Organochlorine Pesticides in Yucatan Coastal Waters: Health Risks
Organochlorine pesticides contamination on the coastal waters of Yucatan, Mexico. Risk for health
Durcy Verenice Ruiz-Ciau1, Ángel Gabriel Polanco-Rodríguez1, Shirley Guadalupe Gómez-Manzanilla1
- Grupo de Investigación en Química Analítica y Ambiental. Laboratorio de Cromatografía. Facultad de Química de la Universidad Autónoma de Yucatán. Calle 43 # 613 por 90, Colonia Inalámbrica. C. P. 97069. Mérida, Yucatán, México.
E-mail: [email protected]
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Ruiz-Ciau, DV., Polanco-Rodríguez, ÁG., et al., 2025. Organochlorine pesticides contamination on the coastal waters of Yucatan, Mexico. Risk for health. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6361
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6361
ISSN 2375-1924
ABSTRACT
Coastal waters are currently a sink for pesticides and pollutants coming from inland due to agricultural and industrial activities, which impact marine ecosystems and pose high risks to human health. The objective of the present study was to evaluate the concentration of organochlorine pesticides in the rainy and dry seasons in seawater, estuary water and drinking water from 6 coastal municipalities of Yucatán, Mexico: Progreso, Chelém, Chuburná, Celestún, Dzilám de Bravo and Ría Lagartos. In the study, α-lindane, β-lindane, ɣ-lindane, δ-lindane, heptachlor, aldrin, heptachlor epoxide, endosulfan I, dieldrin, 4,4´-DDE, endrin, endosulfan II, 4,4´-DDD, endrin aldehyde, endosulfan sulfate, and 4,4´DDT were identified and quantified. Pesticide determination was carried out by gas chromatography with an electron capture detector. The most frequently found pesticides at the sampling sites were α-lindane, β-lindane, heptachlor, and endosulfan sulfate. The highest concentrations of organochlorine pesticides in rainy season were 0.573 mg/L of endosulfan sulfate, 0.487 mg/L of Aldrin, 0.468 mg/L of dieldrin, 0.446 mg/L of ɗ-lindane; in dry season 0.604 mg/L of Aldrin, 0.462 mg/L of dieldrin, 4.52 mg/L of heptachlore, 0.405 mg/L of endosulfan. Zones with more high levels detected on the coasts were Dzilám de Bravo and Progreso. The high levels of organochlorine pesticides detected in coastal municipalities are widespread, representing risks to the public health and marine species.
Keywords: Organochlorine pesticides, public health, seawater, drinking water, estuarine water.
1. Introduction
Organochlorine pesticide pollution remains an emerging international threat to human health and coastal ecosystems. In coastal areas, there are aquatic and terrestrial interactions since, on the one hand, there is a drag of contaminants such as pesticides from inland, and they flow directly into coastal waters; the contaminants come from different sources such as urban, industrial or agricultural. On the other hand, coastal environments, especially semi-enclosed ones such as bays, estuaries, or coastal lagoons, have a great aptitude to accumulate pesticides and other contaminants because dilution is limited by the long residence times of the water and also because the levels of adsorbent materials, primarily organic carbon, are generally high. The spatial distribution of OCP in water, sediments and species in coastal areas internationally, such as South America, is widespread, representing potential risks to human health and coastal ecosystems.
Organochlorine pesticides are categorized as Persistent Organic Pollutants (POPs) and Contaminants of Emerging Concern (CEC). These substances cause cancer risks in humans and organisms in coastal waters. Cancer risks show the urgent need to address the management of persistent organic pollutants to prevent impacts on marine species and human health. These pollutants represent risks to human health due to their neurotoxic and endocrine-disrupting effects as a result of the mismanagement of waste from anthropogenic activities in coastal ecosystems, which requires monitoring, as well as the design of management strategies for the protection of coastal areas, their human populations and marine species that depend on them.
In developing countries, in particular, drinking water sources are contaminated with pesticides due to agricultural runoff, industrial wastewater and a population lacking sanitary infrastructure. Pesticides are carried mainly during rainfall, filtering into groundwater and eventually reaching the coasts. Agrochemicals present in coastal waters contaminate and affect the biota of the food chain that serves as food for marine species, producing an ecological imbalance since it contaminates organic matter as a primary source of food for aquatic organisms, with effects on human health in epigenetic, developmental and immune system aspects. It has been confirmed that agrochemicals can be related to various diseases, such as neurological problems, heart disease, reproductive problems, and various cancers.
Organochlorine pesticides are of global concern due to their effects on the environment and humans. In the case of Yucatan, it is a karstic system with highly stony soils that easily filter contaminants into the underground aquifer, transporting them to coastal areas. In the particular case of coastal areas, the presence of these pesticides causes various species of fish to bioaccumulate them, hurting their life cycle and, on the other hand, affecting the human health of people who ingest them. Some studies show that these compounds are toxic and can induce mutagenesis (alteration of DNA or chromosomes), teratogenesis (malformations in the embryo) and alterations in various human metabolic and reproductive functions.
To date, various studies have generally demonstrated the concentration of pesticides in different regions of Yucatan, in cenotes, wells, and drinking water. However, it is important to monitor organochlorine pesticides in coastal waters such as seawater, estuarine water, and drinking water in coastal communities. Detailed knowledge of the presence, distribution, and effects of these pollutants in the Yucatan coastal ecosystem is essential to taking appropriate mitigation and control measures.
On the other hand, qualitative socio-environmental studies have been carried out to understand the use and management of agrochemicals in the state of Yucatan, the point of view of Maya communities, reporting excessive use of organochlorine pesticides and glyphosate.
1.1 COASTAL AREA OF YUCATAN
In Yucatan, groundwater serves as the primary source of drinking water for various activities. Although the region lacks surface water bodies such as rivers or lakes, it contains underground rivers and is subject to seawater inflows. The high permeability of the geological materials in this area makes the aquifer particularly vulnerable to contamination, especially along the coast. The regional groundwater flow map of the Yucatan Peninsula illustrates the different outlet routes of the karst aquifer to the Yucatan coasts, revealing the transport of various contaminants resulting from agricultural-livestock activities and population growth.
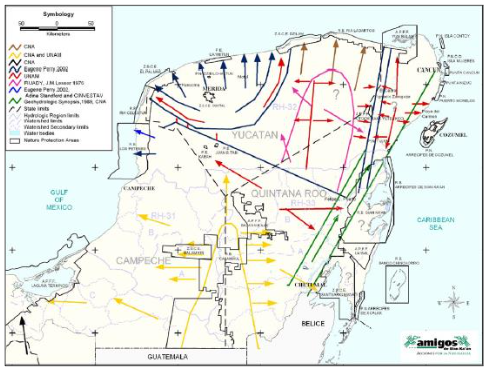
1.2 Vulnerability of the coastal zone
Among the productive activities that affect groundwater are agriculture and livestock, which use pesticides and fertilizers, and the bottling industry due to the over-extraction of water and saline intrusion processes.
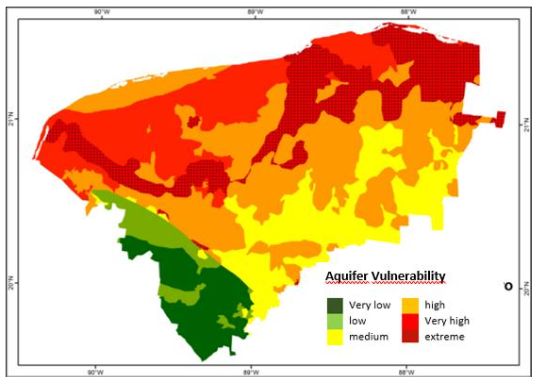
1.3 IMPACT ON THE COASTAL WATER
Organochlorine pesticides affect the development of organisms in the marine and estuarine environment. Because they are hydrophobic, they can accumulate in the adipose tissue of organisms through contaminated biota. Pesticides in the marine environment from inland farming activities can have severe consequences for biota and marine ecosystems of ecological importance, even in protected natural areas. In addition to influencing reproduction, some organochlorine pesticides cause damage to different organs of fish, such as the liver, kidney, brain, gills, muscle, intestine and gonads. These pesticides affect marine species that serve as bioindicators. Assessing organochlorine pesticide levels in seafood allows organochlorine pesticides (OCPs) to enter the fish’s body, negatively impacting public health.
1.4 BIOMONITORING OF PESTICIDES IN THE BLOOD OF MAYAN WOMEN WITH CANCER IN YUCATAN
A study of bioaccumulation of organochlorine pesticides in the blood of Mayan women with cervical cancer in 18 municipalities with highest mortality of cancer in Yucatan, revealed the detection of pesticides in more than 70% of the women, and concentrations of endosulfan I (7.35 µg/mL), aldrin (3.69 µg/mL), 4,4′ DDD (2.33 µg/mL), and 1.39 and 1.46µ g/mL of -HCH. In another study of bioaccumulation of pesticides in the breast milk of Mayan women were detected high levels such as 18 mg/kg of heptachlore epoxide, 1.92 mg/kg of endrin, 2.10 m/kg of dieldrin, 0.103 mg/kg of heptachlor, 0.127 mg/kg of endrin aldehyde, indicate a impact on women and their newborn children from the gestational period, as well as in their first year of life when fed with breast milk with organochlorine pesticides, which implies high risks for the health of children related to congenital malformations and neurological disorders.
2. Materials and Methods
2.1 WATER SAMPLING
Water samples were collected during the dry season (march) and the rainy season (november) of 2022, obtaining 3 samples per zone (one zone from the east and one from the west), one from drinking water, one from the estuary and one from the seawater, giving a total of 6 samples for each port and for each season.
| Place | Coordinates |
|---|---|
| Progreso | 21.289392, -89.654211 |
| Chelem | 21.274424, -89.729872 |
| Chuburná | 21.254855, -89.817692 |
| Celestún | 20.867773, -90.397925 |
| Dzilam de Bravo | 21.394841, -88.888130 |
| Río Lagartos | 21.614266, -88.175551 |
The sample type was simple and punctual. The samples were collected in 1liter amber glass bottles previously washed with reagent-grade hexane. They were transported and stored in the laboratory at 4° C until analysis.
2.2 REAGENTS USED
The reagents used for this investigation were HPLC-grade methanol (MeOH), HPLC-grade methyl tert-butyl ether (MTBE), and MilliQ-grade deionized water. For the internal standard, 4-chlorophenyl-4-chlorobenzenesulfonate (Ovex) from Fluka® Analytical brand was used, standard grade, at a concentration of 2000 mg/L. J.T. Baker C18 solid phase extraction cartridges with 500 mg of stationary phase. 200 mg/L standard solution containing the 16 OCs calibrated with MTBE, from which working solutions were prepared with concentrations of 0.5, 0.75, 1, 1.5 and 2 mg/L, which were maintained at temperatures of 4°C and Ovex (internal standard) was added at a concentration of 20 mg/L.
2.3 SAMPLE PREPARATION
The solid-phase extraction (SPE) technique was employed to separate organochlorine pesticides from potential interferences, thereby reducing factors that could introduce uncertainty in the analysis. Initially, the samples were gravity-filtered using Whatman® filter paper with a diameter of 24 cm. Next, the extraction cartridge was conditioned by rinsing it with 5 mL of methanol, 5 mL of Methyl Tertiary Butyl Ether (MTBE), and 3 mL of water. Following this, 500 mL of the sample was passed through the cartridge at a flow rate of 1 mL/min. The cartridge was then rinsed with 10 mL of water, and finally eluted with 2 mL of MTBE after allowing it to dry for 30 minutes.
The determination was carried out using the Gas Chromatography with Electron Capture Detector (GC-ECD) TRACE 1310, chromatography equipment equipped with an auto sampler model AS 1310, in which the analyte pass in gaseous form through the column dragged by a gaseous mobile phase (helium BIP) with a splitless injection mode with an injection volume at 7 gas flow carried at 1 mL/min. An HP-5MS column (95% dimethyl, 5% difficult polysiloxane) of 30 m x 0.250 mm ID and 0.25 micrometres film thickness is used; the brand used was Agilent Technologies.
2.4 QUALITY CONTROL PARAMETERS OF THE METHOD
Linearity
For this study, a calibration curve was prepared with concentrations of 0.5, 0.75, 1, 1.5 and 2 mg/L of the reference standard of the OCs with an original concentration of 2000 mg/L. The analysis was performed in triplicate and with the chromatographic conditions mentioned above. Once the data was obtained, a statistical analysis was performed to determine the equation of the regression line, the correlation coefficient and the coefficient of determination.
Various equations were used to determine the parameters:
Slope (b)
𝑏 = ∑ 𝑥𝑦 − ∑ 𝑥 ∑ 𝑦 / 𝑛 ∑ 𝑥² − (∑ 𝑥)² / 𝑛
Independent term (a)
𝑎 = ∑ 𝑦 − 𝑏 ∑ 𝑥 / 𝑛
Correlation coefficient (R) and determination (R²)
𝑅 = ∑(𝑥 − 𝑥𝑚)(𝑦 − 𝑦𝑚) / √∑(𝑥 − 𝑥𝑚)² ∑(𝑦 − 𝑦𝑚)²
Residual variance (S²y,x)
𝑆y,x² = ∑(𝑦 − 𝑦𝑚)² / 𝑛 − 2 (1 − 𝑟²)
Slope variance (S²b)
𝑆b² = 𝑆y,x² ∑ 𝑥² − (∑ 𝑥)² / 𝑛
Variance of the independent term (S²a)
𝑆a² = (𝑆b²)(∑ 𝑥² / 𝑛)
Where:
x: independent variable.
𝑥m: total mean of the independent variable.
y: dependent variable.
𝑦m: total mean of the dependent variable.
n: total number of tests performed.
Detection Limit (LD) and Quantification Limit (QL)
The calculation was performed using the ordinate at the origin obtained from the calibration line, obtained from the linearity. To calculate the LD, the following equation was used:
LD=3.3σ/m
The equation used to calculate the QL for each pesticide was:
LD=10σ/m
Where
σ: standard deviation of the average concentrations for each period.
m: slope of the equation of the straight line.
3. Results
Regardless of the season, the average concentration of organochlorine pesticides in coastal waters showed a lower concentration of β-lindane (0.025 mg/L) and a higher concentration of heptachlor (5.64 mg/L).
The highest concentrations of organochlorine pesticides were heptachlor in seawater (3.90 mg/L), estuary water (5.64 mg/L), the highest concentration in the entire study, and drinking water (3.14 mg/L).
Aldrin levels were high in seawater (0.540 mg/L), estuary water (0.467 mg/L), and drinking water (0.540 mg/L); endosulfan II also showed high pesticide concentrations in seawater (0.407 mg/L), estuary water (0.401 mg/L), and drinking water (0.422 mg/L); heptachlor in estuary water (5.64 mg/L); as well as 4,4′-DDD in seawater (0.407 mg/L), estuary water (0.403 mg/L), and drinking water (0.403 mg/L).
The pesticides detected in all the coastal municipalities sampled were α-lindane and endosulfan sulfate. Although pesticides are banned and restricted, high concentrations continue to be found in various municipalities in Yucatan.
| Pesticide | Type of water (mg/L) | Seawater | Estuarine Water | Drinking Water |
|---|---|---|---|---|
| α-lindane | 0.12390 | 0.2692 | 0.0001 | |
| β-lindane | 0.0315 | 0.0540 | 0.0255 | |
| ɣ-lindane | 0.3651 | 0.3798 | ND | |
| δ-lindane | 0.4238 | 0.4228 | 0.4234 | |
| Heptachlore | 3.9013 | 5.6443 | 3.1440 | |
| Aldrín | 0.5408 | 0.4677 | 0.5405 | |
| Heptachlore epoxide | 0.3172 | 0.3167 | 0.3226 | |
| Endosulfán I | ND | 0.0217 | ND | |
| Dieldrín | 0.4548 | 0.4621 | 0.4535 | |
| 4,4´-DDE | 0.2518 | 0.2579 | 0.3549 | |
| Endrín | 0.1817 | 0.1892 | 0.1924 | |
| Endosulfán II | 0.4072 | 0.4010 | 0.4225 | |
| 4,4´-DDD | 0.4072 | 0.4036 | 0.4031 | |
| Endrín aldehído | 0.3326 | 0.3353 | 0.3266 | |
| Endosulfán sulfate | 0.3180 | 0.3660 | 0.5909 | |
| 4,4´DDT | 0.0001 | 0.0382 | ND |
Table 5 shows the mean concentration of pesticides, with heptachlor having the highest concentration in both seasons.
| Pesticides | Ports sampled (N=6) | Detection Frequency ±DST (mg/L) |
|---|---|---|
| α-lindane | 12 | 0.214±0.009 |
| β-lindane | 12 | 0.031±0.005 |
| ɣ-lindane | 11 | 0.376±0.004 |
| δ-lindane | 12 | 0.432±0.009 |
| Heptachlore | 10 | 0.809±0.410 |
| Aldrín | 12 | 0.489±0.026 |
| Heptachlore epoxide | 11 | 0.321±0.004 |
| Endosulfán I | 7 | 0.130±0.157 |
| Dieldrín | 6 | 0.461±0.003 |
| 4,4´-DDE | 5 | 0.267±0.018 |
| Endrín | 5 | 0.294±0.0.109 |
| Endosulfán II | 12 | 0.411±0.005 |
| 4,4´-DDD | 11 | 0.390±0.015 |
| Endrín aldehído | 4 | 0.343±0.015 |
| Endosulfán sulfate | 12 | 0.449±0.037 |
| 4,4´DDT | 7 | 0.329±0.043 |
3.1 QUANTIFICATION OF PESTICIDES IN DRY SEASONS.
The mean concentration and standard deviation during the dry season are given in Table 6.
| Pesticides | Sampled ports (N=6) | ±DST |
|---|---|---|
| α-lindane | N=6 | 0.214±0.019 |
| β-lindane | N=6 | 0.044±0.029 |
| ɣ-lindane | N=6 | 0.371±0.005 |
| δ-lindane | N=6 | 0.424±0.005 |
| Heptachlore | N=6 | 3.7±0.615 |
| Aldrín | N=6 | 0.529±0.055 |
| Heptachlore epóxide | N=6 | 0.319±0.009 |
| Endosulfán I | N=6 | 0.249±0.007 |
| Dieldrín | N=6 | 0.459±0.003 |
| 4,4´-DDE | N=6 | 0.28±0.042 |
| Endrín | N=6 | 0.187±0.0.008 |
| Endosulfán II | N=6 | 0.411±0.013 |
| 4,4´-DDD | N=6 | 0.402±0.018 |
| Endrín aldehído | N=6 | 0.343±0.015 |
| Endosulfán sulfate | N=6 | 0.031±0.102 |
| 4,4´DDT | N=6 | 0.015±0.048 |
Heptachlore was the pesticide with the highest concentration during the dry season, and its presence was predominant in Dzilám de Bravo. Table 7 shows the concentration of pesticides in the dry season by municipality.
| Pesticides | Ports sampled (N=6) | Progreso | Chelem | Chuburná | Celestún | Dzilam de Bravo | Río Lagartos |
|---|---|---|---|---|---|---|---|
| α-lindane | N=6 | 0.232 | 0.186 | 0.24 | 0.453 | 0.5 | 0.366 |
| β-lindane | N=6 | 0.024 | 0.025 | 0.354 | 0.4 | 0.26 | 0.4 |
| ɣ-lindane | N=6 | 0.367 | 0.368 | 0.363 | 0.189 | 0.22 | 0.35 |
| δ-lindane | N=6 | 0.425 | 0.42 | 0.418 | 0.382 | 0.32 | 0.384 |
| Heptachlore | N=6 | 4.09 | 2.5 | 0.493 | 0.5 | 4.52 | 0.366 |
| Aldrín | N=6 | 0.516 | 0.604 | 0.493 | 0.5 | 0.5 | 0.4 |
| Heptachlore epoxide | N=6 | 0.31 | 0.24 | 0.257 | 0.31 | 0.3 | 0.3 |
| Endosulfán I | N=6 | ND | ND | ND | ND | ND | ND |
| Dieldrín | N=6 | 0.454 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| 4,4´-DDE | N=6 | ND | ND | ND | ND | ND | ND |
| Endrín | N=6 | ND | ND | ND | ND | ND | ND |
| Endosulfán II | N=6 | 0.411 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| 4,4´-DDD | N=6 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Endrín aldehído | N=6 | ND | ND | ND | ND | ND | ND |
| Endosulfán sulfate | N=6 | ND | ND | ND | ND | ND | ND |
| 4,4´DDT | N=6 | ND | ND | ND | ND | ND | ND |
3.2 QUANTIFICATION OF PESTICIDES IN THE RAINY SEASON.
The results obtained for the mean concentration and standard deviation are recorded in Table 8.
| Pesticides | Ports sampled (N=6) | ±DST |
|---|---|---|
| α-lindane | N=6 | 0.213±0.012 |
| β-lindane | N=6 | 0.025±0.001 |
| ɣ-lindane | N=6 | 0.376±0.007 |
| δ-lindane | N=6 | 0.437±0.019 |
| Heptachlore | N=6 | 0.383±0.03 |
| Aldrín | N=6 | 0.47±0.041 |
| Heptachlore epoxide | N=6 | 0.322±0.006 |
| Endosulfán I | N=6 | 0.225±0.015 |
| Dieldrín | N=6 | 0.465±0.002 |
| 4,4´-DDE | N=6 | 0.229±0.015 |
| Endrín | N=6 | 0.278±0.127 |
| Endosulfán II | N=6 | 0.377±0.013 |
| 4,4´-DDD | N=6 | 0.342±0.062 |
| Endrín aldehído | N=6 | ND |
| Endosulfán sulfate | N=6 | 0.537±0.12 |
| 4,4´DDT | N=6 | 0.377±0.013 |
The pesticide with the highest concentration observed during this period is endosulfan sulfate, with an average concentration of 0.537 mg/L detected at all sampling sites. Most pesticides were found at all sampling sites, except for 4,4′-DDE and endrin aldehyde, which were the least frequently detected in the coastal area of this study.
| Pesticides | Coastal municipalities | Progreso | Chelem | Chuburná | Celestún | Dzilam de Bravo | Río Lagartos |
|---|---|---|---|---|---|---|---|
| α-lindane | N=6 | 0.206 | 0.026 | 0.369 | 0.446 | 0.37 | 0.5 |
| β-lindane | N=6 | 0.026 | 0.024 | 0.36 | 0.423 | 0.36 | 0.4 |
| ɣ-lindane | N=6 | 0.369 | 0.025 | 0.376 | 0.37 | 0.42 | 0.4 |
| δ-lindane | N=6 | 0.446 | 0.423 | 0.37 | 0.4 | 0.4 | 0.4 |
| Heptachlore | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Aldrín | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Heptachlore epoxide | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Endosulfán I | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Dieldrín | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| 4,4´-DDE | N=6 | ND | ND | ND | ND | ND | ND |
| Endrín | N=6 | ND | ND | ND | ND | ND | ND |
| Endosulfán II | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| 4,4´-DDD | N=6 | 0.37 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Endrín aldehído | N=6 | ND | ND | ND | ND | ND | ND |
| Endosulfán sulfate | N=6 | ND | ND | ND | ND | ND | ND |
| 4,4´DDT | N=6 | ND | ND | ND | ND | ND | ND |
3.3 COMPARISON OF CONCENTRATIONS DURING DRY AND RAINY SEASONS.
It is important to compare the rainy and dry seasons to analyze which season has the highest concentration of organochlorine pesticides and which season is most frequent to find a variety of these.
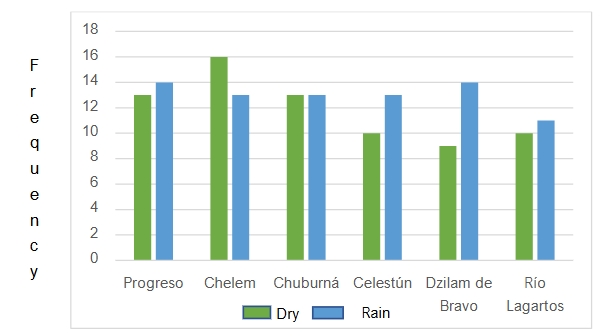
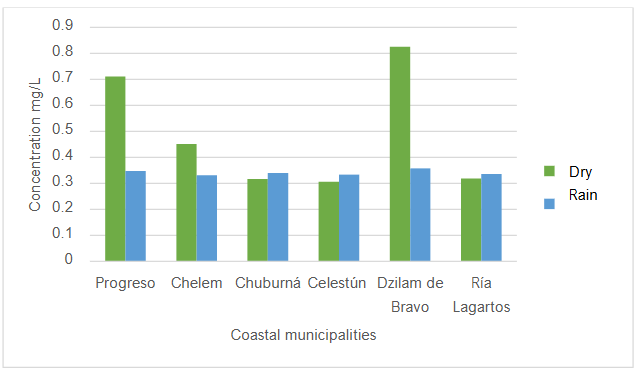
In most cases, greater amounts of pesticides are found in rainy seasons. This may be due to the drag of contaminants from the soil to the underground aquifer generated by the rains in the region. Regarding the average global concentrations of pesticides, Figure 5 shows that the dry season had higher concentration values than the rainy season.
4. Discussion
The levels of organochlorine pesticides detected on the coasts of Yucatan during the dry and rainy seasons, in seawater, estuary water and drinking water, are above the maximum permissible limits of the Mexican Official Standard (NOM-127-SSA1-2021). The global average of organochlorine pesticide load in the dry season was from highest to lowest: Dzilám Bravo (0.824 mg/L), Progreso (0.711 mg/L), Chelém (0.451 mg/L), Ría Lagartos (0.318 mg/L), Chuburná (0.317 mg/L), Celestún (0.305 mg/L). These concentrations are above the Mexican official standard and even higher than international standards.
The global average of organochlorine pesticide load during the rainy season was, from highest to lowest: Dzilám Bravo (0.357 mg/L), Progreso (0.347 mg/L), Chuburná (0.339 mg/L), Ría Lagartos (0.336 mg/L), Celestún (0.333 mg/L), and Chelém (0.331 mg/L). These concentrations are also above the official Mexican standard and even higher than international standards.
The results of this study indicate high levels of organochlorine pesticides in both periods on the Yucatecan coasts, which puts human health and marine biodiversity at risk.
During the dry season, higher levels of heptachlor were also obtained in the municipality of Progreso (4.09 mg/L), as well as high concentrations of aldrin (0.604 mg/L in Chelém), (0.516 mg/L in Progreso), (0.493 mg/L in Chuburná), (0.50 mg/L in Celestún), (0.60 mg/L in Dzilám Bravo), (0.455 mg/L in Ría Lagartos); as well as 0.425 mg/L of ɗ-lindane in Progreso. During the rainy season, the pesticide detected with the highest concentration was endosulfan sulfate, with 0.759 mg/L in Chuburná (northwest coastal area), 0.573 mg/L in Progreso (central coastal area), 0.441 mg/L in Ría Lagartos (eastern coastal area), which indicates high geographically widespread contamination by endosulfan sulfate in coastal waters of Yucatán, implying risks.
In the dry season, as the temperature constantly increases the water evaporates, decreasing the amount of water and thus causing a higher concentration of pesticides. The concentration of pesticides may depend on factors such as evaporation, adsorption, and leaching, depending on the region’s environmental conditions. In the rainy season, because the sea level and estuary level rise and there is also a constant movement of currents, the concentrations are dispersed and decrease as they are more distributed.
The results indicate that productive activities such as agriculture and livestock from inland areas in Yucatan are applying organochlorine pesticides that end up on the Yucatan coast, putting human health, marine biodiversity, and ecosystems at risk. The levels of pesticides detected in marine water in coastal municipalities put human health at risk due to the ingestion of contaminated marine species.
In a study in Yucatán to determine organochlorine pesticides in cenotes water, in the area of the Cenote Ring, the highest concentration detected was for heptachlor in the municipality of Dzilám González during the dry season (more than 13 mg/L). This municipality is adjacent to the coastal municipality of Dzilám Bravo, where the highest concentration of heptachlor (4.52 mg/L) was also obtained during the dry season in the coastal study. The results in this area coincide with the geographic distribution of the Karstic Aquifer Pollution Index (IVAKY), which shows high and extreme vulnerability to contamination of the Karstic Aquifer. Karst aquifers are highly vulnerable to groundwater contamination due to the easy filtration of contaminants such as organochlorine pesticides.
These contaminants are prohibited and/or restricted for use globally due to their high impact on public health. They are classified as carcinogenic and neurotoxic endocrine disruptors and have been linked to diseases such as congenital malformations, Attention Deficit Hyperactivity Disorder (ASD), autism, and Parkinson’s disease, among others.
On the other hand, this area is one of high deforestation and a high density of cenotes (surface water bodies), which are a route for contaminants to enter the underground aquifer. The area also has a density of faults and fractures in the soil, and agricultural activities are conducted using agrochemicals. Both municipalities of Yucatan are located in the northeast area of groundwater discharge from the Cenote Ring to the coastal area of Yucatan, which involves a drag of contaminants, so it can be inferred that the levels of contamination during the dry season are high in these adjacent municipalities.
The National Institute of Statistics, Geography and Informatics in Mexico (INEGI) reports that in the coastal municipality of Progreso, which includes the ports of Chelém, Chuburná and Chicxulub, the percentage of the population with a disability due to the limitation in activity was 13.40%. In the municipality of Celestún on the west coast, the percentage of the population with a disability due to limitation in activity in 2020 was 9.57%. On the east coast of Yucatán, in the municipalities of Dzilám Bravo, the percentage of the population with some disability due to limitation in activity in 2020 was 9.63%, and in Ría Lagartos, the percentage of the population with some disability due to limitation in activity in 2020 was 14.77%.
Organochlorine pesticides as endocrine disruptors have potential toxicological effects on humans, such as mutagenicity and carcinogenicity. In Mexico in 2019, there were 222 thousand cases of cancer and 105 thousand deaths in the general population. The number of new cases and deaths from cancer increased between 10% and 436% from 1990 to 2019. Breast, cervical, and colorectal cancers were the main causes of death among women, while prostate, lung, and colorectal cancers were the main causes of death among men. In Mexico, these cancers were the third leading cause of death in 2019.
In developing countries, cancer in women, such as cervical cancer, is in the first places of prevalence and mortality.
Qualitative socio-environmental studies have been carried out in the agricultural and livestock zones on the use and management of carcinogenic agrochemicals in the Mayan zone of Yucatán. These studies report a high use of agrochemicals and a low perception of health risks in pesticide management. This is similar to a study on pesticide use in farmers in Punjab, Pakistan, where farmers are excessive users of pesticides and have the most perceived low levels of risk. This low-risk perception can be improved with training in educational programs. Exposure to behavioural and environmental risk factors will increase the global cancer burden.
Among the factors that influence the detection of pesticides in aquifers are preferential underground flows, variations in the year’s seasons in dry and rainy periods, types of aquifers, and land use. To date, based on various research groups, it is concluded that there is a clear need to implement monitoring systems for organochlorine pesticides in groundwater in the Latin American and Caribbean regions to reduce the impacts on public health and ecosystems.
In another study, we can see the impact of pesticides on coastal waters, biota and marine species, in which pesticides were measured in marine species and zooplankton, in two fishes (Harpadon nehereus and Pampus argenteus) and shrimps (Penaeus indicus); the samples were collected from three tidal creeks in the region of India. The results detected contamination by organochlorine pesticides in surface water: hexachlorocyclohexane isomers (ΣHCH: 525 1581 ng dichlorodiphenyltrichloroethane metabolites (ΣDDT: 188 377 ng endosulfan metabolite (ΣEND: 687 1474 ng and other organochlorine pesticides (512 1334 ng The zooplankton community showed damage due to pesticide bioaccumulation. Aldrin, heptachlor and α-HCH levels in edible biota may cause cancer. These contaminants disrupt the food chain, damaging the structure of marine biota and various fish species.
The bioaccumulation of contaminants in marine species, which are seafood products consumed by humans, causes potential risks to public health. These can include various cancers and severe effects on pregnant women, such as congenital malformations, cognitive delay, and miscarriages. On the other hand, some studies have shown a relationship between pesticides and congenital malformations due to consuming contaminated food and water around agricultural areas.
Organochlorine pesticides are categorized as chlorinated cyclodienes, chlorinated benzenes, and cyclohexanes, and are of great international concern due to their severe effects on the environment and human health because they are bioaccumulative in living beings. Data from a study on fish from African coasts showed high concentrations of pesticides, which indicates that the regular consumption of these fish may indicate possible development of cancer in humans.
In a study of bioaccumulation of organochlorine pesticides in breast milk in women from the coast of Chelém, Yucatán, located adjacent to the main port of Progreso on the coast of Yucatán, high levels of organochlorine pesticides were detected; 60.53% of samples exceeded the Acceptable Daily Intake (ADI) value for heptachlors, and high levels of p,p′-DDE, γ-chlordane, β-hexacyclohexane were detected. 36% of the samples exceeded the FAO/WHO acceptable daily intake (ADI).
In the extensive study of organochlorine pesticide bioaccumulation in the blood of Mayan women with cervical cancer conducted in Yucatán in the 18 municipalities with the highest mortality, high levels of endosulfan were detected in the blood of Mayan women in the municipality of Tizimín, which is adjacent to the coastal municipality of Dzilám Bravo of this study. This area is highly vulnerable to groundwater contamination. One of the coastal municipalities studied in this study includes the port of Progreso, Yucatán, where levels of 4.09 mg/L of heptachlor were detected.
The effects of pesticides in drinking water and contaminated food on mothers during pregnancy and the prenatal period represent risk to the health of newborns. A study on neural tube defects carried out in Mexico during the period 1980-1997 reported an average mortality of 5.8/10,000 in live births. Anencephaly was the most common malformation with 37.7%, followed by spine bifida with 31.6%. There are various factors related to these defects which should continue to be studied, such as genetic polymorphisms, exposure or bioaccumulation to pesticides, as well as social factors of poverty and marginalization.
The state of Yucatán is in third place nationally in congenital malformations, deformities and chromosomal abnormalities and ranked first with the highest incidence of diseases related to tumors (neoplasms).
The Ministry of Health, in its National Epidemiological Bulletin (2023), reported for Yucatán 448 cases of malignant breast tumours, 112 cases of cervical cancer, 145 cases of leukaemia, 21 cases of microcephaly.
The results of this study indicate that coastal sea waters, estuaries, and drinking water, contain concentrations of organochlorine pesticides above Mexican standards and even higher than international standards, which threatens the sustainability of human health and the marine ecosystem.
Conclusions
The present study shows high levels of organochlorine pesticide contamination in seawater, estuarine water, and drinking water, in coastal municipalities of the state of Yucatan. These pesticides are prohibited and/or restricted for use at an international level because they are carcinogenic endocrine disruptors.
The soils’ and karstic aquifers’ high vulnerability and the high use of pesticides in agricultural activities inland easily filter into the groundwater, which finally flows and empties into coastal waters. It is recommended to establish agroecological programs for sustainable agriculture.
The levels of organochlorine pesticides detected in coastal waters bioaccumulate in marine species for human consumption, implying health and biodiversity risks.
It is recommended that a surveillance program be established to monitor levels of organochlorine pesticides in coastal waters, that infrastructure be improved to manage quality water for human consumption, and that disease prevention programs be established through participatory research methodologies.
Conflict of Interest: None.
Funding Statement: Support of the Facultad de Química de la Universidad Autónoma de Yucatán.
Project Registration: FQUI-2022-0001
Acknowledgements: To the Mayan communities in the coastal area for their willingness and facilities for taking water samples.
References
2. Girones, L., Oliva, A.L., Marcovecchio, J.E. et al. (2020a). Spatial Distribution and Ecological Risk Assessment of Residual Organochlorine Pesticides (OCPs) in South American Marine Environments. Curr Envir Health Rpt 7, 147–160 (2020). https://doi.org/10.1007/s40572-020-00272-7
3. Girones, Lautaro; Palacios, Pilar; Oliva, Ana Laura; Marcovecchio, Jorge Eduardo; Arias, Andres Hugo (2020b). Organochlorine pesticides (OCPs) in coastal marine environments: levels, fate, behavior, and effects on biota; Nova Science Publishers, Inc.; 2020; 143-197. https://ri.conicet.gov.ar/handle/11336/146285
4. Islam Md. Ariful, Nurul Amin S.M., Rahman M. Aminur, Juraimi Abdul Shukor, Uddin Md. Kamal, Brown Christopher L., Arshad Aziz (2022). Chronic effects of organic pesticides on the aquatic environment and human health: A review, Environmental Nanotechnology, Monitoring & Management, Volume 18, 2022, 100740, ISSN 2215-1532, https://doi.org/10.1016/j.enmm.2022.100740.
5. Syafrudin M, Kristanti RA, Yuniarto A, Hadibarata T, Rhee J, Al-onazi WA, Algarni TS, Almarri AH, Al-Mohaimeed AM. (2021). Pesticides in Drinking Water—A Review. International Journal of Environmental Research and Public Health. 2021; 18(2):468. https://doi.org/10.3390/ijerph18020468
6. Synan F. AbuQamar, Mohamed T. El-Saadony, Samar S. Alkafaas, Mohamed I. Elsalahaty, Sara S. Elkafas, Betty T. Mathew, Amal N. Aljasmi, Hajar S. Alhammadi, Heba M. Salem, Taia A. Abd El-Mageed, Rashed A. Zaghloul, Walid F.A. Mosa, Ahmed Ezzat Ahmed, Ahmed S. Elrys, Ahmed M. Saad, Fatimah A. Alsaeed, Khaled A. (2024). El-Tarabily, Ecological impacts and management strategies of pesticide pollution on aquatic life and human beings, Marine Pollution Bulletin, Volume 206, 2024, 116613, ISSN 0025-326X, https://doi.org/10.1016/j.marpolbul.2024.116613.
7. Calderón-Garcidueñas, Ana L., Martínez-Valenzuela, Ma. del Carmen, & Waliszewski-Kubiak, Stefan M. (2023). Pesticide exposure and its effects on intrauterine and postnatal development. Perinatología y reproducción humana, 37(1), 23-30. Epub 05 de junio de 2023. https://doi.org/10.24875/per.22000014
8. Noman, M.A., Feng, W., Zhu, G. et al. (2022). Bioaccumulation and potential human health risks of metals in commercially important fishes and shellfishes from Hangzhou Bay, China. Sci Rep 12, 4634 (2022). https://doi.org/10.1038/s41598-022-08471-y
9. Polanco Rodríguez, A. G., Navarro Alberto, J. A., Solorio Sánchez, J. Mena Rejón, G. J., MarrufoGómez, J.,& Del Valls, C. A. (2015). Contamination by organochlorine pesticides in the aquifer of the ring of Cenotes in Yucatán, México. Water and Environment Journal. https://doi.org/10.1111/wej.12080.
10. Polanco Rodríguez, A. G., Araujo León, J. A., López Cetz, R., Long, D., Álvarez Cervera, F. J., Barache, U., & Rosas Sánchez, D. H. (2022). Organochlorine pesticides in the drinking water of Merida and its Metropolitan Zone, a Karst Region. Urban Water Journal, 19(1), 40–50. https://doi.org/10.1080/1573062X.2021.1955279
11. Polanco Rodríguez Ángel Gabriel, Arankowsky Sandoval Gloria, Góngora Alfaro José Luis & Salgado Burgos H. (2024). Exposure to Contaminants in Karst Groundwater. In: Brears, R. (eds). The Palgrave Encyclopedia of Sustainable Resources and Ecosystem Resilience. Palgrave Macmillan, Cham. https://doi.org/10.1007/978-3-030-67776-3_20-1
12. Polanco Rodríguez Angel G., Riba López M. Inmaculada, Del Valls Casillas T Angel, Quattrocchi Patrizia, Alvarez Cervera Fernando J, Solorio Sánchez Francisco J & Jorge A. Navarro Alberto (2015). Risk perception and chronic exposure to organochlorine pesticides in Maya communities of Mexico. Human and Ecological Risk Assessment: An International Journal DOI:10.1080/10807039. 2015.1004159.
13. Polanco Rodríguez Angel Gabriel, Magaña Castro Teresa Virginia, Iuit Jorge Cetz, Quintal López Rocío (2019). Uso de agroquímicos cancerígenos en la región agrícola de Yucatán, México. Centro Agrícola, Cuba, Vol.46, No.2, abril-junio, 72-83, 2019
14. Bauer-Gottwein, P., Gondwe, B.R.N., Charvet, G. et al. Review: The Yucatán Peninsula karst aquifer, Mexico. Hydrogeol J 19, 507–524 (2011). https://doi.org/10.1007/s10040-010-0699-5
15. Aguilar-Duarte Y., Bautista F., Mendoza M. E., Frausto O., Ihl T., Delgado C. (2016). IVAKY: Index of Vulnerability to Pollution of Yucatecan Karstic Aquifer. Revista Mexicana de Ingeniería Química 2016, 15 (3).
16. Laicher Dylan, Benkendorff Kirsten, White Shane, Conrad Steve, Woodrow Rebecca L., Butcherine Peter, Sanders Christian J. (2022). Pesticide occurrence in an agriculturally intensive and ecologically important coastal aquatic system in Australia, Marine Pollution Bulletin, Volume 180, 2022, 113675, ISSN 0025-326X, https://doi.org/10.1016/j.marpolbul.2022.113675.
17. Hussein MA, Hammad OS, Tharwat AE, Darwish WS, Sayed-Ahmed A, Zigo F, Farkašová Z, Rehan IF. (2022). Health risk assessment of organochlorine pesticide residues in edible tissue of seafood. Front Vet Sci. 2022 Nov 28;9:1042956. doi: 10.3389/fvets.2022.1042956. PMID: 3654455 2; PMCID: PMC9761600.
18. Polanco Rodríguez Angel G., Riba López M. Inmaculada, Del Valls Casillas T Angel (2017). Monitoring of organochlorine pesticides in blood of Maya women with uterine cervix cancer from Yucatan, Mexico. Environmental Pollution, Volume 220, Part B, January 2017, Pages 853–862.
19. Polanco Rodríguez, Á. G., Riba López, M. I., DelValls Casillas, T. A., Araujo León, J. A., Kumar Prusty, B. A., & Álvarez Cervera, F. J. (2017b). Levels of persistent organic pollutants in breast milk of Maya women in Yucatán, México. Environmental Monitoring and Assessment, 189, 59 https://doi.org/10.1007/s10661-017-5768-y
20. Campanale Claudia Massarelli, Carmine, Losacco Daniela, Bisaccia Donatella, Triozzi Mariangela, Uricchio Vito Felice (2021). The monitoring of pesticides in water matrices and the analytical criticalities: A review, TrAC Trends in Analytical Chemistry, Volume 144, 2021, 116423, ISSN 0165-9936, https://doi.org/10.1016/j.trac.2021.116423.
21. Blackstone, N.T., Battaglia, K., Rodríguez-Huerta, E. et al. (2024). Diets cannot be sustainable without ensuring the well-being of communities, workers and animals in food value chains. Nat Food 5, 818–824 (2024). https://doi.org/10.1038/s43016-024-01048-0
22. Mnif, W., Hassine, A. I. H., Bouaziz, A., Bartegi, A., Thomas, O., & Roig, B. (2011). Effect of Endocrine Disruptor Pesticides: A Review. International Journal of Environmental Research and Public Health, 8(6), 2265-2303. https://doi.org/10.3390/ijerph8062265
23. INEGI (2020a). Instituto Nacional de Estadística, Geografía e Informática. México en cifras. Discapacidad, población con limitación en la actividad. Municipio de Progreso. https://www.inegi.org.mx/app/areasgeograficas/#tabMCcollapse-Indicadores
24. INEGI (2020b). Instituto Nacional de Estadística, Geografía e Informática. México en cifras. Discapacidad, población con limitación en la actividad. Municipio de Celestún. https://www.inegi.org.mx/app/areasgeograficas/?ag=31059#collapse-Indicadores
25. INEGI (2020c). Instituto Nacional de Estadística, Geografía e Informática. México en cifras. Discapacidad, población con limitación en la actividad. Municipio de Dzilám Bravo. https://www.inegi.org.mx/app/areasgeograficas/?ag=31059#tabMCcollapse-Indicadores
26. INEGI (2020d). Instituto Nacional de Estadística, Geografía e Informática. México en cifras. Discapacidad, población con limitación en la actividad. Municipio de Ría Lagartos. https://www.inegi.org.mx/app/areasgeograficas/?ag=31059#tabMCcollapse-Indicadores
27. Thakur, J.S., Rao1, B.T., Arvind, Rajwanshi, Parwana, H.K., Rajesh, Kumar1 (2008). Epidemiological study of high cancer among rural agricultural community of Punjab in northern India. Int. J. Environ. Res. Public Health 5 (5), 399e407.
28. Tabrez, S., Ahmad, M. (2009). Toxicity, biomarker, genotoxicity and carcinogenicity of trichloroethylene (TCE) and its metabolites: a review. J Env Sci. Health, Part C 27 (3), 178e196.
29. Tabrez, S., Priyadarshini, M., Priyamvada, S., Khan, M.S., Arivarasu, N.A., Zaidi, S.K. (2014). Gene-environment interactions in heavy metal and pesticide carcinogenesis. Mutat. Res. 760, 1e9.
30. Beltran-Ontiveros SA, Contreras-Gutierrez JA, Lizarraga-Verdugo E, Gutierrez-Grijalva EP, Lopez-Lopez K, Lora-Fierro EH, Trujillo-Rojas MA, Moreno-Ortiz JM, Cardoso-Angulo DL, Leal-Leon E, Zatarain-Lopez JR, Cuen-Diaz HM, Montoya-Moreno M, Arce-Bojorquez B, Rochin-Teran JL, Cuen-Lazcano DE, Contreras-Rodriguez VA, Lascurain R, Carmona-Aparicio L, Coballase-Urrutia E, Gallardo-Vera F, Diaz D., 2023. National Burden and Trends for 29 Groups of Cancer in Mexico from 1990 to 2019: A Secondary Analysis of the Global Burden of Disease Study 2019. Cancers (Basel). 2023 Dec 28;16(1):149. doi: 10.3390/cancers16 010149. PMID: 38201576; PMCID: PMC10778521.
31. Ferlay, J., Soerjomataram, I., Dikshit, R., Eser, S., Mathers, C., Rebelo, M., et al. (2015). Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 136, E359eE386.
32. Khan Muhammad, Mahmood Hafiz Zahid, Damalas Christos A. (2015). Pesticide use and risk perceptions among farmers in the cotton belt of Punjab, Pakistan, Crop Protection, Volume 67, 2015, Pages 184-190, ISSN 0261-2194, https://doi.org/10.1016/j.cropro.2014.10.013.
33. Tran KB, Lang JJ, Compton K., Xu R., Acheson AR, Henrikson HJ, Kocarnik JM, Penberthy L., Aali A., Abbas Q. (2022). La carga mundial de cáncer atribuible a factores de riesgo, 2010-2019: un análisis sistemático para el estudio de la carga mundial de enfermedad 2019. Lancet, 2022;400: 563–591. doi: 10.1016/S0140-6736(22)01438-6.
34. Grondona Sebastian Iván, Lima María Lourdes, Massone Héctor Enrique, Miglioranza Karina Silvia Beatriz (2023). Pesticides in aquifers from Latin America and the Caribbean, Science of The Total Environment, Volume 901, 2023, 165992, ISSN 0048-9697, https://doi.org/10.1016/j.scitotenv.2023.165992.
35. Basu Sanghamitra, Chanda Abhra, Gogoi Pranab, Bhattacharyya Subarna (2021). Organochlorine pesticides and heavy metals in the zooplankton, fishes, and shrimps of tropical shallow tidal creeks and the associated human health risk, Marine Pollution Bulletin, Volume 165, 2021, 112170, ISSN 0025-326X, https://doi.org/10.1016/j.marpolbul.2021.112170.
36. Muñoz-Armenta, G., Pérez-González, E., Rodríguez-Meza, G.D. et al. (2022). Health risk of consuming Sphoeroides spp. from the Navachiste Lagoon complex due to its trace metals and organochlorine pesticides content. Sci Rep 12, 18393 (2022). https://doi.org/10.1038/s41598-022-22757-1
37. Melanda VS, Galiciolli MEA, Lima LS, Figueiredo BC, Oliveira CS. (2022). Impact of Pesticides on Cancer and Congenital Malformation: A Systematic Review. Toxics. 2022 Nov 9;10(11):676. doi: 10.3390/toxics10110676
38. Taiwo Adewale Matthew (2019). A review of environmental and health effects of organochlorine pesticide residues in Africa, Chemosphere, Volume 220, 2019, Pages 1126-1140, ISSN 0045-6535, https://doi.org/10.1016/j.chemosphere.2019.01.001.
39. Rodas-Ortíz, J.P., Ceja-Moreno, V., González-Navarrete, R.L., Alvarado-Mejía Jorge, Rodríguez-Hernández Marissa E. & Gold-Bouchot Gerardo (2008). Organochlorine Pesticides and Polychlorinated Biphenyls Levels in Human Milk from Chelem, Yucatán, México. Bull Environ Contam Toxicol 80, 255–259 (2008). https://doi.org/10.1007/s00128-007-9356-7
40. Rohweder Ricardo, Salcedo Arteaga Shirley, da Silva Gomes Vithória Luz, Casanova Schulze Paulo Alfredo, and Schuler-Faccini Lavinia (2024). Pesticide Exposures during Pregnancy and Health Outcomes in Latin America and the Caribbean: A Scoping Review of Human Observational Studies. Journal of Health and Pollution, Volume 12, Issue 1-4. https://ehp.niehs.nih.gov/doi/10.1289/JHP1043.
41. La influencia de factores asociados a estos defectos, como determinados polimorfismos genéticos, la deficiencia de ácido fólico, la obesidad materna, la exposición laboral a plaguicidas y la pobreza deberán evaluarse mediante estudios específicos.
42. Yucatán Voluntary Subnational Report (2020). Government of the State of Yucatan. https://www.local2030.org/pdf/vlr/yucatan-2020-min.pdf
43. Ministre of Health (2024). Epidemiological National Bulletin, Yucatan, Mexico. https://www.gob.mx/cms/uploads/attachment/file/964745/sem52.pdf




