Breast Cancer Trends in Young Women in Brazil (2014-2022)
Temporal Trend of Breast Cancer Diagnoses in Young Women in Different Regions of Brazil from 2014 to 2022
Gabriele de Sousa Silva 1, Lara Marcon Dandolini 1, Maria Eduarda Calil Netto 1, Jessica da Fontoura 1, Fabiana Oenning da Gama 2, Rebeca Heinzen 3
- Student of the Medicine Program. Universidade do Sul de Santa Catarina – UNISUL – Campus Pedra Branca – Palhoça (SC) Brazil.
- Nurse. Master’s in Psychopedagogy. Specialist in Intensive Care. Faculty member of the Undergraduate Medicine Program. Universidade do Sul de Santa Catarina – UNISUL – Campus Pedra Branca – Palhoça (SC) Brazil.
- Obstetrician and Gynecologist. Specialist in Mastology and Doctorate in Surgical Clinics from the Universidade de São Paulo. Faculty member of the Undergraduate Medicine Program Universidade Federal de Santa Catarina – UFSC – Campus Florianópolis – Florianópolis (SC) Brazil.
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION: Silva, GS., Dandolini, LM., et al., 2025. Temporal Trend of Breast Cancer Diagnoses in Young Women in Different Regions of Brazil from 2014 to 2022. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6441
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6441
ISSN 2375-1924
ABSTRACT
Introduction: Breast cancer is the leading cause of female mortality in Brazil, and in young women, it generally presents more aggressively, with unfavorable characteristics, advanced stages, and a higher risk of recurrence.
Objective: To analyze the temporal trend of diagnosed breast cancer cases in young women in different regions of Brazil from 2014 to 2022.
Methods: An ecological time-series study using data from the Cancer Information System (SISCAN) of the Department of Informatics of the Unified Health System from 2014 to 2022. Reported cases of breast cancer according to ICD-10 C50 were included. For statistical analysis, standardized coefficients and simple linear regression were used.
Results: An increasing trend in the overall diagnosis rate of breast cancer in young women (β=0.719; p<0.001). Increase in diagnoses in all regions, especially in the North region (β=0.888; p<0.001). Increment in all age groups, particularly among those aged 25 to 29 years, with the age group 40 to 44 years (β=1.817; p<0.001) having the highest rates. A major increase in cases of Carcinoma In Situ (β=0.047; p<0.001), with Ductal Carcinoma (β=0.394; p<0.001) being the most incident during the period.
Conclusion: The data show an increase in the rate of breast cancer in young women in Brazil, across all regions, age groups, and histological types. This highlights the need for a better epidemiological understanding of the disease to develop strategies that reduce the associated morbidity and mortality.
Keywords
Cancer. Breast cancer. Young women. Epidemiology.
INTRODUCTION
Breast cancer is a disease that currently has a significant psychological, functional and social impact on women. It is a disease that develops from the uncontrolled proliferation of abnormal breast cells and is caused by family history, medical history, advanced age, prolonged exposure to female hormones and numerous environmental factors. Moreover, genetic changes are also related to the development of this pathology, such as mutations in the BRCA1 and BRCA2 genes, which are responsible for repairing the genetic material of cells and preventing the emergence of neoplasms.
It is the most common malignant tumor in women in Brazil and worldwide, with the exception of non-melanoma skin cancer. With 2.3 million new cases in 2022, it becomes the most commonly diagnosed cancer worldwide, surpassing lung cancer. In Brazil, 94,728 new cases of breast cancer were estimated for 2022, corresponding to an adjusted incidence rate of 43.74 cases per 100,000 women. The estimated diagnosis for the triennium 2023-2025 corresponds to an estimated risk of 66.54 new cases per 100,000 women. In terms of mortality, according to the National Cancer Institute José Alencar Gomes da Silva (INCA), breast cancer is the leading cause of death among Brazilian women. When analyzing the trends in the country’s macro-regions, the data show significant regional differences, with the South and Southeast regions having the highest rates.
The age group with the highest incidence of breast cancer is concentrated between 50 and 65 years old. In this group of women, the disease tends to present more favorable molecular characteristics, which contributes to a better prognosis and a higher chance of treatment success. Additionally, women in this age group are covered by the mammographic screening program, which enables the detection of the disease in its early stages. On the other hand, the occurrence of this neoplasm in younger women is less common, representing about 7 to 10% of cases in women under 40 years old. In this population, the condition tends to be more aggressive, with unfavorable clinicopathological characteristics, diagnosis at advanced stages, and a higher risk of recurrence when compared to older women.
An important factor that contributes to the poorer prognosis in young women is the difficulty in early diagnosis. Several studies show that mammography (MMG), the main method of population screening, has low sensitivity in younger women due to the high density of breast tissue, which makes it difficult to detect suspicious changes. Furthermore, this age group is not included in public health system screening programs, which further exacerbates the situation.
In summary, recent studies have shown that due to the continental expansion and significant regional inequalities, the number of MMGs performed and the breast cancer mortality rate in Brazil are disproportionately high in the macro-regions. However, there is a lack of studies that comprehensively describe the epidemiologic profile of young women with malignant neoplasms of the breast in Brazil, comparing the five regions (South, Southeast, North, Northeast and Central-West). Therefore, the analysis of trends over time will help to collect epidemiological data that can serve as a basis for research and intervention programs for early cancer detection.
In this context, this study aims to analyze the temporal evolution of breast cancer cases diagnosed in young women in the different regions of Brazil between 2014 and 2022, in order to contribute to the planning of actions and prevention strategies.
METHODS
Ecological time series study of the temporal evolution of breast cancer cases diagnosed in young women in different regions of Brazil, based on data from the Cancer Information System database (SISCAN) provided by the Department of Informatics of the Unified Health System (DATASUS), a publicly available resource. The study analyzed breast cancer cases diagnosed in young Brazilian women between the ages of 20 and 44. All cases of malignant neoplasms of the breast registered according to the international classification code C50 (ICD-10) were included.
To determine the rate of diagnosed cases, the publicly available DATASUS database was used. Population size information for the calculation of breast cancer notification rates was obtained from the Brazilian Institute of Geography and Statistics (IBGE) based on population estimates by state, age and sex for the years 2000 to 2030.
The analysis of the temporal evolution of diagnosed cases was performed using records of breast cancer cases reported from 2014 to 2022 in Brazil and its regions. The dependent variables were the rates of total and specific cases by sex (female), age group (20 to 44 years), type of lesion and regions of the country, each expressed per 100,000 women. The independent variable was the year in which the data were collected. The period from 2014 to 2022 was selected because it represents the most recent and available data at the time of the research, ensuring an up-to-date analysis of the temporal trend.
Data organization and primary processing were performed using the TABWIN software available at DATASUS, and the data were then exported to Microsoft Excel. Data analysis was performed using the Statistical Package for the Social Sciences (SPSS), version 18.0. [Computer program]. Chicago: SPSS Inc; 2009.
For each year of the study period, the coefficients for breast cancer cases, both crude and specific, were calculated for Brazil as well as for its regions, according to the dependent variables. The temporal evolution of breast cancer cases was analyzed using standardized coefficients with the simple linear regression method. The average rate for the period was calculated, along with the percentage deviation (PV) between the rates of the first (2014) and last year (2022), the β-value by the average annual change (AAC), with a 95% confidence interval (CI) and statistical significance for p<0.05. In this method, the standardized coefficients for breast cancer cases were considered as the dependent variable and the calendar years of the study as the independent variable. The estimated model was determined using the formula Y = b0 + b1X, where Y = standardized coefficient, b0 = average coefficient for the period, b1 = average annual change, and X = year.
The study adhered to National Health Council Resolutions 466/12 and 510/16. As the study used publicly available data, it was not submitted to the Research Ethics Committee for review. The authors declare that they have no conflict of interest.
RESULTS
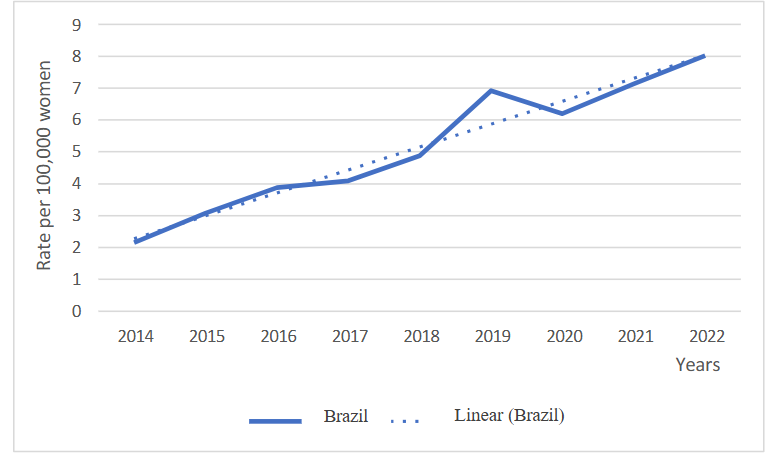
A total of 19,506 new diagnoses of breast cancer in young women aged 20 to 44 years in Brazil between 2014 and 2022 were analyzed. A trend towards an increase in the overall diagnosis rate was observed (β 0.719; p < 0.001), with an average rate of 5.15 cases per 100,000 women and an increase of 272.76% when comparing the rates of the first and last year.
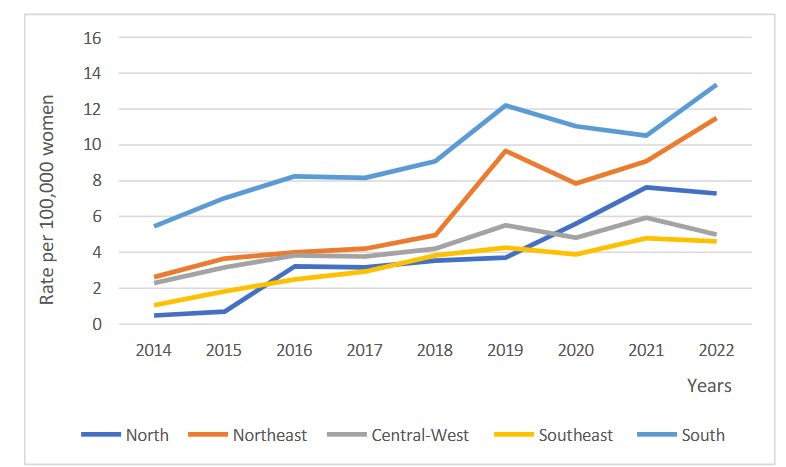
The same trend of increase was observed in all regions of Brazil. The North (β 0.888; p < 0.001), Northeast (β 1.082; p < 0.001), Central-West (β 0.382; p < 0.001), Southeast (β 0.454; p < 0.001) and South (β 0.862; p < 0.001) regions had average rates of 3.30 to 9.45 diagnoses per 100,000 women, with increases between the first and last year of 1,393.11% to 117.48%. The highest rates were observed in the South and Northeast regions, while the largest increase was seen in the North.
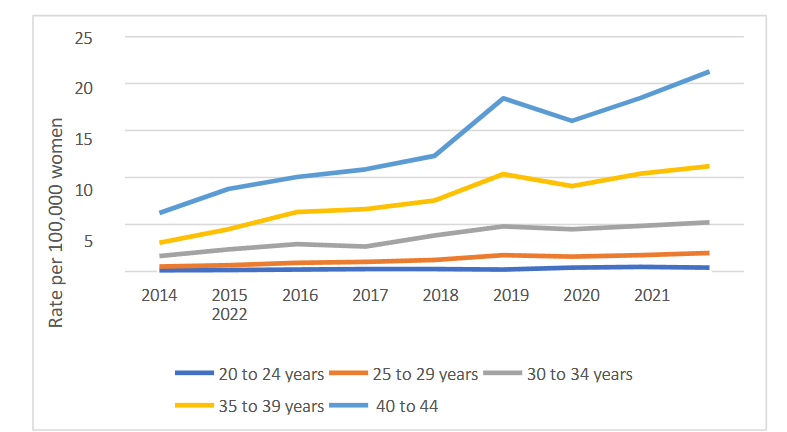
An increase was observed in all female age groups analyzed (p < 0.05) from 20 to 44 years, with average rates varying from 0.27 to 13.60 diagnoses per 100,000 women and increases ranging from 210.53% to 274.27% when rates were compared between 2014 and 2022. An increasing trend in the average annual change (AAC) was observed with increasing age. The highest percentage change (PV) was observed in the 25-29 age group (274.27%), while the highest rate and increase were seen in the 40-44 age group.
Regarding lesion types, an increase were observed in lesions classified as Carcinoma In Situ (β 0.047; p < 0.001), Ductal Carcinoma (β 0.394; p < 0.001), Invasive Lobular Carcinoma (β 0.026; p 0.007) and other classifications (β 0.243; p < 0.001), with average rates of 0.31, 3.32, 0.20 and 1.19 per 100,000 women and increases of 499.38%, 215.01%, 282.11% and 481.50%, respectively, between the first and last year analyzed. The “special” classifications remained stable during the period.
| Variables | Average Rate * | PV (%) † | AAR ‡(β) | 95% CI of AAR§ | P-value || | Trends |
|---|---|---|---|---|---|---|
| Brazil | 5.15 | 272.76 | 0.719 | 0.577 to 0.860 | <0.001 | Increase |
| North Region | 3.93 | 1393.11 | 0.888 | 0.658 to 1.118 | <0.001 | Increase |
| Northeast Region | 6.40 | 338.05 | 1.082 | 0.715 to 1.448 | <0.001 | Increase |
| Central-West Region | 4.28 | 117.48 | 0.382 | 0.217 to 0.546 | 0.001 | Increase |
| Southeast Region | 3.30 | 335.43 | 0.454 | 0.331 to 0.576 | <0.001 | Increase |
| South Region | 9.45 | 144.92 | 0.862 | 0.557 to 1.168 | <0.001 | Increase |
| 20 to 24 years | 0.27 | 210.53 | 0.041 | 0.021 to 0.060 | 0.002 | Increase |
| 25 to 29 years | 1.26 | 274.27 | 0.184 | 0.145 to 0.222 | <0.001 | Increase |
| 30 to 34 years | 3.64 | 218.99 | 0.454 | 0.339 to 0.569 | <0.001 | Increase |
| 35 to 39 years | 7.69 | 266.74 | 0.991 | 0.751 a 1.230 | <0.001 | Increase |
| 40 to 44 years | 13.60 | 241.64 | 1.817 | 1.387 a 2.246 | <0.001 | Increase |
| Carcinoma In Situ | 0.31 | 499.38 | 0.047 | 0.033 to 0.062 | <0.001 | Increase |
| Ductal Carcinoma | 3.32 | 215.01 | 0.394 | 0.252 to 0.536 | <0.001 | Increase |
| Invasive Lobular Carcinoma | 0.20 | 382.11 | 0.026 | 0.010 to 0.042 | 0.007 | Increase |
| Specials | 0.08 | 41.80 | 0.005 | -0.002 to 0.011 | 0.139 | Stability |
| Others | 1.19 | 481.50 | 0.243 | 0.169 to 0.317 | <0.001 | Increase |
DISCUSSION
Breast cancer stands out as the most common type of cancer, both in Brazil and worldwide. According to GLOBOCAN data, in 2022, 94,728 new cases were diagnosed in Brazil and 2,296,840 cases worldwide. Furthermore, in the same year, breast cancer was identified as the most prevalent in 158 countries, with Brazil ranking as the leader in Latin America regarding the incidence of this disease. These figures highlight the importance of an in-depth analysis of the factors contributing to this high incidence and the measures required to address it.
The results of this study reveal a concerning upward trend in the number of breast cancer diagnoses in young women in Brazil and across all regions of the country from 2014 to 2022. Specifically, it was found that the North region exhibited the largest percentage change in cases over this period, while the South region recorded the highest average incidence rate of this malignancy. These findings underscore the urgent need for a more detailed analysis of the factors contributing to this alarming increase, along with the implementation of effective strategies for the prevention and control of breast cancer in young women across the country.
Research has focused on understanding the factors contributing to this alarming trend, with key findings highlighting the influence of exposure to risk factors such as high-dose ionizing radiation, pesticides, organochlorines, long-term oral contraceptive use, alcohol consumption, and smoking. Genetic and hormonal factors, such as age at menarche, genetic mutations like BRCA1 and BRCA2, and family history of breast cancer, should also be considered due to their impact on the risk of breast cancer in young women.
A study involving 1,663 Brazilian patients, conducted between 2015 and 2017, demonstrated a high prevalence of pathogenic/possibly pathogenic mutations in genes such as BRCA1, BRCA2, and TP53 in young women. This reinforces the need for differentiated guidelines for monitoring and risk reduction strategies for women with hereditary breast cancer (BC) in Brazil and their family members carrying the same genetic variants.
It is also crucial to reflect on the impact of changes in lifestyle patterns, such as long working hours influencing the preference for fast food and promoting sedentary habits, which contribute to the development of obesity, a known risk factor for breast cancer. Furthermore, advances in the early detection and diagnosis of breast cancer, driven by campaigns such as Pink October, promoted by the Primary Health Care Network, may have an impact by encouraging more women to seek early information about the disease, resulting in an apparent increase in cases, with more young women being diagnosed at earlier stages.
Given Brazil’s vast regional diversity, including socio-economic, social, cultural, and healthcare access factors, significant differences in the epidemiological landscape of diseases are expected. Disparities between the South and North regions result in divergences in the incidence of diagnosed breast cancer cases, influenced by a variety of epidemiological and socio-economic factors. While the South region stands out for its more favorable socioeconomic indicators, directly impacting disease identification, such as access to preventive exams and more effective screening programs, the North faces challenges such as inadequate healthcare infrastructure, vast geographic distances, and a lack of information on prevention and healthcare, leading to later diagnoses.
One possible explanation for the sharp increase in the percentage change in case numbers in the North region may be attributed to the lack of access to adequate diagnoses for many women in previous years. Thus, with improved access to healthcare, these women gained better access to diagnoses, resulting in a higher incidence of this type of cancer. Furthermore, the highest rate of cases is found in the South region, which may be explained by its greater healthcare infrastructure and higher socioeconomic indicators. Consequently, the population in this region has better access to exams and medical consultations, both through the public system and private or health insurance providers.
When analyzing different age groups, a worrying increase in the incidence of breast cancer was observed across all examined female age groups. Specifically, in the 40 to 44-year-old group, there was a significant increase in both the rate of new cases and the percentage variation between 2014 and 2022. Additionally, it was found that the 25 to 29-year-old age group showed the largest percentage variation in the incidence of breast cancer during the study period. These findings align with the concerning trend identified in the AMAZONA III/GBECAM 0115 study, a prospective cohort study conducted in Brazil involving 2,950 women from 22 health centers distributed across nine states, diagnosed with tumors between January 2016 and March 2018. Of the participants, 43% were under 50 years old at the time of diagnosis, and 36.9% of these were at stage 3 of the disease, considered locally advanced.
The findings mentioned may be related to several factors, including late pregnancies, reduced breastfeeding, prolonged use of oral contraceptives, exposure to carcinogens, and greater reporting of diagnosed cases. It is also important to highlight that the observation of a significant proportion of women under 50 years of age diagnosed with advanced-stage breast cancer suggests the need for a review of early detection strategies and education on breast health for younger women.
Regarding histologic type, the current investigation revealed a relevant increase in the percentage change in the number of diagnosed cases of Carcinoma In Situ and identified that Ductal Carcinoma had the highest incidence rate during the study period. This phenomenon may be linked to the increasing performance of mammograms in younger women and greater access to information through technology and prevention campaigns. Additionally, the diagnosis of cancer in a woman triggers a chain reaction, motivating family members and women close to them to undergo preventive exams earlier, which may contribute to overdiagnosis of Ductal Carcinoma In Situ.
The finding that Ductal Carcinoma is the most common histologic type across different Brazilian regions corroborates previous research, which shows it as the most common pattern worldwide, both in young women and older women, with prevalence rates varying between 78% and 95.6% depending on the study.
It is important to note that the overdiagnosis rates of in situ carcinomas in screening exams vary across different studies, ranging from 10% to 52%. Some studies concluded that, for every 2,000 women screened over 10 years, only one would have her life saved by early screening, while 10 would experience overdiagnosis and overtreatment, and 200 would face psychological stress due to false positives. Consequently, the effects may encompass not only a false familial risk, generating a psychosocial impact that extends to subsequent generations, as daughters and sisters of women with breast cancer believe they are at greater risk of developing the disease, but it is also estimated that half of overdiagnosed women will suffer chronic pain due to overtreatment.
In light of this scenario, the Ministry of Health (MS) maintains the recommendation for mammography screening only for women aged 50 to 69, with the possibility of screening at younger ages in cases of high risk, such as a family history of early breast cancer or genetic mutations. This guidance is based on concerns about potential harms of the procedure in younger women, including an increase in false positive results and the harms associated with screening.
On the other hand, Brazilian medical organizations such as the Brazilian Federation of Associations of Gynecology and Obstetrics (FEBRASGO), the Brazilian Society of Mastology (SBM), and the Brazilian College of Radiology and Diagnostic Imaging (CBR) present a counterpoint to this guideline, advocating for annual mammograms for asymptomatic women aged 40 to 74, provided there are no comorbidities that reduce life expectancy by at least seven years. This recommendation is based on literature reviews that show an increase in breast cancer incidence in young women, as well as a reduction in mortality associated with mammography screening, especially among women aged 39 to 49. Moreover, Brazil, like other developing countries, has a higher proportion of breast cancer patients in this age group compared to developed countries.
Several factors limit the analysis of the results of this study. One of the issues relates to the high underreporting of diseases in Brazil, which, although significantly improved in recent years, is still influenced by demographic and socio-economic infrastructure factors in various regions. An additional challenge in the reporting and registration of breast cancer in young women lies in several factors, such as social and cultural barriers, limited access to healthcare services, socio-economic inequalities, lack of awareness and education about the disease, and the fear and stigma associated with cancer. Another factor to consider is the lack of recent scientific research on the trends in rates across different regions of Brazil, which could enable more in-depth comparative studies of the results.
In conclusion, considering the epidemiological and etiological aspects of breast cancer in young women, it is clear that it is a complex disease with various manifestations and unfavorable clinical evolutions. Despite its incidence rate showing variations across regions, it increases gradually in all of them, especially due to challenges in early diagnosis. This is exacerbated by barriers to healthcare access, such as mammograms and specialized medical consultations, and the scarcity of effective screening programs targeting younger age groups. Therefore, a better regionalized and epidemiological understanding of the disease’s progression over time becomes an essential tool for defining health promotion strategies and the appropriate allocation of resources, as well as raising awareness about the clinical picture, prognostics, and mitigation of modifiable risk factors to reduce the morbidity and mortality associated with breast neoplasms in young women.
CONCLUSION
A significant upward trend in the overall breast cancer diagnosis rate among young women in Brazil is observed. Regarding the regions of Brazil, the trend of increasing average rates was consistent across all regions, with the highest rates in the South and Northeast regions, and the greatest increase in the North region. Regarding the analyzed female age groups, all showed an increase in diagnosis rates, with the highest percentage variations observed in the 25 to 29-year-old age group and the highest incidence in the 40 to 44-year-old age group. In terms of lesion types, there was a trend towards higher diagnosis rates for carcinoma in situ, ductal carcinoma, invasive lobular carcinoma and other classifications, while cases classified as special remained stable throughout the period analyzed, with ductal carcinoma having the highest incidence.
CONFLICTS OF INTEREST STATEMENT
The authors have no conflicts of interest to declare.
ACKNOWLEDGEMENTS
We would first like to thank our advisors who were essential to the completion of this project. We would also like to thank our parents, who provided the foundation for us to reach this point today, as well as everyone who contributed in any way to our education and the completion of this work. Our sincere thanks go to all of them.
REFERENCES
- Pinheiro AB, Lauter DS, Medeiros GC, et al. Câncer de Mama em Mulheres Jovens: Análise de 12.689 Casos. Revista Brasileira de Cancerologia. 2013;59(3):351-359. doi:https://doi.org/10.32635/2176-9745.RBC.2013v59n3.500
- Thuler LC. Considerações sobre a prevenção do câncer de mama feminino. Revista Brasileira de Cancerologia. 2003;49(4):227-238. doi:https://doi.org/10.32635/2176-9745.RBC.2003v49n4.2076
- Yoshida K, Miki Y. Role of BRCA1 and BRCA2 as regulators of DNA repair, transcription, and cell cycle in response to DNA damage. Cancer Sci. 2004;95(11):866-871. doi:10.1111/j.1349-7006.2004.tb02195.
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: a Cancer Journal for Clinicians. 2021;71(3):209-249. doi:https://doi.org/10.3322/caac.21660
- Alcantara LL de M, Tomazelli JG, Zeferino FRG, Oliveira BFA de, Silva GA e. Tendência Temporal da Cobertura de Mamografias no Sistema Único de Saúde, Brasil, 2010-2019. ninhoincagovbr. 2022;68(2176-9745). https://ninho.inca.gov.br/jspui/handle/123456789/10229
- Ministério da Saúde (Brasil), Instituto Nacional de Câncer José Alencar Gomes da Silva – INCA. Estimativa 2022: Incidência de câncer no Brasil. Acessado March 20, 2023. Available at: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//dados_e_numeros_site_cancer_mama_setembro2022.pdf.
- Ministério da Saúde (Brasil), Instituto Nacional de Câncer José Alencar Gomes da Silva – INCA. Estimativa 2023: Incidência de câncer no Brasil. Acessado March 20, 2023. Available at: https://www.inca.gov.br/sites/ufu.sti.inca.local/files/media/document/estimativa-2023.pdf.
- Ministério da Saúde (Brasil), Instituto Nacional de Câncer José Alencar Gomes da Silva – INCA. Coordenação Geral de Prevenção e Vigilância, Divisão de Detecção Precoce e Apoio à Organização de Rede. Ficha técnica de indicadores relativos às ações do câncer de mama; 2014. Acessado March 20, 2023. Available at: https://www.inca.gov.br/sites/ufu.sti.inca.local/files/media/document/ficha-tecnica-indicadores-mama-2014.pdf.
- Pecinato V, Jacobo A, Silva SG da. Tendência temporal de mortalidade por neoplasia maligna de mama e de colo de útero em Passo Fundo, Rio Grande do Sul: uma análise segundo faixa etária e escolaridade, 1999-2019. Epidemiologia e Serviços de Saúde. 2023;31(2237-9622):e2022440. doi:https://doi.org/10.1590/s2237-9622202200030002.
- Dutra MC, Rezende MA, Andrade VP de, et al.. Imunofenótipo e evolução de câncer de mama: comparação entre mulheres muito jovens e mulheres na pós-menopausa. Rev Bras Ginecol Obstet. 2009;31(2):54-60. doi:10.1590/S0100-72032009000200002.
- Stival RS, Prestes AL, Mansani FP. Câncer de mama em mulheres jovens: uma análise do estadiamento clínico inicial e dos subtipos moleculares dos tumores. Rev Bras Mastologia. 2014;24(1):17-22. doi:10.5327/z201400010004rbm.
- Pollán M. Epidemiology of breast cancer in young women. Breast Cancer Res Treat. 2010;123 Suppl 1:3-6. doi:10.1007/s10549-010-1098-2.
- Yamashita H, Iwase H, Toyama T, et al. Estrogen receptor-positive breast cancer in Japanese women: trends in incidence, characteristics, and prognosis. Ann Oncol. 2011;22(6):1318-1325. doi:10.1093/annonc/mdq596.
- Anders CK, Hsu DS, Broadwater G, et al. Young age at diagnosis correlates with worse prognosis and defines a subset of breast cancers with shared patterns of gene expression. J Clin Oncol. 2008;26(20):3324-3330. doi:10.1200/JCO.2007.14.2471.
- Soares PB, Quirino Filho S, Souza WP et al. Características das mulheres com câncer de mama assistidas em serviços de referência do Norte de Minas Gerais. Rev Bras Epidemiologia. 2012;15(3):595-604. doi:10.1590/s1415-790×2012000300013.
- Soerjomataram I, Louwman MW, Ribot JG, Roukema JA, Coebergh JW. An overview of prognostic factors for long-term survivors of breast cancer. Breast Cancer Res Treat. 2008;107(3):309-330. doi:10.1007/s10549-007-9556-1.
- Rossi L, Mazzara C, Pagani O. Diagnosis and Treatment of Breast Cancer in Young Women. Current Treatment Options in Oncology. 2019;20(12). doi:https://doi.org/10.1007/s11864-019-0685-7.
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer. 2015;136(5):E359-E386. doi:https://doi.org/10.1002/ijc.29210.
- Villarreal-Garza C, Aguila C, Magallanes-Hoyos MC, et al. Breast cancer in young women in Latin America: an unmet, growing burden. The Oncologist. 2013;18 Suppl(S2):26-34. doi:https://doi.org/10.1634/theoncologist.18-S2-26.
- Eugênio DSG, Souza JA, Chojniak R, Bitencourt AGV, Graziano L, Souza EF. Breast cancer features in women under the age of 40 years. Revista da Associação Médica Brasileira. 2016;62(8):755-761. doi:https://doi.org/10.1590/1806-9282.62.08.755.
- Franco-Marina F, López-Carrillo L, Keating NL, Arreola-Ornelas H, Marie Knaul F. Breast cancer age at diagnosis patterns in four Latin American Populations: A comparison with North American countries. Cancer Epidemiology. 2015;39(6):831-837. doi:https://doi.org/10.1016/j.canep.2015.10.004.
- Garicochea B, Morelle A, Ana Elisa Andrighetti, Cancella A, Ângelo Bós, Werutsky G. Idade como fator prognóstico no câncer de mama em estádio inicial. Revista de saúde pública/Revista de Saúde Pública. 2009;43(2):311-317. doi:https://doi.org/10.1590/s0034-89102009005000009.
- Crippa CG. Câncer de Mama Em Mulheres Jovens: Estudo de Probabilidade de Sobrevida Livre de Doença.; 2002. Accessed January 23, 2025. https://repositorio.ufsc.br/bitstream/handle/123456789/84048/PMED0033-D.pdf?sequence=1.
- Hindle WH, Davis L, Wright D. Clinical value of mammography for symptomatic women 35 years of age and younger. American Journal of Obstetrics and Gynecology. 1999;180(6):1484-1490. doi:https://doi.org/10.1016/s0002-9378(99)70043-8.
- Sirovich BE, Sox HC. Breast Cancer Screening. Surgical Clinics of North America. 1999;79(5):961-990. doi:https://doi.org/10.1016/S0039-6109(05)70056-6.
- King MC, Rowell S, Love SM. Inherited breast and ovarian cancer. What are the risks? What are the choices? JAMA. 1993;269(15):1975-1980. doi:https://doi.org/10.1001/jama.269.15.1975.
- Kelsey JL, Gammon MD, John EM. Etiology of breast cancer. Cancer. 1990;65(5):1242-1251. doi:10.1002/1097_0142(19900301)65:5<1242::AID-CNCR2820650535>3.0.CO;2-1.
- Gonzaga CMR. Mortalidade por câncer de mama nas mulheres brasileiras no período de 1980 a 2009. Biblioteca digital de teses e dissertações. Published September 30, 2011. Accessed March 20, 2024. http://repositorio.bc.ufg.br/tede/handle/tde/1750.
- Rocha-Brischiliari SC, Oliveira RR de, Andrade L, et al. The Rise in Mortality from Breast Cancer in Young Women: Trend Analysis in Brazil. Ahmad A, ed. PLOS ONE. 2017;12(1):e0168950. doi:https://doi.org/10.1371/journal.pone.0168950.
- Barbosa IR, Costa I do CC, Pérez MMB, Souza DLB de. Mortalidade por câncer de mama nos estados do nordeste do Brasil: Tendências atuais e projeções até 2030. Revista Ciência Plural. 2015;1(1):04-14. https://periodicos.ufrn.br/rcp/article/view/7318.
- Soares P dos R. Tendência Temporal de Mortalidade Por Câncer de Mama Em Mulheres No Brasil de 2000 a 2017.; 2020. Accessed March 20, 2023. https://repositorio.animaeducacao.com.br/bitstream/ANIMA/15444/1/artigo%20cient%c3%adfico%20P%c3%a2mela%20dos%20Reis%20Soares.pdf.
- Fitzmaurice C, Dicker D, Pain A, et al. The Global Burden of Cancer 2013. JAMA oncology. 2015;1(4):505-527. doi:https://doi.org/10.1001/jamaoncol.2015.0735.
- The International Agency for Research on Cancer (IARC). Global Cancer Observatory. gco.iarc.fr. Published 2022. https://gco.iarc.fr/en.
- Azevedo e Silva G, Bustamante-Teixeira MT, Aquino EML, Tomazelli JG, dos-Santos-Silva I. Acesso à detecção precoce do câncer de mama no Sistema Único de Saúde: uma análise a partir dos dados do Sistema de Informações em Saúde. Cadernos de Saúde Pública. 2014;30(7):1537-1550. doi:https://doi.org/10.1590/0102-311×00156513.
- Guindalini RSC, Viana DV, Kitajima JPFW, et al. Detection of germline variants in Brazilian breast cancer patients using multigene panel testing. Scientific Reports. 2022;12(1):4190. doi:https://doi.org/10.1038/s41598-022-07383-1.
- Kluthcovsky ACGC, Faria TNP, Carneiro FH, Strona R. Female breast cancer mortality in Brazil and its regions. Revista Da Associacao Medica Brasileira. 2014;60(4):387-393. doi:https://doi.org/10.1590/1806-9282.60.04.019.
- Silva GA e, Souza-Júnior PRB de, Damacena GN, Szwarcwald CL. Early detection of breast cancer in Brazil: data from the National Health Survey, 2013. Revista de Saúde Pública. 2017;51(suppl 1). doi:https://doi.org/10.1590/s1518-8787.2017051000191.
- Vieira RA da C, Formenton A, Bertolini SR. Breast cancer screening in Brazil. Barriers related to the health system. Revista da Associação Médica Brasileira. 2017;63(5):466-474. doi:https://doi.org/10.1590/1806-9282.63.05.466.
- Franzoi MA, Rosa DD, Zaffaroni F, et al. Advanced Stage at Diagnosis and Worse Clinicopathologic Features in Young Women with Breast Cancer in Brazil: A Subanalysis of the AMAZONA III Study (GBECAM 0115). Journal of Global Oncology. 2019;5(5):1-10. doi:https://doi.org/10.1200/jgo.19.00263.
- Silva MM da, Silva VH da. Envelhecimento: importante fator de risco para o câncer. Arquivos Médicos do ABC. 2005;30(1).
- Pinheiro AB, Lauter DS, Medeiros GC, et al. Câncer de Mama em Mulheres Jovens: Análise de 12.689 Casos. Revista Brasileira de Cancerologia. 2013;59(3):351-359. doi:https://doi.org/10.32635/2176-9745.RBC.2013v59n3.500.
- Bleyer A, Welch HG. Effect of Three Decades of Screening Mammography on Breast-Cancer Incidence. New England Journal of Medicine. 2012;367(21):1998-2005. doi:https://doi.org/10.1056/nejmoa1206809.
- Independent UK Panel on Breast Cancer Screening. The benefits and harms of breast cancer screening: an independent review. The Lancet. 2012;380(9855):1778-1786. doi:https://doi.org/10.1016/s0140-6736(12)61611-0.
- Jorgensen KJ, Gotzsche PC. Overdiagnosis in publicly organised mammography screening programmes: systematic review of incidence trends. BMJ. 2009;339(jul09 1):b2587-b2587. doi:https://doi.org/10.1136/bmj.b2587.
- Gøtzsche PC. Screening for breast cancer with mammography. The Lancet. 2001;358(9299):2167-2168. doi:https://doi.org/10.1016/s0140-6736(01)07198-7.
- Gøtzsche PC, Jørgensen KJ. Screening for breast cancer with mammography. Cochrane Database of Systematic Reviews. 2013;6(6). doi:https://doi.org/10.1002/14651858.cd001877.pub5.
- Tesser CD, d’Ávila TL de C. Por que reconsiderar a indicação do rastreamento do câncer de mama? Cadernos de Saúde Pública. 2016;32(5). doi:https://doi.org/10.1590/0102-311×00095914.
- Migowski A, Silva GA e, Dias MBK, Diz MDPE, Sant’Ana DR, Nadanovsky P. Diretrizes para detecção precoce do câncer de mama no Brasil. II – Novas recomendações nacionais, principais evidências e controvérsias. Cadernos de Saúde Pública. 2018;34(6). doi:https://doi.org/10.1590/0102-311×00074817.
- Urban LABD, Chala LF, Bauab SDP, et al. Radiol Bras. Jul/Ago. 2017;50(4):244-249. doi:https://doi.org/10.1590/0100-3984.2017-0069.
- Gonzaga CMR, Freitas-Junior R, Souza MR, Curado MP, Freitas NMA. Disparities in female breast cancer mortality rates between urban centers and rural areas of Brazil: Ecological time-series study. The Breast. 2014;23(2). https://www.thebreastonline.com/article/S0960-9776(14)00007-1/fulltext.
- Badan GM, Roveda Júnior D, Ferreira CAP, Junior OA de N. Complete internal audit of a mammography service in a reference institution for breast imaging. Radiologia Brasileira. 2014;47(2):74-78. doi:https://doi.org/10.1590/s0100-39842014000200007.
- Tabár L, Vitak B, Chen THH, et al. Swedish two-county trial: impact of mammographic screening on breast cancer mortality during 3 decades. Radiology. 2011;260(3):658-663. doi:https://doi.org/10.1148/radiol.1111.

