MMP-2 in Bone Marrow Metastasis Post-FOLFOX in Colon Cancer
Decreased expression of matrix metalloproteinase-2 in bone marrow micro-metastasis after FOLFOX adjuvant chemotherapy for stage III colon cancer is association with an increased progression free survival and the elimination of circulating tumour cells
Murray N.P. ¹, Villalon R. ², Hartmann D. ³
¹ Faculty of Medicine, University Finis Terrae, Santiago, Chile.
² Department of Coloproctology, Clinica Davila, Santiago Chile.
³ Faculty of Medicine, Universidad de Chile, Santiago Chile.
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION Murray, NP., Villalon, R., et al., 2025. Decreased expression of matrix metalloproteinase-2 in bone marrow micro-metastasis after FOLFOX adjuvant chemotherapy for stage III colon cancer is association with an increased progression free survival and the elimination of circulating tumour cells. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6365
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i3.6365
ISSN 2375-1924
ABSTRACT
Introduction: FOLFOX adjuvant chemotherapy is standard practice after curative surgery for stage III colon cancer. Even so there a significative number of patients who relapse. This study investigates the expression of metallo-proteinase (MMP-2) in bone marrow micro-metastasis and the progression free survival in these patients.
Methods: Bone marrow and blood samples were taken before and after chemotherapy to detect CEA expressing cancer cells and the expression of MMP-2.
Results: Patients with bone marrow micro-metastasis expressing MMP-2 after chemotherapy had a lower progression free survival rate and a shorter time to relapse.
Conclusions: The detection of MMP-2 positive micro-metastasis or CTCs may provide useful information of the prognosis of stage III colon cancer patients.
Keywords: colon cancer; micro-metastasis; MMP-2
INTRODUCTION
After curative surgery for stage III colon cancer, adjuvant chemotherapy is recommended, FOLFOX (5-flurouracil, oxaliplatin and folinic acid) is one such treatment protocol (NCCN guidelines). Without adjuvant therapy approximately 30% of patients treated with curative surgery as monotherapy will relapse within 5 years. According to the report by Salvatore et al adjuvant chemotherapy reduced the relative risk of relapse by 33% and in 10-15% of treated patients the overall survival increased by 10-15%. Adjuvant chemotherapy is used to try to eliminate minimal residual disease (MRD) which is defined as microscopic foci of disseminated tumours not detected by imaging techniques such as positron emission tomography-computerised tomography or magnetic resonance imaging. Three types of MRD have been described for patients with colon cancer; Group 1 includes patients negative for bone marrow micro-metastasis and circulating tumour cells, Group 2 those patients with only bone marrow micro-metastasis and Group 3 positive for both. Group 1 has the best prognosis, followed by Group 2 which a relatively long period of dormancy before relapse occurs and finally Group 3 with a poor prognosis and early relapse.
Matrix metalloproteinase-2 is a type IV collagenase and plays a role in many stages of the metastatic cascade. This has been recently reviewed. In summary, it permits the cancer cells pass through the basement membrane and extra-cellular matrix to enter the circulation and to implant in distant tissues. It also releases cytokines and chemokines that cause immune suppression both in the pre-metastatic and metastatic niches. Cancer cells that express MMP-2 have been associated with the presence of circulating tumour cells and a worse progression free survival. The fact that after FOLFOX chemotherapy patients may relapse implies the presence of minimal residual disease (MRD) has been defined as the presence of cancer cells in distant tissues that are too small to be detected using radiological studies. That the use of FOLFOX improves progression free survival implies that it eliminates these cancer cells or can maintain them in a state of latency for a variable period. Although metastasis to bone, detected using radiological methods are infrequent, it has been reported that up to 30% of patients may have bone marrow micro-metastasis detected.
We present a prospective observational study of the changes in the expression of MMP-2 in bone marrow micro-metastasis before and after FOLFOX chemotherapy and its association with progression free survival.
MATERIALS AND METHODS
A single centre prospective observational study of consecutive patients with stage III colon cancer treated with FOLFOX adjuvant chemotherapy, evaluated for MRD between January 2007 and December 2014. The STROBE guidelines for cohort studies were used and included all patients with pathological Stage III colon cancer who had negative surgical margins and no evidence of metastasis based on CT images. Patients who had received neo-adjuvant chemotherapy or had previous malignancies were excluded from the study group.
CTC detection:
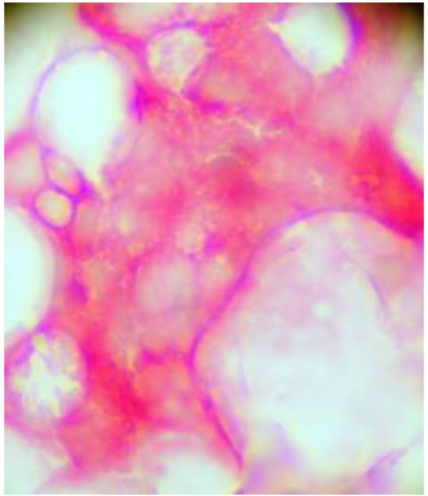
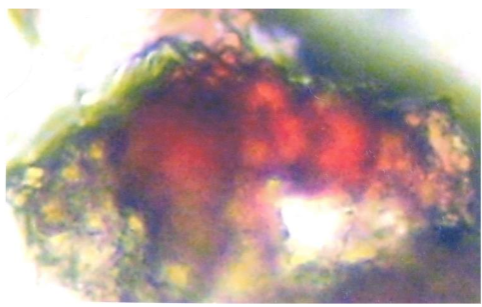
Three tubes containing EDTA (Becton, Vacutainer USA) were drawn from the cubital vein, the first tube was discarded to avoid contamination by epithelial cells. The samples were processed within 24 hours while being maintained at room temperature. To obtain mononuclear cells the sample was centrifuged with Histopaque 1.077® (Sigma-Aldrich, USA) according to the instructions of the manufacturers and resuspended in 100µl of autologous plasma. Using 25 µl of this cell suspension four slides were prepared using sialinized slides, (DAKO, USA). These were air dried for 24 hours and fixed using a solution of 70% ethanol, 5% formaldehyde and 25% of phosphate buffered saline (PBS) for five minutes, then washed twice in PBS. The slides were then incubated one hour later at room temperature with the monoclonal antibody anti-CEA clone 11-7 (DAKO, USA) in a humified chamber for one hour. To detect CEA positive cells a system based on alkaline phosphatase-anti-alkaline phosphatase was used (DAKO, USA) and neofuschin used as the chromogen to stain the positive cells red. If the cells stained positive for CEA a second stage was carried out using anti-MMP-2 clone 1B4 (Novacastra Laboratories, UK) with a peroxidase-based system for identifying MMP-2 positive cells (LSAB2, DAKO, USA) and with 3,3 diaminobenzidine tetrahydrochloride (DAB) as the chromogen.
Bone marrow micro-metastasis detection:
From the posterior iliac crest, a bone marrow biopsy was taken prior to the chemotherapy and one-month after completing FOLFOX. Four touch preparations were made for the detection of CEA expressing cancer cells, the slides were processed as processed in the same way as for CTCs. Cells expressing CEA were defined as micro-metastasis and positive samples underwent a second process to detect the co-expression of MMP-2 as described previously.
Classification of patients:
The patients were classified into three groups; those patients who were negative for both CTCs and micro-metastasis; a second group of patients who were only positive for bone marrow micro-metastasis expressing CEA with or without the expression of MMP-2 and finally a third group who were positive for CTCs and bone marrow micro-metastasis.
Follow-up:
Three monthly check-ups were performed: with a colonoscopy and a thoracic, abdominal and pelvic computerised tomography being performed. If these proved to be negative, the same controls were performed yearly. At the same time blood and bone marrow samples were taken to evaluate if there were changes in the MRD classification. A relapse was defined as a positive finding either by colonoscopy or CT imaging. The time from finishing the chemotherapy to a positive result by either method defined the progression free survival time.
MMP-2 classification:
The expression of MMP-2 was classified according to the criteria of Trudel et al. If 10% or more of detected CEA cells were positive for the co-expression of MMP-2, the sample was considered as being MMP-2 positive. Samples which were CEA positive, but only 1-9% of these cells co-expressed MMP-2 were classified as weakly positive and samples which showed CEA positive cells but were negative for the expression of MMP-2 were classified as negative. The intensity of the staining for MMP-2 of each CEA positive cell was classified using a scale of 0, +1, +2 and +3 and thus a median MMP-2 score could be calculated.
Study endpoints:
- Progression free survival from finishing chemotherapy according to MRD subtypes.
- Changes in the expression of MMP-2 in patients who change MRD sub-type after chemotherapy.
- If there was no change, did this affect progression free survival.
Statistical analysis:
The median values with the inter-quartile range (IQR) as a measure of distribution were used to measure the differing variables. The Chi-squared test was used to compare the different frequencies of these variables and the Mann-Whitney test to compare the median values of these variables. The Kaplan-Meier test was used to determine the progression free survival of the three MRD sub types and the mean restricted progression free survival rate was calculated. A P-value of < 0.05 was used to define statistical significance.
Ethical Considerations:
The study was approved by the local ethics committee and performed in complete conformity with the Declaration of Helsinki and all the patients signed written informed consent forms.
RESULTS
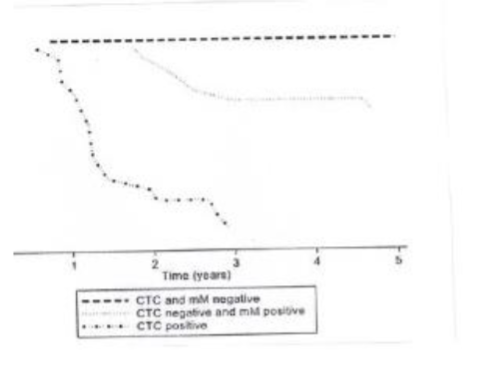
A total of 188 patients participated in the study, none of these patients were lost to follow up with a median follow up time of 4.1 years (IQR 3.8-4.4 years). A total of 82 (43%) patients relapsed after FOLFOX chemotherapy. Of the three subtypes of MRD pre-chemotherapy there were 48 (25.9%) patients that formed Group 1; 52 (28.1%) patients formed Group 2 and finally 88 (46%) of patients formed Group 3. The response to chemotherapy was heterogeneous and the subtype of MRD did not predict the response to treatment. Three months after finishing FOLFOX chemotherapy blood and bone marrow samples classified the patients as the following: 95 (50.5%) of patients were classified as Group 1; 37 (19.7%) of patients were classified as Group 2 and finally 56 (29.8%) were classified as Group 3. The classification of the three sub-groups of MRD determined the progression free survival after chemotherapy as demonstrated in the Kaplan. Meyer analysis. The progression free survival rates are shown in Table 1. Those patients which formed Group 1 had the best progression free survival while those forming Group 3 had a significantly shorter time to the appearance of metastasis; this is shown in Table 1.
| MRD subtype | % Observed progression free survival rate (median and IQR) | 95% CI RMPFST in years | 95% CI |
|---|---|---|---|
| Group 1 | 88% (66%-96%) | 4.6 (4.2-5.0) | |
| Group 2 | 56% (31%-75%) | 4.1 (3.4-4.7) | |
| Group 3 | 6% (0.5%-2.5%) | 1.3 (0.9-1.9) |
After completing FOLFOX adjuvant chemotherapy a total of 72 (40%) changed their MRD sub-group status. Some deteriorating in their subgroup while others improved their MRD sub-group.
| MRD Group | Pre-chemotherapy | Post-chemotherapy |
|---|---|---|
| Group 1 N=48 | Group 1 48 (100%) | Group 1 N= 47 |
| Group 2 N =13 | Group 2 N= 52 | Group 1 N = 29 |
| Group 3 N =8 | Group 3 N= 88 | Group 1 N = 14 |
The results show that even after FOLFOX adjuvant chemotherapy the patient may still be at a higher risk of relapse. Possibly because the chemotherapy is selecting resistant cancer cells. The alternate is that the bone marrow did not harbour cancer cells and the patients relapsed from visceral micro-metastasis.
| Group 2 N =52 | Group 3 N = 88 | |
|---|---|---|
| MMP-2 > 10% | 2 (4%) | 9 (10%) |
| MMP-2 1-10% | 4 (8%) | 53 (60%) |
| MMP-2 0% | 46 (88%) | 26 (30%) |
| Mean +/- SD | 0.29 +/- 0.21 | 1.95 +/- 0.4 |
Table 4 shows in the frequency and median MMP-2 scores who changed their MRD subgroup after chemotherapy. Those patients who improved their MRD subgroup has a decrease in the expression of MMP-2, while those who worsened their MRD sub-group increased the expression of MMP-2 in the bone marrow micro-metastasis.
| Changed to Group 2 N =31 | Changed to Group 3 N =8 | |
|---|---|---|
| MMP-2 > 10% | 4 (13%) | 8 (100%) |
| MMP-2 1-10% | 6 (19%) | 0 (0%) |
| MMP-2 0% | 21 (68%) | 0 (0%) |
| Mean +/- SD | 0.35 +/- 0.28 | 2.27 +/- 0.51 |
DISCUSSION
The use of FOLFOX adjuvant chemotherapy has improved the progression free survival and overall survival of patients with stage III colon cancer. Chemotherapy not only is able to eliminate proliferating cancer cells but also may cause changes in the host immunological system. Both 5-fluorouacil and oxaliplatin have been reported to improve the anti-tumour immune response. Increasing the function of cytotoxic T-cell and decreasing myeloid derived suppressor cells and regulatory T-cells. This article raises the question does FOLFOX chemotherapy improve the immune function or by decreasing MMP-2 expression improves the immune function to improve the progression free and overall survival rates? 5-fluorouracil is a pyrimidine analogue which inhibits the enzyme thymidylate synthase essential for nucleotide synthesis, whereas oxaliplatin induces tumour cell apoptosis by covalently binding to DNA and thus causing inhibition of DNA synthesis. The micro-metastatic site is not only comprised of cancer cells, but they are found within their own tumour micro-environment. As reported here, FOLFOX adjuvant therapy may eradicate tumour cells, decrease the expression of MMP-2 without eliminating the cancer cells or have no effect. This may be explained in part by the tumour micro-environment (TME), which is composed of tumour cells, immune cells which cause local immunosuppression, via T-regulatory lymphocytes (Tregs) immunosuppression macrophages, chemokines and cytokines and decreased numbers of effector cells such as cytotoxic T-cells and NK cells. This TME is dynamic and changes with time due to the clonal instability of the tumour cells and changes in the immunosuppressive effects within the micrometastasis. The TME may be different in different tissues, explaining the different frequencies of the metastatic sites. Both 5-fluorouracil and oxaliplatin affect the TME; oxaliplatin induces immunogeneic cells death (ICD) by increasing the immunogenicity of the tumour cells. It increases cytotoxic T-cells and decreases myeloid derived stem cells and Tregs. Although the cytotoxicity is non-specific and can decrease the total lymphocyte population for a short time, the changes in the immune TME are longer lasting. Oxaliplatin is one chemotherapeutic drug that may block immunoinhibitory signal pathways, increasing anti-tumour immunity. 5-Fluorouracil induces ICD via the Calreticulin endoplasmic reticulum chaperone. Its translocation to the plasma membrane where it binds to CD91 which starts a process whereby dendritic cells, the release of pro-inflammatory cytokines and activation of T-helper cells are recruited to the micro-metastasis thereby, increasing the anti-tumour immune response. It also decreases the immunosuppression caused by the TME affecting both Tregs and myeloid derived suppressor cells. This a dynamic process and may change with subsequent treatment cycles.
The dual effects of cytotoxicity and changes in the immune response would explain why some patients improved in their MRD status, similarly those tumour cells resistant to chemotherapy or with a higher level of immune function would not be affected, explaining why some patients did not change their MRD subgroup or passed to a more advanced MRD subgroup. What is more difficult to explain is why decreased or the absence of MMP-2 expression changed after FOLFOX chemotherapy. MMP-2 is secreted as a pro-enzyme achieving its active form via MT-MMP-1. Tissue inhibitors of metalloproteinases TIMP-1 and TIMP-2 controlling the activity of MMP-2. Higher levels of TIMP-1 in the plasma and primary tumour before curative surgery have been associated with a worse prognosis. However, it has also been reported that preoperative serum analyses of matrix metalloproteinases (MMP-2, MMP-9) and tissue inhibitors of matrix metalloproteinases (TIMP-1 and TIMP-2) in colon cancer have limited value for determining the prognosis of patients. They may also not reflect the TIMP levels in the micro-metastasis or the ratio MMP-TIMP. It was beyond the scope of this study to detect TIMP 1 and TIMP 2 levels. Even so this would not answer the question of how MMP-2 expression was decreased or absent. TIMP-1, 2 and 4 both promote and inhibit tumour development, while TIMP-3 has been reported to be consistently anti-tumour in its effect.
As mentioned previously, the expression of MMP-2 in bone marrow micro-metastasis is associated with a higher frequency of relapse and the presence of CTCs after FOLFOX chemotherapy for stage III colon cancer. MMP-2 expression and the presence of CTCs was associated with a significantly higher relapse rate and a shorter progression free survival with 6% having a five-year progression free survival rate and time to relapse being only 1.3 years. Group 2 patients had a longer latency period where dormant cancer cells do not proliferate, however due to clonal instability or changes in the tumour micro-environment such as immunosuppression. These changes in the phenotypic characteristics of the cancer cells caused an increased frequency of progression free survival decreases, although the time to relapse was similar to those patients who formed Group 1. Therefore, identifying patients who form Group 2 may provide information and the formation of personalized treatments to maintain the cancer cells dormant.
The key question raised by this study is that if MMP-2 expression could be suppressed or decreased in an asymptomatic patient could the progression free survival be prolonged? Unfortunately, trials using anti-metalloproteinases have been stopped due to the adverse effects caused by these drugs. Batimastat did not reach Phase 1 clinical trials; while marimastat, tanomestat and prinomastat in Phase III trials did not show a survival benefit.
CONCLUSIONS
The expression of MMP-2 in bone marrow micro-metastasis after FOLFOX chemotherapy for stage III colon cancer is associated with the presence of CTCs, a lower and shorter progression free survival time. Further multi-center trials are needed to confirm these results. The mechanisms whereby MMP-2 expression is decreased in some patients after FOLFOX needs to be elucidated so as possibly increase the latency period.
FUNDING
The study was supported by a Western Health Authority Research Grant.
CONFLICTS OF INTEREST
The authors reported no conflicts of interest.
REFERENCES
- NCCN guidelines 2008 http://www.nccn.org/professionals/physicians_gls/pdf/colon.pdf
- Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet 2014: 383; 1490-1502 http://doi.org10.1016/S0140(13)61649-9
- Salvatore I, Imperatori M, Arnold E et al. Management of patients with early-stage colon cancer: guidelines of the Italian Medical Oncology Association. ESMO open 2020 5; e001001 http:/doi.org:10.1136/esmoopen-2020-001001
- Murray NP, Villalon R, Hartmann D, Rodriguez MP, Aedo S (2021) Improvement in immune function after FOLFOX chemotherapy for stage III colon cancer is associated with improved minimal residual disease prognostic subtype and outcome. Colorectal Dis 2021; 23: 2879-2893 http:/doi.org:/10.1111/codi.15899
- Murray NP. (2024) The role of matrix metalloproteinase-2 in the metastatic cascade: a review. Oncologia 26 27-40. xxx
- Lu W, Kang Y. (2019) Epithelial-mesenchymal plasticity in cancer progression and metastasis. Dev Cell 2019; 49: 361-374 http://doi.org:10.1016/j.devcel.2019.04.10
- Bhattacharya R, Panda CK, Nandi S, Mukhopahyay A. An insight into metastasis: Random or evolving paradigms? Pathol Red Pract 2018; 214: 1064-1067 http://doi.org/10.1016/j.prp.2018.06.017
- Celia-Terrassa T, Kang Y. Metastatic niche functions and therapeutic opportunities. Nat Cell Biol 2018;20: 868-877 http://doi.org/10.1038/s41556-018-01459
- Viehl CT, Weixler B, Guller U, Dell-Kuster S, Rosenthal R, Ramser M et al. (2017). Presence of bone marrow micro-metastasis in stage I-III colon cancer patients is associated with worse disease free and overall survival. Cancer Med 2017; 6: 918-927 http://doi.org/10.1002/cM4.1056
- Ramser M, Warchkow R, Viehl CT, Kettelhack C, Zettl A, Lobbes LA et al. The simultaneous presence of isolated tumour cells and bone marrow micrometastasis in Stage I and II colon cancer-challenging the theory of a chronological pathway of tumour cell dissemination. World J Surg 2022; 46: 680-689 http://doi.org/10.1186/s12885-016-2239-8
- Trudel D, Fradet Y, Meyer F, Harel F, Tetu B. (2009) Significance of MMP-2 expression in prostate cancer: an immunohistocehmical study, Cancer Res 2003; 63; 8511-8515
- Royston P, Parmar MK. The use of restricted mean survival time to estimate the treatment effect in randomized clinical trials when the proportional hazards assumption is in doubt. Stat Med. 2011; 30(19):2409-21. http://doi.org/10.1002/sim.1274
- Gmeiner WH. Fluoropyridine modulation of anti-tumor immune-response prospects for improved colorectal cancer treatment. Cancer 2020; 12: 1641 http://doi.org/10.103390/cancers.1206.1641
- Li X, Slaton WB. (2013) Molecular mechanisms of platelet and stem cell rebound after 5-fluorouracil treatment. Exp Hematol 2013; 41: 635-645 http://doi.org/10.1016/j.exphem.2013.03.003
- Stojanoska V, Prakash M, Mc Quade R, Fraser S. Oxaliplatin alters systemic immune response. Biomed Res Int 2019.4650695 http://doi.org/10.1155/2019/4650695
- Wu T, Dai Y. Tumor microenvironment and therapeutic response. (2017) Cancer Lett 387; 61-68 http://doi.org/10.1016/j.canlet.2016-01-043
- Gou HF, Zhou L, Huang J, Chen XC. Intraperitoneal oxaliplatin administration inhibits the tumour micro-environment in an abdominal implantation model of colon cancer. Mol Med Rep 2018; 18: 2335-2341 http://doi.org/10.3892/mmr.2018.9219
- Restifo NP, Dudley ME, Rosenberg SA. Adaptive immunotherapy for cancer: Harnessing the T-cell response. Nat Rev Immunol 2012; 12: 269-281 http://doi.org/10.1038/nri3191
- Dudley ME, Wunderlich JR, Robbins PF, Yang JC, Hwu P, Schwartzentruber DJ et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumour lymphocytes. Science 2002; 298: 850-854 http://doi.org/10.1126/sciebnce.1076514
- Alizadeh D, Larmonier N. Chemotherapeutic targeting of cancer-induced immunosuppressive cells. Cancer Res 2014; 74: 2663-2668 http://doi.org/10.1158/0008-5472.CAN-14-0301
- Ruan H, Leibowitz BJ, Zhang L, Yu J. Immunogenic cell death in colon cancer prevention and therapy. Mol Carcinog 2020; 59: 783-793 http://doi.org/10.1002/mc.23183
- Gong Y, Scott E, Lu R, Xu Y, Oh WK, Yu Q. TIMP-1 promotes accumulation of cancer associated fibroblasts and cancer progression. PlusOne 2013; 8: e77366 hhtp://doi.org/10.1371/journal.pone.0077366
- Bockelman C, Beilmann-Lehtonen I, Kaprio T, Koskensalo S, Tervahartiala T, Mustonen H et al. Serum MMP-8 and TIMP-1 predict prognosis in colorectal cancer. BMC Cancer 2018; 18: 679 http://doi.org/10.1186/s12885-018-458x
- Oberg A, Hoyhtya M, Tavelin R, Stenling R, Lindmark G. Limited value of preoperative serum analyses of matrix metalloproteinases (MMP-2, MMP-9) and tissue inhibitors of matrix metalloproteinases (TIMP-1 and TIMP-2) in colon cancer. Anticancer Res 2000; 20 (2B): 1085-1091
- Rai GP, Baird SK. Tissue inhibitor of matrix metalloproteinase-3 has both anti-metastatic and anti-tumourigenic properties. Clin Exp Metastasis 2020; 37: 69-76 http://doi.org/10.1007/s10585-019-10017-y
- Teng MW, Swan JB, Koebel CM, Schrieber RD, Smyth MJ. Immune mediated dormancy: an equilibrium with cancer. J Leukoc Biol 2008; 84: 988-993 http://doi.org/10.1189/jlb.1107774
- Rasmussen HS, Mc Cann PP. Matrix metaproteinase inhibition as a novel cancer agent: a review with special focus on batimastat and marimastat. Pharmacol Ther 1997; 75: 69-75 http://doi.org/10.1016/s0163-7258(97)00023-5
- Bramhall S, Hallissey M, Whiting J, Tierney G, Stuart R, Hawkins R et al. Marimastat as maintenance therapy for patients with advanced gastric cancer- a randomized trial. Br J Cancer 2002; 86: 1864-1870 http://doi.org/10.1038/sj.bjc.6600816
- Hirte H, Vergote IB, Jeffery JR, Grimshaw S, Coppieters B, Schwartz B et al. A phase III randomized trial of BAY12-9566 (tanomastat) as maintenance therapy in patients with advanced ovarian cancer responsive to primary surgery and paclitaxel/platinum containing chemotherapy: a National Cancer Institute of Canada Clinical Trials Group Study. Gynecol Oncol 2006, 102: 300-308 http://doi.org/10.1016/j.ygyno.2005.12.020
- Bissett D, O´Bryne KJ, von Pawel J, Gatzmeier U, Price A, Nicolson M et al. (2005) Phase III study of matrix metalloproteinase inhibitor prinomastat in non-small cell lung cancer. J Clin Oncol 2005; 23: 842-849 http://doi.org/10.1200/jco.2005.03.170
- Bain B. Bone marrow biopsy morbidity: review of 2003. J Clin Pathol 2005; 58: 406-408 http://doi.org/10.1136/jcp.2004.0221178

