB., et al., 2025.CORONARY AND AORTIC CALCIFICATION AND COGNITIVE PERFORMANCE CHANGE IN ELSA-BRASIL: A LONGITUDINAL STUDY. Medical Research Archives, [online] 13(3).https://doi.org/10.18103/mra.v13i3.6314
COPYRIGHT:© 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6314
ISSN
2375-1924
ABSTRACT
Backgrounds and aims:
Evidence of an association between arterial calcification and worse cognitive performance comes, for the most part, from cross-sectional studies focusing solely on coronary arteries and, even so, with some conflicting results. We sought to investigate whether the presence and severity of calcification in the coronary artery (CAC), total thoracic aorta (TAC) and its segments [ascending thoracic aorta (ATAC), aortic arch (AAC) and descending thoracic aorta (DTAC)] are longitudinally associated with changes in cognitive performance at 4 years of follow-up in Brazilian adults from the ELSA-Brasil cohort, independently of major cardiovascular risk factors.
Methods:
Our study included 1,331 individuals (mean age 62.4 years) from the ELSA-Brasil cohort in the state of Minas Gerais who participated in two subsequent waves (2012-2014 and 2017-2019). Measurements of arterial calcification (CAC, TAC, ATAC, DTAC) were obtained by computed tomography during our study baseline and analyzed dichotomously (present or absent) and in levels (0, 1-399UA and ≥ 400UA); cognitive performance was evaluated in tests (trail B test, verbal fluency, memory test and G-factor) applied in baseline and follow-up visit. Sociodemographic parameters (gender, education, race/color), behavioral (smoking and leisure-time physical activity) and clinical parameters (hypertension, diabetes and body mass index) were ascertained through face-to-face interviews at baseline. For the statistical analysis, linear regression models with mixed effects were used.
Results:
In the dichotomous analyses of calcification, only the presence of any DTAC was associated with worse cognitive performance at baseline. When levels of calcification were considered, higher values (≥ 400UA) of CAC, TAC and AAC were also associated with worse cognitive performance either in trail B test or G-factor at baseline. In regard to DTAC, not only both medium (1-399UA) and higher values were associated with worse cognitive performance at baseline in several cognitive tests at baseline, but higher DTAC was found to be related to an accelerated decline in performances in memory tests and G-factor.
Conclusion:
Although higher levels of CAC and total and segmental thoracic aorta calcification were both independently associated with worse cognitive performance in this cohort of Brazilian adults, only higher DTAC was implied in a decline in cognitive performance over approximately 4 year of follow-up.
Introduction
One of the hallmarks of atherosclerosis is calcification, which can be easily quantified using computed tomography (CT) scans by Agatston score.¹ A high coronary arteries calcium (CAC) score is a well-established independent marker of risk of cardiovascular events, cardiac mortality, and all cause-mortality.² Calcification in the thoracic aorta (TAC), on the other hand, has not undoubtedly demonstrated prognostic value for cardiac events, independent of risk factors and CAC,³ although it seems to be associated with non-cardiovascular disease-related mortality, probably through a pathway that is unrelated to atherosclerosis per se (most likely, through inflammation).⁴ It has been shown, however, that calcification in each of the thoracic aorta segments (ascending thoracic aorta, aortic arch and descending thoracic aorta) in separate presents peculiarities, as it seems to be related to different risk factors⁵ and to different clinical outcomes as well.⁶ ⁷
In recent years, there has been a growing interest in verifying whether arterial calcification in different vessel beds also relates to cognitive function and dementia, since atherosclerosis and cognitive decline are both chronic subclinical processes that appear to share many risk factors.⁸ ⁹ Most studies on this topic focused on the burden of calcification in the coronary arteries and its association to cognitive function and/or risk of dementia in cross-sectional analyses.¹¹ ¹⁷ In general, even though positive associations between a greater severity of CAC and cognitive function have been found, in some studies CAC and cognitive function were only weakly correlated.¹¹ ¹⁷ Beyond that, the associations between CAC and worse cognitive function do not appear to be strictly related to the same cognitive domains in the different studies that have been carried out. Moreover, when associations between CAC and risk of or diagnosis of dementia were prospectively evaluated, results were somehow controversial.¹⁸ ¹⁹
In that sense, associations between CAC and cognitive function over the years should be more extensively investigated. Additionally, seeking whether calcification in other vessel beds can more reliably be considered a risk factor for cognitive decline is imperious, as this could aid towards revealing strategies of early intervention against cognitive impairment and dementia.
To our knowledge, to this date, there are no studies on the possible association between thoracic artery calcification and cognition. As evidence supports, for instance, that increasing TAC scores increases by themselves the risk of conversion of CAC=0 to CAC>0 over the years, it is not unreasonable to question if TAC might also be associated with other atherosclerotic mediated process, including longitudinal changes in cognition.²⁰
Thus, the present study aims to prospectively evaluate whether the presence of any calcification and/or the severity of arterial calcification in coronary arteries and total thoracic aorta artery, ascending thoracic aorta artery, aortic arch and/or descending thoracic aorta artery are associated, independently of traditional cardiovascular risk factors, with changes in performance in three cognitive function tests in about 4-year follow-up of a sample of middle-aged adults and elderly participants of the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil).
Materials and methods
STUDY POPULATION
The ELSA-Brasil study is a large multicenter cohort started in 2008–2010 with 15,105 active or retired civil servants (35–73 years old) from higher education and research institutions in 6 Brazilian capitals (São Paulo, Belo Horizonte, Porto Alegre, Salvador, Rio de Janeiro and Vitória). The institutional review boards of all sites approved the study and all participants signed informed consent. To this date, the ELSA-Brasil cohort has completed three visits of interviews and examinations. Detailed information on ELSA-Brasil study can be found elsewhere.²¹ ²²
The present study used data obtained exclusively in the ELSA-Brasil research center of Minas Gerais at visits 2 and 3, as only participants from this center
underwent CT scans of the coronary arteries and thoracic aorta. Because CT scans were performed only at visit 2 (2012–2014), this visit serves as the baseline of the present study. The follow-up (visit 3) occurred between 2017 and 2019.
In total, 2,516 individuals were scanned. Cognitive tests were applied in all visits, but only to participants aged ≥55 years old at visit 2; therefore, 1,161 participants were excluded. Participants with a history of stroke were also excluded from the analyses (N=24). Thus, our final sample was restricted to participants from Belo Horizonte center aged ≥55 years old at baseline who underwent complete coronary arteries and thoracic aorta CT scans at visit 2 (N=1,331).
CORONARY ARTERY AND THORACIC AORTA CALCIUM SCORES
All participants in our sample underwent the same 64-slice MSCT scanner (Lightspeed, General Electric, Chicago, IL, USA). The scanogram encompassed from 1 cm above the top of the aortic arch to the heart apex. 2.5 mm thick slices were acquired with 20 × 0.62 mm collimation, 120 kVp, 100 mAs and prospective ECG triggering at 70% of the cardiac cycle. The reconstruction algorithm used body filter. The media of effective dose calculated was 1.75 mSv.²³
All images were analyzed by an experienced radiologist and calcium was identified using semiautomatic software (Smart Score v4.0), and by applying a threshold for calcium of 130 Hounsfield Unit (HU) to obtain the Agatston score.²⁴ A second experienced radiologist analyzed a random sample of 50 CT scans and a correlation study was performed, resulting in intraclass correlation coefficients for all arteries scored higher than 0.99 for intraobserver and interobserver analysis.⁵
Coronary artery calcification consisted of any calcified lesions identified within left main, left anterior descending, left circumflex, and right coronary arteries. Anatomical references to evaluate the thoracic aorta by segments were as follow: the ascending thoracic aortic calcium (ATAC) was considered from the sinotubular junction until the lower edge of pulmonary artery bifurcation, therefore calcium from Valsalva sinus and aortic valve was not included. The descending thoracic aortic calcium (DTAC) was defined from the level of the lower edge of pulmonary artery bifurcation to the heart apex. Consequently, the aortic arch calcium (AAC) was found above ATAC and DTAC using the same anatomical level as reference (the lower edge of pulmonary artery bifurcation).²⁵
Measurements of arterial calcification (CAC, TAC, ATAC, AAC, DTAC) were categorized as a dichotomous (present/absent) variable, and as ordinal variable [level 1 – no calcification (=0), level 2 – medium calcification (=1–399AU) and level 3 – high calcification (≥400 AU)]. These threshold levels were chosen based on clinical recommendations for cardiovascular risk stratification for coronary artery calcium.²⁶
COVARIATES
The covariates in our study comprised participants’ sociodemographic characteristics (sex, level of education, race/skin color), health behaviors (smoking, leisure physical activity), and medical history factors (body mass index (BMI), total cholesterol/HDL (high-density lipoprotein) ratio, diabetes and hypertension).
The covariates were obtained by face-to-face interviews at the time of the CT-scan (baseline). Relevant medical outcomes were also monitored by phone calls at least once a year. Level of education was classified in three categories: (1) university degree, (2) complete secondary school and (3) complete or incomplete elementary school. Race/skin color was self-reported and categorized into white, brown or “pardo”, black and indigenous and Asian. Due to small number, we chose to group white and Asian as a single category, and brown or “pardo” along with black and indigenous as other. Age was used in the analysis to index time. Thus, at baseline, age was the starting point, and in follow-up age corresponded to the initial age plus the interval (in years) between the 2 visit dates.
The health behaviors included smoking (non-smoker or current smoker) and leisure physical activity. Physical activity was evaluated by the long version of the International Physical Activity Questionnaire and grouped according to the number of MET (metabolic equivalent of task) into: light (<600 MET min/week), moderate (600–3000 MET min/week), and strong (≥3000 MET min/week).²⁷ For the purpose of this study, moderate and strong physical activities were grouped as a single category.
BMI was obtained by anthropometric measures of weight and height and was expressed in kg/m² as a continuous variable. Total cholesterol/HDL (high-density lipoprotein) ratio, determined by the enzymatic colorimetric method (ADVIA Chemistry; Siemens) was also assessed as continuous variable. Diabetes was evaluated as dichotomous (yes/no) and was defined by self-reported diagnosis or any of the following criteria: use of antidiabetic drugs, or fasting glucose ≥126 mg/dL, or glucose tolerance test ≥200 mg/dL, or glycated hemoglobin ≥6.5%. Hypertension, defined by systolic blood pressure (SBP) ≥140 mmHg and/or diastolic blood pressure (DBP) ≥90 mmHg and/or use of anti-hypertensive drugs, was also taken as a dichotomous variable.²⁸
COGNITIVE EVALUATION
The ELSA-Brasil study uses a combination of the Brazilian version of the Consortium to Establish a Registry for Alzheimer’s Disease (CERAD)²⁹,³⁰ to assess cognitive function. It comprises three parts: Memory test, Verbal fluency test and Trail B test.
In combination, the three tests assess all major cognitive domains: memory tests evaluate declarative memory; verbal fluency tests evaluate mainly language and executive function, but also explicit memory and semantics; finally, trail B test evaluates attention, concentration, psychomotor speed, visual scanning and mental flexibility.³¹
In Memory tests, the subjects were assessed for their ability to recall a specific list of 10 words immediately after an initial exposure in which the words were presented in three different paper sheets in random order each time (word list learning). Then, participants were asked to recall the same 10 words about 5 minutes after performing other activities (delayed word recall). Finally, they were asked to recognize the initial list of 10 words among 10 other distracting words (word recognition). The final score was defined by the sum of words correctly recalled each time, worth mentioning that, in recognition test, an incorrect word eventually recalled was subtracted from the total score.²⁹
In verbal fluency tests, the subjects were asked, at baseline, to name vegetables for 60 seconds and then to name words beginning with letter A also for 60 seconds. At follow-up, they were asked to name animals and words beginning with the letter F for the same amount of time. The final score was the sum of correct words spoken for 60 seconds.²⁸ The categories of the verbal fluency test varied between visits to reduce the learning effects. Because the 2 versions of the verbal fluency test can be parallel, but not necessarily equivalent, the test scores on baseline were converted into equivalent scores, comparable to the scores of the verbal fluency tests on follow-up visit.³²
In the Trail Making Test Part B (TMT-B), participants were asked to draw lines connecting letters and numbers disposed on a paper sheet. Subjects were instructed that the lines should be drawn alternating numbers and letters and in ascending order (1 – A – 2 – B – 3 etc.). Performance was measured in seconds to accomplish the test.³⁴ Therefore, for the TMT-B, higher scores indicated poorer performance, which contrasts to the aforementioned Memory and Verbal Fluency Test. Only individuals who completed the trail A test performed the trail B test. Thus, for individuals who did not complete the trail B test (N=17), the time of accomplishment was estimated as being the maximum time (+ one second) taken for individuals of the same sex, age group (5 years), and schooling.³⁴ To allow direct comparisons, we inverted the trail-making test B scores. Because the final score was not normally distributed, it was log transformed for the statistical analysis.
The global cognitive score, measured by the global cognitive factor (G-factor), was obtained from factor analysis with standardized scores (Z score) of memory, fluency, and trail B tests of participants who performed all the tests. G-factor was the first factor and explained 64% of the total variance of cognitive tests. This proportion is a typical value of variance explained by g-factor.³⁵
In order to compare cognitive performance scores between the two visits, the final scores of all tests were standardized to Z-scores [based on the mean and standard deviation (SD) of this study baseline]. Therefore, a reduction in participant’s Z score in any test at follow-up visit signals a relative decline in participant’s performance.
Statistical analyses
Categorical variables were described as proportions and continuous variables as means and SD.
Mixed linear regression models with random intercept and slope were used to evaluate associations of all calcium measurements with longitudinal changes in cognitive tests performance between baseline and follow-up visit.³⁶ ³⁷
The fixed effects (β) and variancecomponents (α) of the mixed linear models were estimated using maximum restricted likelihood methods.
Separate regression models were run for each cognitive test and for the global cognitive score, as well as for each explanatory variable (CAC, TAC, ATAC, AAC, DTAC). All explanatory variables and covariates (sex, level of education, race/skin color, smoking, leisure physical activity, BMI, total cholesterol/HDL ratio, diabetes and hypertension) were included in the models as fixed effects, and age was modeled as random effect to index time.
First, we modeled each explanatory variable of interest to level of education and sex. Then, the other covariates (race/skin color, smoking, leisure physical activity, BMI, total cholesterol/HDL ratio, diabetes and hypertension) were included in the models. Next, the longitudinal change in standardized cognitive scores was evaluated by including interaction terms between the explanatory dichotomous variables and age (CAC × age, TAC × age, ATAC × age, AAC × age, DTAC × age), and ordinal variables and age [CAC (level 2) × age, CAC (level 3) × age, TAC (level 2) × age, TAC (level 3) × age, ATAC (level 2) × age, ATAC (level 3) × age, AAC (level 2) × age, AAC (level 3) × age, DTAC (level 2) × age, DTAC (level 3) × age] and the results of the fully adjusted models presented. Statistical significance was defined as P<0.05 and 95% CI. The analyses were performed using Stata software version 14 (Stata Corporation, College Station, United States).
Results
Participants’ characteristics at baseline are presented in Table 1. Of 1,331 participants included, 54,5% were female and mean age was 62.4 (SD 5,7) years. 63.9% of individuals had university degree education. The overwhelming majority of participants were non-smokers (90.1%) and did not have diabetes (77.2%).
The prevalence of CAC, TAC, ATAC, AAC and DTAC among participants in our final sample is shown in table 2. Among thoracic aorta segments, higher calcium scores were more prevalent in aortic arch. Most participants did not have any calcification in descending thoracic aorta.
Table 1: Characteristics of the study population at baseline (2012–2014). (N = 1,331)
| Characteristics | Baseline |
|---|---|
| Age, y [average (SD)] | 62.4 (5.7) |
| Sex, women (%) | 54.5 |
Level of education (%)
-
University degree — 63.9
-
Incomplete — 24.5
-
Fundamental — 11.6
Race or skin color (%)
-
White/Asian — 55.5
-
Black or “pardo” — 44.5
Smoking (%)
-
Non-smoker — 90.1
-
Current smoker — 9.9
Leisure-time physical activity (%)
-
Low — 68.9
-
Moderate/vigorous — 31.1
| Other Variables | Baseline |
|---|---|
| Body mass index, kg/m² [average (SD)] | 27.0 (4.7) |
| Total cholesterol/HDL cholesterol ratio [average (SD)] | 3.7 (0.9) |
| Hypertension (%) | 50.0 |
| Diabetes (%) | 22.8 |
| Memory test score (number of correct words)ᵃ [average (SD)] | 38.9 (5.9) |
| Verbal fluency test score (number of correct words)ᵇ [average (SD)] | 29.6 (8.2) |
| Trail B test score (seconds)ᶜ [average (SD)] | 109 (69–171) |
Notes
ELSA-Brasil (N = 1,331). Data given as percentage, mean (SD), or median (range).
HDL indicates high-density lipoprotein.
-
ᵃ Score ranging from 0 to 50 correct words.
-
ᵇ Score ranging from 0 to infinite correct words remembered in a time interval of 1 min.
-
ᶜ Score ranging from 1 to 1368 seconds.
Table 2: Prevalence of calcification in different vessel beds in Agatston Units (AU) among participants at baseline
| Artery | 0 AU | 1–399 AU | ≥400 AU |
|---|---|---|---|
| Coronary arteries (CAC) | 42.69 | 47.26 | 10.05 |
| Total thoracic aorta (TAC) | 15.08 | 57.69 | 27.23 |
| Ascending thoracic aorta (ATAC) | 64.89 | 33.91 | 1.20 |
| Aortic arch (AAC) | 20.41 | 57.16 | 22.43 |
| Descending thoracic aorta (DTAC) | 53.71 | 37.66 | 8.63 |
ELSA-Brasil (N = 1,331). Data given in percentage.
After considering all covariates in the analyses, we found that only the presence of any DTAC was associated with a worse cognitive performance in Verbal Fluency test, in Trail B test and in global cognitive score at baseline (Table 3) and this association was sustained to follow-up visit. The interaction term calcification × age was not statistically significant for any cognitive test, which implies that the mere presence of any calcification in any of the analyzed vessel beds did not accelerate cognitive decline over the study time.
Table 3 – Association between presence of any calcification in different vessel beds at baseline and cognitive performance test in Brazilian adults, over about four years follow-up. ELSA-Brasil, 2012–14 and 2017–19. (N = 1,331)
| Cognitive function test | Memory tests (Z score) (N = 1,200) | Verbal fluency tests (Z score) (N = 1,202) | Trail B test (Z score) (N = 1,331) | G-factor (N = 1,199) |
|---|
Calcification = 0
Ref. | Ref. | Ref. | Ref.
Calcification > 0
Coronary arteries (CAC)
-
Memory: 0.0253 (-0.0732 to 0.1239)
-
Verbal: –0.0270 (-0.1179 to 0.0638)
-
Trail B: –0.05149 (-0.1422 to 0.0392)
-
G-factor: –0.0580 (-0.1249 to 0.0088)
Total thoracic aorta (TAC)
-
Memory: 0.0727 (-0.0544 to 0.1999)
-
Verbal: 0.025581 (-0.0952 to 0.1464)
-
Trail B: –0.0958 (-0.2163 to 0.0247)
-
G-factor: –0.0634 (-0.1511 to 0.0261)
Ascending thoracic aorta (ATAC)
-
Memory: –0.0242 (-0.1244 to 0.0759)
-
Verbal: 0.0605 (-0.0309 to 0.1521)
-
Trail B: –0.0132 (-0.1044 to 0.0780)
-
G-factor: –0.0299 (-0.0976 to 0.0377)
Aortic arch (AAC)
-
Memory: 0.0340 (-0.0797 to 0.1477)
-
Verbal: 0.0341 (-0.0727 to 0.141)
-
Trail B: –0.0614 (-0.1682 to 0.0453)
-
G-factor: –0.0501 (-0.1282 to 0.0279)
Descending thoracic aorta (DTAC)
-
Memory: –0.0208 (-0.1186 to 0.0770)
-
Verbal: –0.0986 (-0.1888 to –0.0083)ᶜ
-
Trail B: –0.1332 (-0.2230 to –0.0435)ᵇ
-
G-factor: –0.1486 (-0.2146 to –0.0826)ᵃ
Interaction term: calcification × age
Coronary arteries (CAC)
-
Memory: –0.0005 (-0.0138 to 0.0126)
-
Verbal: 0.0009 (-0.0112 to 0.0131)
-
Trail B: –0.0054 (-0.0168 to 0.0059)
-
G-factor: –0.0045 (-0.0127 to 0.0036)
Total thoracic aorta (TAC)
-
Memory: 0.0162 (-0.0046 to 0.0372)
-
Verbal: 0.0009 (-0.0190 to 0.0209)
-
Trail B: –0.0052 (-0.0236 to 0.0131)
-
G-factor: –0.0064 (-0.0194 to 0.0063)
Ascending thoracic aorta (ATAC)
-
Memory: –0.0042 (-0.0175 to 0.0091)
-
Verbal: –0.0028 (-0.0150 to 0.0093)
-
Trail B: 0.0021 (-0.0092 to 0.0136)
-
G-factor: –0.0040 (-0.0123 to 0.0043)
Aortic arch (AAC)
-
Memory: 0.0018 (-0.0154 to 0.0191)
-
Verbal: 0.0088 (-0.0073 to 0.0250)
-
Trail B: –0.0059 (-0.0211 to 0.0091)
-
G-factor: –0.0039 (-0.0146 to 0.006)
Descending thoracic aorta (DTAC)
-
Memory: –0.0016 (-0.0148 to 0.0114)
-
Verbal: –0.0029 (-0.0150 to 0.0091)
-
Trail B: –0.0002 (-0.0116 to 0.0110)
-
G-factor: –0.0074 (-0.0156 to 0.0007)
Model note:
Final model obtained by linear mixed effects regression adjusted for age, sex, education level, race/skin color, smoking, leisure-time physical activity, body mass index, hypertension, diabetes, total cholesterol/high-density cholesterol and interaction term “calcification × age”.
P value:
ᵃ <0.001
ᵇ <0.01
ᶜ <0.05
Age (age at baseline + time to follow-up visit) was modeled as a random effect to index time.
In regards to the levels of calcification, we found that higher CAC were associated with worse global cognitive score (Table 4). Associations between higher AAC and TAC and a worse performance in Trail B test were also statistically significant. We observed statistically significant associations between both medium and higher DTAC and worse cognitive performance in Trail B test performance and global cognitive scores. Intriguingly, medium DTAC, but not higher DTAC, was associated with worse performance in Verbal Fluency test. Although a positive association between greater DTAC and memory test performance was seen at baseline, the interaction term “levels of calcification” × age was negative and statistically significant indicating greater acceleration in the speed of memory decline between visits for individuals with calcifications ≥400AU in this vessel bed.
Table 4 – Association between severity of calcification of different vessel beds (in Agatston) at baseline and cognitive performance over 4 years of follow-up, estimated by linear mixed effects regression.
ELSA-Brasil, 2012–2014 and 2017–2019
| Cognitive function test | Memory tests (Z score) (N = 1,200) | Verbal fluency tests (Z score) (N = 1,202) | Trail B test (Z score) (N = 1,331) | G-factor (N = 1,199) |
|---|
Calcification = 0
Ref. | Ref. | Ref. | Ref.
Coronary arteries (CAC)
1 to 399
-
Memory: 0.0317 (−0.0686 to 0.1321)
-
Verbal: −0.0220 (−0.1146 to 0.0705)
-
Trail B: −0.0274 (−0.1196 to 0.0648)
-
G-factor: −0.0420 (−0.1101 to 0.0260)
≥400
-
Memory: −0.0242 (−0.2026 to 0.1541)
-
Verbal: −0.0642 (−0.2243 to 0.0959)
-
Trail B: −0.2249 (−0.3827 to −0.0670)
-
G-factor: −0.1771 (−0.2962 to −0.0579)ᵇ
Thoracic aorta (total) (TAC)
1 to 399
-
Memory: 0.0591 (−0.0701 to 0.1884)
-
Verbal: 0.0112 (−0.1114 to 0.1338)
-
Trail B: −0.0804 (−0.2029 to 0.0419)
-
G-factor: −0.0557 (−0.1449 to 0.0335)
≥400
-
Memory: 0.1256 (−0.0302 to 0.2814)
-
Verbal: 0.0798 (−0.0655 to 0.2252)
-
Trail B: −0.1520 (−0.2965 to −0.0074)ᶜ
-
G-factor: −0.0915 (−0.1976 to 0.0145)
Ascending thoracic aorta (ATAC)
1 to 399
-
Memory: −0.0263 (−0.1273 to 0.0746)
-
Verbal: 0.0665 (−0.0257 to 0.1587)
-
Trail B: −0.0020 (−0.0939 to 0.0898)
-
G-factor: −0.0225 (−0.0907 to 0.0456)
≥400
-
Memory: 0.0485 (−0.3955 to 0.4926)
-
Verbal: −0.1310 (−0.5210 to 0.2588)
-
Trail B: −0.3680 (−0.7530 to 0.0169)
-
G-factor: −0.2677 (−0.5594 to 0.0239)
Aortic arch (AAC)
1 to 399
-
Memory: 0.0171 (−0.0991 to 0.1333)
-
Verbal: 0.0169 (−0.0922 to 0.1261)
-
Trail B: −0.0403 (−0.1494 to 0.0686)
-
G-factor: −0.0447 (−0.1245 to 0.0350)
≥400
-
Memory: 0.1011 (−0.0476 to 0.2499)
-
Verbal: 0.1004 (−0.0366 to 0.2374)
-
Trail B: −0.1409 (−0.2772 to −0.0046)ᶜ
-
G-factor: −0.0706 (−0.1714 to 0.0300)
Descending thoracic aorta (DTAC)
1 to 399
-
Memory: −0.0599 (−0.1606 to 0.0407)
-
Verbal: −0.0960 (−0.1891 to −0.0030)ᶜ
-
Trail B: −0.1165 (−0.2092 to −0.0239)ᶜ
-
G-factor: −0.1426 (−0.2107 to −0.0744)ᵃ
≥400
-
Memory: 0.2336 (0.0457 to 0.4215)ᶜ
-
Verbal: −0.1139 (−0.2788 to 0.0508)
-
Trail B: −0.2315 (−0.3943 to −0.0687)ᵇ
-
G-factor: −0.1859 (−0.3095 to −0.0622)ᵇ
Interaction term: calcification × age
Coronary arteries (CAC)
1 to 399
-
Memory: −0.0020 (−0.0158 to 0.0117)
-
Verbal: −0.00002 (−0.0127 to 0.0126)
-
Trail B: −0.0047 (−0.0166 to 0.0071)
-
G-factor: −0.0045 (−0.0130 to 0.0040)
≥400
-
Memory: 0.0081 (−0.0143 to 0.0305)
-
Verbal: 0.0071 (−0.0132 to 0.0276)
-
Trail B: −0.0022 (−0.0211 to 0.0165)
-
G-factor: −0.0007 (−0.0148 to 0.0132)
Thoracic aorta (TAC)
1 to 399
-
Memory: 0.0173 (−0.0042 to 0.0389)
-
Verbal: 0.00336 (−0.0172 to 0.0239)
-
Trail B: −0.0024 (−0.0213 to 0.0165)
-
G-factor: −0.0032 (−0.0165 to 0.0099)
≥400
-
Memory: 0.0120 (−0.0111 to 0.0351)
-
Verbal: −0.0062 (−0.0281 to 0.0156)
-
Trail B: −0.0081 (−0.0283 to 0.0120)
-
G-factor: −0.0117 (−0.0259 to 0.0025)
Ascending thoracic aorta (ATAC)
1 to 399
-
Memory: −0.0026 (−0.0161 to 0.0108)
-
Verbal: −0.0018 (−0.0142 to 0.0103)
-
Trail B: 0.0026 (−0.0088 to 0.0142)
-
G-factor: −0.0030 (−0.0114 to 0.0053)
≥400
-
Memory: −0.0631 (−0.1265 to 0.0003)
-
Verbal: −0.0260 (−0.0848 to 0.0327)
-
Trail B: 0.0068 (−0.0460 to 0.0597)
-
G-factor: −0.0235 (−0.0629 to 0.0158)
Aortic arch (AAC)
1 to 399
-
Memory: −0.0016 (−0.0195 to 0.0163)
-
Verbal: 0.0088 (−0.0079 to 0.0256)
-
Trail B: −0.0053 (−0.0209 to 0.0103)
-
G-factor: −0.0040 (−0.0151 to 0.0070)
≥400
-
Memory: 0.0076 (−0.0131 to 0.0283)
-
Verbal: 0.0056 (−0.0135 to 0.0248)
-
Trail B: −0.0041 (−0.0220 to 0.0137)
-
G-factor: −0.0030 (−0.0159 to 0.0098)
Descending thoracic aorta (DTAC)
1 to 399
-
Memory: −0.0002 (−0.0141 to 0.0136)
-
Verbal: −0.0005 (−0.0133 to 0.0123)
-
Trail B: 0.0009 (−0.0111 to 0.0129)
-
G-factor: −0.0050 (−0.0136 to 0.0035)
≥400
-
Memory: −0.0258 (−0.0502 to −0.0014)ᶜ
-
Verbal: −0.01401 (−0.0361 to 0.0080)
-
Trail B: 0.0001 (−0.0204 to 0.0207)
-
G-factor: −0.0175 (−0.0326 to −0.0024)ᶜ
ELSA-Brasil (N = 1,331). Final model adjusted for age, sex, education level, race or skin color, smoking, leisure-time physical activity, body mass index, hypertension, diabetes, total cholesterol/high-density lipoprotein ratio, and interaction: severity of calcification × age. β indicates β coefficient.
P value:
ᵃ <0.001
ᵇ <0.01
ᶜ <0.05
Age (age at baseline + follow-up time) was modeled as a random effect to index time.
Also, the interaction term shows that calcifications ≥400AU in the descending aorta was also related to faster decline in global cognitive performances between study visits in our final sample, irrespective of covariates, as compared to individuals without calcification in this vessel bed. (Figure 1)
-
Background
-
Methods
-
Results
-
Conclusion
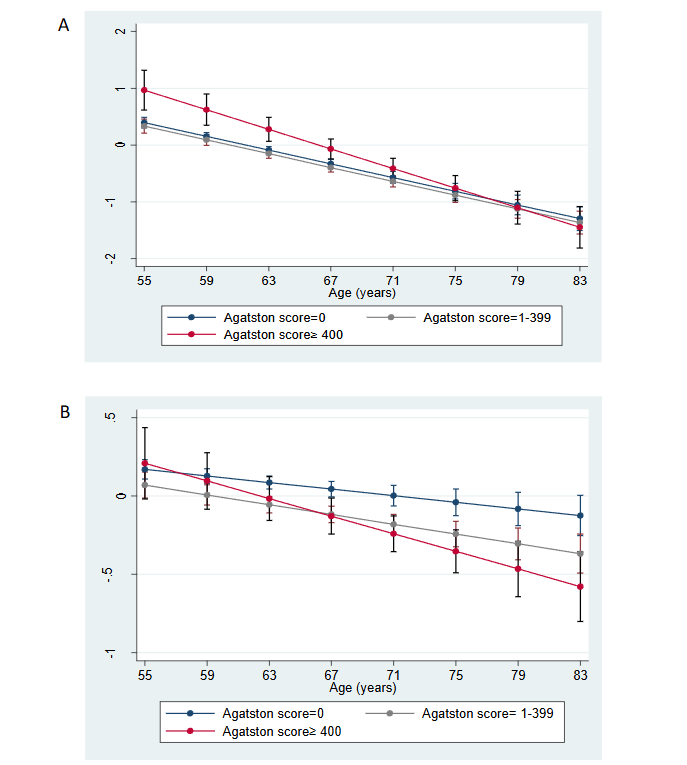
Figure 1: Predicted values (95% CI) for Memory Test (A) and for Factor G (B) scores, according to calcification levels in Agatston Units (AU) in the descending thoracic aorta between two study visits, after adjustments. ELSA-Brasil, 2012–2014 and 2017–2019.
As age was modeled as a random effect in data analysis, this figure shows changes in cognitive performance over time (ie, as individuals age).
Predicted values are values of the dependent variable based on the estimated regression coefficients (mean ratios observed in the regression) and a prediction about the values of the independent variables.
Discussion
In this large cohort of middle-aged and older adults, we examined whether calcification in the coronary artery and the thoracic aorta was associated with cognitive performance in about four-year follow up. Results indicate that the presence of any calcification in the descending aorta, but not in other vessel beds, was associated with worse cognitive performances in verbal fluency and trail B tests as well as global cognition at baseline, with no indication that it changed the pace of cognitive decline in these domains in the follow up period. The analysis considering the level of calcification at distinct vessel beds, however, shows that calcifications ≥ 400 AU in the coronary arteries, thoracic aorta, aortic arch and descending aorta were associated to worse performance either in Trail B test or in global cognition at baseline, independently of major cardiovascular risk factors, but had no effect on the speed of cognitive decline in these domains, except for greater calcification in the descending aorta that appear to speed up the performance decline in global cognition in the study period. Finally, greater calcification in the descending aorta was associated with better performance in memory test at baseline, but with greater decline in this domain during follow up.
Previous studies focusing on this topic show some consistency in regard to the severity of CAC and worse cognitive function, but also present conflicting results in regard to CAC and affected cognitive domains. For instance, a cross-sectional study of the ELSA-Brasil cohort in São Paulo center found that participants (mean age 50.9 ± 8.8 years) with higher CAC scores, defined as CAC >100 AU, had slightly poorer performance in the executive function test.¹¹ Xia et al. reported that in a Dutch population of ≥45 years, increased CAC severity was associated with worse performance of working memory, independent of classical cardiovascular risk factors, but not with other cognitive domains and, notably, the inverse relationship of CAC score with working memory was strongest in participants aged 45 to 54 years.¹⁵
A cross-sectional study of the CARDIA cohort (mean age 43–55 years) found a graded association between a greater burden of CAC and worse performance on tests of processing speed, attention, and working memory in midlife irrespective of demographic characteristics, major vascular risk factors, and depressive symptoms.³⁸ But different from our study, the AGES-Reykjavik Study found that increasing calcification of the coronary arteries was associated with decreased cognitive speed of processing, executive function, and increase overall risk of dementia in a cohort with an average age of 76.¹⁶ The Rotterdam Study (mean age 69.5 years) investigated relations between arterial calcification in various vessel beds (coronaries, aortic arch, extracranial, and intracranial carotid arteries), having both cognitive decline and dementia as clinical endpoints, and found that larger calcification volumes in coronary arteries were related to greater decline in global cognition, but not with a higher risk of dementia.¹⁸ The discrepancies observed in these studies may reflect different methodological strategies, different measurements of cognitive function and different population sample in regard, especially, to age range, sample size and duration of follow up. The herein ELSA-Brasil sample was younger, and the follow-up duration smaller than those included in the Rotterdam and AGES-Reykjavik studies. Because cognitive decline is an insidious, age-related process, it is safe to assume, as a general rule, that in cohorts aiming at evaluating cognitive performance over time, the longer the observation time, the greater the chances of detecting cognitive decline in tests designed for this purpose. In parallel to that, the older the sample population, the more pronounced the decline in cognition would be expected to be. In that sense, we believe that longitudinal associations between CAC burden and cognitive decline would probably be found if our population sample was older and was followed for a longer observation time.
In our study, we also observed an association between higher AAC and TAC and a worse performance in Trail B test (which mainly reflects psychomotor speed, attention, concentration, visual scanning and mental flexibility), but found no evidence of a longitudinal effect. Studies focusing on aortic arch calcification and cognition are scarce.¹⁸ ³⁹ However, Saleh et al. reported that increased aortic stiffness assessed by aortic arch pulse wave velocity was associated with measurements of incipient brain injury detected by Diffusion Tensor Imaging (DTI) in participants with high blood pressure, irrespective of age, sex, body mass index, smoking, and white matter hyperintensity volume (WMHV).⁴⁰ Arterial stiffness is regulated by a number of factors, including age, hypertension, diabetes and obesity and it is well known that arterial calcification and arterial stiffness encompass distinct pathological entities; nonetheless, it is hypothesized that arterial calcification might play an important role in mediating arterial stiffness.⁴¹
We believe that the main finding in our study, however, concerns the DTAC and its association with cognitive function over time. The mere presence of calcification in this segment, in opposition to no calcification, was associated to a worse cognitive performance in Verbal Fluency test, in Trail B test and in global cognitive score. Furthermore, we found a graded association between a greater burden of DTAC and worse performances in Trail B test and global cognitive scores. Only medium DTAC, but not high DTAC, probably due to small numbers, was associated to worse performance in Verbal Fluency test, but this finding requires further investigation. We also observed that higher DTAC accelerated cognitive decline in memory test and in global cognitive performances in our study time, independently of covariates. No other analyzed vessel bed calcification burden had such significant impact in predicting worse cognitive performance between visits in any of the cognitive domains assessed in our study.
The results regarding descending thoracic aorta are somehow in line with previous cross-sectional evidence from the Framingham Offspring cohort, in which descending aorta calcification prevalence and burden, evaluated by cardiovascular magnetic resonance imaging (MRI), were associated to decreased total cerebral brain volume (TCBV) and to increased WMHV detected by brain MRI in a sample of 1,527 participants (mean age 59.9 years).⁴² As previously reported, decreased TCBV and increased WMHV are inflicted in accelerated brain aging and, presumably, in cognitive decline.⁴³ ⁴⁴
Descending aorta calcification may reflect more generalized atherosclerosis than calcification in the other vessel beds analyzed in our study, since hemodynamics, vessel caliber and mechanical shear stress secondary to blow flow in descending aorta seem to be distinct and separate from those in the coronaries, ascending thoracic aorta and aortic arch, from which arteries supply blood to the brain emerge.⁴² In accordance to this assumption DTAC has been shown to be more closely associated with stroke than calcification in neighboring thoracic aorta segments.⁴⁵ It is relevant to notice that vascular smooth muscle cells (VSMC) that compose descending thoracic aorta derive from mesoderm and VSMC that compose the neighboring thoracic aorta segments derive from neural crest.⁴⁶ VSMC are largely implicated in atherosclerosis physiopathology.⁴⁷ It has been suggested that each vascular bed exhibits its own distinctive response to the atherogenic process, possibly due to genetic differences in the composition of intimal and subintimal tissues in different vessel beds, and, in that sense, a distinct embryological origin in VSMC might interfere in the observed differences.⁴⁷ ⁴⁸ In line with that hypothesis, it has been shown, perhaps counterintuitively to common sense assumptions, that the extent of impact of diverse cardiovascular risk factors in the calcification in thoracic aorta segments differ in regard to each separate segment of this vessel bed. Thus, it is not unreasonable to presume that thoracic aortic segments calcification perhaps encompass distinct pathological process and may be implicated in distinct cardiovascular and even non-cardiovascular outcomes.
Strengths of this study include large sample from a middle-income country, a longitudinal design, comprehensive measurements of calcifications in total thoracic aorta and in its segments (separately), adjustments for most risk factors for cardiovascular diseases and evaluation of different cognitive abilities.
Limitations of our study should also be addressed. Our study population has a high level of education, and has been followed-up for a relatively short time (about 4 years). We imagine that results of our study could be different if participants were followed for a longer period. The high schooling of our cohort, due to mechanisms involving greater cognitive reserve, may contribute to delayed cognitive decline in the follow-up period. Apart from that, ELSA-Brasil does not account with APOE-4 gene allele testing, precluding adjustments for this possible confounding factor.⁴⁹
Conclusion
Our study adds to current evidence that atherosclerosis is indeed related to cognitive decline and brings new insights into the conjecture that calcification in different vessel beds is distinctively associated to cognitive function. Among thoracic aorta segments, DTAC in particular seems to reflect a more generalized atherosclerotic pathology and appears to signal a greater risk of cognitive decline over the years.
Conflict of interest
Authors have no conflict of interest to declare.
Financial support
ELSA Brazil is funded by the Brazilian Ministry of Health (Science and Technology Department) and the Brazilian Ministry of Science, Technology and Innovation (FINEP and CNPq). Baseline grant numbers:
01 060010.00 and 01.10.0643.03 (RS);
01 06 0212.00 and 01.0742-00 (BA);
01 06 0300.00 and 01.12.0284.00 (ES);
01 06 0278.00 and 01 10 0746 00 (MG);
01 06 0115.00 and 01.10.0773-00 (SP);
and 01 06 0071.00 and 01.11.0093.01 (RJ).
SMB and LG are research fellows from the Brazilian Research Council (CNPQ).
Credit authorship contribution statement
Igor Vasconcelos Barros Cronemberger: Formal analysis, writing – original draft.
Jesiana Ferreira Pedrosa: Formal analysis.
Daniel Boss: Writing – original draft.
Luana Giatti Gonçalves: Formal analysis, writing – original draft.
Sandhi Maria Barreto: Conceptualization, formal analysis, writing – original draft.
References
2. Neves PO, Andrade J, Monção H. Coronary artery calcium score: current status. Radiol Bras. 2017; 50(3):182–189. doi:10.1590/0100-3984.2015.0235
3. Desai MY, Cremer PC, Schoenhagen P. Thoracic Aortic Calcification. JACC: Cardiovascular Imaging. 2018;11(7):1012–1026. doi:10.1016/j.jcm g.2018.03.023
4. Thomas IC, Thompson CA, Yang M, Allison MA, Forbang NI, Michos ED, et al. Thoracic Aorta Calcification and Noncardiovascular Disease–Related Mortality: The Multi-Ethnic Study of Atherosclerosis. Arterioscler Thromb Vasc Biol. 2018;38(8):1926–1932. doi: 10.1161/ATVBAHA.118.310850
5. Pedrosa JF, Brant LCC, de Aquino SA, Ribeiro AL, Barreto SM. Segmental Evaluation of Thoracic Aortic Calcium and Their Relations with Cardiovascular Risk Factors in the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil). Cells. 2021;18;10(5). doi:10.3390/cells10051243
6. Rodrıguez-Palomares JF, Masip AE. Aortic Calcium Score and Vascular Atherosclerosis in Asymptomatic Individuals: Beyond the Coronary Arteries. Rev Esp Cardiol. 2016;69(9):813-816. doi:10.1016/j.rec.2016.05.006
7. Hermann DM, Lehmann N, Gronewold J, Bauer M, Mahabadi AA, Weimar C, et al. Thoracic aortic calcification is associated with incident stroke in the general population in addition to established risk factors. European Heart Journal – Cardiovascular Imaging. 2015;16(6):684–690. doi:10.1093/ehjci/jeu293
8. van Oijen M, Jan de Jong F, Witteman JCM, Hofman A, Koudstaal PJ, Breteler MMB. Atherosclerosis and risk for dementia. Ann Neurol. 2007;61(5):403–410. doi: 10.1002/ana.21073
9. Chu Z, Cheng L, Tong Q. Carotid artery calcification score and its association with cognitive impairment. Clinical Interventions in Ageing. 2019; 14:167–177. doi: 10.2147/CIA.S192586
10. Nordestgaard LT, Christoffersen M, Frikke-Schmidt R. Shared Risk Factors between Dementia and Atherosclerotic Cardiovascular Disease. Int. J. Mol. Sci. 2022;23(17):1-30. doi:10.3390/ijms23179777
11. Suemoto CK, Bittencourt MS, Santos IS, Benseñor IM, Lotufo PA. Coronary artery calcification and cognitive function: cross-sectional results from the ELSA-Brasil study: CAC and cognition. Int J Geriatr Psychiatry. 2017;32(12):e188–194. doi: 10.1002/gps.4698
12. Dempsey RJ, Vemuganti R, Varghese T, Hermann BP. A Review of Carotid Atherosclerosis and Vascular Cognitive Decline: A New Understanding of the Keys to Symptomology. Neurosurgery. 2010; 67(2):484–494. doi: 10.1227/01.NEU.0000371730.1 1404.36
13. Xia C, Vonder M, Sidorenkov G, Oudkerk M, de Groot JC, van der Harst P, et al. The Relationship of Coronary Artery Calcium and Clinical Coronary Artery Disease with Cognitive Function: A Systematic Review and Meta-Analysis. J Atheroscler Thromb. 2020;27(9):934–958. doi: 10.5551/jat.52928
14. Kuller LH, Lopez OL, Mackey RH, Rosano C, Edmundowicz D, Becker JT, et al. Subclinical Cardiovascular Disease and Death, Dementia, and Coronary Heart Disease in Patients 80+ Years. Journal of the American College of Cardiology. 2016;67(9):1013–1022. doi: 10.1016/j.jacc.2015.1 2.034
15. Xia C, Vonder M, Sidorenkov G, Ma R, Oudkerk M, van der Harst P, et al. Coronary Artery Calcium and Cognitive Function in Dutch Adults: Cross‐Sectional Results of the Population‐Based ImaLife Study. J Am Heart Assoc. 2021;10(4):1-12. doi: 10.1161/JAHA.120.018172
16. Vidal JS, Sigurdsson S, Jonsdottir MK, Eiriksdottir G, Thorgeirsson G, Kjartansson O, et al. Coronary Artery Calcium, Brain Function and Structure: The AGES-Reykjavik Study. Stroke. 2010;41(5):891–897. doi: 10.1161/STROKEAHA.11 0.579581
17. Rossetti HC, Weiner M, Hynan LS, Cullum CM, Khera A, Lacritz LH. Subclinical atherosclerosis and subsequent cognitive function. Atherosclerosis. 2015;241(1):36–41.doi: 10.1016/j.atherosclerosis.2 015.04.813
18. Bos D, Vernooij MW, Bruijn RFAG, Koudstaal PJ, Hofman A, Franco OH, et al. Atherosclerotic calcification is related to a higher risk of dementia and cognitive decline. Alzheimer’s & Dementia. 2015;11(6):639-647. doi: 10.1016/j.jalz.2014.05.1758
19. Fujiyoshi A, Jacobs DR, Fitzpatrick AL, Alonso A, Duprez DA, Sharrett AR, et al. Coronary Artery Calcium and Risk of Dementia in MESA (Multi-Ethnic Study of Atherosclerosis). Circ. Cardiovascular Imaging. 2017;10(5):1-7. doi: 10.1161/CIRCIMAGIN G.116.005349
20. Brodov Y, Gransar H, Rozanski A, et al. Extensive thoracic aortic calcification is an independent predictor of development of coronary artery calcium among individuals with coronary artery calcium score of zero. Atherosclerosis. 2015;238 (1):4-8. doi:10.1016/j.atherosclerosis.2014.10.100
21. Aquino EML, Barreto SM, Bensenor IM, Carvalho MS, Chor D, Duncan BB, et al. Brazilian Longitudinal Study of Adult Health (ELSA-Brasil): Objectives and Design. American Journal of Epidemiology. 2012;175(4):315–324. doi: 10.1093/a je/kwr294
22. Schmidt MI, Duncan BB, Mill JG, Lotufo PA, Chor D, Barreto SM, et al. Cohort Profile: Longitudinal Study of Adult Health (ELSA-Brasil). International Journal of Epidemiology. 2015;44(1):68–75. doi: 10.1093/ije/dyu027
23. Pedrosa JF, Ribeiro ALP, Santana PC, Araújo LF, Barreto SM. Relation of Thoracic Aortic and Coronary Artery Calcium to Cardiovascular Risk Factors (from The Brazilian Longitudinal Study of Adult Health [ELSA-Brazil]). The American Journal of Cardiology. 2019;124(11):1655–1661. doi: 10.1016/j.amjcard.2019.08.029
24. Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. Journal of the American College of Cardiology. 1990;15(4):827–832. doi: 10.1016/0735-1097(90)90282-T
25. Pedrosa JF, Barreto SM, Bittencourt MS, Ribeiro ALP. Anatomical References to Evaluate Thoracic Aorta Calcium by Computed Tomography. Curr Atheroscler Rep. 2019;21(12):51-62.
doi: 10.1007/s11883-019-0811-9
26. Greenland P, Blaha MJ, Budoff MJ, Erbel R, Watson KE. Coronary Calcium Score and Cardiovascular Risk. Journal of the American College of Cardiology. 2018;72(4):434–447. doi: 10.1016/j.jacc.2018.05.027
27. Craig CL, Marshall AL, Sj??Str??M M, Bauman AE, Booth ML, Ainsworth BE, et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity: Medicine & Science in Sports & Exercise. 2003;35(8):1381–1395. doi: 10.1249/01.M SS.0000078924.61453.FB
28. Mill JG, Pinto K, Griep RH, Goulart A, Foppa M, Lotufo PA, et al. Afericoes e exames clinicos realizados nos participantes do ELSA-Brasil. Rev Saúde Pública. 2013;47(suppl 2):54–62.
doi: 10.1590/S0034-8910.2013047003851
29. Passos VM de A, Caramelli P, Benseñor I, Giatti L, Barreto SM. Methods of cognitive function investigation in the Longitudinal Study on Adult Health (ELSA-Brasil). Sao Paulo Med J. 2014;132 (3):170–177. doi: 10.1590/1516-3180.2014.1323646
30. Silverman JM, Raiford K, Edland S, Fillenbaum G, Morris JC, Clark CM, et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD): Part VI. Family history assessment: A multicenter study of first‐degree relatives of Alzheimer’s disease probands and nondemented spouse controls. Neurology. 1994;44(7):1159–1165. doi: 10.1212/w nl.44.7.1253
31. Mistridis P, Egli SC, Iverson GL, Berres M, Willmes K, Welsh-Bohmer KA, et al. Considering the base rates of low performance in cognitively healthy older adults improves the accuracy to identify neurocognitive impairment with the Consortium to Establish a Registry for Alzheimer’s Disease-Neuropsychological Assessment Battery (CERAD-NAB). Eur Arch Psychiatry Clin Neurosci. 2015;265(5):407–417. doi: 10.1007/s00406-014-0571-z
32. Bertola L, Benseñor IM, Gross AL, Caramelli P, Barreto SM, Moreno AB, et al. Longitudinal measurement invariance of neuropsychological tests in a diverse sample from the ELSA-Brasil study. Braz J Psychiatry. 2021;43(3):254–261. doi: 10.1590/1516-4446-2020-0978
33. Bertola L, Benseñor IM, Goulart AC, Brunoni AR, Caramelli P, Barreto SM, et al. Normative Data for the ELSA-Brasil Neuropsychological Assessment and Operationalized Criterion for Cognitive Impairment for Middle-Aged and Older Adults. J Int Neuropsychol Soc. 2021;27(3):293–303. doi: 10.1017/S1355617720000880
34. Menezes ST, Giatti L, Colosimo EA, Ribeiro ALP, Brant LCC, Viana MC, et al. Aortic Stiffness and Age With Cognitive Performance Decline in the ELSA‐Brasil Cohort. J Am Heart Assoc. 2019;8 (24):1-12. doi: 10.1161/JAHA.119.013248.
35. Deary IJ. Intelligence. Annu Rev Psychol. 2012;63(1):453–482. doi: 10.1146/annurev-psych-120710-100353
36. Cnaan A, Laird NM, Slasor P. Using the general linear mixed model to analyse unbalanced repeated measures and longitudinal data. Statist Med. 1997;16(20):2349–2380. doi: 10.1002/(sici)1097-0258(19971030)16:20<2349::aid-sim667>3.0.co;2-e
37. Fausto MA, Carneiro M, Antunes CM de F, Pinto JA, Colosimo EA. O modelo de regressão linear misto para dados longitudinais: uma aplicação na análise de dados antropométricos desbalanceados. Cad Saúde Pública. 2008;24(3): 513–524. doi: 10.1590/S0102-311X2008000300005
38. Reis JP, Launer LJ, Terry JG, Loria CM, Zeki Al Hazzouri A, Sidney S, et al. Subclinical atherosclerotic calcification and cognitive functioning in middle-aged adults: The CARDIA study. Atherosclerosis. 2013;231(1):72–77. doi: 10.1016/j.atherosclerosis.2013.08.038
39. Bos D, Vernooij MW, Elias-Smale SE, Verhaaren BFJ, Vrooman HA, Hofman A, et al. Atherosclerotic calcification relates to cognitive function and to brain changes on magnetic resonance imaging. Alzheimer’s & Dementia. 2012;8(5S):104-111.doi: 10.1016/j.jalz.2012.01.008
40. Sala M, van den Berg-Huysmans A, van der Grond J, Huisman M, Brandts A, Westenberg JJ, et al. Aortic Arch Stiffness Is Associated With Incipient Brain Injury in Patients With Hypertension. American Journal of Hypertension. 2016;29(6):705–712. doi: 10.1093/ajh/hpv161
41. McEniery CM, McDonnell BJ, So A, Aitken S, Bolton CE, Munnery M, et al. Aortic Calcification Is Associated With Aortic Stiffness and Isolated Systolic Hypertension in Healthy Individuals. Hypertension. 2009;53(3):524–531. doi: 10.1161/HYP ERTENSIONAHA.108.126615
42. Aparicio HJ, Petrea RE, Massaro JM, Manning WJ, Oyama-Manabe N, Beiser AS, et al. Association of descending thoracic aortic plaque with brain atrophy and white matter hyperintensities: The Framingham Heart Study. Atherosclerosis. 2017;265: 305–311. doi: 10.1016/j.atherosclerosis.2017.0 6.919
43. Geerlings MI, Appelman APA, Vincken KL, Algra A, Witkamp TD, Mali WPTM, et al. Brain volumes and cerebrovascular lesions on MRI in patients with atherosclerotic disease. The SMART-MR study. Atherosclerosis. 2010;210(1):130–136. doi: 10.1016/j.atherosclerosis.2009.10.039
44. Ma YN, Xie WX, Hou ZH, An YQ, Ren XS, Ma YJ, et al. Association between coronary artery calcification and cognitive function in a Chinese community-based population. J of Geriatr Cardiol. 2021;18(7): 514–522. doi:10.11909/j.issn.1671-5411.2021.07.002
45. Hermann DM, Lehmann N, Gronewold J, et al. Thoracic aortic calcification is associated with incident stroke in the general population in addition to established risk factors. Eur Heart J Cardiovasc Imaging. 2015;16(6):684-690. doi:10.1093/ehjci/jeu293
46. Majesky MW. Developmental Basis of Vascular Smooth Muscle Diversity. Arterioscler Thromb Vasc Biol. 2007;27(6):1248–1258. doi: 10.1161/ATVBAH A.107.141069
47. Libby P, Bornfeldt KE, Tall AR. Vascular Smooth Muscle Cells in Atherosclerosis. Circ Res Research. 2016; 118: 692-702. doi: 10.1161/CIRC RESAHA.115.306361
48. Allison MA, Criqui MH, Wright CM. Patterns and Risk Factors for Systemic Calcified Atherosclerosis. Arterioscler Thromb Vasc Biol. 2004;24(2):331–336. doi: 10.1161/01.ATV.0000110786.02097.0c
49. Caselli RJ, Reiman EM, Locke DEC, Hutton ML, Hentz JG, Hoffman-Snyder C, et al. Cognitive Domain Decline in Healthy Apolipoprotein E ε4 Homozygotes Before the Diagnosis of Mild Cognitive Impairment. Arch Neurol. 2007;64(9): 1306-1311. doi: 10.1001/archneur.64.9.1306




