Ocular Tuberculosis: Diagnosis and Treatment Insights
Ocular Tuberculosis: Unilateral, Asymmetric, and Varied in Presentation
Lester H. Lambert, MD1; Carli M. Deitel, MD2; Caleb Choi, MD2; Brittany E. Powell, MD2
- Marine Air Support Squadron-3, Marine Air Control Group-38, First Marine Expeditionary Force, Camp Pendleton, CA 92058.
- Department of Ophthalmology, Naval Medical Center San Diego, San Diego, CA 92134.
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Lambert, LH., Deitel, CM., et al., 2025. Ocular Tuberculosis: Unilateral, Asymmetric, and Varied in presentation. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6356
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6356
ISSN 2375-1924
ABSTRACT
Tuberculosis is an infectious illness that can causes substantial morbidity and mortality globally. It is a “great masquerader,” as it can affect any organ system, in a variety of manifestations. The eye is no exception. Ocular tuberculosis can affect any part of the eye: the anterior segment, posterior segment, or adnexa. A common ocular manifestation is choroiditis. However, it may also present as panuveitis, retinal vasculitis, and neuroretinitis. Involvement of the posterior segment can produce substantial inflammation, increasing the risk of damage to the neurosensory retina, optic nerve, and surrounding tissues, resulting in irreversible vision loss. Thus, it is imperative to identify ocular tuberculosis as early as possible to initiate vision-saving treatment. Here, we present two cases of tuberculous chorioretinitis that demonstrate the wide spectrum of clinical presentation of posterior segment ocular involvement and its characteristic of being asymmetric or unilateral. These cases exemplify the importance of maintaining TB on a broad differential diagnosis of ocular inflammation.
Keywords: Ocular tuberculosis; Chorioretinitis; Mycobacterium; Vitritis; Neuroretinitis
Introduction:
Tuberculosis (TB) is an infectious illness with extremely high morbidity and mortality rates worldwide. Mycobacterium tuberculosis is the primary cause of the illness. It belongs to the Mycobacteriaceae family alongside other pathogenic bacterial species, M. bovis and M. africanum, which also cause clinical TB infection. Nearly a third of the world’s population is latently infected with TB, and more than nine million new cases are diagnosed annually. A disproportionally large disease burden is carried by developing countries, up to 95% of new cases by some estimates. Reported incidence of ocular TB varies widely, based on population and diagnostic criteria. For example, ocular disease has been described in 1.4% to 10.5% of TB cases in high-TB incidence countries, but is less common in low-TB incidence countries. Accordingly, TB occurs in <1% of suspected infectious uveitis cases in the United States.
TB is an airborne communicable disease that most commonly affects the lungs. However, as a “great masquerader” it may affect any organ in a variety of ways. The eye is no exception. TB can affect any part of the eye, and may present unilaterally or with marked asymmetry. Posterior uveitis is the most common presentation of intraocular TB. Even this single umbrella category of posterior uveitis includes many manifestations: choroidal tubercles, multifocal serpiginous-like choroiditis, chorioretinitis, diffuse retinitis, neuroretinitis, occlusive vasculitis, and vitritis. Many of these presentations are similar to other infectious, inflammatory, and neoplastic posterior segment conditions, such as syphilis, toxoplasmosis, fungal infection, sarcoidosis, white dot syndromes, and malignancy. Therefore, a high index of suspicion for TB is needed for any ocular inflammation.
Early diagnosis is critical, as treatment is available for ocular TB. Prompt treatment can prevent devastating destruction to vision, and potentially other organ systems, as ocular involvement may be the earliest sign of disseminated disease. Here, we present two distinct cases in the United States of asymmetric ocular TB as the only manifestation of the disease.
Case presentation 1:
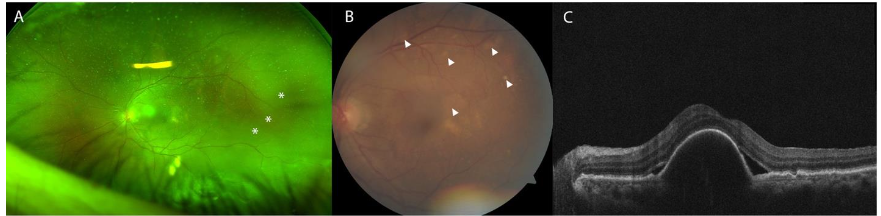
A 53-year-old female without a past ophthalmic history and no relevant medical history presented to the clinic for two weeks of worsening photopsias in the right eye associated with headaches. A recent evaluation by the emergency department for her symptoms resulted in a diagnosis of ocular migraines, and she was discharged with sumatriptan. However, her symptoms progressed despite this treatment and she was subsequently referred to ophthalmology. On presentation to ophthalmology, the patient denied floaters, photophobia, eye pain, or recent illness. She had no history of autoimmune disease. On evaluation, the patient’s best corrected visual acuity (BCVA) was 20/20 in each eye. The ocular surface and anterior segment were normal. Fundoscopic exam of the right eye was notable for peripheral gray-white choroidal lesions superonasally and inferiorly.
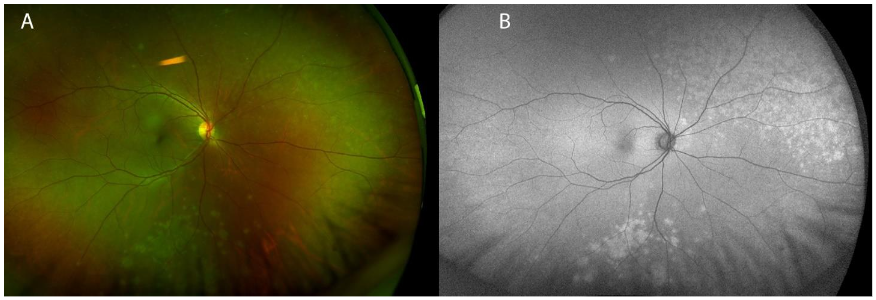
No vitreous inflammation was evident on the exam, which was otherwise normal. Fundus autofluorescence (FAF) showed confluent hyperautofluorescence corresponding to the choroidal lesions on the exam.
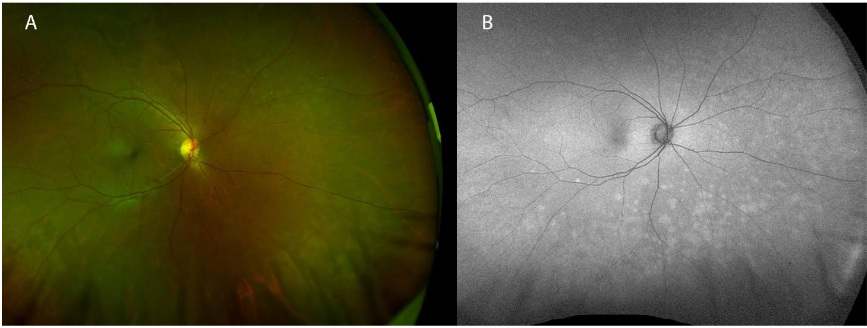
At presentation, the differential diagnosis included various infectious and inflammatory etiologies: syphilis, tuberculosis, sarcoidosis, white dot syndromes (e.g. multiple evanescent white dot syndrome (MEWDS) was highest on the differential given the unilateral presentation, but others include acute posterior multifocal placoid pigment epitheliopathy (APMPEE), multifocal choroiditis, and panuveitis (MCP), punctate inner choroidopathy (PIC), and birdshot chorioretinopathy). A laboratory evaluation was performed and notable for a positive interferon gamma release assay (IGRA), indicating the presence of tuberculosis infection. Subsequent exams were notable for the progression of ocular symptoms and headache, corresponding to an increase number of lesions on exam. The left eye remained normal. Evaluation by the Infectious Disease service concluded her only risk factor was that she grew up in the Philippines. Further testing included chest imaging and a lumbar puncture, which was negative for pulmonary and central nervous system involvement, respectively. Treatment was initiated with a regimen of rifapentine, isoniazid, pyrazinamide, and ethambutol (RIPE). The patient’s symptoms resolved within two weeks of starting RIPE therapy, and her exam returned to baseline. The patient remained symptom-free, and her exam remained normal following the completion of RIPE therapy a year following completion of treatment.
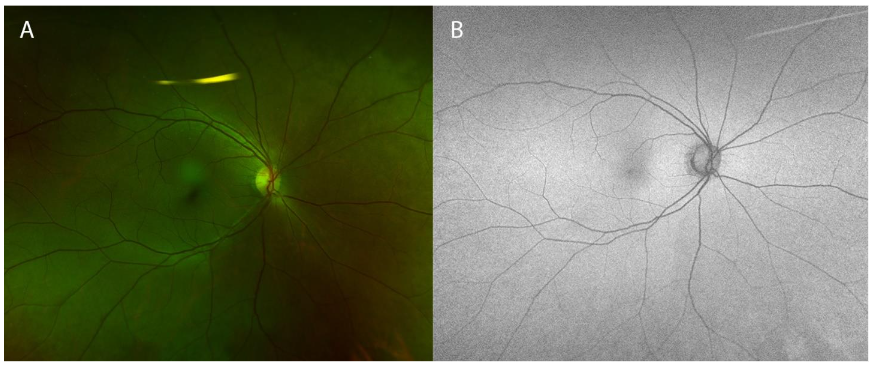
Case presentation 2:
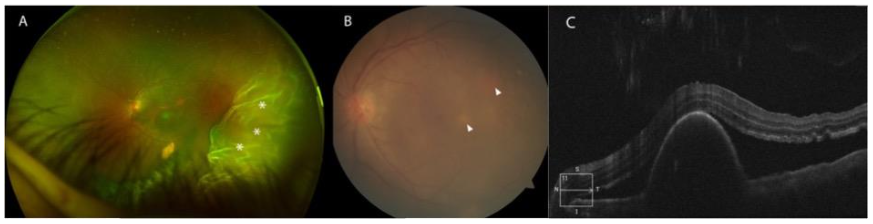
A 64-year-old female with diabetes and hypertension presented to the clinic complaining of two weeks of left eye pain, redness, and worsening vision. The patient’s ocular history was notable for idiopathic polypoidal choroidal vasculopathy (IPCV) complicated by macular hemorrhage in the left eye, successfully treated with pneumatic displacement and intravitreal injection of anti-vascular endothelial growth factor (anti-VEGF) and tissue plasminogen activator (tPA). She presented for a routine appointment for continued intravitreal anti-VEGF injections. BCVA in the affected eye following the macular hemorrhage was 20/70. At the time of her routine injection appointment, she had reported two weeks of worsening left eye pain, redness, and decreased vision. The BCVA of the left eye was 20/80, and she had severe anterior chamber inflammation with 270 degrees of posterior synechiae. Posteriorly, exam was notable for vitreous inflammation, exudative retinal detachment, and new gray-white choroidal lesions in the posterior pole.
The patient was started on oral and topical steroids with a resultant improvement of the exudative detachment, and her BCVA improved to 20/60. Despite this, the previously noted gray-white choroidal lesions increased. Fluorescein angiography was relatively unremarkable, without vasculitis or optic disc inflammation. A laboratory evaluation revealed positive IGRA, confirming tuberculosis infection. The patient urgently returned for a sudden change in vision and new photopsias, after two weeks of significant improvement on oral and topical steroids and cycloplegia. Her BCVA worsened to 20/100, and the exam showed increased number of choroidal lesions, new small retinal holes, and a new rhegmatogenous retinal detachment.
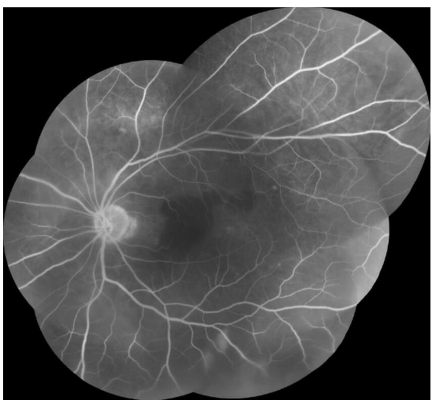
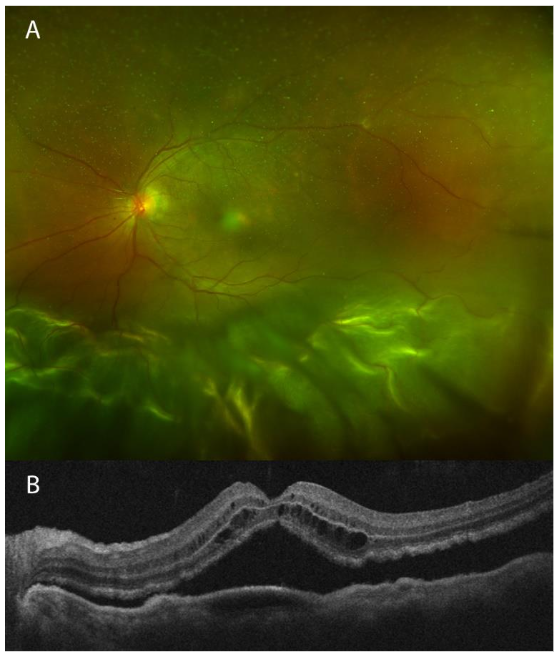
Infectious Disease specialists evaluated the patient. This patient too was from the Philippines, which was the only identified risk factor. No pulmonary or other extrapulmonary involvement was present. RIPE therapy was initiated before ophthalmic surgical intervention. Subsequently, the patient underwent repair of the complex retinal detachment with combined cataract extraction and retinal detachment repair with a silicone oil tamponade. Following surgery and RIPE therapy, the patient’s visual acuity remained stable at 20/200, and her retina remained flat with a resolution of the choroidal lesions.
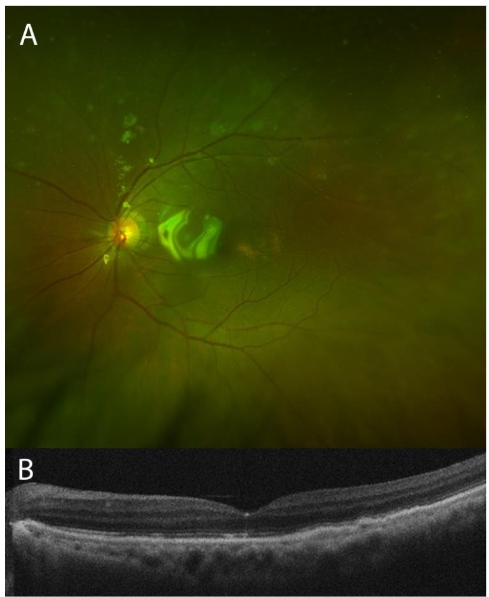
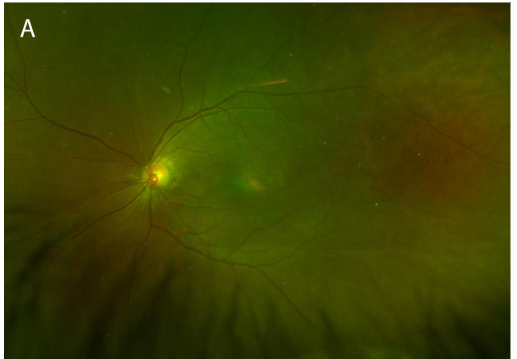
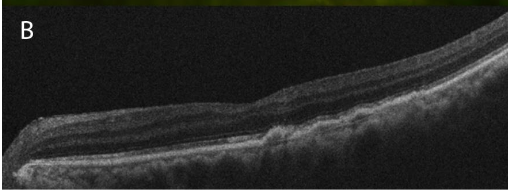
Discussion:
While TB most commonly affects the lungs, extrapulmonary disease is common. In the US in 2019, 30% of patients diagnosed with TB had extrapulmonary disease including two thirds without pulmonary involvement. Ocular disease has been described in as many as 10.5% of TB cases in high-TB incidence countries, and may be the earliest sign of disseminated disease. The incidence of ocular TB among patients presenting with uveitis is high in high-TB incidence countries, but occurs in <1% of suspected infectious uveitis cases in the US. Filardo et al published epidemiology of ocular TB in the United States between 1993-2019. This article found that in the US, ocular TB is found most commonly, and about in equal rates in Asians, Blacks, and Latinos, at 25%, 28%, and 29%, respectively. Ocular TB occurs more commonly in those born outside of the US (65%), compared to those born in the US (35%). The vast majority of ocular TB cases (93%) do not have concomitant pulmonary disease. Finally, 52% of ocular TB cases had no epidemiologic risk factors (e.g. known prior untreated latent TB infection, homelessness, correctional or long-term facility resident, contact with TB-infected person within two years, and occupation as a healthcare worker). Our two patients with ocular TB therefore represent a rare disease in the US. However, as Asians born outside of the US without pulmonary involvement or epidemiologic risk factors, they both represent the most common epidemiologic categories of ocular TB in the US.
Ocular transmission can occur in three ways: primary ocular TB occurs as the primary site of inoculation, and is characteristic of disease on the ocular surface. Secondary ocular TB occurs via hematogenous spread from a distant infectious focus, and is characteristic of uveal disease. Finally, a hypersensitivity reaction occurs when the immune system reacts to tuberculosis bacteria elsewhere, resulting in ocular inflammation. Any ocular structure may be affected by tuberculosis, even without pulmonary involvement. Furthermore, ocular infection can present unilaterally or with marked asymmetry. Some examples of relatively superficial manifestations, though not an exhaustive list, include: on the eyelid as an abscess, chronic blepharitis, atypical chalazion, or reddish-brown nodules that blanch to an “apple jelly” color with pressure; on the ocular surface and anterior segment as a mucopurulent conjunctivitis with regional lymphadenopathy, a phlyctenule (an inflammatory nodule at the junction of the cornea and sclera), infectious keratitis, interstitial keratitis (inflammation within the cornea stroma), infectious scleritis, or anterior uveitis. These presentations are more straightforward to diagnose as tissue is easily accessible for biopsy and culture. Orbital disease can also occur, but is uncommon.
Uveitis, and particularly posterior uveitis, is the most common presentation of intraocular TB. This category includes a wide array of presentations affecting the vitreous, retina, retinal vasculature, choroid, and optic disc. Of these, the choroid is most often affected, which is thought to result from the bacteria’s predilection for highly oxygenated tissue. Indeed, our patients presented with choroidal tubercles. In the first case, these were isolated. In the second, they were accompanied by vitritis and retinal detachment, though this was in the setting of prior retinal disease (IPCV).
A classic and visually devastating manifestation of posterior uveitic TB is serpiginous-like choroiditis. It is distinct from idiopathic serpiginous choroiditis, as lesions in the latter usually extend from the optic nerve to macula, whereas in 85% of serpiginous-like choroiditis the lesions are not contiguous with the optic disc. This distinction is paramount, as disease associated with TB may be treatable, while the idiopathic form is not and carries a poor visual prognosis.
The gold standard for diagnosis is identification of Mycobacterium species in culture. Such studies can be particularly difficult in cases of ocular tuberculosis, as obtaining tissue samples for culture is invasive. PCR of ocular fluid is a newer alternative to cultures. Often, presumed ocular TB is the working diagnosis when the clinical picture is right: risk factor history, ocular findings, chest radiography results, and the Mantoux tuberculin skin test (TST) or Interferon Gamma Release Assay (IGRA). Negative testing does not rule out ocular tuberculosis.
If tissue can be obtained, bacteriological staining techniques can be diagnostic. Ziehl-Neelson acid-fast stained bacilli and/or caseous necrosis with epithelioid cells and Langerhans giant cells suggest ocular tuberculosis. Bacteria in multinucleated giant cells are inconsistently detected by histologic staining. Thus, the results are highly variable, and biopsies can be unreliable. A biopsy may be taken for histopathologic analysis in rare external disease or anterior segment involvement cases. However, intraocular tissue biopsy can be risky and impractical.
Treatment of tuberculosis is the similar, whether the eye or lungs are affected. Ocular tuberculosis is treated with a multidrug regimen of antitubercular antibiotics (e.g. RIPE therapy), and steroid medication is frequently necessary. Multidrug resistant TB must be approached with different treatment, and indeed represents a significant global public health and safety concern, that is beyond the scope of this discussion. In rare instances, administration of ethambutol may cause optic neuropathy, which resolves with discontinuation of the drug. Nonetheless, high-risk patients, such as those taking ethambutol at a high dose or for an extended period or those with impaired renal function, should undergo regular visual examination. Treatment can halt the progression, and often reverse, much of the visual morbidity caused by TB, as was seen in our cases.
Conclusion:
Ocular TB is an infection by Mycobacterium, that can occur with or without pulmonary involvement. Disease manifestation is varied and often asymmetric between the eyes. Involvement of the posterior segment can be particularly damaging to vision. TB must be on the broad differential for inflammation in any ocular tissue. Early diagnosis is important, as treatment exists, and ocular TB may be the earliest sign of disseminated disease. Therefore, treatment may save vision and improve the prognosis of other potentially affected organs.
Conflict of Interest Disclosures:
The authors have no conflicts of interest to declare.
Disclosures:
The authors report having no proprietary or commercial interest in any materials discussed in this letter. The views expressed in this correspondence are those of the author(s) and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the United States Government. This work was prepared as part of our official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the United States Government.” Title 17 U.S.C. 101 defines a United States Government work as a work prepared by a military service member or employee of the United States Government as part of that person’s official duties.
Funding Statement:
None declared.
Clinical Trial Registration:
Not applicable.
Institutional Review Board (Human Subjects):
Not applicable.
Institutional Animal Care and Use Committee (IACUC):
Not applicable.
Acknowledgements:
The authors thank the hospital corpsmen and support staff of Naval Medical Center San Diego for the care of the patients. We thank the patients for granting permission to publish this information.
Author Contributions:
All authors contributed to the writing of the manuscript. Dr. Lambert (LT, MC, USN), Dr. Powell (CDR, MC, USN), and Dr. Choi (LT, MC, USN) wrote the original manuscript. Dr. Deitel (LT, MC, USN) and Dr. Powell (CDR, MC, USN) revised it.
References:
- Kochi A. The global tuberculosis situation and the new control strategy of the World Health Organization. Tubercle. 1991 Mar;72(1):1-6. doi: 10.1016/0041-3879(91)90017-m.
- Biswas J, Badrinath SS. Ocular morbidity in patients with active systemic tuberculosis. Int Ophthalmol. 1995;19(5):293-8. doi:10.1007/BF00130924
- Donahue HC Ophthalmologic experience in a tuberculosis sanatorium. Am J Ophthalmol 1967;64742- 748
- Islam SM, Tabbara KF. Causes of uveitis at The Eye Center in Saudi Arabia: a retrospective review. Ophthalmic Epidemiol 2002;9239- 249
- Zhang Y, et al. Incidence, prevalence, and risk factors of infectious uveitis and scleritis in the United States: a claims-based analysis. PLoS One 2020;15:e0237995.
- Alli HD, et al. Global prevalence and clinical outcomes of tubercular uveitis: a systematic review and meta-analysis. Surv Ophthalmol 2022;67:770-92.
- Wang EW, Okwesili CN, Doub JB. Tuberculosis the great masquerader. ID Cases. Jun 2022; 29:e01541. doi: 10.1016/j.idcr.2022.e01541.
- Thompson MJ, Albert DM. Ocular Tuberculosis. Arch Ophthalmol. 2005;123(6):844–849. doi:10.1001/archopht.123.6.844
- Shah JS, Shetty N, Shah SK, Shah NK. Tubercular Uveitis with Ocular Manifestation as the First Presentation of Tuberculosis: A Case Series. J Clin Diagn Res. Mar 2016;10(3):NR01-3. doi:10.7860/JCDR/2016/16219.7375
- Centers for Disease Control and Prevention. Reported tuberculosis in the United States, 2019.
- Filardo TD, et al. Epidemiology and Clinical Characteristics of Ocular Tuberculosis in the United States, 1993–2019. Open Forum Infectious Diseases, Sep 2024;11(9):1-8. https://doi.org/10.1093/ofid/ofae476
- Albert DM and Raven ML. Ocular Tuberculosis. Microbiol Spectr. 2016 Nov;4(6):10.1128. doi: 10.1128/microbiolspec.TNMI7-0001-2016
- Alvarez GG, Roth VR, Hodge W. Ocular tuberculosis: diagnostic and treatment challenges. Int J Infect Dis. Jul 2009;13(4):432-5. doi:10.1016/j.ijid.2008.09.018
- Kalia NP, Singh S, Hards K, et al. M. tuberculosis relies on trace oxygen to maintain energy homeostasis and survive in hypoxic environments. Cell Rep. May 30 2023;42(5):112444. doi:10.1016/j.celrep.2023.112444
- Srivastav S, Biswas A, Anand A. Interplay of niche and respiratory network in shaping bacterial colonization. J Biol Chem. Dec 9 2024;301(1):108052. doi:10.1016/j.jbc.2024.108052
- Bansal R, Gupta A, Gupta V, Dogra MR, Sharma A, Bambery P. Tubercular serpiginous-like choroiditis presenting as multifocal serpiginoid choroiditis. Ophthalmology. Nov 2012;119(11):2334-42. doi:10.1016/j.ophtha.2012.05.034
- Carreno E, Portero A, Herreras JM, Calonge M, Foster CS. Distinctive clinical features of idiopathic versus infectious serpiginous choroidopathy. Ocul Immunol Inflamm. Dec 2012;20(6):448-52. doi:10.3109/09273948.2012.723236
- Chawla R, Singh MK, Singh L, et al. Tubercular DNA PCR of ocular fluids and blood in cases of presumed ocular tuberculosis: a pilot study. Ther Adv Ophthalmol. Jan-Dec 2022;14:25158414221123522. doi:10.1177/25158414221123522
- Putera I, Widodo E, Riasanti M, et al. Ocular tuberculosis with Mycobacterium tuberculosis DNA presence in ocular fluid: will post-COVID era bring a difference? Eye (Lond). Oct 2024;38(15):3027-3029. doi:10.1038/s41433-024-03200-3
- Vasconcelos-Santos DV, Zierhut M, Rao NA. Strengths and weaknesses of diagnostic tools for tuberculous uveitis. Ocul Immunol Inflamm. Sep-Oct 2009;17(5):351-5. doi:10.3109/09273940903168688
- Dinnes J, Deeks J, Kunst H, et al. A systematic review of rapid diagnostic tests for the detection of tuberculosis infection. Health technology assessment. 2007;11(3)
- Ang M, Htoon HM, Chee SP. Diagnosis of tuberculous uveitis: clinical application of an interferon-gamma release assay. Ophthalmology. Jul 2009;116(7):1391-6. doi:10.1016/j.ophtha.2009.02.005
- Arora D, Dhanashree B. Utility of smear microscopy and GeneXpert for the detection of Mycobacterium tuberculosis in clinical samples. Germs. Jun 2020;10(2):81-87. doi:10.18683/germs.2020.1188
- Rasool G, Khan AM, Mohy-Ud-Din R, Riaz M. Detection of Mycobacterium tuberculosis in AFB smear-negative sputum specimens through MTB culture and GeneXpert((R)) MTB/RIF assay. Int J Immunopathol Pharmacol. Jan-Dec 2019;33:2058738419827174. doi:10.1177/2058738419827174
- Madhavan HN, Therese KL, Gunisha P, Jayanthi U, Biswas J. Polymerase chain reaction for detection of Mycobacterium tuberculosis in epiretinal membrane in Eales’ disease. Invest Ophthalmol Vis Sci. Mar 2000;41(3):822-5.
- Zhang YK, Fu HY, Guan Y, Li YJ, Bai HZ. Concurrent tuberculous chorioretinitis with choroidal neovascularization and tuberculous meningitis: a case report. BMC Ophthalmol. Jun 12 2020;20(1):227. doi:10.1186/s12886-020-01504-y
- Chamberlain PD, Sadaka A, Berry S, Lee AG. Ethambutol optic neuropathy. Curr Opin Ophthalmol. Nov 2017;28(6):545-551. doi:10.1097/ICU.0000000000000416
- Saxena R, Singh D, Phuljhele S, et al. Ethambutol toxicity: Expert panel consensus for the primary prevention, diagnosis and management of ethambutol-induced optic neuropathy. Indian J Ophthalmol. Dec 2021;69(12):3734-3739. doi:10.4103/ijo.IJO_3746_20

