High Frequency Chest Wall Oscillation in COPD Management
Effects of High Frequency Chest Wall Oscillation (HFCWO) on Clinical Symptoms in Chronic Obstructive Lung Disease
Bruner, Meaghan M.¹, Bazan, Clarissa¹, Liu, Bo¹, Cheng, Christina¹, Marion, Chad², Sievert, Chet³, Edwards, Lloyd J¹, Solomon, George M.¹**
- University of Alabama at Birmingham, Birmingham, AL USA.
- Wake Forest University School of Medicine, Winston-Salem, NC USA.
- Clinical Research Associates LLC, Mahtomedi, MN USA.
- Denotes co-senior authorship
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Meaghan M., B., Clarissa, B., et al., 2024. Effects of High Frequency Chest Wall Oscillation (HFCWO) on Clinical Symptoms in Chronic Obstructive Lung Disease. Medical Research Archives, [online] 12(11).
https://doi.org/10.18103/mra.v12i11.5805
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.5805
ISSN 2375-1924
ABSTRACT
Background: Mucociliary clearance plays a critical role in pulmonary host defense. Abnormalities in mucociliary clearance contribute to the pathogenesis of pulmonary disorders, including COPD. In bronchiectasis, treatment options are limited, and patients often experience significant morbidity. High-frequency chest wall oscillation (HFCWO) is a technique designed to enhance mucociliary clearance.
Objective: This study aimed to evaluate the impact of HFCWO on clinical symptoms in patients with COPD as measured by using the St. George’s Respiratory Questionnaire for COPD Patients (SGRQ-C), which is a disease-specific instrument designed to measure the impact on overall health, daily life, and perceived well-being in patients with COPD.
HYPOTHESIS: Based on this background, we hypothesized that HFCWO, when used as an adjunct therapy, would improve clinical symptoms in patients with COPD.
Introduction
The lung is continuously exposed to particles, toxins, and pathogens dealt with by the localized innate system for clearance, a key aspect of host defense. Mucociliary clearance plays a critical role in a multilayered defense system and abnormalities in mucociliary clearance contribute to the pathogenesis of many common pulmonary disorders. In Chronic Obstructive Pulmonary Disease (COPD) mucociliary clearance is impaired by various mechanisms¹. One proposed mechanism responsible for excessive mucus production is the overproduction and hypersecretion by goblet cells²˒³ and mucus hyperviscosity and hyper-concentration⁴. COPD patients also experience difficulty in clearing secretions due to distal airway occlusion, poor ciliary function, and ineffective cough².
Mucus burden is also a major contributing factor in the morbidity of COPD patients. The rate of decline in lung function, the leading predictor of morbidity and mortality for COPD, as well as health care utilization, is higher in patients with mucus hypersecretion⁵˒⁶. One study looked at cases where COPD was an underlying or contributory cause of death and found that patients with COPD and chronic mucus hypersecretion are more likely to die from pulmonary infections compared to patients without chronic mucus hypersecretion⁷. A recent landmark paper examined the effects of mucous plugs on all-cause mortality in patients with COPD and found that patients with mucous plugs occluding medium-to-large sized airways had an increased all-cause mortality⁸. There are various methods to improve mucociliary clearance, however little is known about the effect of mucus-targeted therapies in COPD as many of the approved pharmacologic therapies are primarily anti-inflammatory or bronchodilators. In general, however, recent investigations have found that healthcare providers are aware of methods and agree with a potential benefit in some patients with COPD⁹˒¹⁰. There are several therapies that target elements of impaired mucociliary clearance including goblet cell hyperplasia. These investigational therapies include: bronchial rheoplasty which shows promise to reduce the production of mucus from secretory (goblet cells) in chronic bronchitis and has had early success in clinical trials¹¹˒¹²; metered cryospray and targeted lung denervation¹³˒¹⁴.
In other airway diseases such as CF and non-CF bronchiectasis (NCFB), many treatments target improving mucus obstruction in the airways. Pharmacological interventions include expectorants, mucolytics, and mucokinetics¹⁰˒¹⁵. Non-pharmacological modalities include chest physiotherapy, postural drainage, chest wall percussion and vibration, forced expiration, positive expiratory pressure devices, and high frequency chest wall oscillation (HFCWO)¹⁵. HFCWO therapy is delivered to the patient’s external chest wall via an inflatable vest that imparts oscillatory air pulses that impart compression forces while also generating high-velocity expiratory airflow that are reported to shear and mobilize mucus from the airway walls thereby improving airway mucus clearance¹⁶. HFCWO therapy is often used in conjunction with mucolytics and antibiotics in bronchiectasis and CF patients to improve mucus burden. Several studies in CF patients found HFCWO therapy as effective as or superior to other airway clearance methods¹⁵˒¹⁷. It has been previously shown in a prospective study that in patients with non-cystic fibrosis bronchiectasis (NCFB), the initiation of HFCWO was associated with reductions in patient-reported exacerbation rates, hospitalizations, antibiotic use, and improvements in respiratory symptoms and quality of life¹⁵˒¹⁸.
More recently, patients with COPD have gained reimbursement from Medicare to utilize HFCWO. Although this isn’t yet a guideline-based therapy, HFCWO can be used in addition to other guideline-based therapies for COPD. Given the burden of mucus in many patients with COPD, we hypothesized that HFCWO therapy used regularly
would improve patients’ quality of life. Thereby, we designed this quality of life–based study to investigate the clinical outcomes of using HFCWO as an adjunctive therapy for COPD as measured by using the St. George’s Respiratory Questionnaire for COPD Patients (SGRQ-C) which is a disease-specific instrument designed to measure the impact on overall health, daily life, and perceived well-being in patients with COPD¹⁹. This has been an extensively validated questionnaire and often used to evaluate the advantage of a new treatment for COPD¹⁹˒²⁰.
HYPOTHESIS: Based on this background, we hypothesized that HFCWO, when used according to intended and indications for use will improve the quality of life for those patients with COPD and the symptomatic improvements will be sustained.
Study Design and Methods
STUDY DESIGN
This non-randomized, prospective, open-label, observational study (NCT04271969, Registration Date 18/02/2020). Patients with COPD without bronchiectasis were recruited after HFCWO therapy was prescribed by their attending physician for their clinical condition. Patients were selected by their primary physician for enrollment. Patients were enrolled based on perceived mucus hypersecretion and were felt to be at high risk for exacerbation. The COPD diagnosis was made by the participant’s physician. Patients were excluded that had evidence of bronchiectasis on high-resolution CT scans done within 12 months of the study, based on prescribing physician assessment. The study was done during the Covid-19 pandemic, plus the decentralized method and nature of the study design limited some of the information obtained regarding patients’ COPD diagnosis.
To achieve enrollment, a list comprising the continental United States was collected from the study device (SmartVest) manufacturer. The list of patients was reviewed by study-trained Respiratory Therapists (RT) from Electromed, Inc and an independent clinical research associate and from an independent clinical research organization firm to minimize bias. The CRA determined if the participants met study inclusion/exclusion criteria and were willing to participate in the study. Other inclusion criteria included a confirmed COPD diagnosis accompanied by a SmartVest prescription in patients at least 18 years old. Exclusion criteria excluded those currently using HFCWO therapy and aforementioned physician assessment of concomitant bronchiectasis. If enrollment criteria were met, the Electromed RT delivered the device, performed training and obtained an informed consent and the patient was enrolled as a participant for phone calls by the independent CRA. Participants were asked to continue their baseline concomitant medications related to COPD throughout the study.
STUDY ENDPOINTS
The SGRQ-C questionnaire was administered by phone by the CRA according to SGRQ-C guided instructions with each subject. SGRQ-C responses were recorded at baseline (day 0), day 30, and day 90 (final follow-up). The primary efficacy outcome(s) are changes from baseline to days 30 and 90 in the SGRQ-C total score.
DATABASE MANAGEMENT
Clinical Research Associates validated all study data to ensure data integrity and accuracy. All subjects’ deidentified and validated data were then transferred via a secure transfer protocol process to the lead investigator for analysis and review. Data included demographics and SGRQ-C responses at each time point (baseline, 30, and 90 days). All data were converted to quantitative analysis at each timepoint using a Redcap-based scoring algorithm for SGRQ-C for both total and individual domain scores. The scoring algorithm provided by SGRQ-C was used to calculate questionnaire responses.
STATISTICAL ANALYSIS
Descriptive statistics were computed for all study variables at each time point and aggregated across time points.
The primary efficacy endpoints and comparisons were changes from baseline to days 30 and 90 days for the quality-of-life questionnaire SGRQ-C (continuous measures). A paired t-test was used as primary efficacy analysis to assess changes from baseline for each of the outcomes. Dunnett’s multiple comparisons test was assessed to compare paired group means of SGRQ-C to baseline results at each timepoint.
To adjust for select independent predictors (age, gender, and others), multi-variable linear regression was used with day 30 or day 90 as the dependent variable and baseline and selected independent variables as predictors.
A linear mixed model (LMM) for the analysis of longitudinal data was used as a secondary analysis method to model continuous outcomes at baseline (day 0), day 30, and day 90 while simultaneously adjusting for gender, age, race, GOLD stage (i.e. E or B versus A), time, gender × time, and race × time variables. The models were used to assess longitudinal changes across time and allows adjusting for correlations between the repeated observations while adjusting for fixed and random effects. The fixed effects consisted of gender, age, race, GOLD stage, time, gender × time, and race × time. A random intercept was used to represent the subject-specific effect. The model building process was assessed in a 2-step fashion as outlined below.
-
Are there differences in slopes with respect to time?
To answer this question a “full model’’ with gender, age, race, GOLD stage, time, gender × time, race × time was used. -
Then non-significant interaction terms were removed and a “reduced model’’ was used to assess main effects of gender, age, race, GOLD stage and time.
Finally, to assess the effect of gender and racial differences compared to baseline, a two-way ANOVA was analyzed with multiple comparisons test analyzed for statistical significance for gender differences (Tukey’s multiple comparisons test) and race (Dunnett’s Multiple Comparison test) at each time point compared to baseline.
ETHICS APPROVALS
The study’s sponsor received a written opinion from Western Institutional Review Board’s (WIRB) IRB Affairs Department that the study’s design and protocol requirements, “meets the conditions for exemption under 45 CFR 46.101(b)(4)”. WIRB’s conditions were that, “all of the data are in existence and the information will be recorded in such a manner that the subjects cannot be identified, directly or through identifiers linked to subjects and, that the results of the study will not be submitted to the FDA for marketing approval. Informed consent for participation was obtained from all patients. The protocol was also reviewed for reliance with the Institutional Review Board at the University of Alabama at Birmingham.
PERMISSIONS
A copyright license was obtained for the usage of the SGRQ-C questionnaire and its calculator scoring algorithm from the copyright owners at St. George’s University of London.
Results
DEMOGRAPHICS
We enrolled 102 subjects in the study. Baseline demographic data are summarized in Table 1. The Table indicates a representative distribution of age, gender, racial and ethnic variation amongst the overall cohort and indicates that appropriate sampling in this cohort was achieved to represent the usual COPD population. In addition, the patient population is mostly patients with advanced stage COPD (majority GOLD B or E) and many required oxygen therapy. As expected most patients were former or current smokers. Most patients (>88%) reported a singular pulmonary diagnosis of COPD only. The remainder reported physician diagnosis of asthma, former NTM infection, or “other dyspnea”. 100% of patients carried a diagnosis of COPD and no patients carried a physician assessed diagnosis of bronchiectasis based on CT scans reviewed by referring physicians before referral.
METHODS
ETHICS APPROVALS
The study protocol received a written approval from Western Institutional Review Board’s (WIRB) IRB Affairs Department that the study’s design and protocol requirements, “meets the conditions for exemption under 45 CFR 46.101(b)(4).” WIRB’s conditions were that, “All of the data are in existence and the information will be recorded in such a manner that the subjects will not be identified directly or through identifiers linked to them.” The protocol was also reviewed and approved by the FDA for marketing approval. Patients bseline symptoms were recorded on the initial questionnaire and 77% of the patients reported cough most days of the week, 66% reported increased phlegm most days of the week, 74% reported having shortness of breath most days of the week, and 47% reported wheezing most days of the week. This indicates a relatively significant symptom burden amongst the study population prior to starting HFCWO therapy. Concomitant medication at baseline are summarized in Supplemental Table 1. Most patients were not using mucolytic or airway clearance methods at baseline. However, the majority of patients were on guideline-based therapies for advanced stage COPD. Compliance estimates indicate >90% compliance with the device.
CHANGES IN QOL (OVERALL COHORT)
The primary endpoint of the study was the change in SGRQ-C score compared to baseline (72.84 [69.25, 76.43 95% CI]). The overall cohort of patients demonstrated a significant reduction in the SGRQ-C at 30 days (65.90 [62.13, 69.67 95% CI]) that was sustained at 90 days (63.26 [59.36, 67.16 95% CI]) compared to baseline (Fig. 1A). In addition, individual components of the SGRQ-C i.e., Symptoms, Activity and Impacts were analyzed (Table 3) and the changes were plotted indicating significant reductions at 90 days in the “Symptoms’’ and “Impacts’’ components compared to baseline (Fig. 1 B-D).
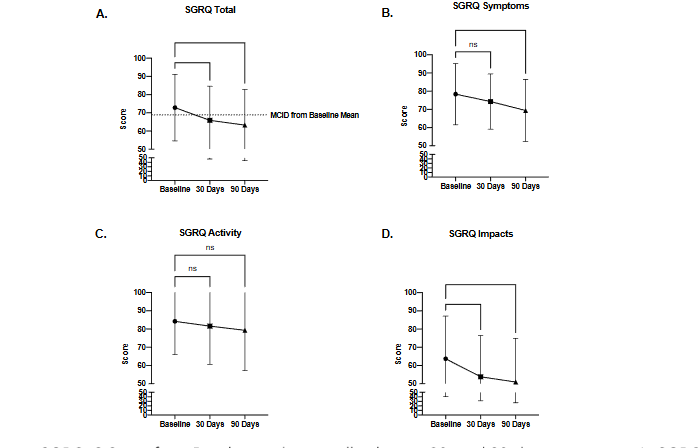
Figure 1. Change in SGRQ-C Score from Baseline in the overall cohort at 30- and 90-day timepoints. A. SGRQ “Total” Score change. Dashed line indicates MCID for this cohort from baseline (based on MCID of -4 points). Scoring of individual score components of the SGRQ-C was also analyzed for changes in B. SGRQ “Symptoms” score, C. Change in SGRQ “Activity” score, and D. Change in SGRQ “Impacts” Score. *p<0.05, **p<0.01, ***p<0.001 by one-way ANOVA with Dunnett’s multiple comparisons tests.
Table 3: SGRQ-C Total and Sub-Scores
| Baseline (N=102) | 30 days (N=97) | 90 days (N=100) | |
|---|---|---|---|
| SGRQ Total | 72.84 [69.25, 76.43 95% CI] | 65.90 [62.13, 69.67 95% CI] | 63.26 [59.36, 67.16 95% CI] |
| SGRQ Symptoms | 78.38 [75.05, 87.71 95% CI] | 74.31 [71.27, 77.35 95% CI] | 69.41 [66.05, 72.77 95% CI] |
| SGRQ Impacts | 63.67 [59.04, 68.30 95% CI] | 53.79 [49.33, 58.25 95% CI] | 50.99 [46.29, 55.69 95% CI] |
| SGRQ Activity | 84.19 [80.53, 87.85 95% CI] | 81.60 [77.47, 85.73 95% CI] | 79.29 [74.91, 83.67 95% CI] |
GENDER OR RACE EFFECTS IN CHANGES IN QOL
Since the overall cohort demonstrated clinically and statistically significant improvements in QOL based on the SGRQ-C at 30 and 90 days, we sought to determine if race and/or gender were independent factors in the changes observed.
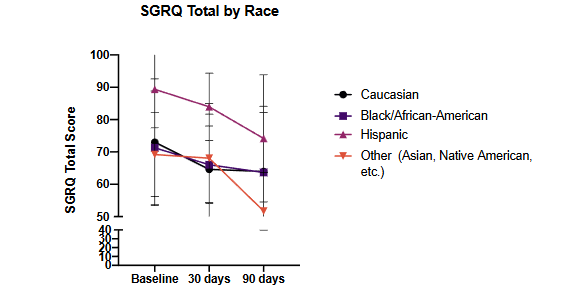
We first assorted the overall cohort based on self-reported race and ethnic classification and analyzed the differences in baseline and 30- and 90-day QOL timepoints as summarized in Figure 2.
Finally, after removing interaction effects, we used a reduced multivariable model to assess the main effects of age, race/ethnicity, gender, and time on therapy on change in QOL assessments from baseline to 90 days. When the models were reduced, the only significant variable was “time on therapy’’ further indicating that the improvement of SGRQ-C Total, Activity, and Impact Scores, from baseline to 90 days is not based on gender, age, or racial effects in the cohort. For SGRQ-C Symptom Scores, both time and age were significant. These findings are summarized in Table 2 for the “Total” Score and for sub-scores in the data supplement (Supplementary tables 2–5).
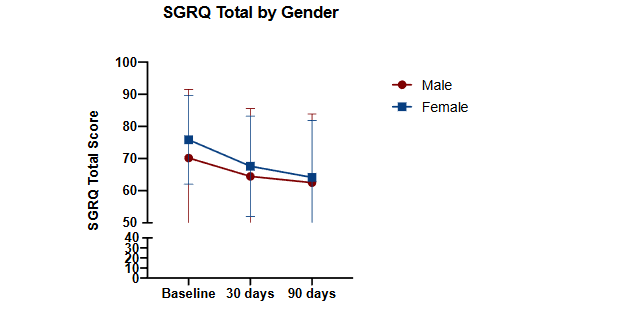
Notably, GOLD stage (B or E versus A) did not have an effect in the overall LMM on total SGRQ-C score at any timepoint. We then assessed the mean changes in SGRQ-C scores across the 30- and 90-day timepoints. Notably, no significant differences were seen in changes of QOL scores when comparing self-reported “female” vs “male” gender. No participants reported “non-binary” or “no gender”. This data is summarized in Figure 3.
To further confirm there is no interaction of gender, age or ethnicity and time, we analyzed the longitudinal outcomes to assess the effects of gender, age, race/ethnicity, time, gender × time, and race × time interactions in the observed QOL improvements in this cohort. were any differences in slopes in change of QOL data with respect to time. To answer that a “full model’’ multivariable model with gender, age, race, time, gender × time, race × time were analyzed. The interaction terms were not significant for any of the outcomes in the SGRQ-C “Total” or sub-scores (see Figures 2–3 and Table 2).
Figure 2: Assessment of SGRQ-C Total score at 30- and 90-day time points compared to baseline assorted on the basis of self-reported race and ethnicity
Figure 2. Assessment of SGRQ-C Total score at 30- and 90-day timepoints compared to baseline assorted on the basis of self-reported race and ethnicity. p = NS for all comparisons of racial and ethnic differences in change of score compared to baseline, despite the relatively higher baseline values for the self-reported “Hispanic’’ cohort ethnicity. All p-values assessed by 2-way ANOVA with Dunnett’s Multiple Comparisons test.
Figure 3: Assessment of SGRQ-C total score at 30- and 90-day time points compared to baseline assorted on self-reported gender classification
Figure 3. Assessment of SGRQ-C total score at 30- and 90-day timepoints compared to baseline assorted based on self-reported gender classification. p = NS for all comparisons of gender difference in change of score compared to baseline. All p-values assessed by 2-way ANOVA with Tukey’s Multiple Comparisons test.
Table 2: SGRQ-C total score final LMM results
| Effect | Estimate | Standard Error | DF | t Value | Pr > |t| |
|---|---|---|---|---|---|
| Intercept | 95.8389 | 13.7954 | 101 | 6.95 | <.0001 |
| Gender | 2.1524 | 3.5178 | 98.6 | 0.61 | 0.5421 |
| Age | -0.3440 | 0.1832 | 102 | -1.88 | 0.0633 |
| Race | -0.4104 | 1.9891 | 251 | -0.21 | 0.8367 |
| GOLD Stage | -0.3319 | 2.1354 | 102 | -0.32 | 0.5698 |
| Time | -0.1037 | 0.01510 | 196 | -6.87 | <.0001 |
Discussion
This open-label observational study demonstrates a significant improvement in QOL based on SGRQ-C scoring after using HFCWO as an adjunctive therapy for patients with a primary diagnosis of COPD without a physician diagnosis of concomitant bronchiectasis. Prior data have reported significant efficacy in improvements in lung function and inflammatory cells but have not explicitly examined the effect of HFCWO on quality of life in COPD²¹. In the same study, only a small cohort of patients had analysis of effects of HRQOL (CAT scores) when using HFCWO²¹. A recent meta-analysis concluded that prior investigation of HFCWO was limited to conclude no significant effect on outcomes aside from the volume of sputum production and found no definitive efficacy in lung function or QOL²². Subsequently, a randomized controlled trial investigated sixty patients with one arm on HFCWO and showed improvements in FEV₁²³.
Our study addresses these limitations in complete assessment of the effect of HFCWO on QOL. We demonstrate significant improvements in several component scores of the SGRQ-C including the “Impacts” and “Symptoms” score both of which were clinically significant (>MCID). This indicates that the improvement in quality of life is broader and may suggest benefits beyond clearance of pathological mucus alone, although this cannot be confirmed in this limited trial due to confounding.
Thus this study adds to the literature the finding of initial improvement of QOL using HFCWO in patients broadly with COPD in a real-world pragmatic setting.
Furthermore, the study indicates that the initial effect of QOL was sustained at 90 days. This suggests meaningful longer-term response to HFCWO therapy. However, due to the limited study duration, we cannot conclude that HFCWO will have longer term effects. Furthermore, the open-label nature of the study creates some biases towards positive clinical responses, which cannot be controlled for in the analysis nor accounted for in the confounding. The controlled nature and patient characterization does indicate that the sustainment of symptom improvement is evidence that the treatment should be analyzed further.
Our study further demonstrates that the initial and longer-term improvements in QOL were observed in patients in all self-reported racial/ethnic groups and when divided based on self-reported gender. While we cannot definitively conclude that there are no racial, ethnic, or gender differences in response to HFCWO, this generates an intriguing hypothesis that this therapy may universally improve QOL in COPD patients. Finally, multivariable regression modelling of the data suggests that time on therapy is the key variable for improvements in QOL, while also supporting the hypothesis that HFCWO therapy may work across a large subset of patients. Furthermore these responses were not unique to patients with more severe GOLD stage (B or E). This contrasts
with other recent therapies with more limited indications such as macrolides, frequent exacerbators, or those with the chronic bronchitis phenotype²⁴˒²⁵.
Mucus clearance is a critical and protective function of the airways and lungs. Failure of this function results in repeated lung infections and eventual respiratory insufficiency leading to increased morbidity and mortality²⁶. COPD is the third leading cause of death in the United States and is increasing in incidence worldwide, therefore finding effective therapies targeted at improving mucus obstruction is vital to improving COPD morbidity and mortality²⁷. Improving quality of life for the COPD patients has great implications. Unfortunately, COPD symptoms reduce the patient’s ability to perform activities of daily living potentially causing personal relationships and activities to become problematic²⁸.
While this study was intended to serve as a pragmatic study that recruited subjects who had a previous COPD diagnosis and were prescribed HFCWO airway clearance therapy, we again acknowledge several limitations in the study. First, the study was conducted as a small, open-label study. Therefore, we cannot definitively assess whether HFCWO initiation was the only variable resulting in improvements in QOL scores nor account for bias in a subjective patient reported outcome measure. Furthermore, baseline exacerbation rate was not assessed so we cannot use this data to further assess efficacy. In addition, variations in the timing of other therapies was not accounted for in this study, but patients were asked to continue their baseline regimen of inhaled medications and other mucus clearance therapy. However, the symptoms burden was relatively high in many patients as evidenced by the mean baseline “Total’’ and “Symptoms’’ score. Therefore, we can reasonably conclude that many patients had productive cough and other symptoms that may be associated with mucus hypersecretion.
Future studies should randomize patients in a larger cohort to determine if HFCWO initiation is definitively associated with improved QOL. This study design would be aided by capturing baseline exacerbation rates to assess this key endpoint in trials aimed at improving mucus clearance.
Interpretation
This open-label observational study demonstrated initial and sustained improvements in quality of life when HFCWO was used as add-on therapy for patients with a primary diagnosis of COPD without concomitant bronchiectasis. Further randomized trials are necessary to assess the full effect of HFCWO on symptoms and outcomes in COPD.
Take home points
Use of HFCWO vest therapy as add-on therapy for patients with a primary diagnosis of COPD without concomitant bronchiectasis may improve QOL and is worthy of future studies to confirm efficacy on QOL.
Conflict of Interest:
No conflicts of interest.
Funding Statement:
This study was funded by the NIH (P30 DK072482-14 to GMS), (1K08HL138153-01A1 to GMS) and electromed inc. to GMS.
Acknowledgements:
We thank the patient and their families for their participation in the trials.
Notation of prior abstract presentation:
This data was presented at the American Thoracic Society Conference, May 2023, Washington D.C.
Author contributions:
GMS, CS, LJE developed the study design.
GMS, CS, MMB, BL participated in experimental conduct and acquisition of data.
MMB, CB, BL, CC, LJE, CM, and GMS performed data analysis.
MMB and GMS prepared the manuscript.
All authors provided necessary revisions and approved a final draft prior to submission.
Clinical trial registration:
NCT04271969, Registration Date 18/02/2020
Declaration:
ETHICS APPROVALS
The study’s sponsor received a written opinion from Western Institutional Review Board’s (WIRB) IRB Affairs Department that the study’s design and protocol requirements, “meets the conditions for exemption under 45 CFR 46.101(b)(4)”. WIRB’s conditions were that, “all of the data are in existence and the information will be recorded in such a manner that the subjects cannot be identified, directly or through identifiers linked to subjects and, that the results of the study will not be submitted to the FDA for marketing approval. Informed consent for participation was obtained from all patients. The protocol was also reviewed for reliance with the Institutional Review Board at the University of Alabama at Birmingham.
Participants all provided consent to participate and consent to publish.
Consent to Publication:
Not applicable
Data Availability
All data is housed in a locked database by the principal investigator. Data will be made available upon written request to the corresponding author.
Permissions
A copyright license was obtained for the usage of the SGRQ-C questionnaire and its calculator/scoring algorithm from the copyright owners at St. George’s University of London.
Abbreviations:
-
COPD – Chronic Obstructive Pulmonary Disease
-
CF – Cystic Fibrosis
-
HFCWO – High Frequency Chest Wall Oscillation
-
QOL – Quality of Life
-
SGRQ-C – The St. George’s Respiratory Questionnaire for COPD Patients
References
2. Ramos FL, Krahnke JS, Kim V. Clinical issues of mucus accumulation in COPD. International Journal of Chronic Obstructive Pulmonary Disease. 2014;9:139-150.
3. Cerveri I, Brusasco V. Revisited role for mucus hypersecretion in the pathogenesis of COPD. European Respiratory Review. 2010;19(116):109.
4. Button B, Anderson WH, Boucher RC. Mucus Hyperconcentration as a Unifying Aspect of the Chronic Bronchitic Phenotype. Ann Am Thorac Soc. 2016;13 Suppl 2(Suppl 2):S156-162.
5. Chakravorty I, Chahal K, Austin G. A pilot study of the impact of high-frequency chest wall oscillation in chronic obstructive pulmonary disease patients with mucus hypersecretion. Int J Chron Obstruct Pulmon Dis. 2011;6:693-699.
6. Holland AE, Button BM. Is there a role for airway clearance techniques in chronic obstructive pulmonary disease? Chron Respir Dis. 2006;3(2):83-91.
7. Prescott E, Lange P, Vestbo J. Chronic mucus hypersecretion in COPD and death from pulmonary infection. European Respiratory Journal. 1995;8(8):1333.
8. Diaz AA, Orejas JL, Grumley S, et al. Airway-Occluding Mucus Plugs and Mortality in Patients With Chronic Obstructive Pulmonary Disease. JAMA. 2023;329(21):1832-1839.
9. Alghamdi SM, Alzahrani A, Alshahrani YM, et al. Perception and clinical practice regarding mucus clearance devices with chronic obstructive pulmonary disease: a cross-sectional study of healthcare providers in Saudi Arabia. BMJ Open. 2023;13(9):e074849.
10. Yan X, Song Y, Shen C, et al. Mucoactive and antioxidant medicines for COPD: consensus of a group of Chinese pulmonary physicians. Int J Chron Obstruct Pulmon Dis. 2017;12:803-812.
11. Valipour A, Fernandez-Bussy S, Ing AJ, et al. Bronchial Rheoplasty for Treatment of Chronic Bronchitis. Twelve-Month Results from a Multicenter Clinical Trial. Am J Respir Crit Care Med. 2020;202(5):681-689.
12. Sciurba FC, Dransfield MT, Kim V, et al. Bronchial rheoplasty for chronic bronchitis: 2-year results from a US feasibility study with RheOx. BMJ Open Respir Res. 2023;10(1).
13. Hartman JE, Garner JL, Shah PL, Slebos D-J. New bronchoscopic treatment modalities for patients with chronic bronchitis. European Respiratory Review. 2021;30(159):200281.
14. Pison C, Shah PL, Slebos DJ, et al. Safety of denervation following targeted lung denervation therapy for COPD: AIRFLOW-1 3-year outcomes. Respir Res. 2021;22(1):62.
15. Barto TL, Maselli DJ, Daignault S, et al. Real-life experience with high-frequency chest wall oscillation vest therapy in adults with non-cystic fibrosis bronchiectasis. Ther Adv Respir Dis. 2020;14:1753466620932508.
16. Leemans G, Belmans D, Van Holsbeke C, et al. The effectiveness of a mobile high-frequency chest wall oscillation (HFCWO) device for airway clearance. Pediatr Pulmonol. 2020;55(8):1984-1992.
17. Nicolini A, Cardini F, Landucci N, Lanata S, Ferrari-Bravo M, Barlascini C. Effectiveness of treatment with high-frequency chest wall oscillation in patients with bronchiectasis. BMC Pulmonary Medicine. 2013;13(1):21.
18. Powner J, Nesmith A, Kirkpatrick DP, Nichols JK, Bermingham B, Solomon GM. Employment of an algorithm of care including chest physiotherapy results in reduced hospitalizations and stability of lung function in bronchiectasis. BMC Pulm Med. 2019;19(1):82.
19. Jones PW. St. George’s Respiratory Questionnaire: MCID. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2005;2(1):75-79.
20. Alma H, de Jong C, Jelusic D, et al. Baseline health status and setting impacted minimal clinically important differences in COPD: an exploratory study. J Clin Epidemiol. 2019;116:49-61.
21. Nicolini A, Grecchi B, Ferrari-Bravo M, Barlascini C. Safety and effectiveness of the high-frequency chest wall oscillation vs intrapulmonary percussive ventilation in patients with severe COPD. Int J Chron Obstruct Pulmon Dis. 2018;13 :617-625.
22. Huang HP, Chen KH, Tsai CL, et al. Effects of High-Frequency Chest Wall Oscillation on Acute Exacerbation of Chronic Obstructive Pulmonary Disease: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Int J Chron Obstruct Pulmon Dis. 2022;17:2857-2869.
23. Pestelli MT, Grecchi B, Vottero E, et al. Effectiveness of a new “focused pulse” high-frequency chest wall oscillation in patients with moderate to severe COPD. Minerva Med. 2024; 115(1):4-13.
24. Cui Y, Luo L, Li C, Chen P, Chen Y. Long-term macrolide treatment for the prevention of acute exacerbations in COPD: a systematic review and meta-analysis. Int J Chron Obstruct Pulmon Dis. 2018;13:3813-3829.
25. Albert RK, Connett J, Bailey WC, et al. Azithromycin for Prevention of Exacerbations of COPD. New England Journal of Medicine. 2011;365(8):689-698.
26. Livraghi A, Randell SH. Cystic fibrosis and other respiratory diseases of impaired mucus clearance. Toxicol Pathol. 2007;35(1):116-129.
27. Lin VY, Kaza N, Birket SE, et al. Excess mucus viscosity and airway dehydration impact COPD airway clearance. European Respiratory Journal. 2020;55(1):1900419.
28. Duiverman ML, Wempe JB, Bladder G, Kerstjens HAM, Wijkstra PJ. Health-related quality of life in COPD patients with chronic respiratory failure. European Respiratory Journal. 2008;32(2):379.