Hip Muscle Geometry After Transfemoral Amputation
Hip muscle geometry and function following a transfemoral amputation – a cross-sectional study protocol
Reslin Schelhaas1,2, Klaske van Kammen1, Han Houdijk3, Richard Reijmes4, Paul C Jutte5, Rienk Dekker1, Jan HB Geertzen1
- University of Groningen, University Medical Center Groningen, Department of Rehabilitation Medicine, Groningen, The Netherlands.
- Hanze University of Applied Sciences, Occupational Therapy, Groningen, The Netherlands.
- University of Groningen, University Medical Center Groningen, Department of Human Movement Sciences, Groningen, The Netherlands.
- Hanze University of Applied Sciences, Department of Medical Imaging and Radiation Therapy, Groningen, The Netherlands.
- University of Groningen, University Medical Centre Groningen, Department of Orthopedic Surgery, Groningen, the Netherlands.
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Schelhaas, R., van Kammen, K., et al., 2025. Hip muscle geometry and function following a transfemoral amputation – a cross-sectional study protocol. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6120
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i2.6120
ISSN: 2375-1924
ABSTRACT
Background: Individuals with a transfemoral amputation often experience limited walking abilities and difficulties with balance. These difficulties may arise not only from the loss of a limb but also from changes in the muscles in the residual limb post-amputation, such as muscle atrophy, increased intermuscular fat, and alterations in muscle architecture. This can result in loss of muscle function. Individuals with a transfemoral amputation indeed show decreased muscle strength in the residual limb compared to the intact limb. Although research has shown that muscle strength deficits in individuals with a transfemoral amputation are associated with difficulties in daily functioning, residual limb control, and walking performance, the specific role of residual muscle adaptations in contributing to these strength deficits and functional challenges remains unclear. Therefore, this paper presents a cross-sectional study protocol to investigate hip muscle geometry and function following transfemoral amputation, with the goal of understanding muscle adaptations and their functional consequences. Additionally, the influence of various amputation related factors, including the surgical technique, will be investigated.
Methods: Individuals aged 18 years or older who have undergone a unilateral transfemoral amputation within the past 1-5 years and are actively using a prosthesis (K-level ≥ 2) will be recruited. Participant characteristics such as age, sex, weight, height, cause, and date of amputation will be collected via a questionnaire. Study measurements will include a 1,5T Magnetic Resource Imaging scan to gather data on muscle geometry and femur length for both the residual and intact limbs, and a dynamometer strength test (isokinetic and isometric) to assess the hip muscle strength for each muscle group. The L Test of Functional Mobility will be performed to evaluate physical function. If possible, participants will be grouped based on the surgical technique used (myodesis vs. myoplasty) to allow group comparisons. Within-subjects differences will be assessed as the differences between the intact and residual limb, and Pearson’s correlation coefficients will be used to examine the relationships between muscle geometry and hip muscle strength. Ethical approval has been obtained by the Medical Ethical committee of the University Medical Center Groningen (METc 2024/392).
Keywords
transfemoral amputation, hip muscle geometry, muscle strength, rehabilitation, myodesis, myoplasty
INTRODUCTION
Individuals with a transfemoral amputation (TFA) often face challenges with walking and maintaining balance. These functional limitations could be attributed to the anatomical changes that occur following amputation. A TFA results not only in the loss of structures below the amputation level, but also in adaptations in the residual structures above amputation level. Understanding how these adaptations affect functionality is crucial for optimal rehabilitation, making informed surgical decisions, and understanding the possibilities of regaining mobility.
Studies have shown that following TFA, there is muscle atrophy and an increase in intermuscular fat in the residual limb, both of which can potentially impair muscle functioning. In addition to these changes in muscle volume and fat composition, the architecture of muscles is also altered following TFA. Jaegers et al. observed that some transected hip muscles were reattached in different positions – either more ventral, dorsal, medial, or lateral – relative to the femur. These changes in muscle reattachment altered the muscle’s line of action compared to the muscles on the intact side. This shift in the line of action alters the muscle’s moment arm and the direction of the force relative to the hip joint, thereby influencing its function around the hip. Also, the change of the once biarticular muscles into monoarticular muscles affect their function and recruitment.
In addition to alterations in muscle geometry – which encompasses both muscle morphology and architecture – individuals with a TFA demonstrate decreased muscle strength in the residual limb compared to the intact limb. Isometric and isokinetic peak torques are reported to be 22% to 33% lower in the residual limb compared to the intact limb, in individuals 1 month to 10 years post-amputation. However, one study found no significant differences in isometric hip abduction and adduction torque, and another more recent study reported significantly higher isometric peak torques in the residual limb compared to the intact limb after normalizing to body mass and thigh length, following a median of 12 years post-amputation. Although research has shown that muscle strength deficits in individuals with a TFA are associated with difficulties in daily functioning, residual limb control, and walking performance, the specific role of residual muscle adaptations in contributing to these strength deficits and functional challenges remains unclear.
Understanding the muscle adaptations following TFA and their functional consequences is important not only for enhancing rehabilitation but also for optimizing surgical techniques. In TFA surgery, two surgical techniques can be used to reattach the dissected muscles in the residual limb. First, with a myodesis, the distal end of the dissected muscles are sutured to the end of the femur, while, secondly, with a myoplasty, muscle groups are sutured to the antagonistic muscle group, without an attachment to the femur. Evidence on how these two methods affect muscle adaptations and functionality of the residual limb is limited, although some studies suggest that myodesis may provide better muscle function, improved rehabilitation outcomes, and a better maintenance of the normal femoral anatomical alignment compared to myoplasty. Another surgical aspect that can influence muscle geometry and function is the residual limb length, as it determines which muscle insertions remain intact and affects the length of the muscles’ lever arms. A prior mixed-method study concluded that a longer residual limb length has advantages for gait parameters, preventing flexion and abduction contractures, and controlling the prosthesis. Furthermore, a Magnetic Resonance Imaging (MRI) study reported more atrophy in shorter residual limbs, also highlighting the benefits of a longer residual limb length. On the other hand, a modelling study found that femur length had little effect on muscle capacity as long as muscle tension was preserved, suggesting that muscle stabilization may be more important than preserving limb length.
Understanding the relationship between the surgical procedure and the geometrical and functional muscle changes following TFA can provide valuable insights into the benefits of different surgical techniques and residual limb lengths. This knowledge can assist surgeons in making evidence-based choices and can help clinicians to optimize and individualize rehabilitation care, focusing on restoring function. The golden standard to evaluate muscle volumes and three-dimensional muscle properties is MRI. Previous studies already used MRI to examine the muscle geometry in individuals with a TFA. While a relationship between residual limb length and the amount of atrophy has been reported, only two studies have attempted to compare the changes in muscle geometry after TFA between myodesis and myoplasty. Both studies were unable to draw conclusions because of small sample sizes (respectively n=12 and n=4), and the scarcity of available operation reports. When comparing MRI measurements with functional outcomes following TFA, only one study investigated how muscle volume in the residual limb affected clinical function in individuals with a unilateral TFA using a prosthesis. They concluded that the larger the ratio of hip flexor/hip extensor volume in the residual limb, the lower the oxygen consumption rate during walking. However, only five participants were included, and no correlations were found between muscle volume and gait and balance assessments. Additionally, this study did not assess the relationship between muscle volume and muscle strength. Therefore, information about the relationship between muscle geometry, muscle strength and functional outcomes in individuals with a TFA is still lacking. Since it is unclear how muscle adaptations following TFA affect functionality, further research is needed to describe adaptations in muscle geometry, strength and function following TFA, and to explore their interrelationships. Additionally, to understand how surgical techniques influence these adaptations, the differences between myodesis and myoplasty as well as the impact of residual limb length needs to be explored. This paper presents a cross-sectional study protocol designed to investigate muscle geometry and function following TFA, with the goal of understanding muscle adaptations and their functional consequences.
METHODS
The Medical Ethics Committee of the University Medical Center Groningen (METc) has assessed and ethically approved the study protocol (METc 2024/392). The study will adhere to the principles of the Declaration of Helsinki (October 2013) and comply with the Medical Research Involving Human Subjects Act.
STUDY AIMS
The main goal of the study is to examine and describe the differences in muscle morphology and muscle strength of the hip muscles in the residual limb compared to the intact limb in individuals with a TFA, and to explore how muscle morphology and muscle strength are related. The secondary aims are to explore the potential differences in muscle geometry and muscle strength between surgical techniques (myodesis vs. myoplasty), and to gain insight into the impact of the residual limb length on these outcomes. Additionally, the relationships between the muscle geometry, muscle strength, and functional mobility will be explored.
STUDY DESIGN AND POPULATION
This cross-sectional study will be carried out at the Hanze University of Applied Sciences Groningen in collaboration with the University Medical Center Groningen (UMCG). Individuals who have undergone a TFA within the past 1-5 years and are walking with a prosthesis will be included. The study will be a nationwide study conducted in the Netherlands. Participants will be recruited by healthcare professionals and researchers from or affiliated with the UMCG. In addition, open recruitment efforts will include distributing posters in rehabilitation centers, as well as utilizing social media platforms and networks of patient associations. Based on the performed sample size calculation (See ‘Sample Size Calculation’), the aim is to include a total of 27 participants. Informed consent will be obtained from all participants.
Inclusion criteria:
- The participant has a primary, unilateral transfemoral amputation;
- The transfemoral amputation was performed 1 to 5 years ago;
- The participant is aged 18 years or older;
- The participant is fitted with a prosthesis and is actively using it (≥ K-level 2);
- The participant meets the criteria to be eligible for an MRI, which will be confirmed by checking the MRI screening form the participant fills in.
Exclusion criteria:
- The participant is showing signs of dementia or other cognitive impairments, which limit their ability to read the information letter and fill in the screening form;
- The participant does not have sufficient knowledge of the Dutch language to read the information letter or answer the questions;
- The participant has a bone-anchored prosthesis;
- The participant has a contraindication for MRI after screening.
If possible, participants will be sampled purposefully to match the two groups of myodesis and myoplasty by age, sex, and time since amputation. The participant’s operation report will be obtained and reviewed to determine the surgical technique used (myodesis vs. myoplasty). If this information is missing, it will be obtained from the surgeon.
DATA ACQUISITION
Participant characteristics (e.g., age, sex, body weight, height) and amputation details (cause and date of amputation, details on prosthetic use, K-level) will be collected via a questionnaire. A 1,5T MRI-scanner (Siemens Magnetom Aero Ecoline, 1,5 Tesla, Siemens, Germany) will be used to collect MRI data and obtain information about the muscle geometry and residual limb length. The participant will be positioned in supine position with feet towards the magnet. 3D T1-VIBE DIXON isotropic images of both legs will be acquired using two packages, which will be composed into a single view. The imaging field-of-view for each package will be 450×450 mm, with an axial-plane resolution of 1.2×1.2 mm and a slice thickness of 1 mm. The scan will cover the area from the iliac crest to the caput fibulae of the intact limb. For the full MRI scan protocol, see: https://doi.org/10.17605/OSF.IO/4PZU2. 2D contours of muscles, fat and bone will be subtracted from the MRI using Mimics (Mimics, Materialise, Belgium) or comparable software. The 2D contours will be combined to get 3D images and to obtain information about the muscle geometry.
A HUMAC NORM (CSMi Medical Solutions, Stoughton, MA, USA) isokinetic dynamometer will be used to assess the isokinetic and isometric hip muscle strength per muscle group, for both the residual and intact limb. Strength testing will be conducted according to the measurement protocol outlined by Crozara et al. For the hip abduction-adduction strength participants will be positioned in lateral decubitus with 15° of hip abduction (Figure 1A). For the hip flexion-extension strength, participants will be positioned in a supine position with 60° of hip flexion (Figure 1B). The trunk, hip and contralateral limb will be stabilized by adjustable straps. The HUMAC NORM’s ‘Zero Gravity Mode’ will be used to eliminate the weight of the limb, enabling accurate measurements. After positioning, the hip muscle strength will be assessed following the testing procedures described below.
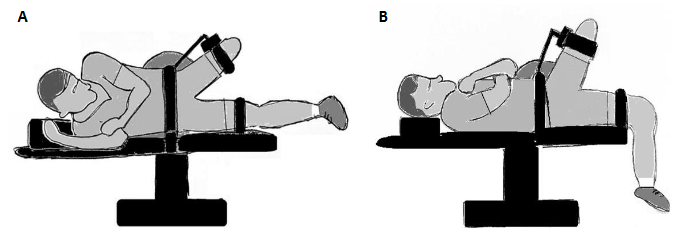
For hip abduction-adduction, isokinetic strength assessments will be conducted at angular velocities of 30°/s and 90°/s over a maximum range of 30 degrees adduction to 45 degrees abduction. Participants will perform three practice submaximal voluntary contractions to familiarize themselves with the movement and velocity, followed by five repetitions at 30°/s and 90°/s. A 2-minute rest will be given after each isokinetic testing sequence. Subsequently, isometric strength testing will be conducted, consisting of three sets of 5-second maximal isometric voluntary contractions at 15° of hip abduction, with a 30-second rest between repetitions and a 1-minute rest after the three sets. Participants will perform one submaximal contraction for practice before the isometric test.
For hip flexion-extension, isokinetic strength assessments will be conducted at angular velocities of 60°/s and 180°/s over a maximum range of 0 to 120 degrees. Participants will perform three practice submaximal voluntary contractions to familiarize themselves with the movement and velocity, followed by five repetitions at 60°/s and 10 repetitions at 180°/s. A 2-minute rest will be given after each isokinetic testing sequence. Subsequently, isometric strength testing will be conducted, consisting of three 5-second maximal isometric voluntary contractions at 60° of hip flexion, with a 30-second rest between repetitions and a 1-minute rest after the three sets. Participants will perform one submaximal contraction for practice before the isometric test. Participants will receive verbal encouragement to perform maximal muscle contractions.
As a third measure, the L Test of Functional Mobility (L Test) will be performed to assess physical function. The L Test provides an assessment of walking over a distance of 10 meters and involves turning in two directions, forming an ‘L’ shape. This measure is used in individuals with a lower-limb amputation as a replacement of the Timed Up and Go (TUG) to overcome the ceiling effect of the TUG.
OUTCOMES AND DATA ANALYSIS
Table 1 shows the baseline characteristics, primary outcomes, and secondary outcomes that will be collected. The primary outcomes include the muscle volume (cm3) of the hip adductors, abductors, flexors and extensors; the amount of inter- and intramuscular fat (cm3); and the hip muscle strength per muscle group (Nm). Muscle groups that will be assessed are listed in Table 2. Muscle volume and inter- and intramuscular fat will be extracted from MR images and calculated for both the intact and residual limb of each individual. The degree of muscle atrophy in the residual limb will be calculated by determining the muscle volume of the residual limb as a percentage of the muscle volume in the intact limb. The hip muscle strength will be determined by calculating the highest muscle torque among repetitions for each movement trial, under both isometric and isokinetic conditions. Strength measurements for the adductors, abductors, flexors, and extensors of the hip will be compared between the residual and intact limbs. The torque-angle relationship will be visualized to demonstrate how much torque can be delivered throughout the entire range of motion during isokinetic testing. Within-subject differences will be analyzed to compare the muscle morphology and function between the intact and residual limbs. Additionally, where appropriate, muscle morphology and strength measurements will be normalized to body dimension metrics and the measurements of the intact limb. Both absolute and normalized values will be reported.
Secondary outcomes include the femur length (cm) and the time score on the L Test (s) (Table 1). Femur length will be used to assess the residual limb length. For the residual limb, this will be measured from the greater trochanter to the most distal point of the femur. For the intact limb, this will be measured from the greater trochanter to the lateral femoral epicondyle. These measurements can also be taken without imaging facilities and therefore facilitate comparisons with other literature. Functional mobility will be assessed using the L Test score, which records the time a participant takes to complete the test. On a more qualitative basis, the muscle architecture will also be explored.

STATISTICAL CONSIDERATIONS
Descriptive statistics will be used to summarize participant characteristics, amputation details, muscle volume, inter- and intramuscular fat, femur length, hip muscle strength and the score on the L Test. Continuous variables will be presented as the mean and standard deviation or as the median and interquartile range, when appropriate depending on normality. Categorical variables will be presented as frequencies and percentages. Summary statistics will be given for the residual and intact limb and for the myodesis and myoplasty group, when appropriate. The primary outcomes are planned to be compared between the intact and residual limbs using dependent samples t-tests for normally distributed variables or Wilcoxon Signed-Rank tests for skewed distributed variables. Pearson’s correlation coefficients are planned to be used to explore the relationships between muscle morphology outcomes and hip muscle strength. To meet the secondary research objectives, the differences in muscle geometry and hip strength between the myodesis and myoplasty groups will be explored. An independent samples t-test is planned to be used for normally distributed variables, and a Mann Whitney U test for skewed variables. To ensure a fair comparison and eliminate interindividual differences, the values normalized to the intact limb will be used for this analysis. If power allows (depending on distribution in inclusion), multiple linear regression analysis will be used to explore any effects of the residual limb length and surgical technique on the primary outcomes. As a secondary analysis, the relationships between the primary outcomes and functional mobility will also be explored. Statistical significance level is set to p < 0.05. Analysis will be performed using IBM SPSS Statistics software version 28 (IBM Corp., Armonk, New York, USA) and R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria).
SAMPLE SIZE CALCULATION
A power calculation was performed in R to determine the required sample size to address the research question on the correlation between muscle morphology and hip muscle strength. Since there are no known correlations between muscle morphology and hip muscle strength in the respective target group, the expected correlation was based on the relationship between thigh muscle fat infiltration and thigh muscle strength in patients with type 2 diabetes mellitus. The reported correlation between the amount of inter- and intramuscular fat and the produced peak torque and total work was considered for this. The mean correlation was -0.52, which was used as expected correlation. Together with a statistical significance level of α=0.05 and power of 80%, 27 participants will be needed for the current study to detect the relationship between muscle morphology and muscle strength in the residual limb of individuals with a TFA. The R script has been published elsewhere (https://doi.org/10.17605/OSF.IO/4PZU2). Given the limited research on this topic within the target population, interim analyses will be conducted to assess whether the previously mentioned correlations and effects emerge at an earlier stage.
Conflicts of Interest statement: The authors have no conflicts of interest to declare.
Acknowledgements: We would like to thank Floris de Groot and Annemieke van der Heij-Meijer for their valuable support in making this research possible. Their early involvement and guidance were key to getting this project going.
REFERENCES
- Wong CK, Chen CC, Blackwell WM, Rahal RT, Benoy SA. Balance ability measured with the Berg balance scale: a determinant of fall history in community-dwelling adults with leg amputation. J Rehabil Med. 2015;47(1):80-86. doi:10.2340/16501977-1882
- Azuma Y, Chin T, Miura Y. The relationship between balance ability and walking ability using the Berg Balance Scale in people with transfemoral amputation. Prosthet Orthot Int. 2019;43(4):396-401. doi:10.1177/0309364619846364
- Jaegers SMHJ, Arendzen JH, De Jongh HJ. Changes in hip muscles after above-knee amputation. Clin Orthop Relat Res. 1995;319(319):276-284. doi:10.1097/00003086-199510000-00030
- Putz C, Block J, Gantz S, et al. Structural changes in the thigh muscles following trans-femoral amputation. Eur J Orthop Surg Traumatol. 2017;27(6):829-835. doi:10.1007/s00590-017-1929-5
- Roda GF, Awad ME, Melton DH, Christiansen CL, Stoneback JW, Gaffney BMM. The Amputated Limb Gluteus Medius is Biomechanically Disadvantaged in Patients with Unilateral Transfemoral Amputation. Ann Biomed Eng. 2024;52(3):565-574. doi:10.1007/s10439-023-03400-0
- Henson DP, Edgar C, Ding Z, et al. Understanding lower limb muscle volume adaptations to amputation. J Biomech. 2021;125. doi:10.1016/j.jbiomech.2021.110599
- Addison O, Marcus RL, Lastayo PC, Ryan AS. Intermuscular fat: A review of the consequences and causes. Int J Endocrinol. 2014;2014. doi:10.1155/2014/309570
- Ranz EC, Wilken JM, Gajewski DA, Neptune RR. The influence of limb alignment and transfemoral amputation technique on muscle capacity during gait. Comput Methods Biomech Biomed Engin. 2017;20(11):1167-1174. doi:10.1080/10255842.2017.1340461
- Gottschalk F. Transfemoral Amputation. Clin Orthop Relat Res. 1999;361(361):15-22. doi:10.1097/00003086-199904000-00003
- Crozara LF, Marques NR, LaRoche DP, et al. Hip extension power and abduction power asymmetry as independent predictors of walking speed in individuals with unilateral lower-limb amputation. Gait Posture. 2019;70(March):383-388. doi:10.1016/j.gaitpost.2019.03.033
- Rutkowska-Kucharska A, Kowal M, Winiarski S. Relationship between asymmetry of gait and muscle torque in patients after unilateral transfemoral amputation. Appl Bionics Biomech. 2018;2018. doi:10.1155/2018/5190816
- Hewson A, Dent S, Sawers A. Strength deficits in lower limb prosthesis users: A scoping review. Prosthet Orthot Int. 2020;44(5):323-340. doi:10.1177/0309364620930176
- Croisier JL, Maertens De Noordhout B, Maquet D, et al. Isokinetic Evaluation of Hip Strength Muscle Groups in Unilateral Lower Limb Amputees. Isokinet Exerc Sci. 2001;9:163–169.
- Sawers A, Fatone S. Normalization alters the interpretation of hip strength in established unilateral lower limb prosthesis users. Clin Biomech. 2022;97(June):105702. doi:10.1016/j.clinbiomech.2022.105702
- Heitzmann DWW, Leboucher J, Block J, et al. The influence of hip muscle strength on gait in individuals with a unilateral transfemoral amputation. PLoS One. 2020;15(9 September):1-16. doi:10.1371/journal.pone.0238093
- Bevacqua N, Dell Elce G, Intelangelo L. Assessing muscle strength of persons with transfemoral amputation with and without a prosthesis: A cross-sectional study. Prosthet Orthot Int. 2023;47(5):532-536. doi:10.1097/PXR.0000000000000228
- Gottschalk F. Chapter 45: Transfemoral Amputation: Surgical Management. In: Krajbich JI, Pinzur MS, Stevens PM, Potter BK, eds. Atlas of Amputations and Limb Deficiencies : Surgical, Prosthetic, and Rehabilitation Principles. 4th ed. AAOS – American Academy of Orthopaedic Surgeons; 2016. ISBN: 978-1-62-552437-9
- Fabre I, Thompson D, Gwilym B, et al. Surgical Techniques of, and Outcomes after, Distal Muscle Stabilization in Transfemoral Amputation: A Systematic Review and Narrative Synthesis. Ann Vasc Surg. 2024;98:182-193. doi:10.1016/j.avsg.2023.07.105
- Gottschalk FA, Stills M. The biomechanics of transfemoral amputation. Published online 1994:12-17.
- Tintle SM, Keeling JJ, Shawen SB, Forsberg JA, Potter BK. Traumatic and trauma-related amputations: Part I: General principles and lower-extremity amputations. J Bone Jt Surg. 2010;92(17):2852-2868. doi:10.2106/JBJS.J.00257
- Geertzen JHB, de Beus MC, Jutte PC, Otten E, Dekker R. What is the optimal femur length in a trans-femoral amputation? A mixed method study: Scoping review, expert opinions and biomechanical analysis. Med Hypotheses. 2019;129. doi:10.1016/j.mehy.2019.109238
- Pons C, Borotikar B, Garetier M, et al. Quantifying skeletal muscle volume and shape in humans using MRI: A systematic review of validity and reliability. PLoS One. 2018;13(11). doi:10.1371/journal.pone.0207847
- Leijendekkers RA, Marra MA, Ploegmakers MJM, et al. Magnetic-resonance-imaging-based three-dimensional muscle reconstruction of hip abductor muscle volume in a person with a transfemoral bone-anchored prosthesis: A feasibility study. Physiother Theory Pract. 2019;35(5):495-504. doi:10.1080/09593985.2018.1453902
- Geertzen JHB, van der Schans SM, Jutte PC, Kraeima J, Otten E, Dekker R. Myodesis or myoplasty in transfemoral amputations. What is the best option? An explorative study. Med Hypotheses. 2019;124(January):7-12. doi:10.1016/j.mehy.2019.01.008
- Yun DH, Jung IY, Moon CW, Cho KH. Correlation of Femoral Muscle Volume Using Three-Dimensional Modeling and Locomotor Function After Unilateral Trans-femoral Amputation. Ann Rehabil Med. 2022;46(6):303-311. doi:10.5535/arm.22110
- Deathe BA, Miller WC. The L Test of Functional Mobility: Measurement Properties of a Modified Version of the Timed “Up & Go” Test Designed for People with Lower-Limb Amputations. Phys Ther. 2005;85(7):626-635.
- Huang Y, Yan J, Zhu H, et al. Low thigh muscle strength in relation to myosteatosis in patients with type 2 diabetes mellitus. Sci Rep. 2023;13(1). doi:10.1038/s41598-022-24002-1

