Physiological Interpretation of CTG: Intrapartum FIT-CAT
Physiological Interpretation of Cardiotocograph (CTG): The role of the intrapartum “FIT-CAT”
Edwin Chandraharan¹, Shehara Amanti Gunaratne², Yash Kahar³, Mary Edmondson⁴, Mareike Bolten⁵
- Director of Global Academy of Medical Education & Training London, United Kingdom
- International Medical University, Kuala Lumpur, Malaysia
- Final Year Medical Student, University College London, London
- Fetal Monitoring Midwife, Medical and Health Care Educator, Medical and Health Care Education, UK
- Consultant Obstetrician and Gynaecologist, Klinikum Leverkusen, Academic Hospital of Cologne University, Am Gesundheitspark 11, 51375 Leverkusen, Germany
OPEN ACCESS
PUBLISHED: 31 December 2024
CITATION: Chandraharan, E., Gunaratne, S.A., et al., 2024. Physiological Interpretation of Cardiotocograph (CTG): The role of the intrapartum “FIT-CAT”. Medical Research Archives, [online] 12(12). https://doi.org/10.18103/mra.v12i12.6146
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i12.6146
ISSN 2375-1924
ABSTRACT
Physiological interpretation of cardiotocograph (CTG) involves determining the combination or sequence of features of the fetal heart rate to recognise different types of fetal hypoxic or non-hypoxic (e.g., chorioamnionitis) stresses, as well as scrutinising the CTG trace to differentiate features suggestive of fetal compensatory responses from decompensation.
The Fetal Monitoring Checklist was recommended in 2017, and this has been recently endorsed by the International Expert Consensus Statement on Physiological Interpretation of CTG produced by more than 50 CTG experts from over 20 countries. The aim of the Fetal Monitoring Checklist is to exclude or to identify features suggestive of pre-existing fetal compromise by assessing the oxygenation of fetal central organs, placental function and the wider clinical context.
This enables the frontline clinicians to ask the question “Is THIS fetus FIT to undertake the progressively hypoxic journey of human labour?” However, some pre-existing risk factors may manifest after the onset of established labour due to the super-imposed hypoxic stress.
Similarly, intra-amniotic infection following a recent spontaneous prelabour rupture of membranes may be silent during the incubation period, but it may manifest during labour. In such cases, the use of the Fetal Monitoring Checklist may lead to clinicians concluding that the “Fetus is FIT for Labour”, although there is increased risk of fetal compromise with the passage of time and intrapartum super-imposed hypoxic stress.
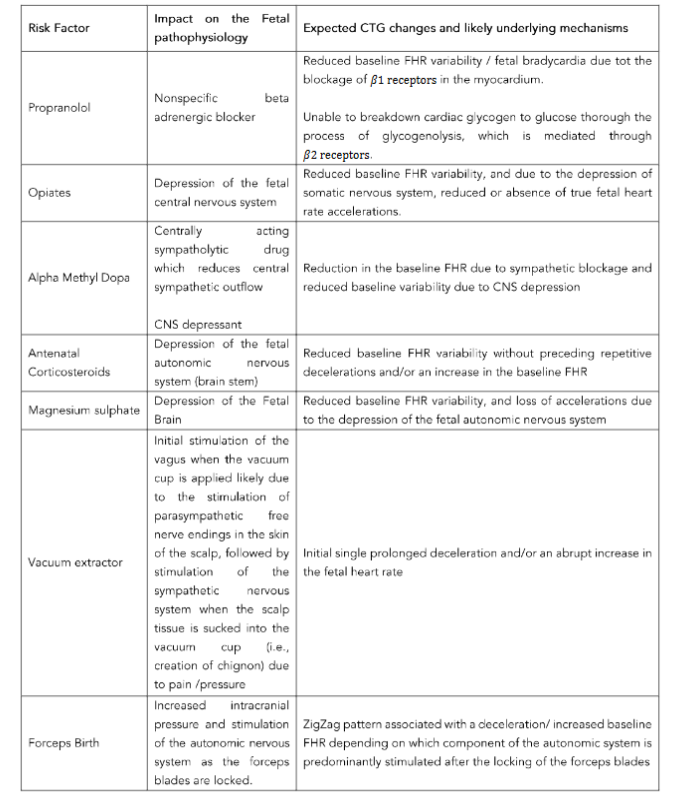
The intrapartum “FIT-CAT” (FIT for labour – Clinical Anticipation Tool) is designed to aid frontline clinicians to anticipate the changes on the CTG which are likely to occur during labour, based on the identified antenatal or intrapartum risk factors.
This intrapartum anticipation tool may enable the timely recognition of features on the CTG which herald the onset of fetal decompensation in fetuses who are deemed “FIT” to commence labour with pre-existing risk factors and/or develop intrapartum risk factors.
Prompt recognition and appropriate management based on individualisation of care and the understanding of the impact of clinical context on the CTG trace may help improve perinatal outcomes.
Key Words: Physiological Interpretation of CTG, Chorio Duck Score, FIT for Labour, “How is THIS Fetus?”, FIT-CAT, Tone Enhancers.
Introduction
Intrauterine fetal well-being depends on good placental function for gaseous exchange as well as transfer of essential nutrients and removal of fetal metabolic waste products. Optimum intrauterine fetal environment with clear and copious amniotic fluid without any microbial invasion as well as a conducive maternal environment that facilitates fetal growth and wellbeing are equally important.
This is because in addition to normal maternal oxygen saturation and normal functioning of the maternal liver, kidneys, lungs, cardiac function with sufficient perfusion pressure to the placenta, the fetus also depends on appropriate “concentration-gradients” for transfer of metabolic byproducts and body heat generated during growth, body movements and metabolism.
Therefore, if there is maternal acidosis or pyrexia, this essential maternal-fetal concentration gradient will be blunted leading to accumulation of metabolic waste products and overheating in the fetal compartment, which may lead to fetal neurological damage.
The intrauterine life consists of approximately 40 weeks during the antenatal period and 6–12 hours, if labour commences spontaneously and progresses normally. In addition to chronic utero-placental insufficiency, several metabolic, haematological, cardiovascular, neurological, endocrine and inflammatory disorders may compromise fetal well-being during the antenatal period.
Such a fetus with an ongoing, pre-existing antenatal compromise may not be able to withstand additional, super-imposed hypoxic stress caused by intermittent and progressively increasing frequency, duration and strength of uterine contractions during established labour.
Moreover, their physiological reserves may have been already exhausted via depletion of the buffering system leading to a shortened “stress to neurological damage” interval. Guidelines which use arbitrary parameters and unscientific time limits may miss such fetuses with pre-existing fetal compromise with blunted protective reflex responses.
In 2017, Pereira & Chandraharan developed the “Fetal Monitoring Checklist” to identify CTG features which were suggestive of pre-existing fetal compromise at the onset of CTG monitoring.
This “Fetal Monitoring Checklist” was aimed to enable the frontline clinicians to answer the question “Is THIS Fetus FIT to undertake the progressively hypoxic journey of labour?”.
This checklist (Table 1) was also recently recommended by the International Expert Consensus Guidelines on Physiological Interpretation of CTG produced by more than 50 CTG experts from over 20 countries.
This approach of applying the “Fetal Monitoring Checklist” as a screening tool at the time of initial admission in labour (or prior to an induction of labour) to exclude the features of ongoing fetal compromise due to pre-existing pathology (antenatal causes) will help avoid subjecting these fetuses to additional, super-imposed hypoxic stress and help prevent further injury.
Table 1. Fetal Monitoring Checklist: Is THIS Fetus Fit for Labour? (Pereira & Chandraharan 2017)
CTG Features / Risk Factors — Assessment (Yes / No)
- Baseline fetal heart rate stable and appropriate for the gestational age — Yes / No
- Normal variability and cycling — Yes / No
- Presence of TRUE accelerations (not in labour or latent phase of labour) — Yes / No
- No shallow / tardy decelerations — Yes / No
- Consider the wider clinical picture: meconium, pyrexia, fetal growth restriction, reduced fetal movements, gestational DM, pre-eclampsia, induction/augmentation, other … — Yes / No
Overall Impression: Normal / Chronic Hypoxia / Chorioamnionitis / RUPI / Other: __________
Management Plan: __________________________
Date: _______ Time: _______ Name: _______ Signature: _______
However, even fetuses which are deemed to be “FIT for labour” may have antenatal or de novo intrapartum risk factors which may increase the likelihood of fetal compromise.
If there is a delay in recognising these features suggestive of fetal compromise, fetal neurological injury or a terminal myocardial failure may occur.
For example, a fetus with a background history of spontaneous prelabour rupture of membranes (SROM) may display a “normal” CTG at the onset of labour and therefore be deemed “FIT for labour”.
However, bacteria that entered the amniotic cavity may be multiplying at this stage and fetal inflammation/infection due to entry of bacteria and toxins into lungs during fetal breathing movements may result in congenital pneumonia, which may occur several hours later, increasing fetal metabolic rate.
This initial change on the CTG trace (>10% increase in baseline FHR, e.g., from 120 bpm to 135 bpm) may be easily missed because several CTG guidelines continue to provide a large “population-based” range (110–160 bpm) without individualising care.
A clinical management decision must be made based on the wider clinical context which includes parity, cervical dilatation and rate of labour progress.
If a timely delivery is not accomplished when features of increased fetal metabolic rate are observed (i.e., >10% increase in baseline FHR), continued bacterial multiplication and production of bacterial toxins such as lipopolysaccharides (LPS) and inflammatory cytokines such as interleukin-6 (IL-6) may lead to the development of fetal systemic inflammatory response syndrome (FIRS), resulting in multi-organ failure, neonatal encephalopathy (NNE) and terminal myocardial failure.
Recently, it has been shown that features suggestive of fetal inflammation (SOFI) on the CTG trace are associated with approximately four-fold increase in the levels of IL-6 in the umbilical artery at birth compared to the control group and had increased risk of composite adverse perinatal outcomes.
Similarly, a fetus with a pre-existing subclinical placental failure may not display any abnormalities when screened using the “Fetal Monitoring Checklist” because of the absence of any mismatch between oxygen demand and supply prior to the onset of established uterine contractions with sufficient intensity.
However, as the intensity and frequency of uterine contractions increase, due to episodic reduction of placental perfusion from compression of the branches of uterine arteries supplying the placental bed, fetal metabolic and oxygen demands may outstrip the supply.
Such a fetus is likely to mount compensatory responses to ensure adequate perfusion to central organs, to deal with episodes of “relative” utero-placental insufficiency.
The CTG features suggestive of an ongoing relative utero-placental insufficiency during labour (RUPI-L) may be easily missed if clinicians are not aware of these specific changes and may fail to anticipate their occurrence.
Similarly, a macrosomic fetus and a fetus with likely placental malfunction (e.g., gestational diabetes mellitus) may experience progressive hypoxic stress after the onset of established labour as oxygen delivery via the placenta may not be sufficient to meet metabolic requirements.
In addition to such antenatal pathology which may manifest their detrimental effects on the fetus after the onset of established labour, several intrapartum risk factors may also contribute to or hasten fetal compromise.
Passage of meconium during labour leads to reduced phagocytosis because bile salts and bile acids inhibit the activity of neutrophils within the amniotic cavity, leading to increased risk of chorioamnionitis.
Therefore, based on the clinical context (meconium staining of amniotic fluid), it is essential to anticipate and predict specific CTG changes which are likely to occur to ensure timely and appropriate actions are taken to optimise perinatal outcomes.
Conversely, changes in the cardiotocograph (CTG) trace suggestive of fetal compromise may occur secondary to certain medications administered to the mother. For example, administration of opioids
Therefore, if these changes are not anticipated and predicted on the CTG after the administration of these medications, based on the knowledge of fetal pathophysiology, then, it may lead to increased likelihood of unnecessary interventions and their undesirable consequences.
Robust Intrapartum Fetal Surveillance (RIFS) does not solely depend on the use and the correct interpretation of the cardiotocograph. It involves a careful scrutiny of the wider clinical context, which includes both antepartum and intrapartum risk factors.
In addition, the anticipation and prediction of CTG changes based on the observed clinical context are crucial not only to institute timely and appropriate interventions but also to avoid unnecessary obstetric interventions. This should be done after the fetus has been deemed to be “FIT” for labour, and should continue throughout the intrapartum period, and when additional risk factors are observed, or medications are administered to the parturient.
Aim & Scope
The aim and scope of this expert review is to provide a practical tool to aid frontline clinicians to anticipate the CTG changes which are likely to occur based on the identified antepartum and intrapartum risk factors.
What is FIT-CAT?
It is well recognised that a fetus who is deemed to be “FIT” for labour at the time of admission may not remain “FIT” throughout labour.
This is because some of the antepartum risk factors such as entry of bacteria into the amniotic cavity due to spontaneous prelabour rupture of membranes (SROM) may have a delayed manifestation after several hours, and CTG may show changes suggestive of chorioamnionitis in late first stage of labour.
Similarly, passage of meconium in early labour may be associated with a normal CTG on admission (i.e., “FIT for labour”), however, with passage of time due to the blunting of the antibacterial effect of the amniotic fluid by the inactivation of neutrophil phagocytosis, the CTG trace may show changes suggestive of chorioamnionitis in late first stage of labour.
Moreover, a fetus who was previously deemed “FIT for labour” may be exposed to an intrapartum risk factor such as commencement of oxytocin and resultant reduction in utero-placental oxygenation.
However, the features of a gradually evolving hypoxic stress may not be apparent on the CTG trace until a few hours later.
Therefore, anticipation of future CTG changes based on the observed risk factors is essential to timely recognition of the significance of the observed CTG changes so that prompt actions can be taken to mitigate the risks of hypoxic-ischaemic encephalopathy (HIE) and/or perinatal death.
The FIT for Labour – Clinical Anticipation Tool (FIT-CAT) has been developed to aid frontline clinicians to anticipate the expected CTG changes (Table 2) based on the observed clinical context.
It recognises that the human fetus is exposed to a continuum of risk posed by several underlying pathophysiological processes capable of causing neurological or myocardial damage, both during the antenatal period as well as extending throughout the intrapartum period until birth.
It must be appreciated that not all underlying risk factors will immediately manifest the expected changes on the CTG, and some may start their detrimental effects on the fetal metabolic, biochemical, cardiovascular neurological systems hours later.
Some risk factors (e.g., subclinical or relative utero-placental insufficiency) may lead to CTG changes only after the onset of superimposed uterine contractions with sufficient intensity and/or duration.
Therefore, knowledge of specific CTG changes associated with risk factors which are associated with fetal compromise (Table 1) is essential to anticipate and respond to these changes in a timely manner to optimise perinatal outcomes.
The key parameters of the FIT-CAT are given below:
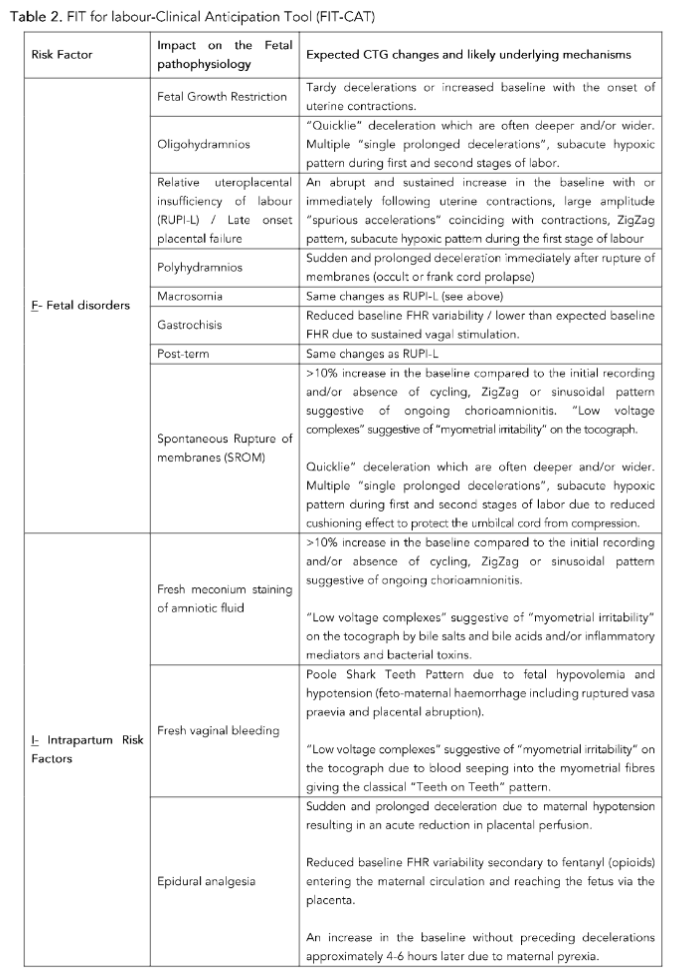
a. FETAL DISORDERS
Several pathophysiological processes affecting the fetus may lead to specific changes in the fetal heart rate, based on the underlying pathology, as the labour progresses.
These changes may manifest at any time during labour.
For example, a fetus with growth restriction with mild to moderate utero-placental insufficiency may tolerate ongoing relatively milder uterine contractions during the latent phase and early active phase of labour due to the availability of sufficient reserves.
CTG changes suggestive of a gradually evolving hypoxic stress may be observed during late first stage or second stage of labour.
Conversely, a fetus SGA with moderate to severe loss of placental function secondary to placental infarction or thrombosis during the antenatal period may show features suggestive of hypoxic stress even during the latent phase of labour. Well grown fetuses with subclinical placental insufficiency (e.g., loss of approximately 10–30% of placental function) or those with late onset placental failure may not demonstrate any abnormalities in the fetal heart rate at the onset of labour.
However, if attempts are made to induce labour or in the presence of established uterine contractions, these fetuses may show CTG changes suggestive of a relative uteroplacental insufficiency of labour (RUPI-L).
Fetuses who are “large for dates” (LFD) or “macrosomic” may have a critical balance between their oxygen supply from the placenta and their (increased) metabolic demands during the antenatal period. This is because relatively larger organs, increased muscle mass and adipose tissue require extra oxygen to meet their metabolic requirements to avoid anaerobic metabolism and development of lactic acidosis.
However, this “critical balance” may be lost with the onset of uterine contractions with sufficient intensity and duration to reduce placental oxygenation leading to the ongoing increase in the fetal metabolic demands outstripping the blood supply.
Therefore, features suggestive of a gradually evolving hypoxic stress may be seen on the CTG trace. However, if there is a rapid evolution of hypoxic stress, then, a ZigZag pattern may be seen.
Post-term fetuses may also demonstrate similar CTG features due to the relative larger size of the fetus as the gestation advances associated with a progressive decrease in the placental function.
Oligohydramnios usually indicates a severe chronic utero-placental insufficiency with renal shutdown (i.e., leading to a reduced fetal urine output). Therefore, most fetuses would show features of “chronic hypoxia” on the CTG trace and would be deemed “Not FIT” for labour when the Fetal Monitoring Checklist is used.
However, if uterine contractions are allowed to continue in fetus with oligohydramnios, then due to the lack of sufficient amniotic fluid “cushion” to protect the blood vessels within the umbilical cord from being compressed during uterine contractions, then features of a gradually evolving hypoxic stress may be observed.
However, in late first stage of labour or if uterotonic agents are used, then, a subacute or acute hypoxic pattern may be observed.
Fetuses with abdominal wall defects would have exposure and irritation of the contents of the peritoneal cavity, especially the intestines, which are richly supplied by the parasympathetic nervous system. Ensuring sustained vagal stimulation may lead to a persistent and sustained drop in baseline FHR and/or a reduction in the baseline variability.
In modern obstetric practice, these fetuses are likely to be diagnosed during antenatal ultrasound scans leading to an elective caesarean section.
However, if these fetuses are exposed to uterine contractions during labour, then “deep and ugly” decelerations may occur due to the ongoing vagal stimulation and compression of the exposed abdominal contents as well as the parietal peritoneum during uterine contractions.
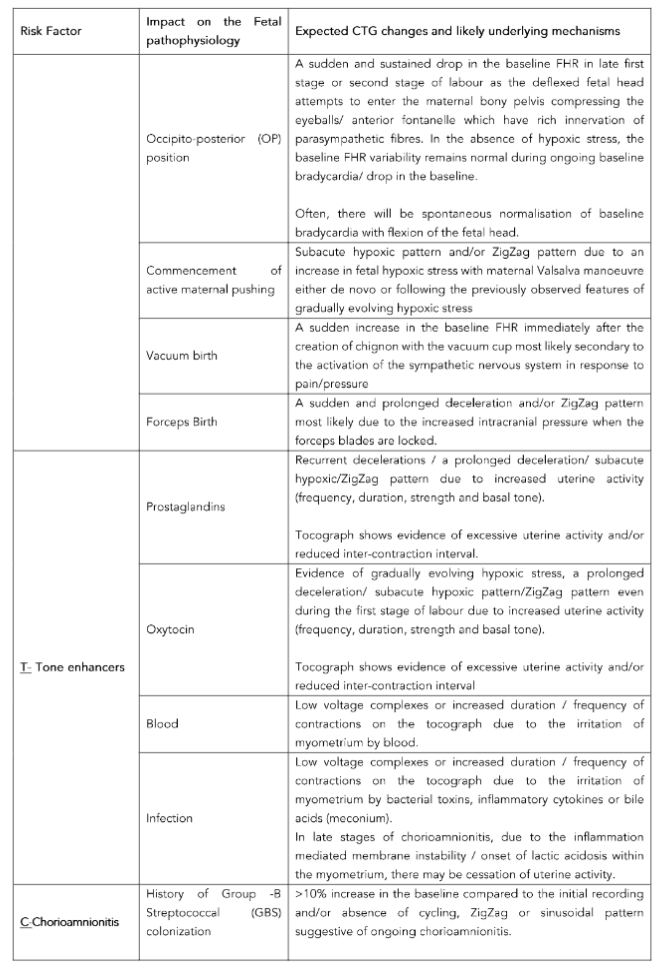
b. INTRAPARTUM RISK FACTORS
Unfortunately, despite the superficial attempts to consider the “wider clinical picture” whilst interpreting intrapartum CTG traces and introducing acronyms such as “DR C BRAVADO”, the same CTG classification tool has been used in all human fetuses.
This illogical practice of “defining the risk” (DR) and then illogically using exactly the same number of contractions and CTG features with the same arbitrarily pre-defined time duration (C BRAVADO) has resulted in disastrous consequences to babies and their families.
One cannot use the same cut-off for the upper limit of the baseline FHR (>160 bpm) with the same range of 110–160 bpm in all human fetuses because a post-term or a fetus with reduced physiological reserves who increases the baseline from 120 bpm to 140 bpm due to ongoing hypoxic or inflammatory stress will be missed.
Classification of these fetuses by using the same range for the baseline (110–160 bpm) will miss these fetuses who have increased the baseline from 120 to 140 bpm leading to disastrous consequences¹⁹. It is regrettable that some CTG guidelines even experimented with human physiology by artificially increasing the upper limit of the baseline FHR to >180 bpm²⁰,²¹ making it impossible for fetuses with poor physiological reserves to ever become “abnormal”.
This unscientific, illogical and avoidable harm-inducing action would lead to an increase in avoidable poor perinatal outcomes²².
The continued use of such illogical CTG guidelines which failed to consider specific CTG changes induced by the intrapartum clinical risk factors has been associated with repetitive publications highlighting avoidable poor perinatal outcomes in approximately 70% of cases²³–²⁸.
In addition, the medico-legal costs due to settling the clinical negligence claims involving avoidable harm in the maternity service have been eye-wateringly high, dwarfing other “high-risk” clinical specialties²²,²⁹.
According to the latest report from NHS Resolution, a body responsible for indemnifying National Health Service from clinical negligence claims, it has highlighted that claims due to cerebral palsy and severe brain damage, the vast majority of which are caused by CTG misinterpretation, cost the tax payer in the United Kingdom approximately £6.7 million, every day³⁰.
Therefore, a deeper understanding of the intrapartum clinical risk factors and the specific anticipated changes in the CTG trace in response to these risk factors is essential to avoid poor perinatal outcomes (Table 1).
A thorough scrutiny of both cardiac (fetal heart rate changes) and toco (uterine activity) parts of the CTG trace are essential whenever an intrapartum risk factor is identified.
For example, intrapartum passage or detection of meconium may lead to the obliteration of the antibacterial activity of the amniotic fluid resulting in an intraamniotic infection³¹,³² characterised by >10% increase in the baseline compared to the initial recording and/or absence of cycling, ZigZag or sinusoidal pattern.
However, in early stages, bile salts and bile acids present in meconium may cause “low voltage complexes” suggestive of “myometrial irritability” on the tocograph (Table 1).
Therefore, lack of knowledge to anticipate these changes based on the understanding of fetal pathophysiology may lead to delays in appropriate action and poor perinatal outcomes.
c. TONE ENHANCERS
It is important to appreciate that intrapartum fetal wellbeing depends on the ability of a fetus to mount an effective compensatory response to slow and a progressively increase in hypoxic stress, with sufficient relaxation time, in between the contractions, to obtain fresh oxygen and to eliminate carbon dioxide and other metabolic byproducts from the fetal compartment.
Therefore, any agent which increases the uterine tone would significantly increase ongoing hypoxic stress, and impairment of fetal oxygenation and may cause rapid decompensation.
Unfortunately, several CTG guidelines erroneously defined uterine hyperstimulation based on the frequency of uterine contractions alone (>5 in 10 minutes)²¹ due to the apparent lack of knowledge of muscle physiology.
Properties of any muscle contraction include frequency, duration, strength and resting tone, and therefore, focusing solely on the frequency >5 in 10 minutes would lead to adverse outcomes in fetuses exposed to <5 contractions in 10 minutes.
If these uterine contractions had longer duration, increased intensity or an enhanced resting tone in between uterine contractions, then, rapid fetal decompensation may occur culminating in hypoxic-ischaemic encephalopathy (HIE).
Therefore, it is not surprising that repetitive Each Baby Counts Reports published by the Royal College of Obstetricians & Gynaecologists (RCOG) has highlighted the association of oxytocin use in cases of intrapartum and early neonatal deaths as well as severe hypoxic-ischaemic brain injuries²⁵–²⁸.
Approximately 70% of medico-legal cases involving cerebral palsy have been reported to be associated with injudicious use of oxytocin³³.
Therefore, it is essential to use the correct definition of uterine hyperstimulation whilst using uterotonic agents, and any increase in uterine activity (frequency, duration, strength and resting tone) associated with changes in the fetal heart rate should be considered as uterine hyperstimulation³⁴.
In addition to the use of prostaglandins and oxytocin infusion to induce and augment labour, respectively, both blood (e.g., placental abruption), as well as inflammatory cytokines and bacterial toxins (chorioamnionitis) can irritate and stimulate the myometrium.
Irrespective of the underlying pathophysiology, increased uterine tone over time will result in progressive reduction in fetal oxygenation.
Therefore, anticipation of specific changes (Table 1) is crucial to optimise perinatal outcomes, and if a spontaneous vaginal birth is not imminent, and the underlying pathology is irreversible (e.g., placental abruption or chorioamnionitis), then, birth should be expedited.
d. CHORIOAMNIONITIS
The spectrum of intraamniotic inflammation, intraamniotic infection and chorioamnionitis occurs in approximately 10% of labour and it is an important cause of fetal compromise³⁵,³⁶.
It has been reported that chorioamnionitis increases the risk of neonatal encephalopathy (NNE) five-fold and approximately 10% of all cases of cerebral palsy were due to chorioamnionitis³⁷,³⁸.
Several animal experimental studies have confirmed that injection of bacterial toxins such as lipopolysaccharides (LPS) are associated with perinatal brain damage³⁹–⁴¹.
Galli et al⁴² analysed 2105 cases of both subclinical and clinical chorioamnionitis and reported specific CTG features associated with chorioamnionitis such as >10% increase in the baseline FHR, absence of FHR cycling, the ZigZag and sinusoidal patterns⁴².
These were subsequently confirmed by Sukumaran et al., who analysed histologically confirmed cases of chorioamnionitis and funisitis⁴³.
Similar features were reported in fetuses exposed to maternal cytokine storm in Covid-19 infection⁴⁴.
Therefore, if these features suggestive of an ongoing fetal inflammation are present, then co-existing or superimposed hypoxic stress should be avoided and delivery should be expedited.
This is because several animal experimental studies have confirmed that in the presence of fetal inflammation and consequent sensitisation of the developing fetal brain, the threshold for hypoxic-ischaemic injury is lowered⁴⁵–⁴⁹.
In other words, if uterine contractions (super-imposed hypoxic stress) are continued in a fetus with ongoing chorioamnionitis, then, due to the synergistic effect whereby ongoing fetal neuroinflammation reduces the threshold at which hypoxic-ischaemic brain injury occurs, the risk of brain injury and cerebral palsy will be potentiated⁵⁰.
Despite this scientific knowledge several CTG guidelines had failed to include the features of chorioamnionitis in their classification system.
For example, repetitive NICE CTG guidelines did not include any of the features suggestive of chorioamnionitis, but also erroneously increased the upper threshold of abnormal FHR baseline from the internationally accepted 160 bpm to 180 bpm²⁰,²¹.
Moreover, they had continued to recommend fetal scalp blood sampling (FBS) until 2022, despite the physiological knowledge that in chorioamnionitis, due to peripheral vasodilation, there would be excessive blood flow to the skin, which is a peripheral non-essential tissue, leading to falsely reassuring, false negative results.
Despite these risks being highlighted by scientific publications²⁵–⁵³, FBS was continued to be recommended in the UK contrary to available scientific evidence until 2022.
Therefore, it is not surprising that due to the lack of due diligence regarding the specific features of chorioamnionitis by the NICE CTG Guideline, NHS Resolution Report in 2019 has highlighted that more than 15% of neonates who required brain cooling for severe brain injury at birth had evidence of infection [30].
Recent scientific evidence has confirmed that fetuses with CTG features suggestive of fetal inflammation (SOFI) such as >10% increase in baseline FHR, absence of FHR cycling, the ZigZag and sinusoidal patterns are associated with approximately a four-fold increase in the levels of interleukin-6 (IL-6), which is a marker of fetal inflammation⁵⁴.
Chandraharan & Bolten published a “Chorio Duck Score” (CDS), and recommended that if the CDS > 5, the overall clinical context should be considered and delivery must be expedited if a spontaneous vaginal birth is not imminent⁵⁵.
Table 3 highlights the rationale for the parameters included in the Chorio Duck Score.
Table 3. Clinical Rationale of the Chorio-Duck Score (CDS)
Parameter | Clinical Rationale: Why do these changes occur?
Increase in the Baseline FHR
An increased demand of oxygen in the presence of infection leads to a rise in the baseline heart rate. This helps to maintain aerobic metabolism by increasing oxygen delivery to tissues and organs to prevent metabolic acidosis; however, it comes at the expense of increased myocardial workload and augmented cardiac oxygen demand.
Features of Neuroinflammation
Reduced variability at a higher-than-expected baseline and absence of accelerations and cycling are a result of neuroinflammation which occurs early in FIRS due to the absence of a blood-brain barrier in the fetus.
Meconium
The presence of meconium can alter antibacterial effect of the amniotic fluid by inhibiting the phagocytic function of neutrophils and phagocytes. Meconium impairs the normal protective effect of cells which fight bacterial infections and therefore predisposes the fetus to ascending infections.
Likewise, chorioamnionitis can cause the passage of meconium by fetal gastroenteritis caused by ingestion of infected amniotic fluid as well as through increasing acidosis in the presence of FIRS.
The developing acidosis when the higher oxygen demand in sepsis is not met triggers the chemoreceptors which alongside other reactions trigger a strong vagal response that activates peristalsis and relaxes the muscles of the anal sphincter.
Myometrial Irritability
The majority of women with subclinical acute chorioamnionitis demonstrate extension of the inflammation into the choriodecidual space and the myometrium, which can result in tissue damage and uncontrolled spasms/irritability of the myometrium.
Maternal Parameters
In the vast majority of cases, chorioamnionitis occurs as a result of an ascending infection through the cervical canal. Only in late stages of FIRS does the infection extend to the mother directly through the umbilical arteries or via the decidual vessels. The mother will then start showing signs of sepsis with an increase in heartbeat and pyrexia which, if untreated, may lead to low blood pressure.
It is indeed very regrettable that even the recently updated NICE CTG Guideline has chosen to disregard the features suggestive of inflammation (SOFI) in the classification tool⁵⁶.
This has serious consequences for fetuses with intraamniotic inflammation or infection and it is very likely that NHS Resolution Reports in the future would continue to report avoidable brain damage due to fetal infection.
Lack of knowledge and disregarding scientific evidence should have a “Zero tolerance” approach in obstetric practice.
Therefore, the authors strongly recommend that chorioamnionitis should be considered as an important intrapartum risk factor which increases the likelihood of fetal compromise.And any features suggestive SOFIor myometrial irritability requires an immediate, careful review and the most appropriate obstetric intervention based on the progress of labour.
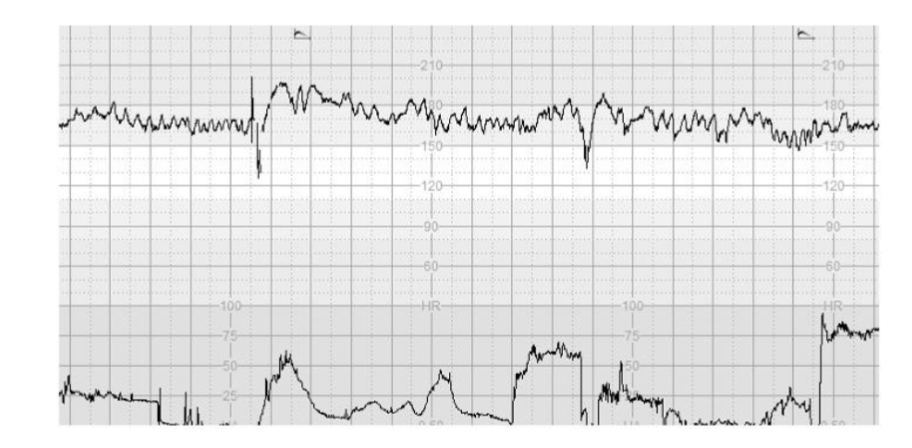
Figure 1 illustrates >10% increase in the baseline FHR without repetitive decelerations, absence of cycling and presence of the “Poole Shark Teeth Pattern” six hours after spontaneous rupture of membranes.
The use of traditional CTG guidelines classifying CTG traces into “Normal, suspicious and Pathological”, focussing on the morphology of decelerations would miss such fetuses.
However, the use of the intrapartum FIT-CAT may help frontline clinicians to anticipate such changes, facilitating timely intervention to avoid super-imposed hypoxic stress.
Figure 1. CTG trace illustrates >10% increase in the baseline FHR without repetitive decelerations, absence of cycling and presence of the “Poole Shark Teeth Pattern”, which are the features of SOFI, following SROM.
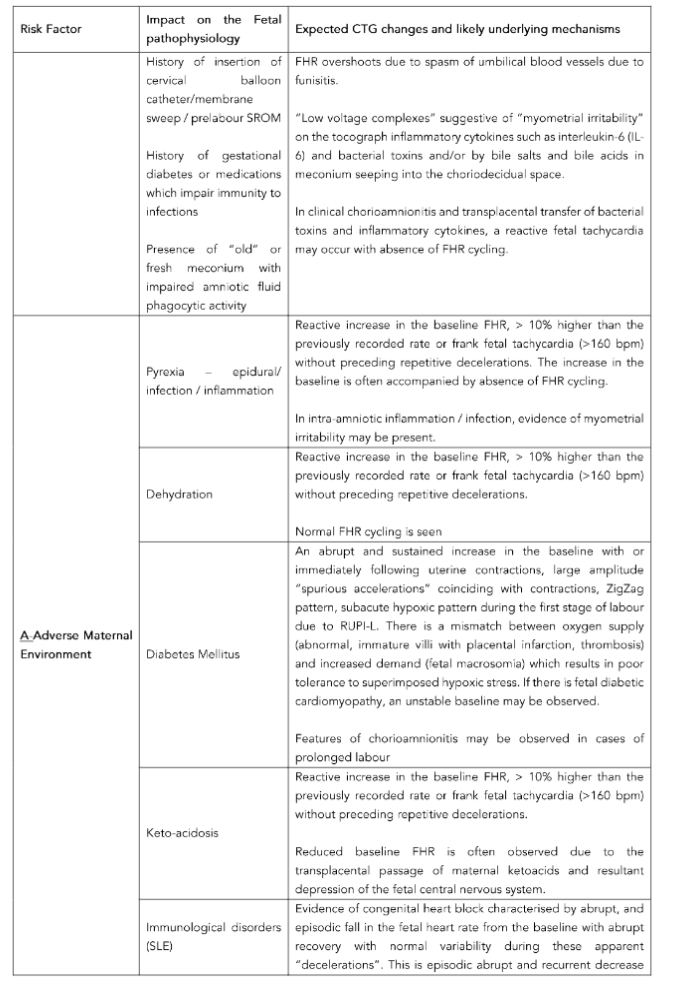
e. ADVERSE MATERNAL ENVIRONMENT
The immediate “external” environment of a fetus is the maternal milieu comprising of haematological, endocrine, metabolic, inflammatory, cardio-respiratory, excretory and immunological components.
Therefore, derangements in any of the above systems can adversely affect intrauterine fetal wellbeing and may result in changes in the CTG (Table 1).
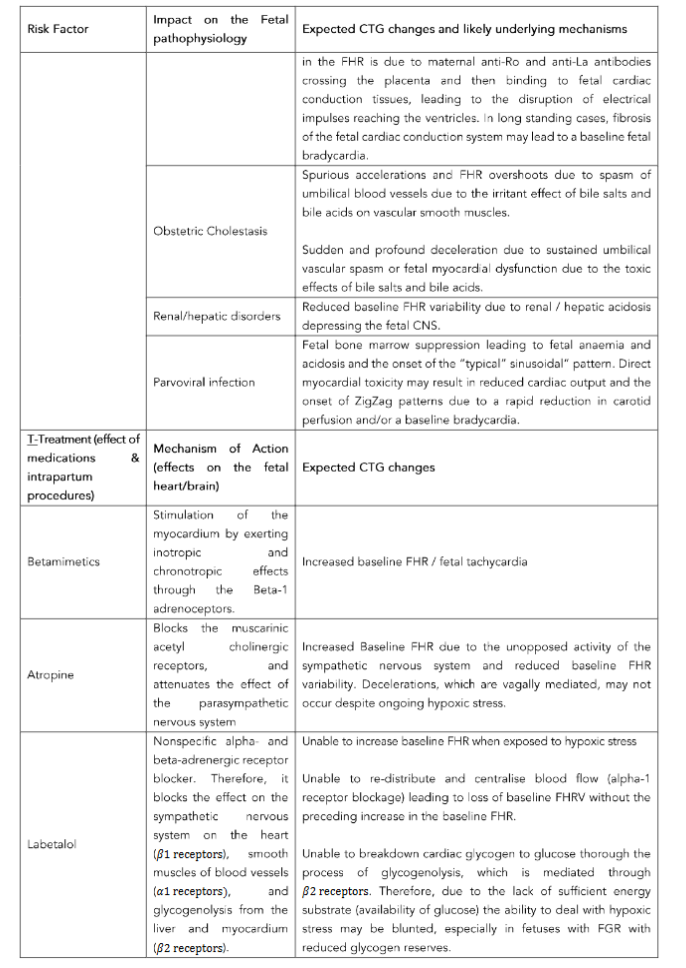
For example, a mother with systemic lupus erythematosus (SLE) may transfer Anti-Ro and Anti-La (or SS-A and SS-B) antibodies via the placenta and the umbilical cord into the fetal compartment.
These abnormal antibodies have a special affinity to the fetal cardiac conductive tissue (Bundle of His, Bundle Branches and Purkinje Fibres) leading to a reduction in transmission of electrical impulses from the SA node to the ventricles resulting in congenital heart block (1:1, 1:2 or 1:3).
However, in some cases, due to prolonged exposure and resulting fibrosis and calcification of the cardiac conducting tissue following the initial inflammatory response can result in a baseline bradycardia.
Similarly, maternal parvovirus infection may lead to the transplacental passage of the parvovirus into the fetal compartment.
Parvovirus preferentially attacks two fetal organs: the bone marrow and the myocardium leading to bone marrow suppression causing chronic fetal anaemia and acidosis culminating in fetal cardiac failure, and direct myocardial suppression.
Therefore, one should carefully scrutinise the CTG trace for the “typical sinusoidal” pattern (chronic fetal anaemia and acidosis) or a progressive reduction in the baseline FHR (direct myocardial depression).
Simply listing “Parvovirus” as the “defined risk” of the illogical “DR C BRAVADO” methodology and then ignoring the specific features due to this clinical risk factor, due to the lack of knowledge of fetal pathophysiology should no longer be acceptable in modern obstetric practice.
Recently, progressive fetal encephalopathy has been reported, and therefore, it is essential to exclude absence of fetal heart rate cycling⁵⁹.
It is also important to appreciate that the fetus depends on the concentration gradient for the transfer of essential nutrients from the mother and metabolic byproducts to the mother via the placenta.
Therefore, in maternal pyrexia and maternal acidosis, this essential concentration gradient would be adversely affected, resulting in accumulation of heat and metabolic acids and other waste products within the fetal compartment.
The CTG trace is likely to show reduced variability in fetal acidosis and absence of cycling due to the disruption of the temperature gradient in maternal pyrexia⁶⁰,⁶¹.
Table 4 illustrates the negative impacts of an adverse maternal environment.
Table 4. Impact of persistent adverse maternal environment with super-imposed intrapartum hypoxic stress
Observed Abnormality | Expected impact on the fetus | Likely outcome
Persistent fever
- Increased metabolic rate and tissue oxygen demands
- Neuronal dysfunction/denaturation of proteins
→ Increased predisposition for hypoxic-ischaemic brain injury and multi-organ failure
→ Neonatal convulsions and neonatal encephalopathy (NNE)
Persistent tachycardia
- Increased oxygen requirement of the myocardium due to increased myocardial workload
- Reduced coronary perfusion due to reduced diastole leading to progressive myocardial ischaemia
→ Fetal “high output” heart failure leading to terminal bradycardia
Persistent hypoxia (bronchial asthma, Covid-pneumonia)
- Reduced placental oxygen transfer leading to hypoxia to fetal tissues and organs
→ Increased predisposition for hypoxic-ischaemic brain injury and multi-organ failure
Persistent maternal hypotension
- Reduced perfusion pressure to the placental bed resulting in rapid development of hypoxia-ischaemia to fetal central organs
→ Increased predisposition for hypoxic-ischaemic brain injury and multi-organ failure
Persistent maternal hepatic/renal acidosis
- Disruption in the materno-fetal concentration gradient required for transfer across the placenta, leading to accumulation of acid in the fetal compartment
→ Neonatal encephalopathy and terminal myocardial failure due to acidosis
Persistent obstetric cholestasis
- Toxicity to smooth muscles leading to spasm of the blood vessels within the umbilical cord and/or depression of fetal myocardium
→ Terminal bradycardia and intrauterine fetal death
f. TREATMENT (EFFECT OF MEDICATIONS & INTRAPARTUM PROCEDURES)
Several medications which are administered to the mother may cross the placenta and affect the fetal cardiovascular and central nervous systems resulting in changes in the CTG trace (Table 2).
Failure to understand the specific CTG features secondary to the medications administered to the mother may lead to over-reaction to observed patterns resulting in unnecessary operative interventions.
For example, reduced baseline FHR variability due to depression of fetal CNS by antenatal corticosteroids may lead to the classification of the cardiotocograph (CTG) trace as “pathological” due to the illogical, arbitrary time limit of 90 minutes stipulated by some CTG guidelines. guideline. This may result in an unnecessary, iatrogenic premature delivery with resultant complications.
Conversely, it must be appreciated that some medications may blunt the anticipated fetal heart rate changes in gradually evolving hypoxia, and failure to appreciate this may lead to disastrous consequences.
For example, nonspecific beta-blockers such as propranolol and labetalol may blunt the fetal catecholamine response and resultant increase in the baseline FHR due to blockage of cardiac β1 receptors.
Therefore, the baseline FHR variability may be reduced immediately following repetitive decelerations without an increase in the baseline FHR.
Labetalol also blocks α-1 receptors which are responsible for peripheral vasoconstriction and centralisation of blood flow.
Therefore, blunting of redistribution due to labetalol may lead to rapid onset of decompensation with the onset of reduced baseline FHR variability without a compensatory increase in the baseline FHR.
Management
Individualisation care is the cornerstone of clinical practice, and in a medical ward, a physician or a nurse would never treat all adults with tachycardia with the same medication.
This is because baseline tachycardia in adults may be caused by thyrotoxicosis, sepsis, haemorrhage, stress, anxiety, anaemia and dehydration.
Therefore, it would be illogical to administer fluids to all adults with tachycardia, and the treatment must be aimed at rectifying the underlying pathology which caused tachycardia.
Unfortunately, several CTG guidelines overlooked this very basic tenet of individualisation of care taught in medical schools, and arbitrarily grouped FHR features into different categories, without any scientific evidence with the overall classification of CTG traces into “Normal, suspicious, Pathological”.
Illogically, these guidelines recommended oral or intravenous fluids for all suspicious CTG traces²⁰,²¹, which increased the risk of fetuses with chorioamnionitis sustaining brain damage or dying due to the delay in definitive treatment.
Therefore, it is not surprising that repetitive Each Baby Counts Reports concluded that in more than 70% of babies who sustained severe brain damage and/or died following intrapartum hypoxia, a different care would have given rise to a different outcome.
Similarly, the NHS Litigation Authority (NHSLA) Study on Stillbirths had concluded that 34% of stillbirths were solely due to CTG misinterpretation²⁸.
The same poor outcomes would be expected in an adult or in a paediatric ward, if the doctors and nurses had treated all cases of tachycardia with intravenous or oral fluids, disregarding the underlying pathology.
Similarly, some CTG guidelines illogically recommended fetal scalp blood sampling (FBS) for all pathological CTGs which resulted in arbitrarily grouping random features into different categories, irrespective of underlying pathology and despite the serious concerns raised by several publications⁵¹–⁵³.
This resulted in fetuses who were well compensating to ongoing hypoxic stress by effectively re-distributing oxygenated blood from the scalp, due to catecholamine-mediated peripheral vasoconstriction being misclassified as “fetal distress”, leading to an increase in the rate of unnecessary intrapartum operative interventions to the mother and resultant potentially serious complications.
Despite FBS being stopped in the USA approximately 30 years ago due to lack of scientific evidence to support the use, and obstetricians from most countries around the world not doing FBS due to basic knowledge of fetal physiology, FBS was continued to be used in the UK until December 2022.
This was despite repetitive Cochrane Systematic Reviews concluding no beneficial effects of FBS⁶⁴, and several publications highlighting increased rate of emergency caesarean sections without improving perinatal outcomes⁶⁵,⁶⁶, including a multicentre trial from the UK concluding lack of improvement in perinatal outcomes, and an approximately 60% increase in the perinatal outcomes⁶⁷.
These poor maternal and perinatal outcomes for treating all fetal heart rate changes with the same treatment (fluids/ FBS/ caesarean sections) should no longer be accepted in contemporary obstetric practice.
The medico-legal and ethical pitfalls of treating all human fetuses with the same pre-defined parameters have been repetitively highlighted¹–³,⁶⁸.
However, no action was taken to remove the error-producing CTG guidelines which were causing avoidable poor perinatal outcomes, resulting in women and their families calling for criminal prosecutions in 2022
(https://www.independent.co.uk/news/health/east-kent-maternity-baby-deaths-b2206143.html)
as well as a public enquiry
(https://www.theguardian.com/society/2023/oct/31/parents-of-babies-who-died-or-were-harmed-in-nhs-care-demand-inquiry).
The unscientific CTG guidelines which failed to individualise care were continued despite continuing poor perinatal outcomes, and 100 cases have been referred to the police in 2024
(https://www.bbc.com/news/articles/cpwre51d22yo).
Therefore, frontline clinicians must individualise care based on the observed clinical context during labour because each fetus is different with different physiological reserves and different frequency, strength, duration and the basal tone of ongoing uterine contractions.
Moreover, the intrapartum risk factors and maternal environment are also different.
Therefore, in addition to the classification and management recommended by the International Expert Consensus Statement on Physiological Interpretation of CTG [5], the expected fetal heart rate changes based on the observed clinical context should be anticipated so that prompt recognition and timely action will help improve outcomes.
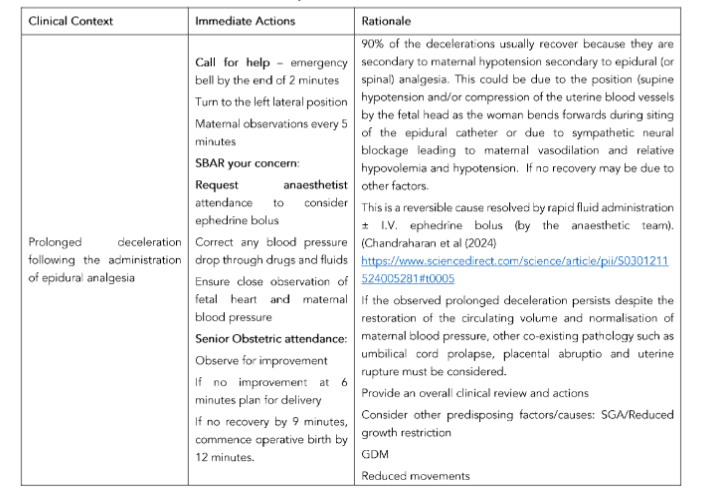
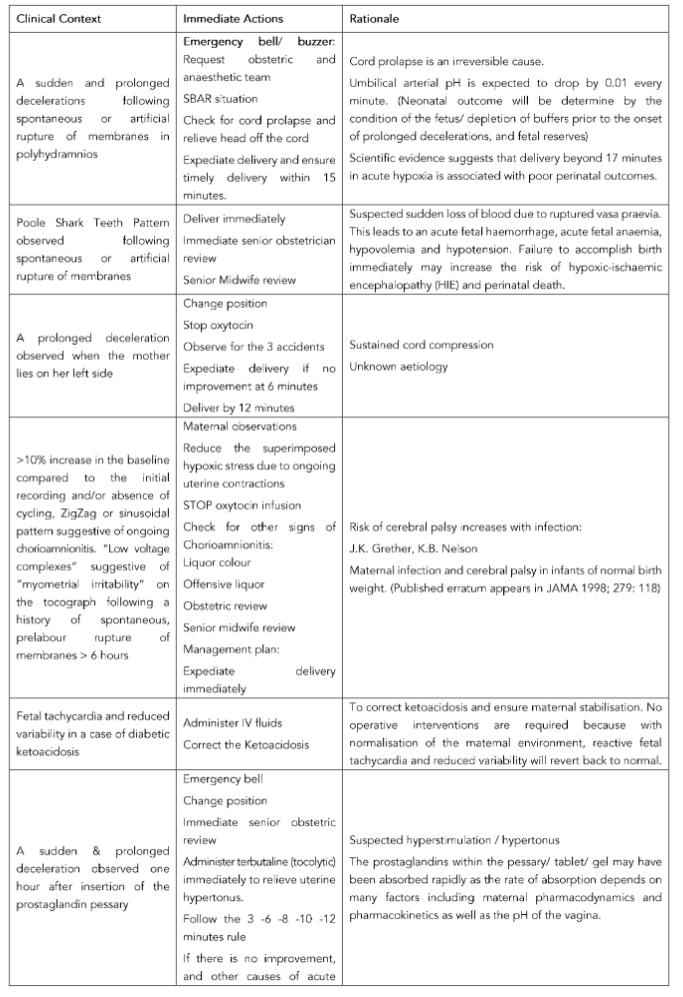
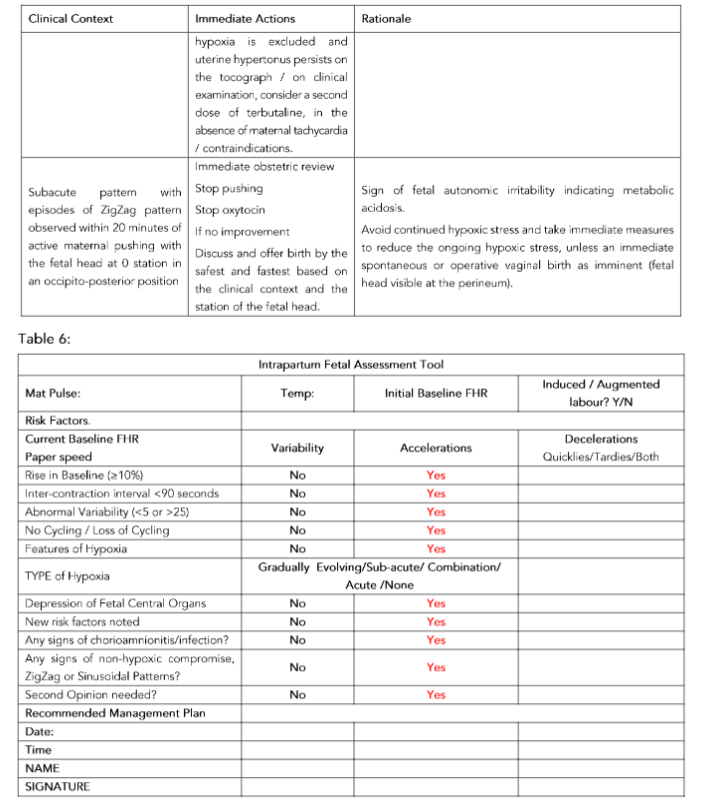
Table 5 provides examples of how management can be tailor-made to the identified clinical context, and this should be used in conjunction with the recommended intrapartum fetal assessment tool (Table 6).
Table 5. Immediate recommended actions by obstetricians and midwives
Conclusion
A fetus who is deemed FIT for labour at the commencement of labour may not remain FIT throughout labour.
This is because several antepartum and intrapartum risk factors may cause fetal compromise during labour, and it is essential to anticipate and timely recognise the specific changes on the CTG trace caused by this underlying pathology.
The blind use of “toolkits” without changing the “Normal, suspicious, Pathological” (or White, Amber, Green) CTG guidelines without considering the types of fetal hypoxia, fetal response to hypoxic and inflammatory stress and the impact of the identified risk factor on the fetal heart rate is likely to worsen maternal and perinatal outcomes.It is hoped that the use of FIT-CAT will aid frontline clinicians to predict the expected changes based on the observed clinical context.
Conflict of Interest
EC, ME and MB are members of the International Expert Consensus Guidelines on Physiological Interpretation of CTG. However, they have no financial interests which conflict with this publication.
Funding Statement
None.
Acknowledgements
None.
Contribution of Authorship
EC conceived the concept of “FIT-CAT”.
SG, YK, ME and MB contributed to the drafting of the manuscript, reviewing and editing the manuscript.
References
1. Chandraharan E, Tahan ME, Pereira S (2016) Each Fetus Matters: An Urgent Paradigm Shift is needed to Move away from the Rigid “CTG Guideline Stickers” so as to Individualize Intrapartum Fetal Heart Rate Monitoring and to improve Perinatal Outcomes. Obstet Gynecol Int J 5(4): 00168. DOI: 10.15406/ogij.2016.05.00168.
2. Chandraharan E (2019) Intrapartum care: An urgent need to question historical practices and ‘non-evidence’-based, illogical foetal monitoring guidelines to avoid patient harm. Journal of Patient Safety and Risk Management 24(5): 210-217.
3. Pereira S, Chandraharan E. Recognition of chronic hypoxia and pre-existing foetal injury on the cardiotocograph (CTG): Urgent need to think beyond the guidelines. Porto Biomed J. 2017 Jul-Aug;2(4):124-129.
4. Chandraharan E. Physiological interpretation of CTG: From Knowledge to Practice. Volumes 1-3. Glob Acad Med Edu Train, London, KDP. 2022 (https://www.amazon.co.uk/s?k=chandraharan&crid=1VKO4VCPZJ5IV&sprefix=chandraharan%2Caps%2C310&ref=nb_sb_noss_1)
5. Chandraharan E, Pereira S, Ghi T, Gracia Perez-Bonfils A, Fieni S, Jia YJ, Griffiths K, Sukumaran S, Ingram C, Reeves K, Bolten M, Loser K, Carreras E, Suy A, Garcia-Ruiz I, Galli L, Zaima A. International expert consensus statement on physiological interpretation of cardiotocograph (CTG): First revision (2024). Eur J Obstet Gynecol Reprod Biol. 2024 Nov;302:346-355.
6. Chandraharan E. Updated NICE Cardiotocograph (CTG) guideline: Is it suspicious or pathological? J Clin MedSurgery. 2023; 3(2): 1129
7. di Pasquo E, Fieni S, Chandraharan E, Dall’Asta A, Morganelli G, Spinelli M, Bettinelli ML, Aloe R, Russo A, Galli L, Perrone S, Ghi T. Correlation between intrapartum CTG findings and interleukin-6 levels in the umbilical cord arterial blood: A prospective cohort study. Eur J Obstet Gynecol Reprod Biol. 2024 Jan 13;294:128-134.
8. Ghi T, Fieni S, Ramirez Zegarra R, Pereira S, Dall’Asta A, Chandraharan E. Relative uteroplacental insufficiency of labor. Acta Obstet Gynecol Scand. 2024 Oct;103(10):1910-1918. doi: 10.1111/aogs.14937.
9. Eidelman AI, Nevet A, Rudensky B, Rabinowitz R, Hammerman C, Raveh D, Schimmel MS. The effect of meconium staining of amniotic fluid on the growth of Escherichia coli and group B streptococcus. J Perinatol. 2002;22(6):467- 71.
10. Bolten M, Chandraharan E. The Significance of ‘Non-Significant’ Meconium Stained Amniotic Fluid (MSAF): Colour versus Contents. J Adv Med Med Res. 2019. doi:10.9734/jammr/2019/v30i530192
11. Pinas A, Chandraharan E. Continuous cardiotocography during labour: Analysis, classification and management. Best Pract Res Clin Obstet Gynaecol. 2016 Jan;30:33-47.
12. Chandraharan E, Arulkumaran S. Prevention of birth asphyxia: responding appropriately to cardiotocograph (CTG) traces. Best Pract Res Clin Obstet Gynaecol. 2007 Aug;21(4):609-24.
13. Chandraharan E. Handbook of CTG interpretation: From patterns to Physiology. First Edition, Cambridge, Cambridge University Press, 2017.
14. Griffiths K, Gupta N, Chandraharan E. Intrapartum fetal surveillance: a physiological approach, Obstetrics, Gynaecology & Reproductive Medicine, Volume 32, Issue 8, 2022,Pages 179-187,
15. Gracia-Perez-Bonfils A, Vigneswaran K, Cuadras D, Chandraharan E. Does the saltatory pattern on cardiotocograph (CTG) trace really exist? The ZigZag pattern as an alternative definition and its correlation with perinatal outcomes. J Matern Fetal Neonatal Med. 2019 Nov 13:1-9.
16. Tarvonen M, Hovi P, Sainio S, Vuorela P, Andersson S, Teramo K. Factors associated with intrapartum ZigZag pattern of fetal heart rate: A retrospective one-year cohort study of 5150 singleton childbirths. Eur J Obstet Gynecol Reprod Biol. 2021 Mar;258:118-125.
17. Chandraharan E. Physiological CTG Masterclass Course Manual. Glob Acad Med Edu Train, London, KDP. 2023. https://www.amazon.co.uk/Physiological-CTG-Masterclasses-Course-Manual/dp/B0C9S99Q3D/ref=sr_1_5?dib=eyJ2IjoiMSJ9.QhJ78JEiv-NVrMd2RUsft6y7dLxSG66qtjE4ZiMw1uhh5iEX7GEsB-a7lDiPAmhL6Zed_7W2FbluQumq2ciYh1zFbk2OQdAb0Kz1DfQDcfI.it1-z6e-mBiAOhDYjgUdOlBltBtCvD3ZkFWCUKzZEkU&dib_tag=se&keywords=chandraharan&qid=1705689835&sr=8-5
18. Royal College of Obstetricians and Gynaecologists. Each Baby Counts: 2020 Final Progress Report. London: RCOG; 2021.( https://www.rcog.org.uk/globalassets/documents/guidelines/research–audit/each-baby-counts/ebc-2020-final-progress-report.pdf)
19. Chandraharan E. Updated NICE Cardiotocograph (CTG) guideline: Is it suspicious or pathological?. J Clin MedSurgery. 2023; 3(2): 112.
20. Evidence-based Clinical Guideline No. 8 The use and interpretation of CTG in intrapartum fetal surveillance (2001). RCOG Press, 2001.
21. National Institute of Clinical Excellence. Intrapartum care: care of healthy women and their babies during labour. NICE Clinical Guideline, December 2014. https://www.nice.org.uk/guidance/cg190/resources/intrapartum-care-for-healthy-women-and-babies-pdf-35109866447557
22. Physiological interpretation of CTG: From Knowledge to Practice. Volumes 3: Avoiding medicolegal pitfalls. Glob Acad Med Edu Train, London, KDP. 2022 (https://www.amazon.co.uk/s?k=chandraharan&crid=1VKO4VCPZJ5IV&sprefix=chandraharan%2Caps%2C310&ref=nb_sb_noss_1)
23. Intrapartum-related Deaths: 500 Missed Opportunities. Chapter In: 2006 Annual Report of the Chief Medical Officer. On the State of Public Health. Department of Health; 2007. (https://webarchive.nationalarchives.gov.uk/ukgwa/20130105021748/http:/www.dh.gov.uk/en/Publicationsandstatistics/Publications/AnnualReports/DH_076817)
24. Study of Stillbirth Claims by NHSLA (2009). https://silo.tips/download/study-of-stillbirth-claims-published-by-nhs-litigation-authority
25. Royal College of Obstetricians and Gynaecologists. Each Baby Counts: 2018 Progress Report. London: RCOG; 2018. (https://www.rcog.org.uk/media/dswjqyin/each-baby-counts-report-2018-11-12.pdf) .
26. Royal College of Obstetricians and Gynaecologists. Each Baby Counts: 2019 Progress Report. London: RCOG; 2020. (https://www.rcog.org.uk/media/qhzlelnc/each-baby-counts-2019-progress-report.pdf) .
27. Royal College of Obstetricians and Gynaecologists. Each Baby Counts: 2020 Final Progress Report. London: RCOG; 2021 (https://www.rcog.org.uk/media/a4eg2xnm/ebc-2020-final-progress-report.pdf).
28. Study of Stillbirth Claims by NHSLA (2009). https://silo.tips/download/study-of-stillbirth-claims-published-by-nhs-litigation-authority
29. NHSLA. Ten years of maternity claims: An analysis of NHS litigation authority data .2012. www.nhsla.com/safety/Documents/Ten%20Years%20of%20Maternity%20Claims%20%20An%20Analysis%20 of%20the%20NHS%20LA%20Data%20-%20October%202012.pdf .
30. NHS Resolution, The Early Notification scheme progress report: collaboration and improved experience for families, September 2019. https://resolution.nhs.uk/wp-content/uploads/2019/09/NHS-Resolution-Early-Notification-report.pdf
31. NHS Resolution. Annual Report & Accounts 2023/24. Published on 23 July 2024. https://resolution.nhs.uk/wp-content/uploads/2024/07/NHS-Resolution-Annual-report-and-accounts_23-24_Access.pdf
32. Bolten M, Chandraharan E. The Significance of ‘Non-Significant’ Meconium Stained Amniotic Fluid (MSAF): Colour versus Contents. J Adv Med Med Res. 2019. doi:10.9734/jammr/2019/v30i530192
33. Mitchell S, Chandraharan E, Meconium-stained amniotic fluid, Obstetrics, Gynaecology & Reproductive Medicine, Volume 28, Issue 4, 2018, Pages 120-124, ISSN 1751-7214.
34. Jonsson M, Nordén SL, Hanson U. Analysis of malpractice claims with a focus on oxytocin use in labour. Acta Obstet Gynecol Scand. 2007;86(3):315-9.
35. Sukumaran S, Jia YJ, Chandraharan E. Uterine Tachysystole, Hypertonus and Hyperstimulation: An urgent need to get the definitions right to avoid Intrapartum Hypoxic-Ischaemic Brain Injury. Glob J Reprod Med. 2021; 8(2): 5556735. DOI: 10.19080 /GJORM.2021.08.555735.
36. Badawi N, Kurinczuk JJ, Keogh JM, Alessandri LM, O’Sullivan F, Burton PR, et al. Intrapartum risk factors for newborn encephalopathy: the Western Australian case-control study. BMJ. 1998 Dec 5;317(7172):1554–8.
37. Sukumaran S, Pereira V, Mallur S, Chandraharan E. Cardiotocograph (CTG) changes and maternal and neonatal outcomes in chorioamnionitis and/or funisitis confirmed on histopathology. Eur J Obstet Gynecol Reprod Biol. 2021 May;260:183-188. doi: 10.1016/j.ejogrb.202 1.03.029. Epub 2021 Mar 30.
38. Wu YW, EscobarGJ, Grether JK, Croen LA, Greene JD, Newman TB. Chorioamnionitis and cerebral palsy in term and near-term infants. JAMA. 2003;290:2677–84.
39. Wu YW, Colford JM Jr. Chorioamnionitis as a risk factor for cerebral palsy: a meta-analysis. JAMA. 2000;284:1417–24.
40. Mallard C, Hagberg H. Inflammation-induced preconditioning in the immature brain. Semin Fetal Neonatal Med. 2007 Aug;12(4):280–6.
41. Hagberg H, Peebles D, Mallard C. Models of white matter injury: comparison of infectious, hypoxic-ischemic, and excitotoxic insults. Ment Retard Dev Disabil Res Rev. 2002;8(1):30–8.
42. Galli L, Dall’Asta A, Whelehan V, Archer A, Chandraharan E. Intrapartum cardiotocography patterns observed in suspected clinical and subclinical chorioamnionitis in term fetuses. J Obstet Gynaecol Res. 2019 Dec;45(12):2343-2350. doi: 10.1111/jog.14133. Epub 2019 Oct 16.
43. Sukumaran S, Pereira V, Mallur S, Chandraharan E. Cardiotocograph (CTG) changes and maternal and neonatal outcomes in chorioamnionitis and/or funisitis confirmed on histopathology. Eur J Obstet Gynecol Reprod Biol. 2021 May;260:183-188. doi: 10.1016/j.ejogrb.20 21.03.029. Epub 2021 Mar 30.
44. Gracia-Perez-Bonfils A, Martinez-Perez O, Llurba E, Chandraharan E. Fetal heart rate changes on the cardiotocograph trace secondary to maternal COVID-19 infection. Eur J Obstet Gynecol Reprod Biol. 2020 Sep;252:286-293.
45. Eklind S,Mallard C, LeverinAL, Gilland E, Blomgren K,Mattsby-Baltzer I, et al. Bacterial endotoxin sensitizes the immature brain to hypoxic–ischaemic injury. Eur J Neurosci. 2001 Mar;13(6) :1101–6.
46. Eklind S,Mallard C, Arvidsson P,Hagberg H. Lipopolysaccharide induces both a primary and a secondary phase of sensitization in the developing rat brain. Pediatr Res. 2005 Jul;58(1):112–6.
47. Yang L, Sameshima H, Ikeda T, Ikenoue T. Lipopolysaccharide administration enhances hypoxic-ischemic brain damage in newborn rats. J Obstet Gynaecol Res. 2004;30(2):142–7.
48. Yang L, Sameshima H, Ikeda T, Ikenoue T. Lipopolysaccharide administration enhances hypoxic-ischemic brain damage in newborn rats. J Obstet Gynaecol Res. 2004;30(2):142–7.
49. Coumans AB, Middelanis JS, Garnier Y, Vaihinger HM, Leib SL, Von Duering MU, et al. Intracisternal application of endotoxin enhances the susceptibility to subsequent hypoxic-ischemic brain damage in neonatal rats. Pediatr Res. 2003 May;53(5):770–5
50. Grether JK, Nelson KB. Maternal infection and cerebral palsy in infants of normal birth weight. JAMA. 1997 Jul;16(278):207–11.
51. Chandraharan E. Fetal scalp blood sampling during labour: is it a useful diagnostic test or a historical test that no longer has a place in modern clinical obstetrics? BJOG 2014;121:1056–60, discussion 1060–2.
52. Chandraharan E, Wiberg N. Fetal scalp blood sampling during labor: an appraisal of the physiological basis and scientific evidence. Acta Obstet Gynecol Scand 2014;93:544–7.
53. Chandraharan E. Should national guidelines continue to recommend fetal scalp blood sampling during labor?. J Matern Fetal Neonatal Med. 2016; 29(22):3682-3685. doi:10.3109/14767058.2016.1140740.
54. di Pasquo E, Fieni S, Chandraharan E, Dall’Asta A, Morganelli G, Spinelli M, Bettinelli ML, Aloe R, Russo A, Galli L, Perrone S, Ghi T. Correlation between intrapartum CTG findings and interleukin-6 levels in the umbilical cord arterial blood: A prospective cohort study. Eur J Obstet Gynecol Reprod Biol. 2024 Jan 13;294:128-134.
55. Chandraharan, E., & Bolten, M. (2024). Recognition of Chorioamnionitis on the Cardiotocograph (CTG): The role of the “Chorio Duck Score”. European Journal of Medical and Health Sciences, 6(1), 1–9.
56. Fetal Monitoring in Labor. NG224. National Institute of Health and Care Excellence (NICE). December 2022. https://www.nice.org.uk/guidance/ng229/resources/fetal-monitoring-in-labour-pdf-66143844065221.
57. Society for Maternal-Fetal Medicine (SMFM). Electronic address: [email protected]; Silver R, Craigo S, Porter F, Osmundson SS, Kuller JA, Norton ME. Society for Maternal-Fetal Medicine Consult Series #64: Systemic lupus erythematosus in pregnancy. Am J Obstet Gynecol. 2023 Mar ;228(3):B41-B60.
58. Giorgio E, De Oronzo MA, Iozza I, Di Natale A, Cianci S, Garofalo G, Giacobbe AM, Politi S. Parvovirus B19 during pregnancy: a review. J Prenat Med. 2010 Oct;4(4):63-6.
59. Kielaite D, Paliulyte V. Parvovirus (B19) Infection during Pregnancy: Possible Effect on the Course of Pregnancy and Rare Fetal Outcomes. A Case Report and Literature Review. Medicina (Kaunas). 2022 May 15;58(5):664.
60. Preti M, Chandraharan E. Importance of fetal heart rate cycling during the interpretation of the cardiotocograph (CTG). Int J Gynecol Reprod Sci. 2018;1(1):10–12.
61. Pereira S, Lau K, Modestini C, Wertheim D, Chandraharan E. Absence of fetal heart rate cycling on the intrapartum cardiotocograph (CTG) is associated with intrapartum pyrexia and lower Apgar scores. J Matern Fetal Neonatal Med. 2021 Jun 22:1-6. doi: 10.1080/14767058.2021.1940130.
62. Chandraharan E. Fetal scalp blood sampling should be abandoned: FOR: FBS does not fulfil the principle of first do no harm. BJOG. 2016 Oct;123(11):1770.
63. O’Heney J, McAllister S, Maresh M, Blott M. Fetal monitoring in labour: summary and update of NICE guidance. BMJ. 2022 Dec 16;379:o2854.
64. Alfirevic Z, Devane D and Gyte G. Continuous cardio- tocography (CTG) as a form of electronic fetal monitor- ing (EFM) for fetal assessment during labour. Cochrane Database Syst Rev 2013; 5: CD006066.
65. Holzmann M, Wretler S, Cnattingius S, Nordström L. Neonatal outcome and delivery mode in labors with repetitive fetal scalp blood sampling. Eur J Obstet Gynecol Reprod Biol. 2015 Jan;184:97-102.
66. Stål I, Wennerholm UB, Nordstrom L, Ladfors L, Wiberg-Itzel E. Fetal scalp blood sampling during second stage of labor – analyzing lactate or pH? A secondary analysis of a randomized controlled trial. J Matern Fetal Neonatal Med. 2022 Mar;35(6):1100-1107.
67. Al Wattar BH, Lakhiani A, Sacco A, et al; AB-FAB Study Group. Evaluating the value of intrapartum fetal scalp blood sampling to predict adverse neonatal out- comes: a UK multicentre observational study. Eur J Obstet Gynecol Reprod Biol 2019; 240: 62–67.
68. Chandraharan E. Physiological interpretation of CTG: From Knowledge to Practice. Volume 3: Avoiding Medico-legal Pitfalls. Glob Acad Med Edu Train, London, KDP. 2022 (https://www.amazon.co.uk/s?k=chandraharan&crid=1VKO4VCPZJ5IV&sprefix=chandraharan%2Caps%2C310&ref=nb_sb_noss_1)

