Leishmania’s Arginine Sensing in Host Macrophages
Leishmania parasite arginine deprivation response pathway influences the host macrophage lysosomal arginine sensing machinery
Evanka Madan¹², Nirmal Kumar Ganguly², Madhu Puri¹, Harsh Pawar³, Angamuthu Selvapandiyan⁴, Rohini Muthuswami⁵, Dan Zilberstein⁶, Rentala Madhubala¹
- Molecular Parasitology Laboratory, School of Life Sciences, Jawaharlal Nehru University, New Delhi-110067, India
- Department of Biotechnology and Research, Sir Ganga Ram Hospital, New Delhi-110060, India
- Faculty of Biology, Technion-Israel Institute of Technology, Haifa – 3200003, Israel
- Department of Molecular Medicine, Jamia Hamdard, New Delhi 110062
- Chromatin Remodelling Laboratory, School of Life Sciences, Jawaharlal Nehru University, New Delhi-110067, India
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Madan, E., Ganguly, N.K., et al., 2024. Leishmania parasite arginine deprivation response pathway influences the host macrophage lysosomal arginine sensing machinery. Medical Research Archives, [online] 12(10).
https://doi.org/10.18103/mra.v12i10.5800
COPYRIGHT: © 2024 European Society of Medicine. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5800
ISSN 2375-1924
Abstract
The outcome of infection is greatly influenced by extensive interaction between the metabolic networks of the host and the pathogen. The intracellular protozoan parasite Leishmania donovani causes competition between the host and the parasite for arginine. L. donovani transports arginine via a high-affinity transporter LdAAP3, encoded by two genes LdAAP3.1 and LdAAP3.2. Earlier reports show that upon arginine starvation, cultured Leishmania parasites promptly activate an Arginine Deprivation Response (ADR) pathway, resulting in the stoichiometric up-regulation of LdAAP3.2 mRNA, protein and activity. Lysosomes, on the other hand, employ a specific sensor and an arginine-activated amino acid transporter, solute carrier family 38 member 9 (SLC38A9), which monitors intra-lysosome arginine sufficiency and subsequently up-regulates cellular mTOR kinase activity. The present study investigates the interaction between Leishmania and macrophage-lysosome arginine sensing machinery. We demonstrate that infection with L. donovani activates SLC38A9 arginine sensing in the human monocyte like-macrophage cell line (THP-1) under physiological concentrations of arginine (0.1 mM). However, THP-1 cells infected with LdAAP3.2 null mutants grown in 0.1 mM arginine exhibited reduced expression of SLC38A9 and mTOR. These results indicate that inside the host macrophage, Leishmania overcome low arginine levels by up regulating the transport of arginine via LdAAP3 and SLC38A9 signalling. Furthermore, while LdAAP3.2 null mutants were impaired in their ability to develop inside THP-1 macrophage, their infectivity and intracellular growth were restored in SLC38A9 silenced macrophages. This study provides the first identification of regulatory role of SLC38A9 in the expression and function of LdAAP3.
Keywords
- Arginine sensing
- Leishmania donovani
- THP-1
- AAP3
- SLC38A9
- mTOR
Introduction
The majority of metabolites are chemically similar or identical between the host and the pathogen; therefore, the crosstalk between their amino acid (AA) metabolic pathways is of paramount importance during infection. The host is dependent on AA metabolism to support defense against pathogens, while the pathogen is known to modulate AA metabolism to suit its own needs. Arginine is a versatile amino acid. It is semi-essential in mammals as it is endogenously synthesized and used for the synthesis of proteins, polyamines, urea, nitric oxide, etc. Leishmania cannot synthesize arginine de novo and is dependent on uptake from macrophages. It is required for the synthesis of polyamines and trypanothione. During infection, Leishmania encounters macrophage defense mechanisms designed to prevent parasite invasion and block their intracellular survival, including the release of reactive oxygen species (ROS) and synthesis of cytotoxic nitric oxide (NO) by nitric oxide synthase (iNOS). However, for macrophages to produce adequate amounts of NO, they must import arginine from the extracellular environment. Leishmania infection promotes the expression of arginine transporters in macrophages, including the cationic acid transporter member CAT-2 (SLC7A2). Invading promastigotes activate macrophage arginase I, thereby restricting the amount of arginine available for NO production. The parasite imports exogenous arginine via a mono-specific amino acid transporter (LdAAP3). Thus, reduction of the host arginine pool becomes a bottleneck for Leishmania in infected macrophages, as they are locked in competition with the host for access to the diminishing arginine supply. Leishmania parasites need the means to sense and respond to changes in arginine availability to be able to play the arginine hunger game successfully.
The lysosome is recognized as an essential intracellular organelle involved in the AA-sensing response mechanism of the mTORC1 pathway. Mammalian cells express four Rag GTPases that form heterodimers, consisting of RagA or RagB in partnership with RagC or RagD. The amino acid-responsive super-complex that includes the pentameric Ragulator complex and the multisubunit Vacuolar ATPase (vATPase) complex promotes the accumulation of Rag heterodimers on the cytoplasmic surface of the lysosome. When Rag heterodimer is active it recruits mTOR to the lysosomal membrane, thereby initiating the activation process of mTOR. The lysosomal membrane represents the site at which the amino acid sensing machinery converges to stimulate mTOR. Solute Carrier family 38 member 9 (SLC38A9), a lysosomal arginine transporter and a sensor, is a 110 amino acid-long, soluble, cytosolic N-terminal domain responsible for its interaction with Rag-Ragulator. Two Rag GTPases and a vacuolar ATPase, along with SLC38A9, transmit information about both cytosolic and lysosomal arginine content by phosphorylating mTOR kinase that subsequently activates a downstream pathway involving p-P70S6K1. Thus, SLC38A9, together with a complex of additional proteins, signals mTOR on arginine sufficiency in the lysosome lumen.
We have previously reported that the Leishmania donovani arginine sensor responds to arginine deprivation by activating a MAPK2-mediated signalling pathway, known as the arginine deprivation response (ADR), resulting in the up-regulation of expression and activity of its amino acid transporter 3 (LdAAP3). Significantly, ADR is also activated during macrophage infection, indicating a role for intra-phagolysosomal arginine sensing in intracellular (amastigote) parasite survival and pathogenesis. In the present study, we have investigated the involvement of the host arginine sensing pathway by which L. donovani can gain access to the macrophage L-arginine pool. Although SLC38A9 and LdAAP3 have emerged as two independent lysosomal arginine transporters, a regulatory link between SLC38A9 and LdAAP3 remains unknown. In the present study, we identified a critical SLC38A9 pathway that is modulated after L. donovani infection. Subsequently, intracellular L. donovani derived from infected THP-1 macrophages whose SLC38A9 expression was silenced maintained in physiological arginine content resulted in reduced expression of LdAAP3. Similarly, we observed that infection of THP-1 cells with LdAAP3.2 null mutants in 0.1 mM arginine resulted in reduced expression of SLC38A9. Therefore, as there exists a positive regulatory loop between LdAAP3 and SLC38A9, high SLC38A9 would lead to further increase in LdAAP3 expression thereby favoring arginine transport inside the parasite. This study identifies the novel interplay of L. donovani and macrophage-lysosome arginine sensing machinery as a means to better understand host-parasite interaction.
Methods
MATERIALS
THP-1 cell line was purchased from NCCS, Pune, India. L-arginine was purchased from Sigma-Aldrich (St. Louis, MO, USA). Fetal bovine serum (FBS), OptiMEM and Lipofectamine 3000 reagent were purchased from Invitrogen, USA. siRNAs for SLC38A9 (L-007337-02-0005) and Control (D-001810-10-05) were purchased from Dharmacon, USA. PCR primers were designed by Eurofins, USA. Antibodies were purchased from the following companies: SLC38A9 (NBP1-69235) from Novus (Littleton, CO, USA); RagA (4357S), mTOR (2983S) and p-P70S6K1 (Ser389) (9206S) from Cell Signaling Technology (Beverly, MA, USA); β-actin (A1978) from Sigma (Sigma-Aldrich, St Louis, MO, USA); HRP-conjugated secondary antibodies from Cell Signaling Technology (Beverly, MA, USA).
LEISHMANIA CELL CULTURE
L. donovani Bob strain (LdBob strain/MHOM/SD/62/1SCL2D) (WT-L. donovani) promastigotes were cultured at 26°C in M199 medium (Sigma-Aldrich, USA), supplemented with 100 units/ml penicillin (Sigma-Aldrich, USA), 100 µg/ml streptomycin (Sigma-Aldrich, USA) and 10% heat-inactivated fetal bovine serum (FBS; Biowest). CRISPR Cas9 mediated AAP3 gene deleted clones of L. donovani that abundantly produce AAP3 protein (Δap3G3-17) and another clone with no increase of AAP3 protein (Δap3D10) were obtained from Prof. Zilberstein group (Technion-Israel Institute of Technology, Israel) and used in the studies.
THP-1 CELL CULTURE AND INFECTION
THP-1 cells, an acute monocytic leukaemia-derived human cell line (202 TM; American Type Culture Collection, Rockville, MD), were maintained in RPMI-1640 (Sigma-Aldrich, USA) medium supplemented with 10% heat-inactivated FBS (Biowest, UK), 100 units/ml penicillin and 100 μg/ml streptomycin at 37°C and 5% CO2. Cells (106 cells/well) were treated with 50 ng/ml phorbol-12-myristate-13-acetate (PMA) (Sigma-Aldrich, USA) for 48 h to induce differentiation into macrophage-like-cells before infection. Cells were washed once with phosphate-buffered saline (PBS) and incubated in RPMI medium (Sigma-Aldrich, USA) containing 0.1 mM arginine, 10% heat-inactivated FBS, 100 units/ml penicillin and 100 μg/ml streptomycin, before infection. To carry out in vitro infection assays, late stationary phase promastigotes (WT and ADR mutants) were used at a ratio of 20 parasites per macrophage. Leishmania-infected macrophages were incubated at 37°C in a 5% CO2-air atmosphere for 4 h to allow the establishment of infection and proliferation of intra-macrophage parasites. The cells were then washed five times with PBS to remove non-adherent extracellular parasites. After that, the cells were incubated in RPMI medium containing 0.1 mM arginine at 37°C in a 5% CO2-air atmosphere for 2 h, 24 h and 48 h.
MACROPHAGE PHAGOSOME ISOLATION
THP-1 cells (3.6 × 106 cells/flask) were cultured in DMEM medium containing 10% FBS. Magnetic beads of 3 μm size were then added to flasks containing THP-1 cells. The isolation of intact macrophage phagosomes was carried out as described previously by Kuhnel et al.
INFECTIVITY ASSAY
THP-1 cells (1 × 106 cells/well), treated with 50 ng/ml of PMA (Sigma-Aldrich, USA) and were seeded on glass coverslips in 6-well plates for 48 h. They were infected and simultaneously transfected with siRNAs as described below, and the intracellular parasite load was visualized by Giemsa staining as described previously by Pawar et al. At least 10 fields were counted manually for each condition to determine the average number of parasites per macrophage.
SMALL INTERFERENCE RNA (siRNA) TRANSFECTION
Macrophage-like THP-1 cells in the exponential phase of growth were plated in a 6-well plate at a density of 1 × 106 cells/well and were cultured overnight. They were then transiently transfected with 60 nM of siSLC38A9 (SMARTpool: ON-TARGETplus SLC38A9 siRNA; Dharmacon, USA) or a non-targeting 60 nM of siControl (ON-TARGETplus Non-targeting Pool; Dharmacon, USA), according to the manufacturer’s protocol. Lipofectamine3000 (Invitrogen, USA) in RPMI media (w/o FBS) (Invitrogen, USA) was used for transfection: 500 μl was added to each well and incubated for 24 h. Then, the supernatant was aspirated, and cells were further incubated in fresh complete growth medium for 12–15 h before they were infected with L. donovani in RPMI medium containing 0.1 mM arginine for 48 h. Confirmation of siRNA-mediated down-regulation of the target gene was evaluated by qPCR and Western blot analysis.
qRT-PCR FOR GENE EXPRESSION ANALYSIS
Total RNA from infected macrophages was isolated using the TRIZOL reagent (Sigma-Aldrich, USA) and its concentration was determined by Nanodrop (Thermo Fischer, USA). cDNA was prepared from two micrograms of RNase-free DNase treated total RNA using first-strand cDNA Synthesis Kit (Thermo Fischer Scientific, USA), as per manufacturer’s instructions, using random hexamer primers. The resulting cDNA was analyzed by quantitative real-time (qRT-PCR) RT-PCR (Applied Biosystems, 7500 Fast Real-Time PCR System, CA, USA) with gene-specific primers using PowerUp SYBR Green PCR Master Mix (Thermo Fisher Scientific, USA). The details of the primers (sequences and annealing temperatures) are given in Table 1. Thermal profile for the real-time PCR was amplification at 50°C for 2 min followed by 40 cycles at 95°C for 15 sec, 60°C for 1 min and 72°C for 20 sec. Melting curves were generated along with the mean CT values and confirmed the generation of a specific PCR product. Amplification of RNU6AP (RNA, U6 small nuclear 1; THP-1 cells) and JW (parasite specific kinetoplast minicircle gene) were used as internal controls for normalization. The results were expressed as fold change of control (Uninfected samples (RNU6AP) and 2 h infected cells (JW) using the 2-ΔΔCT method. All samples were run in triplicates, including a no-template (negative) control for all primers used.
WESTERN BLOTTING
Western blot analysis was done as described previously by Darlyuk et al. Briefly, protein was isolated from the THP-1 cells by resuspending cell lysates in RIPA buffer. Before lysis, adherent macrophages were placed on ice and washed with PBS. Macrophages were scraped in the presence of RIPA lysis buffer containing 1% NP-40, 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM EDTA (pH 8), 10 mM 1,10-phenanthroline and phosphatase and protease inhibitors (Roche). After incubation, lysates were centrifuged for 15 min to remove insoluble matter. The proteins in the lysates were quantified, 80 μg of the lysate was boiled (95˚C) for 5 min in SDS sample buffer and was subjected to electrophoresis on a 10% SDS polyacrylamide gel. Proteins were then transferred onto nitrocellulose (NC) membrane using an electrophoretic transfer cell (Bio-Rad Laboratories, USA) at RT. The membrane was washed with 1× TBST solution three times and blocked with 5% BSA for 2h at RT. The blocked membrane was washed in TBST solution three times. The membrane was then incubated with primary monoclonal antibodies for SLC38A9 (1:500), RagA (1:1000), mTORC1 (1:1000), p-P70S6K1 (1:1000) and β-actin (1:5000) in PBS-Tween 20 containing 5% BSA and incubated overnight at 4°C. The blots were subsequently incubated with the secondary antibody conjugated to horseradish peroxidase at 1:3000 dilution in 5% PBS-Tween 20 for 2h at RT. Enhanced chemiluminescence reaction was used for the detection of the blot. The results were expressed as fold change and quantitated by using AlphaEaseFC image analysis software (Alpha Innotech). The data were expressed as mean ± SD of three independent experiments, and the representative image of one experiment is shown.
IMMUNOPRECIPITATION
At the specified times following infection, for immunoprecipitations, lysates were incubated with 1-2µg of primary antibody: RagA (Cell Signalling) (16 h with rotation, 4°C) and then incubated with pre-blocked Protein G beads (30ul of 50% slurry) (Sigma, USA) for 2h at 4°C. Beads were recovered and washed two times with RIPA lysis buffer before analysis by SDS–PAGE and immunoblotting. Input was used as a positive control and IgG as a negative control.
IMMUNOFLUORESCENCE
THP-1 macrophages were plated on glass coverslips, and after 48 h were infected with L. donovani. After 48 h of infection, Confocal Microscopy was performed as described previously by Singh et al. using anti-SLC38A9 (NBP1-69235, Novus), anti-RagA (D885, Cell Signaling), anti-LdAAP3 (gifted by Prof. Zilberstein) and anti-LAMP1 (D491S, Cell Signaling) antibodies (overnight, 4°C, 2%BSA in PBS). Images were taken with a confocal laser scanning microscope (Olympus FluoViewTM FV1000 with objective lenses PLAPON ×60 O, NA-1.42) at an excitation wavelength of 556 nm.
STATISTICAL ANALYSIS
Fold-expression (qRT-PCR) and intracellular parasite burden were represented as mean ±SD. Each experiment was repeated three times in separate sets. Statistical differences were determined using Student’s unpaired 2-tailed t-test. All statistics were performed using GraphPad Prism Version 5.0 (GraphPad Software, USA). p ≤ 0.05 was considered significant [* (P<0.01 to 0.05), ** (P< 0.001), *** (P< 0.0001), ns (P≥ 0.05)].
Results
L. DONOVANI ADR INFLUENCES SLC38A9-REGULATED ARGININE HOMEOSTASIS IN THE MACROPHAGE LYSOSOME
The first set of experiments was aimed to identify the role if any, of L. donovani infection on SLC38A9 in infected macrophages. THP-1 cells were infected with L. donovani in medium containing 0.1 mM arginine and harvested at the end of each time point for mRNA analysis. At 2 h, 24 h and 48 h post-infection, total RNA was extracted from infected macrophages, and the resulting cDNA was subjected to real-time PCR, using SLC38A9 specific primers as reported in the methods section. Figure 1A shows a time-dependent increase in the expression of SLC38A9 in THP-1 cells after infection with L. donovani. Maximum expression of SLC38A9 was observed at 48 h (~ 5-fold, p ≤ 0.005) post-L. donovani infection in THP-1 cells. As seen in my previous study, the infectivity of L. donovani in THP-1 cells at 2 h, 24 h and 48 h post-infection was between 30–40%. Additionally, the internalization of L. donovani in the host cells in the medium containing 0.1 mM arginine was ~18%.
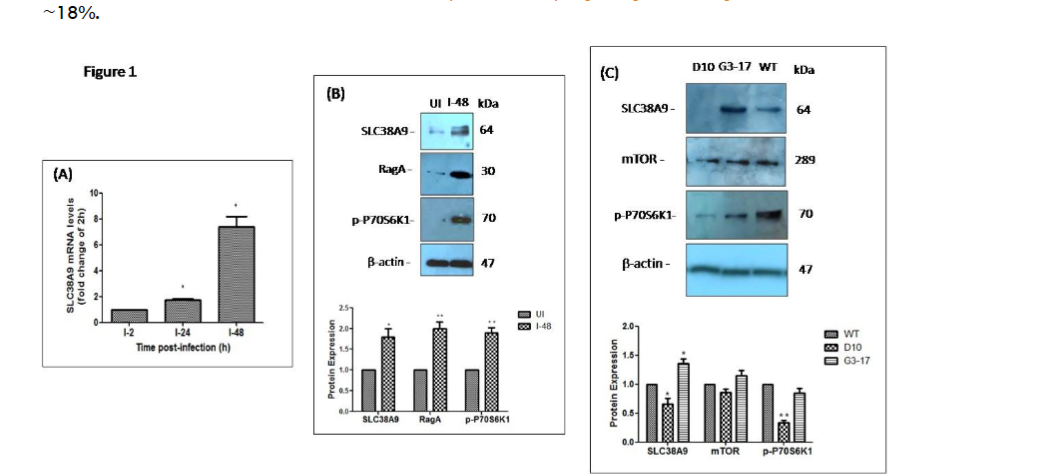
The effect of L. donovani infection on the expression of SLC38A9 in THP-1 cells was analyzed. (A) THP-1 cells maintained in RPMI medium containing 0.1 mM arginine were infected with L. donovani at an MOI of 20 across different time points; for 2 h, 24 h and 48 h. The expression of SLC38A9 measured by real-time PCR has been normalized to RNU6AP gene (RNA, U6 small nuclear 1) as a control gene. The results are expressed as fold-change of 2 h infected cells. Data analysis was performed using the 2-ΔΔCT method. Values are mean ± S.D (n = 3). The results are representative of three independent experiments performed in triplicates.
(B) Total protein was extracted and subjected to Western blot analysis using antibodies specific for SLC38A9, RagA and p-P70S6K1. β-actin was used as a loading control. Cell lysates derived from uninfected THP-1 cultured in 0.1 mM arginine (lane 1), L. donovani-infected THP-1 cultured in 0.1 mM arginine (lane 2), were used. The densitometric analysis shows the fold change in the expression of SLC38A9, RagA and p-P70S6K1 in THP-1 cells infected with L. donovani. Values are mean ± S.D. (n = 3). The results are representative of three independent experiments. (UI: Uninfected THP-1 cells, I-48: THP-1 cells infected for 48 h).
(C) The effect of LdAAP3 in the modulation of arginine sensing by SLC38A9 in L. donovani-infected macrophages was assessed. THP-1 cells maintained in medium containing 0.1 mM arginine were infected with L. donovani deletion mutant (Δap3d10) that lacks AAP3.2, LdAAP3.2 over-expressing strain (Δap3G3-17), or with WT-L. donovani at an MOI of 20 for 48 h. Protein levels of SLC38A9, mTOR and p-P70S6K1 were detected by Western blot in cell lysates using anti-SLC38A9, anti-mTOR and anti-p-P70S6K1 antibodies. Cell lysates derived from THP-1 infected with either Δap3D10 deletion mutant strain (lane 1), LdAAP3 over-expressing L. donovani strain (Δap3G3-17) (lane 2), or WT-L. donovani strain (lane 3), in a medium containing 0.1 mM arginine for 48 h were used. ß-actin was used as a loading control. The densitometric analysis shows the fold change in the expression of SLC38A9, mTOR and p-P70S6K1 in THP-1 cells infected with L. donovani deletion mutant strain, LdAAP3 over-expressing strain, or WT L. donovani. The results are representative of three independent experiments. (I-D10: THP-1 cells infected with L. donovani deletion mutant strain, I-G3-17: THP-1 cells infected with LdAAP3 over-expressing L. donovani strain; Δap3G3-17, I-WT: THP-1 cells infected with WT-L. donovani).
Figure 2
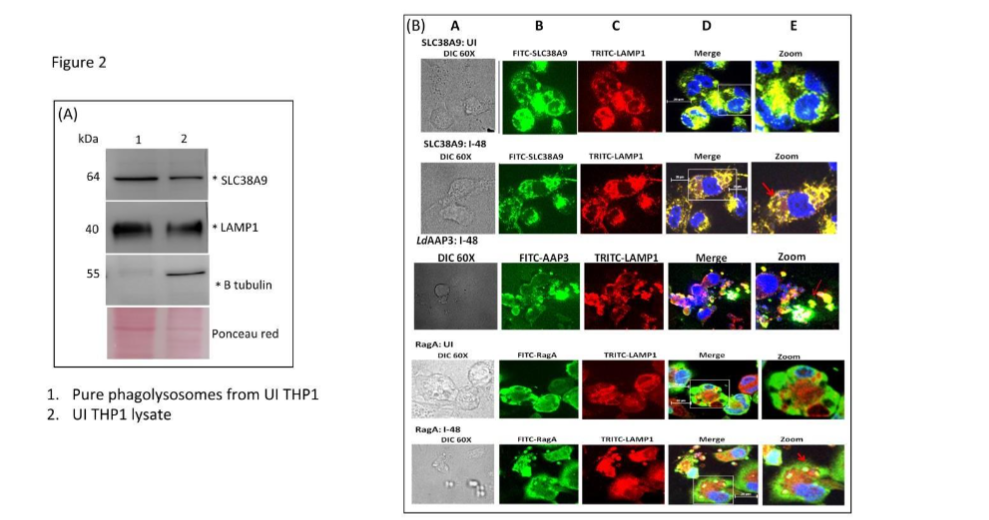
The expression of SLC38A9 protein on phagolysosome membrane was analyzed (A) THP1 macrophages were fed with latex beads (d=3 µm, MOI of 10) for 2 h, washed, and then further incubated for additional 48 h in a CO2 incubator. Subsequently, the cells were harvested and lysed using 27G needle. Phagolysosomes were then isolated using a magnet. Protein from THP-1 lysates and purified phagolysosomes were separated on SDS-PAGE and then subjected to Western blot using antibodies against mammalian SLC38A9, the lysosome specific LAMP1, and β-tubulin. The latter is to show that phagolysosomes are free of cytosol proteins. Lane 1: pure phagolysosomes, lane 2: THP1 cell lysate. (B) The subcellular localization of SLC38A9, LdAAP3 and RagA was investigated. THP-1 cells were infected with L. donovani in RPMI medium containing 0.1 mM arginine for 48 h. Cells were processed and immunostained for SLC38A9, LdAAP3, RagA and LAMP1. Panel A: DIC at 60X. Panel B: anti-SLC38A9/ LdAAP3/RagA antibody detected using FITC (green)-conjugated secondary antibody. Panel C: anti-LAMP1 antibody detected using TRITC (red)-conjugated secondary antibody. Panel D: merged micrographs and Panel E: zoomed images. (UI: Uninfected THP-1, I-48: THP-1 cells infected for 48 h). Scale bar for all is 20µm.
SLC38A9 modulates ADR in intracellular parasites
Several independent reports showed that the lysosomal membrane transceptor SLC38A9 is required for arginine-mediated mTOR activation. We further examined the role of SLC38A9 in mTOR activation (expression of mTOR and p-P70S6K1) in macrophages infected with L. donovani. We silenced SLC38A9 in THP-1 cells by transfecting with siRNA, followed by infection with L. donovani. Uninfected THP-1 transfected with scrambled siRNA was used as control and normalized to 1. At 48 h post-infection, reduced mRNA abundance of SLC38A9 of ~90% (p≤ 0.05) was observed. These SLC38A9 transfected, infected cells expressed lower level of mTOR (~30% decrease, p≤ 0.05) compared to that in control siRNA transfected cells.
Western blot analyses using cell lysates derived from THP-1 cells transfected with SLC38A9 siRNA were performed to confirm the SLC38A9-knockdown-specific regulation of expression of mTOR. As shown in Figure 3B, cell lysates derived from THP-1 cells infected with L. donovani transfected with control siRNA showed higher levels of SLC38A9, mTOR and p-P70S6K1 proteins (lane 2) at 48 h post-infection as compared to the uninfected control. In contrast, THP-1 cells with silenced SLC38A9 (SLCsiRNA), infected by L. donovani showed significant down-regulation of SLC38A9 (p≤ 0.05), mTOR (p≤ 0.05) and p-P70S6K1 (p≤ 0.05) proteins (lane 4) when compared to the levels in infected cells transfected with Control siRNA (CsiRNA). This indicates that SLC38A9 expression affects the abundance and activity of mTOR.
Figure 3
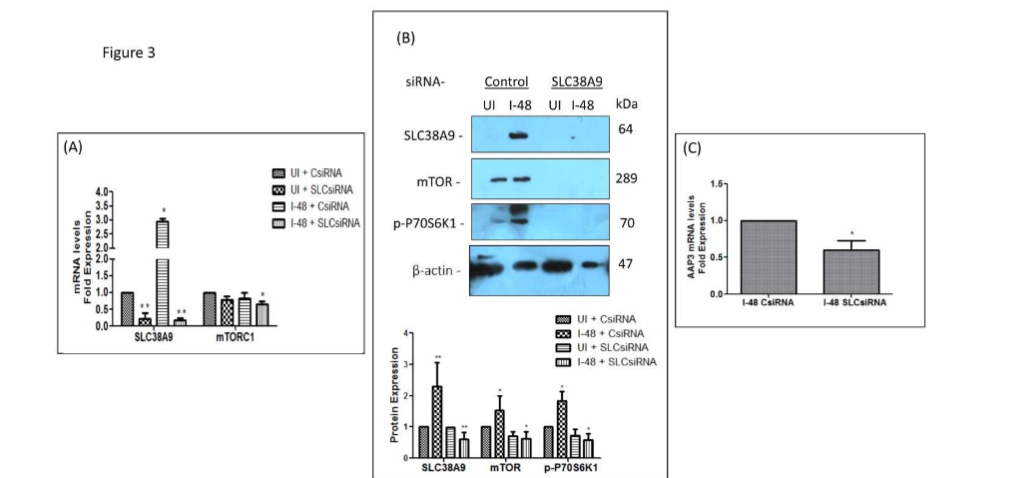
The effect of SLC38A9 in the modulation of mTOR and LdAAP3 in L. donovani infected macrophages was checked. THP-1 cells grown in RPMI medium containing 0.1 mM arginine were transfected with 60 nM SLC38A9 siRNA for 24 h followed by infection with L. donovani at an MOI of 20 for 48 h. Uninfected cells transfected with Control siRNA were used as corresponding controls and set to 1. (A) Total RNA was extracted and cDNA was subjected to real-time PCR analysis using primers specific for SLC38A9 and mTOR. The results are expressed as fold-change of uninfected, control siRNA transfected cells. Here, UI + SLCsiRNA, I-48 + CsiRNA and I-48 + SLCsiRNA were compared to UI + CsiRNA. RNU6A was used as the housekeeping gene. Values are mean ± S.D. (n = 3). The results are representative of three independent experiments performed in triplicates.
(B) The total protein was extracted and subjected to Western blot analysis using antibodies specific for SLC38A9, mTOR and p-P70S6K1. Cell lysates derived from uninfected THP-1 (lane 1), L. donovani infected THP-1 (lane 2), uninfected THP-1 transfected with SLC38A9 siRNA (lane 3), or THP-1 transfected with SLC38A9 siRNA followed by L. donovani infection (lane 4), were used. Immunoblotting was performed using anti-SLC38A9, anti-mTOR and anti-p-P70S6K1 antibodies. Here, UI + SLCsiRNA, I-48 + CsiRNA and I-48 + SLCsiRNA have been compared to UI + CsiRNA. The intensity of the bands was quantified by densitometry using AlphaEase FC Imager software. ß-actin was used as a loading control. The densitometric analysis shows the fold change in expression of SLC38A9, mTOR, p-P70S6K1 in THP-1 cells transfected with SLC38A9 siRNA followed by infection with L. donovani. Values are mean ± S.D. (n = 3). The results are representative of three independent experiments. (UI: Uninfected THP-1 cells, I-48: THP-1 cells infected for 48 h, C: Control, SLC: SLC38A9).
(C) Bar diagrams showing fold expression of LdAAP3 in SLC38A9-transfected infected THP-1 cells as compared to 2 h infected, untransfected control. THP-1 cells grown in RPMI medium containing 0.1 mM arginine were transfected with 60 nM of SLCsiRNA for 24 h followed by infection with L. donovani for 48 h. Infected cells transfected with ON-TARGET plus Control Pool (Dharmacon, USA) was used as a negative control and set to 1. The transfection efficiency was 75–80% as has also been reported by the manufacturer (Dharmacon, USA). The expression pattern was assessed by quantitative real-time qRT-PCR. The qRT-PCR data shown represent mean values obtained from three independent experiments. JW was used as the housekeeping gene. Data analysis was performed using the 2-ΔΔCT method.
We have previously highlighted the regulatory role of arginine in ADR modulation, as arginine starvation in intracellular amastigotes promptly activates the ADR pathway, resulting in up-regulation of the Leishmania arginine transporter (LdAAP3). To validate the role of SLC38A9 in regulating the expression of LdAAP3, THP-1 cells transfected with SLC38A9 siRNA were infected with L. donovani in medium containing 0.1 mM arginine. At 48 h post-infection, total RNA was extracted from transfected macrophages, and the resulting cDNA was subjected to real-time PCR using LdAAP3 specific primers. The mRNA abundance of ADR-induced AAP3 up-regulation decreased in intracellular parasites derived from SLC38A9 siRNA transfected THP-1 cells (~40% decrease, p≤ 0.05). These experiments demonstrate the regulatory role of SLC38A9 on the expression and activity of mTOR and LdAAP3. Thus, our results indicate that ADR activation in intracellular L. donovani is dependent on macrophage SLC38A9 arginine sensing response.
SLC38A9 favours intracellular parasite growth and infection
We further assessed whether increased LdAAP3 and SLC38A9 expression are required for infectivity and virulence in L. donovani. Investigations were carried out to determine the parasite load in SLC38A9 transfected THP-1 cells infected with two different AAP3 mutants: Δap3D10 and Δap3G3-17 at an MOI of 20:1. Control siRNA transfected THP-1 cells infected with WT were used as controls. In the present study, the intracellular parasite burden was determined at 48 h post-L. donovani infection. WT L. donovani infected ~ 50% of THP-1 macrophages, while L. donovani deletion mutant (Δap3D10) had reduced infectivity with only ~30% macrophages (p≤ 0.05) being infected. Upon comparing the parasite load of the Δap3D10 mutant in THP-1 cells transfected with control siRNA, it was observed that Δap3D10 had 50% reduction in the number of parasites (amastigotes/macrophage) as compared to the cells infected with WT L. donovani. Thus, the slow growth phenotype of the ADR mutants compared to the wild type may be partially attributed to the reduced infectivity, which is in agreement with the findings reported by Goldman-Pinkovich et al.
Interestingly, the macrophages that lacked SLC38A9 expression were significantly more susceptible to infection. Infection by WT and Δap3D10 was higher in SLC38A9 siRNA transfected macrophages as compared to that in control siRNA transfected macrophages. Consequently, it was also seen that WT and Δap3D10 had higher parasitemia (amastigotes/macrophage) in SLC38A9 siRNA transfected macrophages as compared to control siRNA transfected macrophages. This points towards the role of induced SLC38A9 favoring intracellular parasite growth and infection.
Figure 4
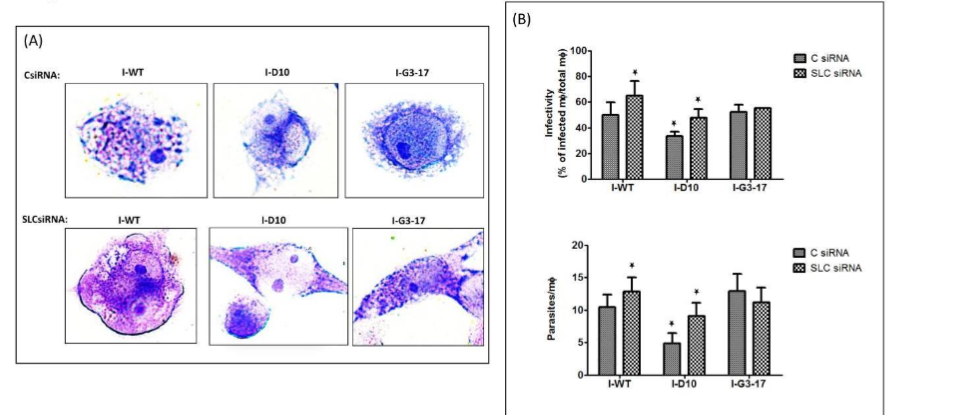
THP-1 cells grown in RPMI medium containing 0.1 mM arginine were transfected with 60 nM SLC38A9 siRNA or 60 nM control siRNA for 24 h followed by infection with L. donovani promastigotes of WT, Δap3D10 and Δap3G3-17 at an MOI of 20:1 for 48 h. (A, B) After 48 h of infection, Giemsa staining was performed to assess the parasite burden. THP-1 cells were fixed, Giemsa stained and amastigotes were counted visually. (A) The respective images of Giemsa-stained infected cells transfected with control/SLC38A9 siRNA can be seen. Scale bar for all is 20µm. (B) Virulence capacity was determined by calculating infectivity and parasitemia. THP-1 cells transfected with control siRNA and infected with WT-L. donovani were used as control. The results are representative of three independent experiments. The results signify mean ± S.D with n = 3, *P < 0.05 statistical difference from the wild-type control.
SLC38A9-RagA-LdAAP3 form a complex during L. donovani infection
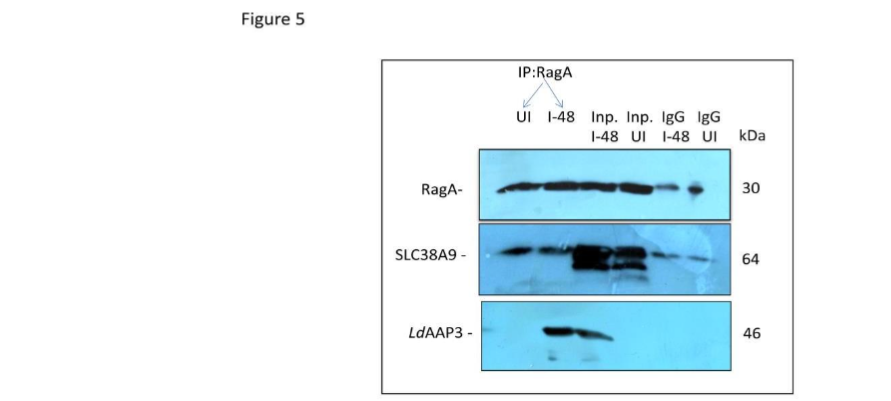
To rule out the possibility that the observed crosstalk between SLC38A9 and LdAAP3.2 is merely an artifact produced by altered levels of lysosomal arginine concentrations, we checked for the direct interaction between LdAAP3.2 and SLC38A9 using Immunoprecipitation assay. To test this, THP-1 cells were infected with L. donovani in medium containing 0.1 mM arginine for 48 h and harvested and immunoprecipitated followed by immunoblotting using SLC38A9, RagA and LdAAP3-specific antibodies. As shown in Figure 5, we found that RagA efficiently pulled down both SLC38A9 and LdAAP3 in THP-1 cells infected with L. donovani. This indicates that the crosstalk between SLC38A9 and LdAAP3.2 is not merely an artifact.
Figure 5
Discussion
The induction of the arginine deprivation response in Leishmania-infected macrophages has been shown to play a key role in providing essential nutrients to the multiplying parasites within parasitophorous vacuoles (PV). This response may also selectively target parasites for neutralization by the innate immune defense mechanisms. Lysosomes have been recognized as a major location of the amino acid pool in mammalian cells, with sensors on the surface that reports lysosome amino acid sufficiency. SLC38A9 is an arginine-activated amino acid transporter localized to the lysosome membrane of mammalian cells. This transporter is also an arginine sensor (transceptor) and together with RagGTPases recruits and activates cytosolic mTOR. Understanding the regulation of the arginine deprivation response in macrophages infected with Leishmania remains largely unclear. The present study provides key insights into the molecular mechanism involved in the arginine sensing response in L. donovani-infected host cells. Additionally, this study identifies a novel role for SLC38A9 as a key regulator of LdAAP3 in intracellular L. donovani and highlights the influence of Leishmania ADR on host SLC38A9. We report that the up-regulation of SLC38A9 begins at 24 h post-infection and increases five-fold at 48 h post-infection, thereby, implying that the activation of SLC38A9 in infected THP-1 cells occurs between 24–48 h post-infection. In infected phagolysosomes the initial arginine concentration is high (~140 μM; the physiological concentration in phagolysosomes) and reduces with time (5 μM; sufficient for ADR activation in axenic L. donovani).
Subsequently, arginine concentration possibly reaches levels at 48 h post-infection, which results in the activation of SLC38A9. However, it could also be possible that induction of SLC38A9 is delayed due to the time taken to deplete intracellular parasite pools of arginine following their phagocytosis. SLC38A9 is a lysosomal membrane transceptor that interacts with the regulator and the Rag GTPases through its N-terminal 119 amino acids (‘Ragulator-binding domain’) and is required for mTOR activation. Several reports have indicated the role of SLC38A9 as a regulator of mTOR, which further regulates protein synthesis through the phosphorylation and activation of the ribosomal S6 kinase (p70S6K1). In line with this, few other studies have shown that SLC38A9 acts on Rag GTPases and could positively regulate mTOR activity, in response to nutrient sufficiency. Furthermore, it is also known that L. donovani infection increases mTOR activity in mouse and human macrophages, including THP-1 cells. However, to our knowledge, no report has studied SLC38A9 mediated mTOR signalling in response to L. donovani infection. Thus, the present study identifies the regulatory role of Leishmania as the key modulator of SLC38A9-mediated mTOR pathway in macrophages maintained in physiological concentration of arginine.
SLC38A9 is an arginine-activated amino acid transporter localized to lysosome membrane of mammalian cells. This transporter upon arginine sufficiency in the lysosome lumen activates leucine efflux from the lysosome to the cytosol. Cytosolic leucine thereby binds to sestrin2 and together with the SLC38A9 complex recruits and activates cytosolic mTOR. mTOR is an activator of M1, the pathway that induces the conversion of arginine to cytotoxic NO. It is already known that following invasion, intracellular L. donovani parasites actively compete for the diminishing arginine pool in the phagolysosome. We have earlier reported that when arginine concentration becomes low, ADR is induced leading to the up-regulation of LdAAP3 expression in intracellular L. donovani. Here, our results demonstrate a perfect temporal correlation between LdAAP3 expression and modulation of SLC38A9 arginine sensing in THP-1 cells infected with L. donovani. We show that when LdAAP3.2 is depleted in the parasites, the expression of SLC38A9 is suppressed and so is the mTOR activity; however, when LdAAP3.2 is overexpressed, the expression of SLC38A9 is induced. We have previously shown that wild type and LdAAP3 overexpressors transport more arginine through LdAAP3 (increased by ADR), while LdAAP3 mutants transport normal arginine, with no further increase. This suggests that SLC38A9 along with LdAAP3 is involved in arginine transport. Therefore, during development inside phagolysosomes, L. donovani may encounter a low level of arginine, and they overcome these low arginine levels by up-regulating arginine transport via LdAAP3 and SLC38A9 arginine sensing. Measuring the arginine pools in lysosomes under these different circumstances in infected macrophages would be interesting follow up experiments.
A delayed response of SLC38A9 in Leishmania-infected macrophages suggested its possible involvement in the repression of parasite LdAAP3. To investigate the biological outcome of infection-specific SLC38A9 induction, we monitored the expression level of LdAAP3 in intracellular L. donovani. There was a marked decrease in the expression of LdAAP3 in intracellular L. donovani derived from infected THP-1 macrophages whose SLC38A9 expression was silenced, maintained in 0.1 mM arginine. Interestingly, it was also observed that macrophages with reduced SLC38A9 expression were significantly more susceptible to infection. Furthermore, the Δap3D10 mutant had reduced infectivity (~30% macrophages [p≤ 0.05] being infected) and was impaired in its ability to develop inside normal THP-1 macrophages. However, upon silencing SLC38A9 in THP-1 cells grown in physiological concentration of arginine, the ability of AAP3 mutant to develop inside these cells was found to be restored to that of the WT, because of the retention of high levels of cellular arginine inside these cells. These experiments indicate that SLC38A9 expression influences parasite ADR in a way that the mutants can resume infectivity in cells with silenced SLC38A9, grown in 0.1 mM arginine, because of retention of high levels of cellular arginine inside these cells. This points towards the role of induced SLC38A9 favoring intracellular parasite growth and infection. This suggests the influence of SLC38A9 on parasite ADR. Hence, we have characterized the role of SLC38A9 during Leishmania infection and show that it is essential for parasite growth or infectivity in vitro. Thus, inside the host macrophage, Leishmania must overcome the arginine “Hunger Games” by activating the SLC38A9 arginine pathway in addition to the transport of arginine via LdAAP3.
The mutual influence of SLC38A9 and AAP3 led us to speculate that mTOR serves as a common theme in the pathways activated by these transporters. To date, a role for mTOR kinase in a Leishmania sensing pathway has not been documented. We hypothesized that upon entry to macrophage phagolysosomes, elements in parasites arginine sensing response, including MAPK2 and the ADR pathway may interact with the macrophage mTOR arginine sensing machinery that thereby induce LdAAP3 up-regulation. Further experiments confirming our hypothesis that the host mTOR pathway may transmit the information to parasite MAPK2 activating the PKA catalytic subunit 1 (PKA-C1) thereby leading to the up-regulation of LdAAP3 expression would be planned in follow up studies.
Based on the current findings, our study demonstrates a host-pathogen interaction mediated through SLC38A9. Upon infection in THP-1 cells, we observed increased levels of SLC38A9, and RagA along with a further increase in p-P70S6K1 abundance. This indicates heightened mTOR activity leading to increased protein synthesis. The up-regulation of LdAAP3 levels observed upon L. donovani infection as previously reported could be attributed to the elevated SLC38A9 levels observed in the present study, thus reflecting the regulatory role of SLC38A9 on LdAAP3 via RagA/mTOR signaling. The reason for LdAAP3 and SLC38A9 sensing systems interacting to assure arginine availability for parasite survival could be due to the formation of a complex among SLC38A9, RagA and LdAAP3 in infected THP-1. As Leishmania-infected phagolysosomes have recently been observed to contain SLC38A9 and LdAAP3 is already been known to be localized on the surface of lysosomes in Leishmania. Thereby, we propose that upon infection under physiological arginine conditions, SLC38A9 transports amino acids and signals their presence at lysosomes via association with cytosolic RagA. This in turn recruits mTOR from the cytosol to the lysosomal surface. Our current data highlight the interaction between host SLC38A9 and LdAAP3. Whether this interaction occurs directly or involves intermediary proteins remains uncertain and requires further investigation. Experiments addressing possible interactions of these proteins can be a part of follow up studies.
The present study is the first to highlight the role of SLC38A9 as a lysosomal amino acid transceptor that regulates LdAAP3 expression in L. donovani. Our results demonstrate a link between SLC38A9 and LdAAP3 and suggest that SLC38A9 may act as a regulator of LdAAP3, in this process. Although we have studied this pathway only in macrophages, the SLC38A9-AAP3 axis may be relevant in other cell types as well. Thus, SLC38A9 could be a novel target for therapeutic intervention in leishmaniasis.
Acknowledgements
This work was funded by a grant from the Indo-Israel Joint Research Programme (F. No. 6-7/2016(IC). Rentala Madhubala is an A. S. Paintal Distinguished Scientist Chair of ICMR and J. C. Bose National Fellow. EM is working as a Consultant and Assistant Professor at Sir Ganga Ram Hospital. Research fellowship to EM was from UGC D.S. Kothari (UGC, India) at JNU. DZ was funded by the Israel Science Foundation (ISF) grant number 887/21. MP and HP received a post-doctoral research fellowship from the Indo-Israel Joint Research Programme. We thank the Central Instrumentation Facility at the School of Life Sciences, Jawaharlal Nehru University, for providing the instrumentation facility.
Author Contributions
RMa, DZ, RMu and EM conceptualized the work. EM designed experiments. EM performed majority of experiments. MP contributed in RT-PCR experiments. HP performed phagolysosome isolation experiment. EM wrote the manuscript. RMa, DZ, NKG and AS revised the manuscript. RMa, DZ and RMu evaluated the work progress. All authors contributed to the article and approved the submitted version.
Conflict of interest
The authors declare no conflict of interest.
Conclusion
Leishmania donovani, the causative agent of kala-azar, exhibits a digenetic life cycle. Following infection of the mammalian host, promastigotes differentiate into intracellular amastigotes within the phagolysosome of macrophages. Arginine is a central point of competition between the host and the pathogen. L. donovani senses lack of arginine in the surrounding micro-environment and activates a unique ADR pathway, thus upregulating the expression of the arginine transporter (LdAAP3). The arginine-activated amino acid transporter SLC38A9 localizes to the lysosome surface of mammalian cells and acts as a sensor that transmits information about arginine levels in the lysosome lumen to the mechanistic target of rapamycin (mTOR) kinase. In the present study, we identified the functional interaction of host SLC38A9 and parasite LdAAP3 in macrophages infected with L. donovani. We report that host SLC38A9 upregulation is critical for enhancing and maintaining high LdAAP3 levels in intracellular L. donovani. Our results decode crucial information regarding the molecular mechanism involved in the arginine sensing response in L. donovani-infected host cells. These findings increase our understanding of the interaction of signalling intermediates during Leishmania infection which may lead to the discovery of novel therapeutic interventions.
References
1. Ahuja K, Arora G, Khade P, Selvapandiyan A*. Selective elimination of Leptomonas from the in vitro co-culture with Leishmania. Parasitol. Intl. 2015; 64(4):1-5.doi: 10.1016/j.parint.2015.01.003
2. Assche T Van, Deschacht M, Inocêncio da Luz R, Maes L, Cos P. Leishmania-macrophage interactions: Insights into the redox biology. Free Radical Biology and Medicine. 2011; 51(2): 337-351. doi:10.1016/j.freeradbiomed.2011.05.011.
3. Barilli A, Rotoli BM, Visigalli R, Bussolati O, Gazzola GC, Asta VD. Arginine transport in human monocytic leukemia THP-1 cells during macrophage differentiation. Journal of Leukocyte Biology. 2011; 90(2). doi:10.1189/jlb.0910510.
4. Buerger C, DeVries B and Stambolic V. Localization of Rheb to the endomembrane is critical for its signaling function. Biochemical and Biophysical Research Communications. 2006; 344(3). doi:10.1016/j.bbrc.2006.03.220.
5. Chaparro V, Leroux LP, Masvidal L, Lorent J, Graber TE, Zimmermann A et al. Translational profiling of macrophages infected with Leishmania donovani identifies mTOR- And eIF4A-sensitive immune-related transcripts. PLoS Pathogens. 2020; 16(6). doi:10.1371/journal.ppat.1008291.
6. Chen Z, Wang T, Liu Z, Zhang G, Wang J, Feng S, Liang J. Inhibition of autophagy by MiR-30A induced by Mycobacteria tuberculosis as a possible mechanism of immune escape in human macrophages. Japanese Journal of Infectious Diseases. 2015; 68(5). doi:10.7883/yoken.JJID.2014.466.
7. Colotti, G. and Ilari, A. Polyamine metabolism in Leishmania: From arginine to trypanothione. Amino Acids. 2011; 40(2):269-85. doi:10.1007/s00726-010-0630-3.
8. Darlyuk I, Goldman A, Roberts SC, Ullman B, Rentsch D, Zilberstein D. Arginine homeostasis and transport in the human pathogen Leishmania donovani. Journal of Biological Chemistry. 2009; 284(30) :19800-7. doi:10.1074/jbc.M901066200.
9. Dibble, C.C. and Manning, B.D. Signal integration by mTORC1 coordinates nutrient input with biosynthetic output. Nature Cell Biology. 2013; 15(6):555-64. doi:10.1038/ncb2763.
10. Ezenyia I, Madan E, Singhal J, Jain R, Chakrabarti A, Ghousepeer GD. Screening of traditional medicinal plant extracts and compounds identifies a potent anti-leishmanial diarylheptanoid from Siphonochilus aethiopicus. Journal of Biomolecular Structure and Dynamics. 2024; 42(5):2449-2463. Doi:10.1080/07391102.2023.2212779
11. Fairlamb AH and Cerami A. Metabolism and functions of trypanothione in the kinetoplastida. Annual Review of Microbiology. 1992; 46:695-729. doi:10.1146/annurev.mi.46.100192.003403.
12. Goldman-Pinkovich A, Balno C, Strasser R, Zeituni-Molad M, Bendelak K, Rentsch D. An Arginine Deprivation Response Pathway Is Induced in Leishmania during Macrophage Invasion. PLoS Pathogens. 2016; 12(4). doi:10.1371/journal.ppat.1005494.
13. Goldman-Pinkovich A, Kannan S, Nitzan-Koren R, Puri M, Pawar H, Bar-Avraham Y. Sensing host arginine is essential for leishmania parasites intracellular development. mBio. 2020; 11(5). doi:10.1128/mBio.02023-20.
14. Gregory DJ and Olivier M. Subversion of host cell signalling by the protozoan parasite Leishmania. Parasitology. 2005; 130 doi:10.1017/S0031182005008139.
15. Jewell JL, Russell RC and Guan KL. Amino acid signalling upstream of mTOR. Nature Reviews Molecular Cell Biology. 2013; 14(3). doi:10.1038/nrm3522.
16. Jung J, Genau HM and Behrends C. Amino Acid-Dependent mTORC1 Regulation by the Lysosomal Membrane Protein SLC38A9. Molecular and Cellular Biology. 2015; 35(14). doi:10.1128/mcb.00125-15.
17. Kühnel M, Anes E and Griffiths G. Isolation of latex bead-and mycobacteria-containing phagosomes. Cell Biology, 2006; Four-Volume Set. doi:10.1016/B978-012164730-8/50080-0.
18. Mandal A, Das S, Roy S, Ghosh AK, Sardar AH, Verma S et al. Deprivation of L-Arginine Induces Oxidative Stress Mediated Apoptosis in Leishmania donovani Promastigotes: Contribution of the Polyamine Pathway. PLoS Neglected Tropical Diseases, 2016; 10(1). doi:10.1371/journal.pntd.0004373.
19. Mandal A, Das S, Kumar A, Roy S, Verma S, Ghosh AK, Singh R et al. L-Arginine uptake by cationic amino acid transporter promotes intra- macrophage survival of Leishmania donovani by enhancing arginase-mediated polyamine synthesis. Frontiers in Immunology. 2017; 8(JUL). doi:10.3389/fimmu.2017.00839.
20. McConville MJ. Metabolic Crosstalk between Leishmania and the Macrophage Host. Trends in Parasitology. 2016; 32(9): 666-668. doi:10.1016/j.pt.2016.05.005.
21. Olive AJ and Sassetti CM. Metabolic crosstalk between host and pathogen: Sensing dapting and competing. Nature Reviews Microbiology. 2016; 14(4):221-34 doi:10.1038/nrmicro.2016.12.
22. Pawar H, Puri M, Weinberger RF, Madhubala R, Zilberstein D. The arginine sensing and transport binding sites are distinct in the human pathogen Leishmania. PLoS Neglected Tropical Diseases. 2019; 13(4). doi:10.1371/journal.pntd.0007304.
23. Rebsamen, M, Pochini L, Stasyk T, de Araújo 3 MEG, Galluccio M, Kandasamy RK. SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1. Nature. 2015; 519(7544). doi:10.1038/nature14107.
24. Ren W, Rajendran R, Zhao Y, Tan B, Wu G, Bazer FW. Amino acids as mediators of metabolic cross talk between host and pathogen. Frontiers in Immunology. 2018; 27:9:319. doi:10.3389/fimmu.2018.00319.
25. Saito K, Araki Y, Kontani K, Nishina H, Katada T. Novel role of the small GTPase Rheb: Its implication in endocytic pathway independent of the activation of mammalian target of rapamycin. Journal of Biochemistry. 2005; 137(3). doi:10.1093/jb/mvi046.
26. Shen, K. and Sabatini, D.M. Ragulator and SLC38A9 activate the Rag GTPases through noncanonical GEF mechanisms. Proceedings of the National Academy of Sciences of the United States of America. 2018; 115(38). doi:10.1073/pnas.1811727115.
27. Showkat M, Beigh MA and Andrabi KI. mTOR Signaling in Protein Translation Regulation: Implications in Cancer Genesis and Therapeutic Interventions. Molecular Biology International. 2014; 115(38):9545-9550 doi:10.1155/2014/686984.
28. da Silva MFL and Floeter-Winter LM. Arginase in Leishmania. Sub-Cellular Biochemistry, 2014; 74:103-17. doi:10.1007/978-94-007-7305-9_4.
29. Singh AK, Pandey RK, Siqueira-Neto JL, Kwon YJ, Freitas-Junior LH, Shaha C, Rentala Madhubala R. Proteomic-based approach to gain insight into reprogramming of THP-1 cells exposed to Leishmania donovani over an early temporal window. Infection and Immunity. 2015; 83(5): 1853-68. doi:10.1128/IAI.02833-14.
30. Singh AK, Pandey RK, Shaha C, Madhubala R. MicroRNA expression profiling of Leishmania donovani-infected host cells uncovers the regulatory role of MIR30A-3p in host autophagy. Autophagy. 2016; 12(10). 1817-1831. doi:10.1080/15548627.2016.1203500.
31. Thomas SA, Nandan D, Kass J, Reine NE. Countervailing, time-dependent effects on host autophagy promotes intracellular survival of Leishmania. Journal of Biological Chemistry. 2018; 293(7). doi:10.1074/jbc.M117.808675.
32. Wanasen N and Soong L. L-arginine metabolism and its impact on host immunity against Leishmania infection. Immunologic Research. 2008;41(1):15-25. doi:10.1007/s12026-007-8012-y.
33. Wang S, Tsun ZY, Wolfson RL, Shen K, Wyant GA, Plovanich ME. Lysosomal amino acid transporter SLC38A9 signals arginine sufficiency to mTORC1. Science. 2015; 347(6218). doi:10.1126/science.1257132.
34. Wang S, Tsun ZY, Wolfson RL, Shen K, Wyant GA, Plovanich ME. The amino acid transporter SLC38A9 is a key component of a lysosomal membrane complex that signals arginine sufficiency to mTORC1. Science. 2015; 347(6218): 188-94. doi: 10.1126/science.1257132
35. Weinstein SL, Finn AJ, Davé SH, Meng F, Lowell CA, Sanghera JS, DeFranco AL. Phosphatidylinositol 3-kinase and mTOR mediate lipopolysaccharide-stimulated nitric oxide production in macrophages via interferon-β. Journal of Leukocyte Biology, 2000; 67(3): 405-14. doi:10.1002/jlb.67.3.405.
36. Wolfson RL, Chantranupong L, Saxton RA, Shen K, Scaria SM, Cantor JR, Sabatini DM. Sestrin2 is a leucine sensor for the mTORC1 pathway. Science. 2016; 351(6268): 43-8. doi:10.1126/science.aab2674.
37. Wu G and Morris SM. Arginine metabolism: Nitric oxide and beyond. Biochemical Journal. 1998; Nov 15;336 (Pt1) (Pt1):1-17. doi:10.1042/bj3360001.
38. Wyant GA, Abu-Remaileh M, Wolfson RL, Chen WW, Freinkman E, Danai LV. mTORC1 Activator SLC38A9 Is Required to Efflux Essential Amino Acids from Lysosomes and Use Protein as a Nutrient. Cell 2017; 171(3) :642-654.e12 doi:10.1016/j.cell.2017.09.046.
39. Xie J and Proud CG. Signaling crosstalk between the mTOR complexes. Translation. 2014; 2(1): e28174. doi:10.4161/trla.28174.
40. Xu, H. and Ren, D. Lysosomal physiology. Annual Review of Physiology, 2015;77: 57-80. doi:10.1146/annurev-physiol-021014-071649.
41. Yang Q, Inoki K, Kim E, Guan KL. TSC1/TSC2 and Rheb have different effects on TORC1 and TORC2 activity. Proceedings of the National Academy of Sciences of the United States of America. 2006; 103(18): 6811-6. doi:10.1073/pnas.0602282103.
42. Zhang N, Prasad S, Huyghues Despointes CE, Young J, Kima PE. Leishmania parasitophorous vacuole membranes display phosphoinositides that create conditions for continuous Akt activation and a target for miltefosine in Leishmania infections. Cellular Microbiology. 2018;20(11). doi:10.1111/cmi.12889.
43. Zoncu R, Bar-Peled L, Efeyan A, Wang S, Sancak Y, Sabatini DM. mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H+-ATPase. Science. 2011; 334(6056): 678-83. doi:10.1126/science.1207056.
44. Zoncu R, Efeyan A and Sabatini DM. MTOR: From growth signal integration to cancer, diabetes and ageing. Nature Reviews Molecular Cell Biology. 2011. 12(1):21-35. doi:10.1038/nrm3025.

