Transcatheter Management of Postoperative Shunt Lesions
Transcatheter interventional management of postoperative residual shunt lesion after congenital cardiac surgery
Dr. Naharuma Aive Hyder Chowdhury¹; Mohammad Sharifuzzaman²; Abul Kalam Shamsuddin³; Dr Jesmin Hossain⁴; Dr. Mohammed Eliyas Patwary⁵; Abul Kalam Azad, MD⁶; Prodip kumer Biswas⁷; Dr Tawfiq Shahriar Haq⁸
- Associate Professor, Pediatric Cardiology, National Heart Foundation Hospital & Research Institute, Dhaka
- Professor, Pediatric Cardiac surgery, National Heart Foundation Hospital & Research Institute, Dhaka
- Professor, Pediatric Cardiac surgery, National Heart Foundation Hospital & Research Institute, Dhaka
- Associate Professor, Pediatric Cardiology, National Heart Foundation Hospital & Research Institute, Dhaka
- Associate Professor, Pediatric Cardiac surgery, National Heart Foundation Hospital & Research Institute, Dhaka
- Assistant Professor, Pediatric Cardiac surgery, National Heart Foundation Hospital & Research Institute, Dhaka
- Assistant Professor, Pediatric Cardiac surgery, National Heart Foundation Hospital & Research Institute, Dhaka
- Professor, Pediatric Cardiology, National Heart Foundation Hospital & Research Institute, Dhaka
OPEN ACCESS
PUBLISHED 31 December 2024
CITATION Chowdhury, NAH., Sharifuzzaman, M., et al., 2024. Transcatheter interventional management of postoperative residual shunt lesion after congenital cardiac surgery. Medical Research Archives, [online] 12(12). https://doi.org/10.18103/mra.v12i12.6005
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i12.6005
ISSN 2375-1924
ABSTRACT
Background: Congenital heart defects can be effectively managed through a variety of approaches, including conservative treatment, surgery, or interventional procedures. Complex defects, such as ventricular or atrial septal defects and patent ductus arteriosus, often necessitate congenital cardiac surgery for correction. Residual lesions refer to defects that persist after surgery or develop later on. Hemodynamically significant residual shunts should be addressed through either transcatheter interventions or redo surgery. Transcatheter closure is increasingly favored as it is less invasive, has a high safety profile, and typically results in lower morbidity, faster recovery times, and reduced costs compared to traditional redo open-heart surgery. This approach allows for effective management of congenital heart defects while minimizing the impact on the patient’s overall health. This study aimed to understand the outcomes of these transcatheter management strategies, providing insight into effective clinical practices for managing post-surgical congenital cardiac shunt lesions.
Methods: The patients who developed residual lesion after cardiac surgical correction of shunt lesion like ventricular septal defect (VSD), arterial switch operation (ASO) with atrial septal defect (ASD), ASD with mitral valve cleft, patent ductus arteriosus (PDA) ligation were included. This is a prospective observational study which was done in National Heart Foundation Hospital & Research Institute during 2014 August to 2024 September.
Results: Among the seven patients with post-operative residual shunt lesions, four were male and three were female, with a median age of 11 years, ranging from 1 to 28 years. Six of these patients (85.7%) underwent elective closures, while one required an emergency intervention (14.3%). On average, the interval between surgery and the subsequent transcatheter intervention was 6.6 years, with a range from 15 days to 13 years. The average hospital stay across all cases was 7 days, varying from 4 to 22 days, and the follow-up duration averaged 5.84 years. Among these patients, one individual with VSD had a residual shunt. Importantly, there were no vascular access-related complications, postprocedural heart block, hemolysis, significant new valvular regurgitation, or procedure-related mortality.
Conclusion: Transcatheter-based interventions are typically the first-line treatment for newly diagnosed cases. Here a few rare interventional techniques have demonstrated successfully resolved complex surgical complications, ensuring optimal patient outcomes with minimal risks.
Keywords: Congenital heart disease, shunt lesion, transcatheter interventions, postoperative complications, ventricular septal defect (VSD), arterial switch operation (ASO), atrial septal defect (ASD), mitral valve cleft, patent ductus arteriosus (PDA) ligation.
INTRODUCTION
Morbidity and mortality-free survival after surgical intervention for congenital heart disease is influenced by presence or residual lesion after surgery. The effect of residual lesions and the timing of reintervention has significant effect on postoperative length of stay, adverse events, and cost. Decision about repairing a residual lesion and its timing is commonly influenced by many factors like patient features, criteria for reintervention, and the complexity of the procedure. It is pertinent to say residual lesions tend to occur in the youngest and smallest patients with the most complex lesions. It is obvious that re-thoracotomy poses significant risks. Transcatheter closure offers a minimally invasive solution for managing residual lesions that may remain after surgery or develop over time, often following repairs of congenital heart defects. These residual lesions, which include valve leaks and defects like atrial septal defects (ASD), ventricular septal defects (VSD), and patent ductus arteriosus (PDA), are relatively common. Residual ASD lesions are seen in about 7-8% of cases. For VSDs, the incidence of residual lesions can vary significantly, ranging from 5% to 25% depending on the defect type and repair technique. In cases of PDA, residual lesions occur in approximately 6%.
Residual lesions after congenital heart defect repair aren’t solely attributed to surgical technique; they can arise from anatomical variations, incomplete preoperative assessment, or even complications, such as infections associated with prosthetic patches. Small, hemodynamically insignificant defects often require only regular medical follow-up, as they generally don’t disrupt blood flow significantly. However, larger residual defects may necessitate reintervention to prevent a range of complications. Potential issues include electrophysiological disturbances, valvular disease, and persistent shunting, as well as more severe consequences like thromboembolic events, vascular disease, myocardial dysfunction, or infection. Early detection and careful monitoring help guide appropriate management, minimizing risks for affected patients.
Transcatheter closure offers an alternative to redo open-heart surgery, providing a less invasive option with a high safety profile, lower morbidity, and faster recovery times. Although rare, potential complications of this procedure, such as device dislodgement or embolization, are generally manageable. The literature on patients undergoing transcatheter closure for postoperative residual defects is limited, making further research valuable in understanding long-term outcomes.
METHODS
The study was done from August 2014 to September 2024 and analyzed a total of 7,597 cardiac catheterization cases, including 4,289 diagnostic catheterizations (56.45%), 3,308 therapeutic interventions (43.54%), 338 emergency interventions (10.21%), and 2,970 elective routine interventions (89.78%). Notably, 7 cases (0.21%) involved postoperative residual shunt lesions requiring intervention. These residual lesions typically arose after congenital heart surgeries such as tetralogy of Fallot repair, arterial switch operation with ASD closure, ASD repair with mitral valve cleft correction, and PDA closure. All patients with residual shunt were clinically assessed and done twelve leads electrocardiogram (ECG), chest X-ray, and complete transthoracic echocardiography (TTE), the pulmonary to systemic flow (QP/QS:≥1.5) ratio, pulmonary arterial pressure, pulmonary and systemic vascular resistance and ratio during catheterization were done for all patients. Procedures are done under general anesthesia for small children especially in risky patients with significant residual shunt infants, and critical patients. Adult patients underwent procedures with conscious sedation or local anesthesia. After vascular sheath insertion, unfractionated heparin 100 IU/kg was given.
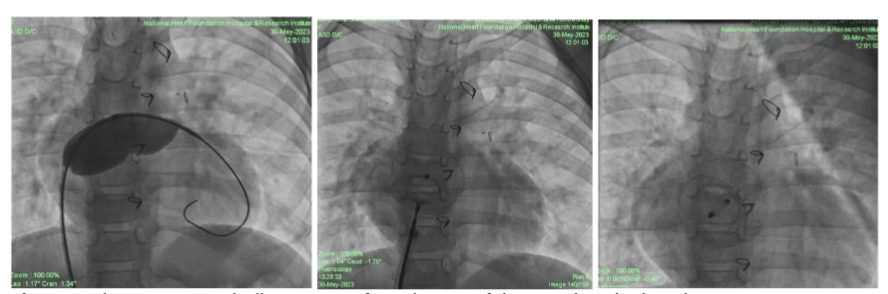
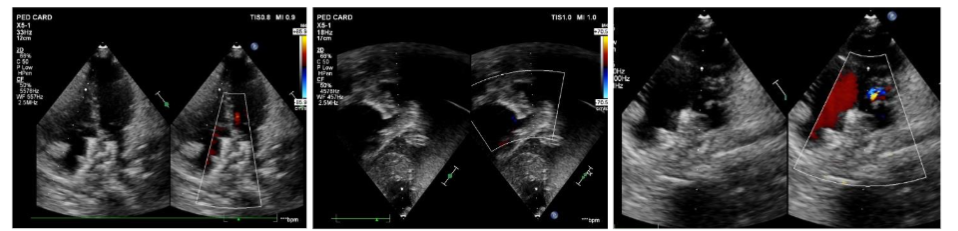
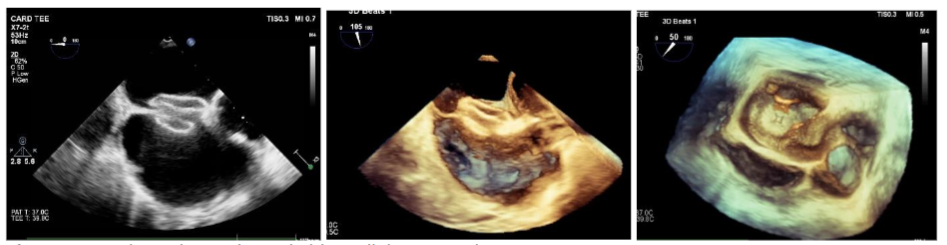
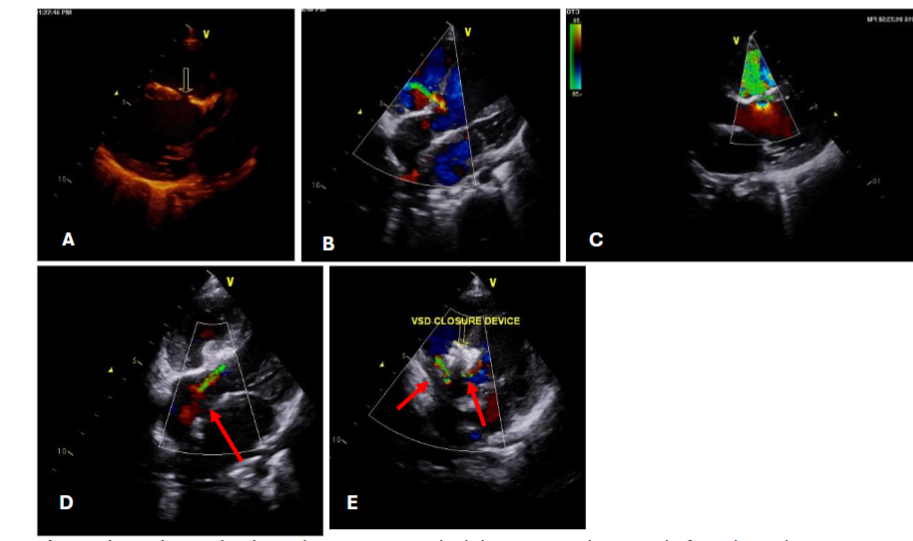
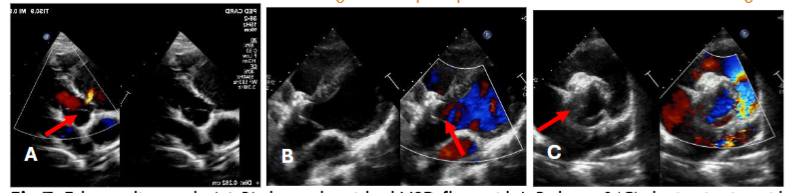
In this case of atrial septal defect (ASD) management, three patients underwent comprehensive evaluations and ASD closure procedures with distinct considerations for each because the normal anatomy was distorted for patch dehiscence in post operative residual case. Transesophageal Echocardiography (TEE) was necessary for one patient to provide enhanced visualization of the septal anatomy. TEE is typically used when more detailed imaging is needed, often in cases where transthoracic echocardiography (TTE) does not provide sufficient clarity or when precise guidance is required during interventions. The other two patients were evaluated using transthoracic echocardiography (TTE). Findings for all three patients included right-sided chamber dilation (likely to be due to volume overload from the left-to-right shunt associated with the ASD) and mild pulmonary arterial hypertension. During catheterization, a significant shunt was confirmed, with a QP/QS ratio ≥1.5, indicating hemodynamically significant ASD requiring intervention. Balloon sizing was performed for all patients to accurately measure the defect and guide device selection for ASD closure. Balloon sizing is crucial for determining the optimal device size and ensuring effective closure with minimal risk of complications. One patient required balloon-assisted device deployment, a technique that provides additional stability and control when deploying the closure device. This approach was chosen for challenging ASD and we were concerned about device positioning. For the other two patients, the device was deployed from the right upper pulmonary vein approach. Fluoroscopy and echocardiography were both utilized to guide device deployment, allowing real-time visualization of device position and septal alignment to ensure optimal closure without residual shunting or encroachment on nearby structures. 3D echocardiography was performed for one patient before device deployment, providing a more detailed and spatial view of the defect. 3D echocardiography allowed us to evaluate the exact shape, location, and surrounding structures of the defect.
In case of VSD, transthoracic echo done to all patient which demonstrated size, location, and proximity to tricuspid and aortic valves. VSD is considered hemodynamically significant if it meets certain criteria indicating it may be affecting the heart’s function such as clinical symptoms of heart failure, left heart chambers dilatation, greater than 33–66% of the diameter of the left ventricular outflow tract (LVOT) and during catheterization, if the QP/QS ratio is ≥1.5. Such VSDs may be selected for percutaneous closure.
In this study, patients presented with hemodynamically significant ventricular septal defects (VSDs), requiring careful assessment of residual shunts following initial repair. Of the three patients, two had a single residual shunt each, both of which were positioned more than 2 mm away from the tricuspid and aortic valves, causing no immediate concern regarding valve interference. The third patient, however, presented with two residual VSD shunts at either end of the patch. While one of these residual shunts was also safely positioned away from the valves, the other was located in proximity to the aortic valve leaflet, potentially posing a risk of interference or other complications. To address this, the treatment plan included closing the residual shunt situated near the tricuspid valve, given its easier accessibility and lower risk to adjacent structures. The other residual shunt, close to the aortic valve, was intentionally left open to avoid potential damage or complications that could arise from interventions near the aortic leaflet. This conservative approach aimed to balance effective shunt management while safeguarding the integrity of the valves.
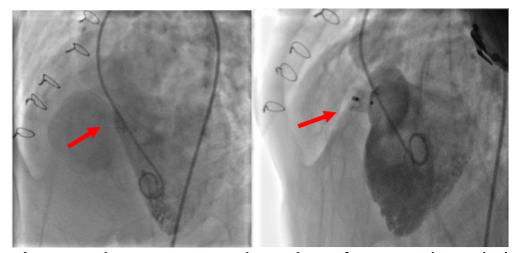
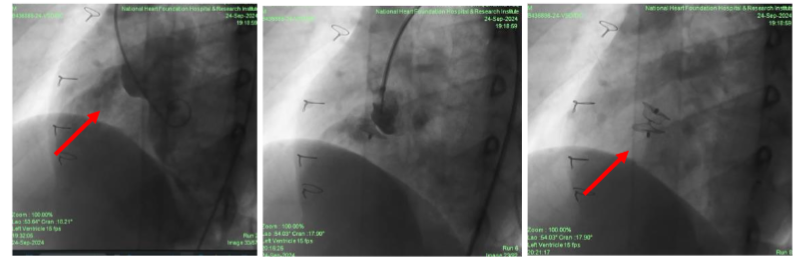
Residual postoperative VSDs of hemodynamic significance carry a high risk of morbidity and mortality, especially after complex congenital cardiac surgery. In one of our complex cases, a patient could not be successfully extubated following surgery, prompting an emergency transcatheter closure of the residual ventricular septal defect (VSD). Factors such as the patch material, geometry, size, and shunt location were carefully considered in determining the feasibility and approach for the transcatheter closure. All patients underwent assessment using fluoroscopy and transthoracic echocardiography to guide the selection of a suitable VSD occluder device based on availability and compatibility. Angiography was performed in 4 chamber projections and a left anterior oblique (LAO) to delineate the residual VSD size, location, and proximity to the aortic valve.
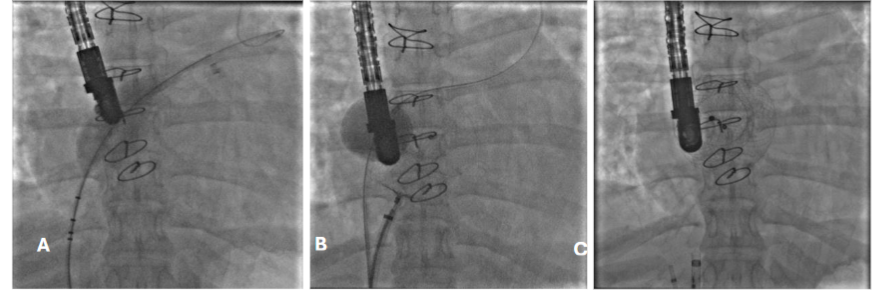
This study utilized a range of devices, including the Amplatzer Duct Occluders (ADO I & II, Abbott Medical, MN, USA) and the Multifunctional Konar VSD occluder (MFO, Lifetech Scientific, Shenzhen, China). The ADO I device was deployed via an antegrade approach in one patient, where an arteriovenous loop was established to facilitate access. For the other two patients, the ADO II and MFO devices were delivered through a retrograde approach. This tailored selection of devices and deployment techniques aimed to optimize outcomes in the management of complex residual VSDs following surgical intervention.
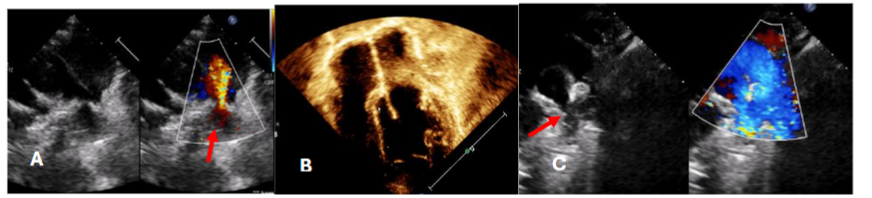
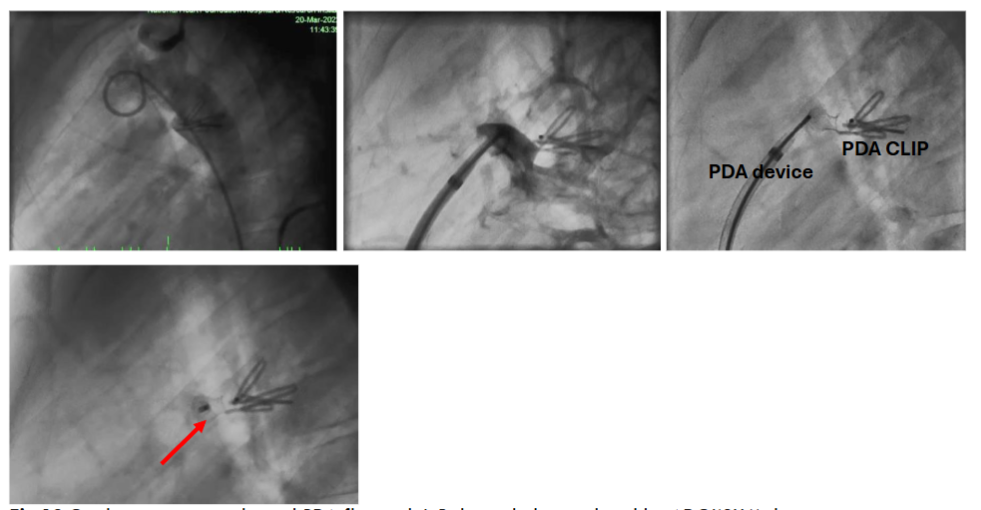
In case of PDA, transthoracic echo demonstrated residual PDA flow with hemodynamic significance and PDA duct was closed by antegrade approach with ADO I(8X6) device. There was considerable resistance while pulling the device into place due to distorted anatomy. Before deployment of device, position was confirmed by fluoroscopy & echocardiography. Next day TTE was done to all patient and discharged with antiplatelet to ASD patients but not to VSD or PDA patient. Discharged after 2-4 days, only emergency patient took longer time than usual. Patients were followed up after one, three, six months and one year. This prospective observational study aimed to understand the outcomes of these transcatheter management strategies, providing insight into effective clinical practices for managing post-surgical congenital heart shunt lesions.
RESULTS
| Case | Age | Sex | Weight | Presentation | Diagnosis | Name of surgery | Time interval between surgery & intervention | Nature of intervention | Type of shunt closure/residual |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 5 yrs | F | 16 kg | Repeated respiratory infection | d-Transposition of Aorta with ASD | Arterial switch with ASD closure | 5 yr 4 m | Elective | Residual ASD closure |
| 2 | 28 yrs | M | 56 kg | Shortness of breath | ASD with cleft in mitral leaflet with moderate MR | ASD closure with mitral valve repair | 2 yrs 3m | Elective | Residual ASD closure |
| 3 | 16 yrs | F | 30 kg | Effort breathing | ASD with mild pulmonary hypertension | ASD patch closure | 1 yr 8 m | Elective | Residual ASD closure |
| 4 | 1 yrs | M | 9.2 kg | Repeated respiratory infection | Double outlet right ventricle with ventricular septal defect with pulmonary stenosis | VSD closure with right ventricular muscle band resection | 1 yr 1 m | Elective | Residual VSD closure |
| 5 | 1 yr | M | 9.5kg | Intubated child for 15 days after surgery | Tetralogy of Fallots | VSD closure with RVOT resection | 15 d | Emergency | Residual VSD closure |
| 6 | 15 yrs | M | 49 kg | Shortness of breath | Double chamber right ventricle with RVOTO | VSD closure with RVOT resection | 13 yrs | Elective | Residual VSD closure |
| 7 | 7 yrs | F | 20 kg | Repeated respiratory infection | PDA | PDA Clipping | 3 yrs 3 m | Elective | Residual PDA closure |
| Case | Echocardiography | Cardiac cath | Type of device | Length of hospital stay | Complication | Duration of Follow up |
|---|---|---|---|---|---|---|
| 1 | S/P ASO with large ASD (17X18 mm) with L-R shunt with mild AR with dilated RA & RV PASP:40/15/29 mmhg Qp:Qs=1.6:1 | ASO (24 mm) | 5 days | No | 1 yr 5 mo | |
| 2 | Secundum ASD (14X12X12) and cleft mitral valve with mild MR PASP: 45/15/30mmhg Qp:Qs=2.9:1 | ASO (20 mm) | 4 days | No | 10 yrs | |
| 3 | Secundum ASD (16X14X12) PASP: 35/15/26mmhg Qp:Qs=1.7:1 | ASO (22 mm) | 4 days | No | 6 yrs | |
| 4 | S/P VSD closure with residual VSD (5X5 mm) right ventricular muscle band resection PASP: 40/15/22mmhg Qp:Qs=1.65:1 | ADO I (10 X 8) | 5 days | Residual flow L-R | 9 yrs | |
| 5 | S/P VSD closure with residual VSD (3X5 mm) right ventricular muscle band resection PASP: 45/15/30mmhg Qp:Qs=2.9:1 | ADOII(6X5) | 22 days | No | 12 yrs | |
| 6 | S/P VSD closure with residual VSD (5X7 mm) with no RVOT PASP: 30/10/13mmhg Qp:Qs=1.6:1 | MFO (12X10) | 5 days | No | 1 m | |
| 7 | S/P PDA ligation with residual PDA flow (3X5 mm) with L-R shunt PASP: 30/10/13mmhg Qp:Qs=1.6:1 | ADOI(8X6) | 4 days | No | 2 yrs 7m |
Among the seven patients with post-operative residual shunt lesions, four were male and three were female, with a median age of 11 years (ranging from 1 to 28 years). Six patients underwent elective closures (85.7%), while one patient required an emergency intervention (14.3%). The average interval between surgery and subsequent transcatheter intervention was 6.6 years, varying from 15 days to 13 years. The emergency case involved a patient with a residual VSD following TOF repair, who failed to be weaned off mechanical ventilation. Hemodynamic assessment revealed a significant shunt, which was closed using an ADO II device and discharged after 7 days of device closure. The average hospital stay across all cases was 7 days (ranges from 4 days to 22 days), though the emergency case had a longer ICU stay post-surgery. The average follow-up duration was 5.84 years, providing insight into long-term outcomes and stability. One VSD patient (14.3%) had a residual shunt lesion which was kept intentionally due to adverse location (near aortic valve). This patient remained stable with ongoing follow-up. There were no reported vascular access-related complications, postprocedural heart block, hemolysis, or significant new valvular regurgitation after the procedure or procedure-related mortality.
DISCUSSION
Postoperative residual lesions often develop in patients with more complex congenital heart conditions and may result from factors like patch dehiscence, suture disruption, incomplete defect closure, or bacterial endocarditis. In patients with intricate anatomical abnormalities or extensive repair needs, these challenges increase the likelihood of residual defects. The complexity of their condition may involve greater tissue tension around the repair site or increased vulnerability to infection.
No intentional fenestrations in the patches or dehiscence secondary to infective endocarditis near the surgical patches were reported in this study. Suture disruption may be a cause for residual lesion. Patch dehiscence most frequently occurs in the posteroinferior and posterosuperior quadrants, regions close to the conductive tissue. This proximity to critical conduction pathways limits the surgeon’s ability to place continuous sutures in these areas, as excessive manipulation could damage the conductive tissue and cause arrhythmias. Consequently, these areas are more prone to dehiscence due to the need for more delicate suturing techniques, which may provide less reinforcement and contribute to the risk of residual lesions.
So these individuals present the greatest challenges in terms of imaging, access, and the technical aspects of surgery, particularly intracardiac repair. However a number of factors including patient features, criteria for reintervention, and the complexity of the procedure play an important role in this decision.
Some residual shunts are restrictive and well-tolerated, but even these can lead to left-to-right shunting, causing persistent ventricular volume overload or pulmonary hypertension over time. Although patients may initially tolerate these residual lesions, the resulting hemodynamic burden on the heart and lungs can gradually lead to more significant issues. This is why certain postoperative residual defects require close monitoring and, in some cases, reintervention to prevent complications such as ventricular strain, progressive pulmonary hypertension, and other adverse effects on cardiac function. Early reintervention can help mitigate these risks and improve long-term outcomes.
Consider the side effects and potential morbidity and mortality associated with prolonged and repeated operations, including extended duration of cardiopulmonary bypass, including myocardial ischemia. Longer ICU stay, greater hospital cost, and worse neurodevelopmental outcomes in different studies. Therefore, deciding what reintervention and when to perform it is of paramount importance. This manuscript reviewed residual lesion treated with transcatheter invention rather than redo surgery avoiding the complication related with redo surgery.
However, concerns remained about whether the tissue around the residual shunt was strong enough to hold an occluder in place. Moreover, the dehisced site had been expected to have deficient rims in ASD, and adjacent to leaflet of tricuspid valve or multiple residual lesion in VSD, clips or suture material on the course of residual PDA duct. As such, prepared for open-heart surgery as a backup.
In residual shunt lesion after surgery especially ASD, balloon-sizing technique which is recognized as the gold standard for estimation of defect diameter and device size. Two-dimensional TTE, TEE, and three-dimensional TEE have been shown to be effective and reliable methods for determination of the defect diameter without balloon sizing. Balloon sizing done to all ASD cases and one VSD case in this study.
Residual defects with hemodynamic significance can complicate the postoperative course due to significant heart failure and low cardiac output; moreover, patients with residual VSDs, especially those related to surgical patches, have a greater risk of infective endocarditis. Most residual defects < 2 mm in size will close spontaneously, while those > 2 mm will not.
The Amplatzer Septal Occluder is the most frequently employed device in transcatheter ASD closure procedures. The Amplatzer muscular VSD occluders, perimembranous VSD occluders, ADOs, and septal occluders have previously been used to close VSDs percutaneously. Improper use of adequate size devices may be associated with a high risk of embolization. In clinical practice, a 1–2 mm larger device is essential to guarantee good closure of the VSD. VSDs related to surgical patches have difficulty estimating the actual size due to the complex geometry of residual VSDs.
The potential major complications associated with this procedure include death, device embolization, heart block, new valvular regurgitation, hemolysis requiring blood transfusion, or the need for surgical or transcatheter re-intervention. The rate of major complications ranges from 0% to 8.6%. None of these complications evident in our patients. A complete atrioventricular (AV) block is indeed one of the more serious complications associated with VSD device closure, though it occurs infrequently. The incidence range of 0%–5.7% reflects variability based on factors like patient age, defect size, and device type. For postoperative residual VSD cases, patches and scar tissue from prior surgical repair can create a buffer that reduces direct pressure on the conduction system, thereby potentially lowering the risk of AV block. The presence of these protective structures helps mitigate the mechanical stress the closure device may otherwise exert on the atrioventricular node and surrounding tissue.
A residual patent ductus arteriosus (PDA) following surgical ligation may occur due to incomplete closure or later recanalization of the ductus. Suboptimal occlusion might result from factors like surgical technique, the ductus’s initial size, or tissue characteristics. Recanalization, on the other hand, involves the reopening of the ductus arteriosus after an initially successful closure, potentially due to vessel wall elasticity or insufficient fibrosis. In these cases, residual shunting can persist, and further intervention may be considered to prevent complications such as left heart volume overload or endarteritis. Since residual PDAs are narrow, coil closure is a common practice. Coil closure is best suited to close a PDA with a minimal internal diameter of less than 2.5 mm. In our patient, the size of PDA was 3.5 mm. So, the device compared to coil, was a better choice.
Interventional catheterization has largely replaced surgery for the closure of all types of Patent Ductus Arteriosus (PDA), except in the newborn period. However, percutaneous closure of a residual PDA after surgical ligation can be more challenging due to the aortopulmonary window-like appearance. Additionally, the management of a tiny or “silent” PDA can be controversial. Residual PDAs can indeed pose additional technical challenges during closure because reactive fibrosis can make them less distensible and more rigid than native PDAs. This reduced flexibility can complicate device positioning and secure placement. To address these challenges, interventionalists often tried for a retrograde approach, entering through the aorta, or use a snare technique to improve control and alignment of the closure device. These methods provide better access and stability for device placement, helping to achieve a successful closure despite the more resistant, fibrotic tissue. In this study PDA closed through antegrade approach and it was a residual lesion followed by a PDA clip and there was no procedure related complications.
CONCLUSION
Transcatheter closure is shown to be effective in closing residual defects that were either missed during initial surgery or developed later. Transcatheter closure provides a minimally invasive, effective solution for managing residual heart lesions post-surgery. It offers a significant benefit in reducing the need for additional open-heart surgeries, with improved patient quality of life, reduced post procedure length of stay, adverse events, and cost.
REFERENCES
- Bol-Raap G, Weerheim J, Kappetein AP, Witsenburg M, Bogers AJ. Follow-up after surgical closure of congenital ventricular septal defect. Eur J Cardiothorac Surg. 2003;24:511–5.
- Murphy JG, Gersh BJ, McGoon MD, et al. Long-term outcome after surgical repair of isolated atrial septal defect. Follow-up at 27 to 32 years. N Engl J Med 1990;323:1645–1650
- Sorensen KE, Kristensen B, Hansen OK. Frequency of occurrence of residual ductal flow after surgical ligation by color-flow mapping. Am J Cardiology. 1991;67:653–654.
- Perlof JK, Warnes CA. Challenges posed by adults with repaired congenital heart disease. Circulation 2001;103:2637- 43.
- Gokalp S, Guler Eroglu A, Saltik L, et al. Relationships between left heart chamber dilatation on echocardiography and left-to-right ventricle shunting quantified by cardiac catheterization in children with ventricular septal defects. Pediatr Cardiol 2014;35:691-8.
- Knauth AL, Lock JE, Perry SB, et al. Transcatheter device closure of congenital and postoperative residual ventricular septal defects. Circulation 2004;110:501-7
- Carminati M, Butera G, Chessa M, et al. Transcatheter closure of congenital ventricular septal defects: results of the European Registry. Eur Heart J 2007;28:2361-8.
- Carminati M, Butera G, Chessa M, et al. Transcatheter closure of congenital ventricular septal defect with Amplatzer septal occluders. Am J Cardiol 2005;96:52L-8L.
- Sugita T, Ueda Y, Matsumoto M, et al. Repeated procedure after radical surgery for tetralogy of Fallot. Ann Thorac Surg. 2000 Nov 1;70(5):1507–1510
- Pedra CA, Pontes SC Jr, Pedra SR, Salerno L, Sousa JB, Miaira MA, et al. Percutaneous closure of postoperative and post-traumatic ventricular septal defects. J Invasive Cardiol. 2007;19:491–5.
- Shuhaiber J, Gauvreau K, Thiagarjan R, et al. Congenital heart surgeon’s technical proficiency affects neonatal hospital survival. J Thorac Cardiovasc Surg 2012; 144(5):1119–1124. [PubMed: 22421402] Nathan et al. Page 10 J Am Coll Cardiol. Author manuscript; available in PMC 2022 May 18.
- Karamichalis JM, Thiagarajan RR, Liu H, Mamic P, Gauvreau K, Bacha EA. Stage I Norwood: Optimal Technical Performance improves outcomes irrespective of preoperative physiologic status or case-complexity. J Thorac Cardiovasc Surg 2010;139(4):962–968. [PubMed: 20074754]
- Gupta SK, Sivasankaran S, Bijulal S, Tharakan JM, Harikrishnan S, Ajit K. Trans-catheter closure of atrial septal defect: balloon sizing or no balloon sizing – single centre experience. Ann Pediatr Cardiol 2011;4:28-33.
- Zhou W, Li F, Fu L, et al. Clinical Experience of Transcatheter Closure for Residual Ventricular Septal Defect in Pediatric Patients. Congenit Heart Dis 2016;11:323-31.
- Kouakou NYN, Song J, Huh J, et al. The experience of transcatheter closure of postoperative ventricular septal defect after total correction. J Cardiothorac Surg 2019;14:104.
- Dua JS, Carminati M, Lucente M, et al. Transcatheter closure of postsurgical residual ventricular septal defects: early and mid-term results. Catheter Cardiovasc Interv 2010;75:246-55.
- Dodge-Khatami A, Knirsch W, Tomaske M, Pretre R, Bettex D, Rousson V, et al. Spontaneous closure of small residual ventricular septal defects after surgical repair. Ann Thorac Surg. 2007;83:902–5.
- Paç A, Polat TB, Çetin İ, Oflaz MB, Ballı S. Figulla ASD occluder versus Amplatzer Septal Occluder: a comparative study on validation of a novel device for percutaneous closure of atrial septal defects. J Interv Cardiol 2009;22:489-95.
- Ramakrishnan S, Saxena A, Choudhary SK. Residual VSD closure with an ADO II device in an infant. Congenit Heart Dis. 2011;6:60–3.
- Yin S, Zhu D, Lin K, An Q. Perventricular device closure of congenital ventricular septal defects. J Card Surg. 2014;29:390–400.
- Egbe AC, Poterucha JT, Rihal CS, Taggart NW, Cetta F, Cabalka AK, et al. Transcatheter closure of postmyocardial infarction, iatrogenic, and postoperative ventricular septal defects: the Mayo Clinic experience. Catheter Cardiovasc Interv. 2015;86:1264–70.
- Sudhakar BG. Percutaneous transcatheter closure of a distorted residual patent ductus arteriosus post surgical ligation using retrograde wiring and femoral arteriovenous loop technique-a case report. IHJ Cardiovascular Case Reports (CVCR). 2018 Oct 1;2(3):208–210. https://doi.org/10.1016/j.ihjccr.2018.04.008
- Baspinar Osman, Kilinc Metin, Kervancioglu Mehmet, Indem Ahmet. Transcatheter closure of a residual PDA after surgical ligation in children. Korean Circ. J. 2011;41(Nov (11)):654–657.
- Lloyd TR, Beekman RH 3rd. Clinically silent patent ductus arteriosus. Am Heart J 1994;127:1664-5.
- Gu X, Zhang Q, Sun H, et al. Transcatheter closure versus repeat surgery for the treatment of postoperative left-to-right shunts: A single center 15-year experience. Cardiol Res. 2017 Dec;8(6):286–292. https://doi.org/10.14740/cr629e

