Idiopathic Acute Exudative Polymorphous Vitelliform Maculopathy
A Case of Idiopathic Acute Exudative Polymorphous Vitelliform Maculopathy
Timothy Lee, MD1,2,3
- Walter Reed National Military Medical Center, Bethesda Maryland 20814
- Naval Hospital Camp Pendleton, Oceanside California 92058
- Naval Medical Center San Diego, San Diego California 92134
OPEN ACCESS
PUBLISHED:28 February 2025
CITATION: Lee, T., Skinner, A., et al., 2025. A Case of Idiopathic Acute Exudative Polymorphous Vitelliform Maculopathy. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6264
DOI https://doi.org/10.18103/mra.v13i2.6264
ISSN 2375-1924
ABSTRACT
Acute exudative polymorphous vitelliform maculopathy is a recently described rare disease that consists of vitelliform lesions and serous macular detachment. Other disorders, including the bestrophinopathies and paraneoplastic polymorphous retinopathy, may share the same vitelliform lesions as acute exudative polymorphous vitelliform maculopathy and could cause potential confusion in attaining the diagnosis. Here we present a case of a 22-year-old male who presented with fundoscopic examination findings of vitelliform lesions that were consistent with the aforementioned disorders. Consequently, the patient underwent an extensive diagnostic work-up, including BEST1 gene testing, electroretinogram/electrooculogram testing, and a paraneoplastic etiology testing, all of which returned negative. With the result of this work-up, the most likely diagnosis for this patient is acute exudative polymorphous vitelliform maculopathy. Given that a number of disorders present with findings of vitelliform lesions, it is important for the clinician to remain cautious and to keep acute exudative polymorphous vitelliform maculopathy in the differential diagnosis. In summary, acute exudative polymorphous vitelliform maculopathy is a rare disease consisting of vitelliform lesions and serous macular detachment that can look similar to other inheritable or acquired retinopathies. Due to this, it is vital to perform thorough testing and evaluation to rule out other potential causes of these examination findings.
Keywords:
Acute exudative polymorphous vitelliform maculopathy (AEPVM), bestrophinopathies, diagnostic work-up, vitelliform lesions
CASE REPORT
A Case of Idiopathic Acute Exudative Polymorphous Vitelliform Maculopathy
Acute exudative polymorphous vitelliform maculopathy (AEPVM) is a rare disease only recently described in literature. AEPVM is characterized by acute visual acuity loss accompanied by yellowish subretinal vitelliform lesions and serous macular detachments, as initially described by Gass et al. Although there may be no evident cause of AEPVM, it is thought that underlying retinal pigment epithelium dysfunction causes the over-accumulation of lipofuscin, owing to the yellowish color of the lesions. While one of the defining features of AEPVM is vitelliform lesions, this feature is also shared with other disorders that may be more common, including best vitreomacular dystrophy (BVMD) and autosomal recessive bestrophinopathy amongst many others. Compared to other disorders with vitelliform lesions, the characteristics of AEPVM include the progression of a serous macular detachment and the presence of vitelliform lesions that are polymorphous. As a result of its infrequent presentation, AEPVM tends to be an idiopathic disease process, with no clearly understood pathophysiology. In some cases, AEPVM may occur as a result of a malignancy, aptly named acute exudative polymorphous paraneoplastic vitelliform maculopathy. In fact, cases of acute exudative polymorphous paraneoplastic vitelliform maculopathy have been described in patients with metastatic or choroidal melanoma. AEPVM may even be the sequela of viral infectious etiologies such as coronavirus disease 2019 (COVID-19) and Ebstein Barr Virus. Interestingly, commonalities in all these etiologies of AEPVM include the aforementioned polymorphous vitelliform lesions and serous macular detachments.
In terms of a clinical course, patients with AEPVM typically have acutely reduced visual acuity due to these vitelliform lesions and serous macular detachments. However, the prognosis of AEPVM is typically good, with most patients having improved or recovered visual acuity over the span of months to years. Regarding potential treatment, the role of steroid medications in patients with AEPVM is not well characterized, although rapid improvements have been described in patients with the use of intravenous methylprednisolone boluses. However, the response to steroids can be variable. Even use of an intravitreal dexamethasone implant did not generate a robust response and improvement in the clinical course of a patient with AEPVM. Even with the potential recovery associated with steroid use, most patients will recover without the use of such interventions. However, even with good outcomes with recovered visual acuity, a number of patients will continue to have persistent yellowish or curvilinear deposits for an indefinite period of time. This is in contrast to some of the other similar presenting retinal disorders. For example, BVMD can present initially with mild to normal vision loss but later stages of the disease can lead to scar tissue formation and geographic atrophy, which lead to irreversible vision loss.
When faced with a patient who presents with vitelliform lesions, it is important to consider AEPVM in the differential diagnosis, as it can be often overlooked due to disorders that present similarly and are more common. Different testing modalities can be used to ultimately exclude these more common disorders and to make the diagnosis of AEPVM. Below we present an interesting case of a young male who presented to clinic with symptoms and findings that were highly suspicious for AEPVM.
Case Presentation
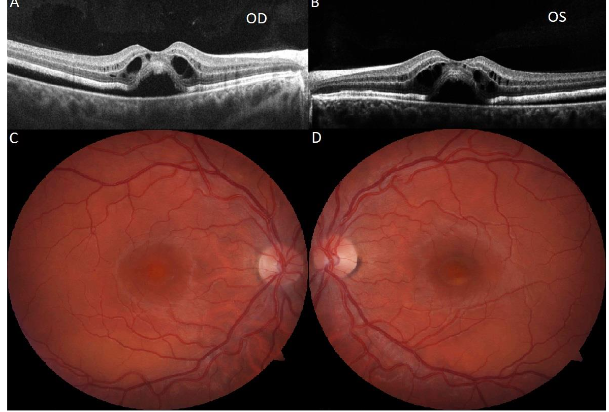
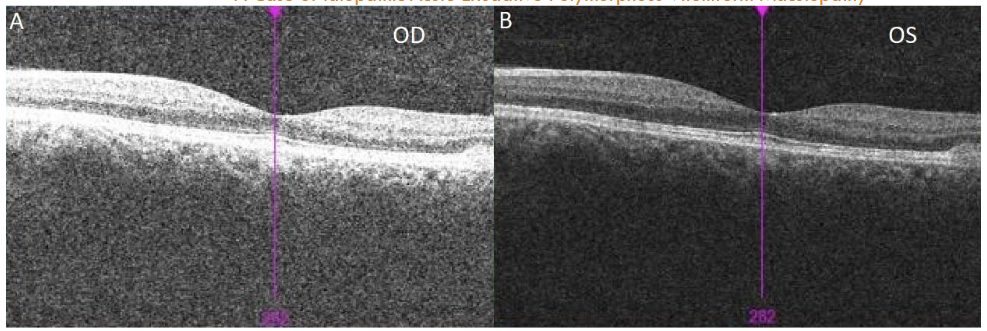
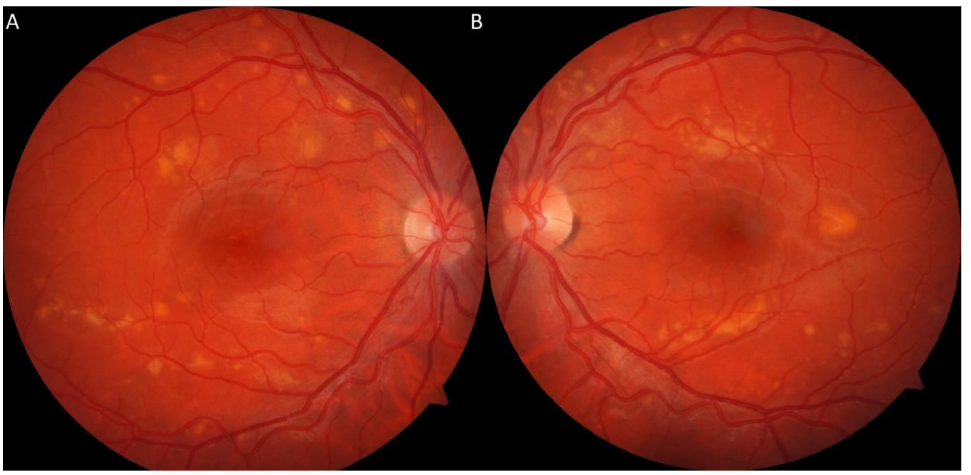
A 22-year-old male with an ocular history of amblyopia, hyperopia, and significant nicotine use presented to clinic with one week of blurry vision and headaches after moving heavy objects. On evaluation, the patient’s best corrected visual acuity (BCVA) was 20/40 in the right eye and 20/80 in the left eye. Fundoscopic exam revealed macular elevations with areas of hypopigmented demarcation inferiorly and vitelliform pseudohypopyons inferior to the fovea with few yellow flecks along the arcades. No vitritis was evident on exam. Ocular Coherence Tomography (OCT) showed subretinal fluid, intraretinal cysts, shallow neurosensory retinal detachment, and a thickened appearance to the retinal pigmented epithelium (RPE) in both eyes with vitelliform deposits inferior to the left fovea.
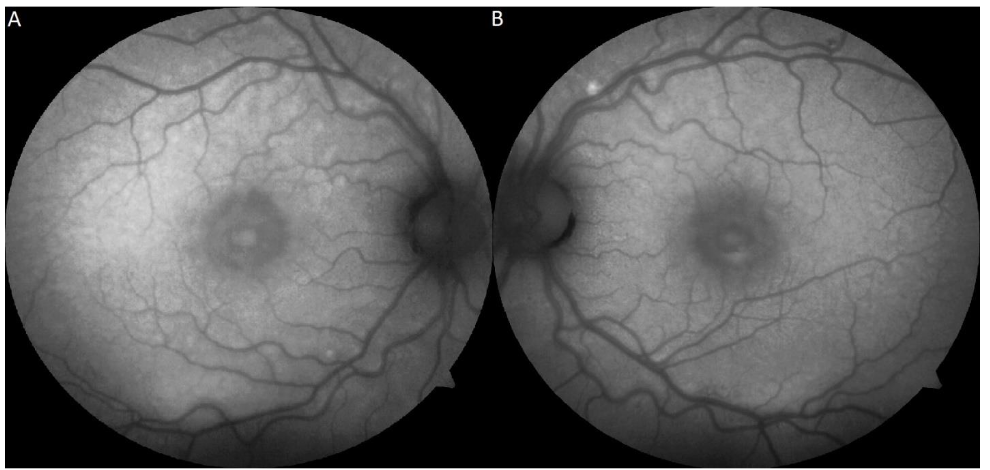
Fundus autofluorescence (FAF) showed patchy areas of hyper- and hypo-autofluorescence predominantly along the vascular arcades and macula in both eyes.
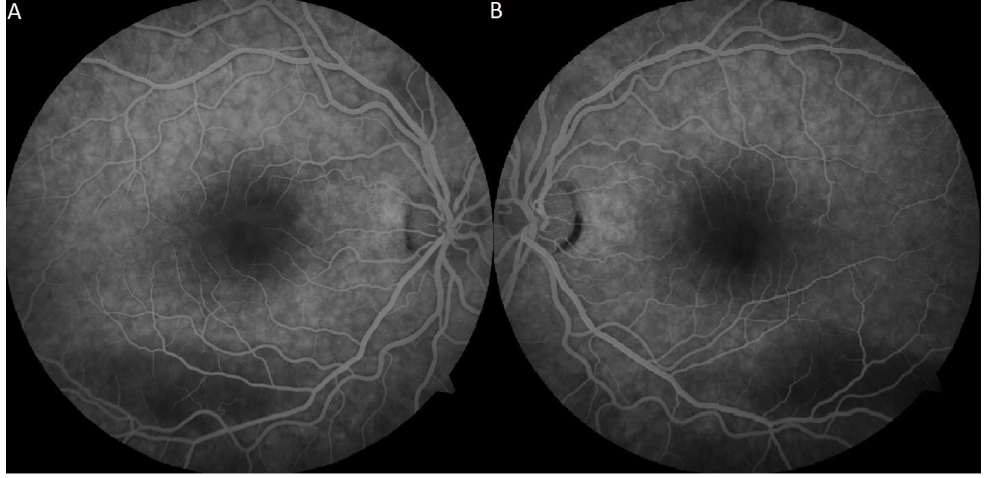
Intravenous fluorescein angiography was notable for patchy choroidal filling, inferior hypofluorescence corresponding to areas of hypopigmented demarcation and thickened RPE in both eyes along with hypofluorescence of the vitelliform lesion in the left eye.
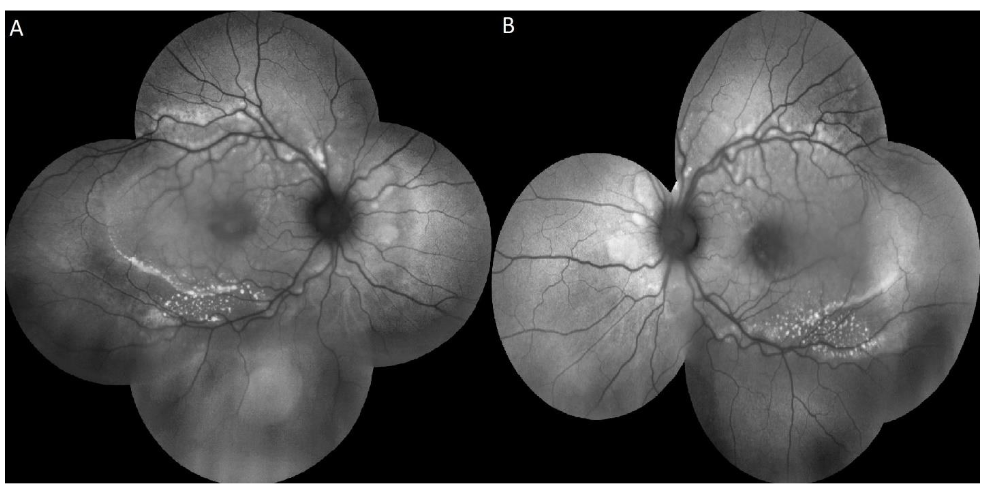
FAF revealed an increased number of hyper-autofluorescent flecks along the arcades and within the posterior pole.
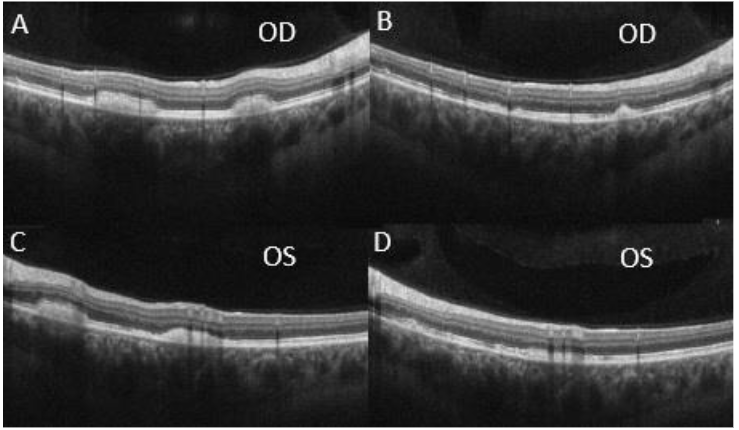
At this time, the differential diagnosis included autosomal recessive Bestrophinopathy, BVMD, Juvenile X-linked Retinoschisis (JXLR or XLRS), toxic retinopathy, as well as paraneoplastic polymorphous retinopathy due to the patient’s significant tobacco history. Further diagnostic work-up was deemed necessary at this time to determine the etiology of this patient’s presentation and the patient was subsequently referred for genetic testing, electroretinogram (ERG)/electrooculogram (EOG), and a paraneoplastic etiology work-up. The patient’s genetic testing for the BEST1 gene returned negative and the patient’s ERG/EOG resulted in a borderline EOG result with normal Arden ratio in the right eye (1.85) and reduced Arden ratio in the left eye (1.51). The paraneoplastic evaluation was notable for a computerized tomography scan which showed a 1cm hilar lymphadenopathy and a subsequent endobronchial ultrasound biopsy was noted to have a benign-appearing morphology with low-risk for malignancy. During this time, interval OCT scans showed improvements with resolution of subretinal fluid at 11 months and reduction of the vitelliform lesions in the peripheral macula from 15 to 17 months at initial presentation.
The patient’s negative genetic test for BEST1 and non-specific ERG/EOG made Bestrophinopathy, BVMD, or other inheritable retinal disorders less likely. The systemic workup for a malignancy also returned negative, making paraneoplastic polymorphous retinopathy less likely as well. Furthermore, the patient lacked evidence of inflammation on fundoscopic exam, making Voyt-Koyanagi-Harada Syndrome and other inflammatory etiologies less likely. With the current work-up, the most probable explanation of the patient’s findings on exam and diagnostic imaging is most likely AEPVM. Separately, smoking cessation counseling was provided and encouraged at each visit and he was referred to his primary care physician for additional intervention.
Discussion
In this case, it was determined that the patient was most likely to have idiopathic AEPVM. However, the patient’s fundoscopic exam showed evidence of vitelliform lesions and subretinal retinal fluid with macular detachment, of which both findings could be found in other retinal disorders. Given the extensive differential diagnoses, other features are important to consider in ruling out other disease entities. On fundoscopic exam, there was an absence of an inflammatory process on exam for this patient, making inflammatory etiologies, including Voyt-Koyanagi-Harada syndrome, less likely. Other genetic disorders can present similarly to AEPVM, so further diagnostic workup and testing were pursued for this patient. For this matter, the patient underwent specific genetic testing and testing for malignancy through CT scan of the chest and a subsequent biopsy. These results were negative for BVMD, autosomal recessive bestrophinopathy, and paraneoplastic polymorphous retinopathy. Based on these negative findings, the suspicion that idiopathic AEPVM remains high considering that no other causes were identified.
In terms of other tools that an ophthalmologist could use to help narrow the differential diagnoses, diagnostic imaging remains an important means of differentiating between different retinal disorders. Fundus autofluorescence (FAF) is a non-invasive diagnostic imaging tool that utilizes lipofuscin to highlight areas in the RPE where there is pathology. Given that the development of AEPVM is marked by lipofuscin overaccumulation, the intensity of the vitelliform lesions in FAF is directly correlated with the amount of lipofuscin. Ocular Coherence Tomography (OCT) is another non-invasive diagnostic imaging test that can show serous detachment, as well as other pathology, present in the macula. This modality can be used to show the extent of the serous detachment and be used to track its improvement or worsening over time. Although these can be used in many other ocular conditions, the utilization of these diagnostic imaging studies can be extremely useful in narrowing the differential diagnoses and determining the degree of the disease when regarding AEPVM.
In terms of treatment, AEPVM does not typically require any medical intervention. Even with acutely decreased visual acuity, patients usually recover or nearly recover most of their visual acuity. Steroid medications, either systemically or intraocularly, can be considered on an individual basis but do not guarantee immediate improvements. In this patient, after determining idiopathic AEPVM was the most likely etiology, in the absence of other explainable causes, no intervention was pursued. Subsequently, the patient’s objective exam improved with resolved subfoveal fluid on OCT and his visual acuity remains stable, only with some fluctuations in his left eye. With these findings, the patient will continue to be followed closely and monitored for any signs of recurrence.
In patients who present with vitelliform lesions, the clinician should remain cautious and keep AEPVM in the differential diagnosis. However, a high number of disorders present with vitelliform lesions and other similar findings which could cause confusion if one is not careful in the approach to a patient. In our case, the diagnosis was made following multiple rounds of negative diagnostic testing. Even with high suspicion of AEPVM, the clinician should continue to perform these diagnostic tests to rule out these more common disorders.
Conclusion
Acute exudative polymorphous vitelliform maculopathy is a rare ophthalmic disorder that is characterized by acute vision loss accompanied by polymorphous, subretinal vitelliform lesions and serous macular detachments. The cause of AEPVM is largely unknown at this time and can be idiopathic, although there are some associations with malignancies and viral etiologies. Since the presentation of AEPVM can be confused with other retinal disorders including more common genetic associations, such as the best vitreomacular dystrophy and autosomal recessive bestrophinopathy, it is vitally important to ensure thorough testing for these conditions. Other similar appearing conditions can include toxic retinopathy and paraneoplastic polymorphous retinopathy. Even with high suspicion of AEPVM, an ophthalmologist should consider and rule out these other disorders through thorough diagnostic testing before considering idiopathic AEPVM as a diagnosis.
Disclaimer
The opinions and assertions expressed herein are those of the author(s) and do not necessarily reflect the official policy or position of the Department of Defense.
Ethical Approval
This case report was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a Health Insurance Portability and Accountability Act (HIPAA)-compliant manner.
Statement of Informed Consent
Informed consent was obtained prior to performing the procedure, including permission for publication of all photographs and images herein.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References:
- Gass JD, Chuang EL, Granek H. Acute exudative polymorphous vitelliform maculopathy. Trans Am Ophthalmol Soc. 1988;86:354-66. PMID: 2979022; PMCID: PMC1298814.
- Barbazetto I, Dansingani KK, Dolz-Marco R, et al. Idiopathic Acute Exudative Polymorphous Vitelliform Maculopathy: Clinical Spectrum and Multimodal Imaging Characteristics. Ophthalmology. 2018;125(1):75-88. Doi:10.1016/j.ophtha.2017.07.020.
- Grunwald L, Kligman BE, Shields CL. Acute Exudative Polymorphous Paraneoplastic Vitelliform Maculopathy in a Patient with Carcinoma, Not Melanoma. Arch Ophthalmol. 2011;129(8):1104–1106. Doi:10.1001/archophthalmol.2011.215.
- Gameiro Filho AR, Sturzeneker G, Rodriguez EEC, Maia A, Morales MC, Belfort RN. Acute exudative polymorphous paraneoplastic vitelliform maculopathy (AEPPVM) associated with choroidal melanoma. Int J Retina Vitreous. 2021;7(1):27. Published 2021 Apr 1. Doi:10.1186/s40942-021-00300-0.
- Osman M, Mehana O, Eissa M, Zeineldin S, Sinha A. Coronavirus Disease 2019-induced Acute Exudative Polymorphous Vitelliform Maculopathy. Middle East Afr J Ophthalmol. 2023;29(4):235-237. Published 2023 Sep 29. Doi: 10.4103/meajo.meajo_61_23.
- Lentzsch AM, Dooling V, Wegner I, et al. ACUTE EXUDATIVE POLYMORPHOUS VITELLIFORM MACULOPATHY ASSOCIATED WITH PRIMARY EPSTEIN-BARR VIRUS INFECTION. Retin Cases Brief Rep. 2022;16(6):740-746. Doi:10.1097/ICB.0000000000001066.
- Grenga PL, Fragiotta S, Cutini A, Vingolo EM. Acute exudative polymorphous vitelliform maculopathy: To bolus or not to bolus? Oman J Ophthalmol. 2018 Sep-Dec;11(3):280-283. Doi: 0.4103/ojo.OJO_12_2017. PMID: 30505125; PMCID: PMC6219324.
- Scupola A, Abed E, Sammarco MG, et al. Intravitreal dexamethasone implant for acute exudative polymorphous vitelliform maculopathy. European Journal of Ophthalmology. 2014;24(5):803-807. Doi:10.5301/ejo.5000477.
- Singh Grewal S, Smith JJ, Carr A-JF. Bestrophinopathies: Perspectives on clinical disease, Bestrophin-1 function and developing therapies. Therapeutic Advances in Ophthalmology. 2021;13. Doi:10.1177/2515841421997191
- Vaclavik V, Ooi KG, Bird AC, Robson AG, Holder GE, Webster AR. Autofluorescence Findings in Acute Exudative Polymorphous Vitelliform Maculopathy. Arch Ophthalmol. 2007;125(2):274–277. Doi:10.1001/archopht.125.2.274.

