Effects of Copaiba Oil on Visceral Fat in Rats
Effects of oral copaiba-oil supplementation on visceral white adipose tissue content and histology of hypothalamic obese and non-obese male and female Wistar rats
Helen de França Kailer¹, Elizangela Stein¹, Marina Helena Forlin¹, Eduarda Felchak Caldas¹, Ellen Carolina Zawoski Gomes¹², Zoe Maria Neves de Carvalho Guareschi¹, Marianela Andrea Díaz Urrutia¹, Sirlei Patrícia de Souza¹, Beatriz Daudt¹, Bruna Schumaker Siqueira¹, Sabrina Grassiolli¹
- Laboratory of Endocrine Physiology and Metabolism – LAFEM. Postgraduate Program in Biosciences and Health. State University of Western Paraná – UNIOESTE. Campus Cascavel – Paraná, Brazil.
- Centro Universitário Fundação Assis Gurgacz – FAG. Cascavel. Paraná. Brazil.
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Kailer, Hd., Stein, E., et al., 2024. Effects of oral copaiba-oil supplementation on visceral white adipose tissue content and histology of hypothalamic obese and non-obese male and female Wistar rats. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5770
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5770
ISSN 2375-1924
ABSTRACT
Background. Obesity involves the excessive expansion of white adipose tissue, typically associated with adipocyte hypertrophy and inflammation, especially in visceral depots, a process influenced by sex. Natural compounds like Copaiba’s oil (CO) exhibit anti-inflammatory and anti-adiposity effects, though their impact on white adipose tissue histology is unknown.
Aims. This study evaluated the effect of oral CO supplementation on the histology and content of visceral WAT depots in hypothalamic obese male and female Wistar rats.
Methods. The litter size was adjusted to 6-8 pups per dam (3-4 males and females) at birth. Hypothalamic obesity was induced in the neonatal period via subcutaneous injection of Monosodium L-glutamate (4g.Kg-1). Non-obese group received equimolar saline (1.25 kg-1). Half of the animals from each group received oral CO-supplementation (0.5mL.kg-1; three times/week) from postnatal days 30 to 90, while non-supplemented groups received saline (0.9%) during the same period, via and frequency. At 92 postnatal days, following 12h of fasting, the animals were euthanized, blood samples were collected, and plasma was used to dosage glucose and triglycerides from which the TyG index was calculated to evaluate insulin resistance. Visceral Perigonadal and Perirenal depots were excised, weighed, and analyzed histologically.
Results. CO-supplementation showed nonsignificant effects in non-obese groups (males or females). Obese male and female animals show higher triglycerides, increased insulin resistance, heavier visceral adipose tissue depots, lower adipocyte numbers, and increased hypertrophy than males and females non-obese. Moreover, obese males displayed hyperglycemia compared to non-obese males. CO supplementation’s effects were sex-dependent in obese males, CO worsened triglyceride levels without affecting visceral adipose tissue content or histology. Conversely, in obese females, CO supplementation improved triglyceride levels, decreased perigonadal weight, and increased adipocyte numbers in perigonadal and perirenal depots.
Conclusion. Chronic oral CO supplementation does not prevent adipose tissue expansion or metabolic dysfunction in male hypothalamic obese rats. In contrast, obese female CO-supplemented showed a slight reduction in adiposity with increased adipocyte proliferation and improved fasting triglycerides levels, indicating greater responsiveness in females to the beneficial effects of CO on obesity.
Keywords: White Adipose Tissue, Obesity, Copaiba, Medicinal Plants.
INTRODUCTION
The adipose tissue exerts an essential role in energy homeostasis, first accommodating excessive calories into lipids deposition and second by secreting a greater variety of signals, nominated of adipokines, which via the central nervous system (CNS) communicate about metabolic body state regulating food intake and energy expenditure. Moreover, adipokines released by white adipose tissue (WAT) also modulate immunological responses and insulin periphery sensibility. Excessive WAT expansion defines obesity, and WAT dysfunctions are related to metabolic abnormalities present in metabolic syndrome (MS), such as dyslipidemia, hyperglycemia, and insulin resistance. Obesity and MS associations elevate the risk of the development of chronic diseases, in particular type 2 diabetes mellitus (T2M) and cardiovascular dysfunctions.
Adipocytes are the most common cell type in WAT of mammals, including humans and rodents, and manage metabolic and endocrine functions in this tissue. The adipocytes show common morphological characteristics such as a large diameter (≥100µm) due to a unilocular lipids deposition (a single and large lipid droplet) resulting in scarce cytoplasm and non-centrally nuclei. Moreover, WAT shows reduced vascularization, innervation, and mitochondria content compared to other tissues. In consequence, WAT presents a lower metabolic rate and a high capacity to store energy.
During high energy surplus conditions, WAT’s adipocytes can get bigger in size (hypertrophy) and number (hyperplasia), expanding adipose mass content next to the visceral organs or subcutaneously. The visceral and subcutaneous WAT depots present different growth patterns and immune and metabolic functions. Thus, visceral WAT expansion occurs preferentially by adipocyte hypertrophy, presenting a higher pro-inflammatory state, reduced vascularization, higher oxidative stress, and elevated lipolysis rate than subcutaneous WAT depots. For this reason, rodents and humans that develop obesity primarily throughout visceral WAT expansion show a more severe deleterious impact on lipids and glucose homeostasis and a higher risk of T2DM development and cardiovascular dysfunctions.
Sexual dimorphism influences energy homeostasis, affecting WAT distribution and determining differences in adipocyte endocrine, metabolic, and immune functions. Consequently, the effect of WAT expansion in obese states exerts a distinct impact on the health of males and females. Visceral WAT deposition is typically obesity-associated in men and postmenopausal women, while peripheral subcutaneous WAT distribution is typical in premenopausal women. Genetic and hormonal levels appear important to WAT distribution and pathological states related to obesity. Thus, obese men and obese male rodents often present a greater prevalence of hyperglycemia and hypertriglyceridemia and a higher risk of MS. A recent study showed that metabolic homeostasis works differently in men and women due to evolutionary adaptations that enable women to resist losing energy stores and protein mass while remaining fertile in times of energy deficit.
Although we currently have different strategies to avoid obesity’s harmful effects on health – bariatric surgery, pharmacological agents, diet, and exercise – as pointed out by epidemiological studies, obesity remains growing worldwide, indicating that none of these mechanisms can preserve body weight control in the long term. Sexual dimorphism also defines the effectiveness of anti-adiposity strategies, and recognizing these effects is fundamental to the treatment of obesity. In this context, many natural compounds present bioactive principles that can attenuate the negative impact of obesity on health.
Plant substances showing potential beneficial effects on health are found in Brazilian forests, including the Copaifera tree, from which copaiba’s oil (CO) is obtained. The use of CO has been reported since the 16th century, and this agent shows several therapeutic properties, such as anti-inflammatory, urinary tract infections, sore throats, stomach ulcers, and infectious diseases. Despite the chemical profile of CO might be slightly different from one species to another, the β-caryophyllene, α-humulene, α-copaene, α-bergamotene, δ-cadiene, and β-bisabolol beside diterpene acids, such as copalic acid, clorechinic acid, and hardwickiic acid are the compounds found in more significant quantities in this oil.
More recently, studies have shown anti-inflammatory properties of CO in the liver, skin, lung, intestine, and neuronal cells. Interestingly, in neuronal cells, the CO-upregulated molecular pathways (pI3K/Akt/mTOR, MAPK, and JAK/STAT) which are also common pathways of the inflammatory process in WAT. However, the effects of CO supplementation on WAT are contradictory. In this sense, de Paula et al., 2023 showed that oral CO supplementation in lean rats did not alter adiposity or WAT inflammation. In contrast, positive anti-adiposity and anti-inflammatory effects of CO supplementation in the WAT were shown in the obesity induced by high sucrose diet.
Herein, we used a hypothalamic obese model induced by high Monosodium L-glutamate (MSG) doses, administered at first post-natal days (PND), to evaluate CO effects on WAT. MSG-obese rodents show massive visceral adiposity, insulin resistance, dyslipidemia, and glucose intolerance, reproducing typical metabolic abnormalities in human obesity. MSG-obese rats develop obesity without hyperphagia, so they avoid diet influences on CO effects. Moreover, until now, no study has evaluated CO supplementation effects on WAT content and histology considering sex influences. Thus, the central aim of the present study was to investigate the effects of oral and chronic CO supplementation on WAT content and histology of hypothalamic MSG-obese and non-obese male and female Wistar rats.
METHODS
ANIMAL AND ETHICAL ASPECTS
In the present study, Wistar male and female rats were neonatally treated with MSG and supplemented with CO for a long life. Animals were maintained at controlled luminosity (dark-light cycle) and temperature (23±2oC), allocated in 3 – 4 rats by cages and receiving chow rodents (Biobase, Brazil) and water ad libitum. The project was approved by the Ethics Committee on Animals Use (CEUA/November 13/2020) of Western Parana State University (UNIOESTE). All procedures respected the guidelines of the National Council for Control of Animal Experiments (CONCEA) and The Arrive Guidelines 2.0.
HYPOTHALAMIC OBESE AND NON-OBESE ANIMALS
Pregnant rats were acquired and maintained in individual cages, with rodent chow and water ad libitum. At birth, the offspring size was adjusted to 3 – 4 pups by the dams of each sex: males and females. Hypothalamic lesions were induced by subcutaneous administration of MSG (4 g.Kg-1) applied from the 2nd to 6th PND. The fragility of the blood-brain barrier in this period allows MSG to cause damage to neurons mainly located in the arcuate nucleus of the hypothalamus, promoting an obese and pre-diabetic phenotype in adult Wistar rats; these male and female animals compose the hypothalamic-obese group (MSG). In the non-obese groups named Control (CT), male and female rats received saline equimolar solution (1.25 g.Kg-1). All animals were weaned and weighed at 21-PND and randomly distributed to subgroups according to the supplementation scheme.
COPAIBA OIL SUPPLEMENTATION
Amazon Oil provides CO donations, and this product are available at: https://amazonoil.com.br/produtos-da-floresta/resina-de-copaiba/. From 22-90 PND, supplemented groups received CO 0.5 mL.Kg-1 (represented by co), orally by gavage method. Non-supplemented groups received saline solution (NaCl 0.9%, represented by ∅). Thus, considering sex, obesity, and supplementation originated in 8 experimental groups (n=6 rats/group). Control non-supplemented (CT∅); Control CO supplemented (CTco); Hypothalamic-obese non-supplemented (MSG∅); Hypothalamic-obese CO supplemented (MSGco) for males and the same groups for females. See experimental design Figure 1.

EUTHANASIA AND ADIPOSITY EVALUATION
At 92 PND, euthanasia was performed, body weight (BW) was measured, and BW gain (∆BW) was calculated between 92 PND BW and 21 PND BW. The serum samples were collected, and white adipose tissue Perigonadal (Pg-WAT) and perirenal (Pr-WAT) depots were excised, weighed, and transferred to a fixation histological solution (Alfac: alcohol 70%; formalin 37-40% and glacial acetic acid) for histological procedures.
FASTING GLUCOSE, TRIGLYCERIDES, AND INSULIN RESISTANCE
Fasting glucose and triglycerides (TG) were obtained from the serum by enzymatic colorimetric commercial kits (Bioloquid, Laborclin Produtos para Laboratórios Ltda, Pinhais, Brazil) following the fabricant’s instructions, and used to perform the insulin resistance evaluation by Triglycerides-glucose (TyG) index, calculated as Ln[fasting TG (mg.dL-1) × fasting glucose (mg.dL-1)/2).
WHITE ADIPOSE TISSUE HISTOLOGY
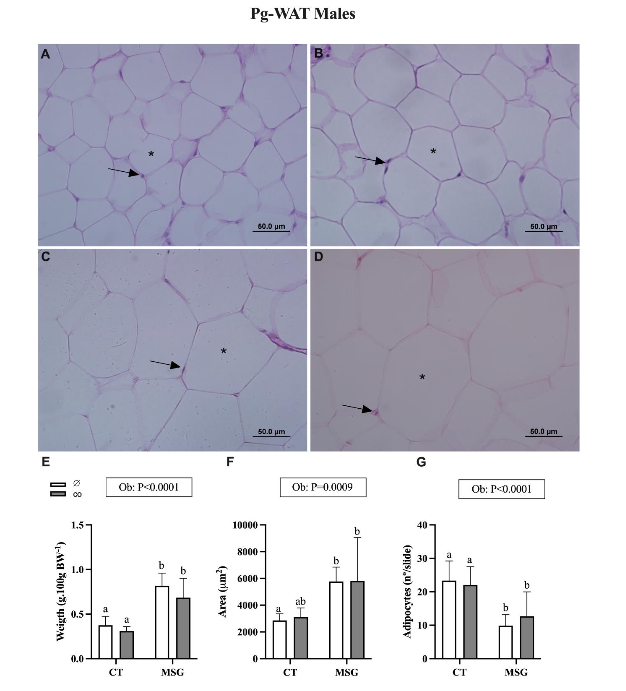
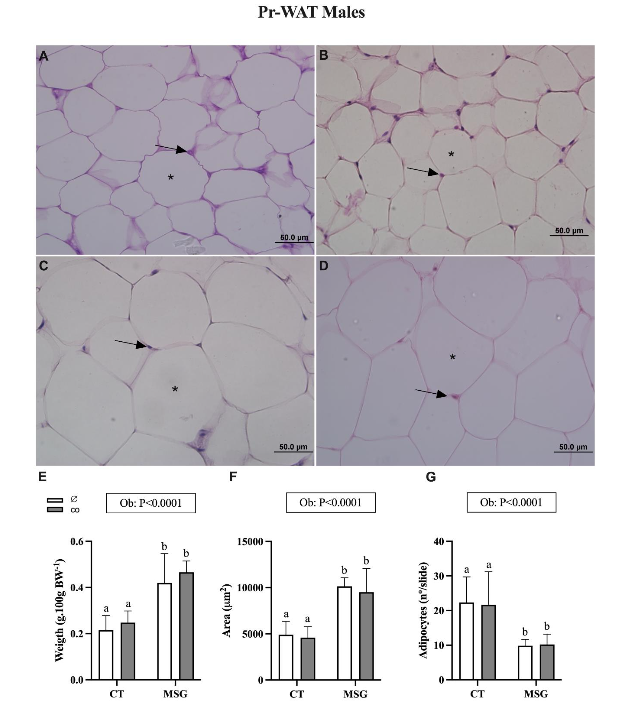
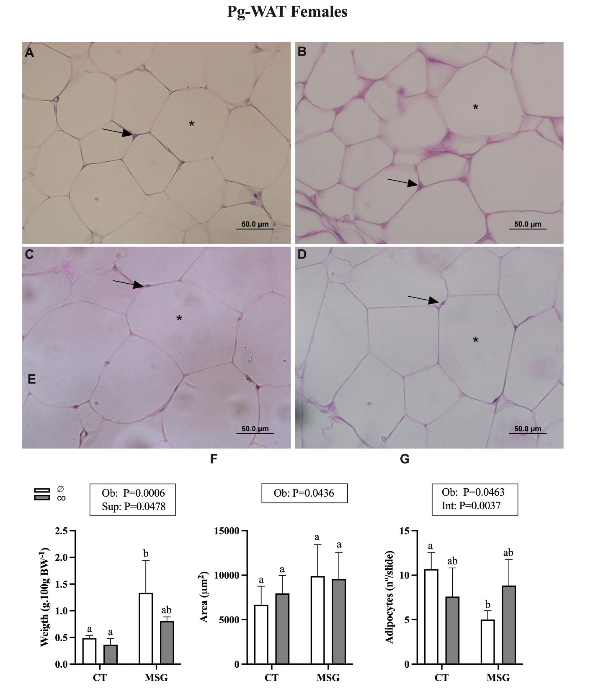
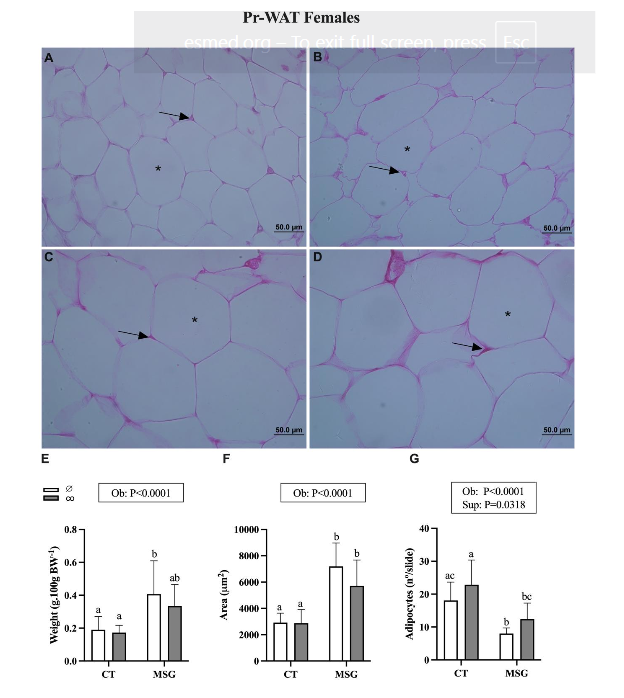
The Pg-WAT and Pr-WAT depots were submitted to the dehydration process in growing alcohol solution after they were diaphanized by xylol and embedded in paraffin (ACS Científica, Brazil). The tissue samples were cut with serial interrupted sectioning (7µm) at microtome (OLYMPUS CUT4055), and at least nine sections were transferred to slides and stained using the hematoxylin and eosin (H&E) technique. The slides were photographed in an Olympus BX60 microscope with an Olympus DP71 camera attached at 40X magnification. The ImageJ software (Version 1.53a, USA) was used to characterize the area (µm2) and number of adipocytes.
STATISTICAL ANALYSIS
Data were shown as mean ± standard deviation (SD). Normality was tested by the Shapiro-Wilk test. Factor influences like obesity (Ob), copaiba’s oil supplementation (Sup), and Interaction (Int) were calculated by ANOVA two-way followed by Tukey’s post hoc (p<0.05); differences between groups are evidenced by different letters.
RESULTS
IMPACT OF ORAL COPAIBA’S OIL SUPPLEMENTATION ON HYPOTHALAMIC OBESE AND NON-OBESE MALE WISTAR RATS (TABLE 1; FIGURES 2 AND 3)
At 90 PND, MSG treatment significantly influenced ∆BW [F(1,20)=0.5070; P<0.0001]; fasting values of glucose [F(1,20)=15.99; P=0.0007]; triglycerides [F(1,20)=28.64; P<0.0001] and TyG index [F(1,20)=44.15; P<0.0001]; in male Wistar rats. Thus, non-supplemented MSG∅ rats showed smaller ∆BW (38%) with elevated values of glucose (18%) and triglycerides (56%) plasma levels than CT∅. As a result, MSG∅ rats were insulin resistant, showing higher TyG values (8%) than CT∅ animals. CO-supplementation no alter ∆BW [F(1,20)=0.2564; P=0.6182], glucose [F(1,20)=0.4382; P=0.5155] or TyG index [F(1,18)=2.812; P=0.1108] in male MSG-obese and CT (Table 1). However, a significant influence of CO on triglyceride levels [F(1,20)=5.343; P=0.0316] in MSGco animals showed hypertriglyceridemia compared to CT∅, CTco, and MSG∅ groups. Interaction effects (Sup versus Ob) were not found in these variables.
| variables | Males | p-value |
|---|---|---|
| ∆ BW (g) | 271a | 0.4847 <0.0001 0.6182 |
| Glucose (mg.dL-1) | 87 a | 0.3141 0.0007 0.5155 |
| Triglycerides (mg.dL-1) | 57 a | 0.2887 <0.0001 0.0316 |
| TyG Index | 7.77 a | 0.5485 <0.0001 0.1108 |
Data are mean (SD) n=6. ∆BW: body weight gain. TyG: Triglycerides-glucose index. CT: control. MSG: obese. ∅non-supplemented and co co-supplemented. P-value shows effects (Ob: obesity; Sup: supplementation and Int: interaction) by Two-way ANOVA test (bold represent p<0.05). Different letters indicate differences in Tukey’s post hoc tests (p<0.05).
Figures 2 and 3 show the effect of MSG treatment and CO supplementation on visceral WAT content and histology of male Wistar rats. The content of visceral Pg-WAT [F(1,20)=50.39; P<0.0001] and Pr-WAT [F(1,20)=42.45; P<0.0001] depots were significantly influenced by MSG-treatment. In this manner, the weight of Pg-WAT and Pr-WAT increased by 118% (Figure 2E; P=0.0001) and 95% (Figure 3E; P=0.0013) in the MSG∅ than in the CT∅ group. Moreover, MSG treatment also changes adipocyte area in Pg-WAT [F(1,20)=15.26; P=0.0009] and Pr-WAT [F(1,20)=63.12; P<0.0001] depots. Thus, adipocytes from MSG∅ rats were significantly higher in the Pg-WAT 102% (Figure 2F; P=0.0435) and Pr-WAT 107% (Figure 3F; P<0.0001) than CT∅ animals. Similarly, the number of adipocytes was affected by MSG treatment in both visceral Pg-WAT [F(1,20)=24.38; P<0.0001] and Pr-WAT [F(1,20)=28.96; P<0.0001] depots. Then, non-supplemented MSG∅ animals showed reduced adipocyte numbers in Pg-WAT 57% (Figure 2G; P=0.028) and Pr-WAT 55% (Figure 3G; P=0.0024) depots compared to the same depots from CT∅ groups. CO supplementation did not modify Pg-WAT or Pr-WAT depots in males obese MSGco or non-obese CTco groups.
IMPACT OF ORAL COPAIBA’S OIL SUPPLEMENTATION ON HYPOTHALAMIC OBESE AND NON-OBESE FEMALE WISTAR RATS (TABLE 2; FIGURES 4 AND 5)
At 90 PND, MSG treatment significantly influenced ∆BW [F(1,20)=56.15; P<0.0001]; fasting triglycerides plasma levels [F(1,20)=26.16; P<0.0001] and TyG index [F(1,20)=30.50; P<0.0001] in female Wistar rats. Thus, non-supplemented MSG∅ rats showed smaller ∆BW (32%) and higher values of triglycerides (60%) and TyG (7%) in comparison to non-supplemented CT∅ (Table 2) without modifying fasting glucose levels. In females, CO supplementation affects ∆BW [F(1,20)=5.452; P=0.0301]. This variable was slightly elevated in the CTco and MSGco groups compared to respective non-supplemented animals (CT∅ and MSG∅). Moreover, we also noted Int (Sup versus Ob) effects on triglyceride plasma levels [F(1,20)=26.16; P<0.0001]). Thus, female MSGco has smaller (21%) triglyceride levels than MSG∅ (table 2). Fasting glucose and TyG index were not significantly altered by CO supplementation in the female groups.
| variables | Females | p-value |
|---|---|---|
| ∆ BW (g) | 144 a | 0.3947 <0.0001 0.0301 |
| Glucose (mg.dL-1) | 96 a | 0.1606 0.4251 0.7038 |
| Triglycerides (mg.dL-1) | 45 a | 0.0179 <0.0001 0.0770 |
| TyG Index | 7.69 a | 0.8969 <0.0001 0.4260 |
Data are mean (SD) n=6. ∆BW: body weight gain. TyG: Triglycerides-glucose index. CT: control. MSG: obese. ∅non-supplemented and co co-supplemented. P-value shows effects (Ob: obesity; Sup: supplementation and Int: interaction) by Two-way ANOVA test (bold represent p<0.05). Different letters indicate differences in Tukey’s post hoc tests (p<0.05).
In the females, the MSG treatment also influenced the content of visceral Pg-WAT [F(1,17)=17.66; P=0.0006] and Pr-WAT [F(1,20)=12.85; P=0.0019] depots. Thus, the weight of Pg-WAT and Pr-WAT were increased by 173% (Figure 4E; P=0.0115) and 114% (Figure 5E; P=0.0019) in the MSG∅ female rats compared to the CT∅ female rats. Herein, CO supplementation influences the Pg-WAT depot’s weight [F(1,17)=4.548; P=0.0478], with MSGco animals presenting a similar weight of Pg-WAT than CT∅. CO supplementation does not alter the weight of Pr-WAT in obese (MSGco) or non-obese (CTco) females. MSG treatment also modifies adipocyte area in visceral Pg-WAT [F(1,20)=4.642; P=0.0436] and Pr-WAT [F(1,26)=41.94; P<0.0001] depots from female groups. Thus, non-supplemented MSG∅ rats presented higher adipocyte size in Pr-WAT 146% (Figure 5F; P<0,0001) but no significant difference in Pg-WAT (Figure 4F; P=0.0657) depots in comparison to CT∅ animals. Similarly, the number of adipocytes was affected by MSG treatment in both visceral Pg-WAT [F(1,18)=4.579; P=0.0463] and Pr-WAT [F(1,24)=25.92; P<0.0001] depots. Then, non-supplemented MSG∅ animals showed reduced adipocyte numbers in Pg-WAT 52% (Figure 4G; P=0.0057) and Pr-WAT 55% (Figure 5G; P=0.0084) depots compared to CT∅ groups. The size of adipocytes in the Pg-WAT and Pr-WAT depots was not altered by CO supplementation in females, obese (MSGco), or non-obese (CTco) groups. However, the number of adipocytes in the Pr-WAT depot was modified by CO supplementation [F(1,24)=5.200; P=0.0318]. Thus, the number of adipocytes in the Pr-WAT depot from MSGco was similar to that observed in CT∅ (Figure 5G, P=0.2205). Moreover, the Int effect (Ob versus Sup) was observed in adipocyte numbers of Pg-WAT depot of female groups [F(1,18)=11.09; P=0.0037] with MSGco rats presenting similar to CT∅ animals (Figure 4G; P=5672).
DISCUSSION
The hypothalamus is an essential neuronal center regulator of energy homeostasis and hypothalamic lesions induced by high doses of MSG are a well-known model for hypothalamic obesity induction. In the present study, we confirm the efficacy of neonatal MSG treatment to induce obesity accompanied by metabolic abnormalities. Thus, we noted that MSG-obese rodents showed, despite reduced BW, high visceral WAT content, hypertriglyceridemia, and insulin resistance in both sexes. We and others have shown that lipogenic effects of insulin have a relevant role in augmented adiposity in MSG-obese rodents.
Hypothalamic obesity MSG-induced in rats is associated with reduced growth hormone (GH) levels, which is known for its well-established lipolytic effect on WAT. Additionally, this obesity model exhibits elevated corticosterone levels, a condition that promotes visceral WAT deposition. Moreover, the autonomic imbalances, typically observed in MSG-obese rodents, contribute to WAT expansion. In this context, vagal hyperactivity induces hyperinsulinemia, which in turn stimulates adipogenesis and lipogenesis, while sympathetic hypofunction diminishes WAT’s lipolytic activity, further contributing to its expansion. Similar endocrine, autonomic, and metabolic disturbances observed in MSG-obese rodents are also found in humans with hypothalamic obesity.
Sexual hormones strongly influence metabolism, determining WAT distribution and its consequences on immune and energy homeostasis. Several studies have proved that MSG treatment exerts different effects on female and male rodents. In agreement, we noted that sexes influence some characteristics in the MSG-obese model. Thus, in our data, male MSG-obese rats showed fasting hyperglycemia, a condition not found in female MSG-obese animals. Similarly, Hernández Bautista et al. (2019) found higher glucose intolerance in male MSG-obese rats. These data also confirm human findings that those men presenting more visceral WAT content present a greater risk of diabetes in comparison to obese women. Reduction in testosterone levels in obese men appears to be responsible for these deleterious effects on glucose homeostasis. Low testosterone levels are associated with increased visceral WAT content, intimately related to energy imbalance, glucose intolerance, insulin resistance, and dyslipidemia.
It is important to notice that after hypothalamic lesions induced by MSG, gonadal atrophy occurs with a significant reduction in testosterone levels. Considering sex influences metabolism, it is common to observe different responses to anti-obesity treatment between males and females and women and men. For this reason, our study evaluated the effects of oral and chronic CO-supplementation on adiposity and WAT histology, exploring sex influences. CO is a natural compound used as an anti-inflammatory, antimicrobial, and cicatrizing agent, especially by Brazilians, but is also freely commercialized in other countries. More recent studies have pointed out that this oil would show anti-adiposity effects, whose mechanisms have yet to be revealed.
As fat mass expansion defines obesity, our first question was whether chronic CO supplementation offered at long life could avoid visceral WAT expansion in male and female hypothalamic obese rodents? In male MSG-obese rats, we found no effects of CO-supplementation in body weight or visceral WAT depot content. Moreover, in the male MSG-obese group, the CO-supplementation does not change adipocyte area or number in the visceral WAT depot, suggesting no preventive effects of this oil on fat mass expansion. In contrast, a study using oral CO supplementation (200 mg.kg-1.day-1 for eight weeks) showed reduced adiposity and inflammatory process in obesity induced by a high sucrose diet. These authors also noted a reduction in visceral WAT depots accompanied by improved lipid profile, fasting glucose levels, and insulin resistance. At present work, none of these data were confirmed in the MSG-obese male rats. In contrast, we noted that chronic oral CO-supplementation worsens triglyceride levels in the male MSG-obese model. Males MSG-obese rodents are responsible for other anti-adiposity strategies, such as exercise, bariatric surgery, and taurine supplementation. We have demonstrated that oral vitamin D supplementation improved metabolic metabolism without altering adiposity in male MSG-obese rats. These data suggest that MSG-obese male rats can be resistant to the anti-adiposity effects of CO supplementation.
Obesity-induced by high carbohydrate or fat diets normally exerts more deleterious effects on the liver and inflammatory processes than the hypothalamic obese model, and these points appear to be more positively improved by oral CO-supplementation. Moreover, it is important to remember that hypothalamic obese rats do not show hyperphagia, present lower GH and testosterone levels, and have higher corticosterone concentrations conditions that could influence CO effects. The absence of anti-adiposity effects in male lean rats was also noted by Paula et al. (2023), corroborating the present study, where males and females who were non-obese (CT) were unresponsive to CO supplementation effects.
Contrary to MSG-obese male rats, we noted influences of oral CO-supplementation in MSG-obese females. Thus, female MSG-obese supplemented with CO showed reduced triglyceride plasma levels and a slight reduction in Pg-WAT content without altering insulin sensibility. Moreover, our data from histological analyses of visceral WAT depots show that oral CO supplementation elevates adipocyte nuclei number in MSG-obese females despite no change in adipocyte size. Thus, our results indicate more health benefits of oral CO supplementation to visceral WAT of MSG-obese females.
Molecular in vitro studies have shown that CO can stimulate phosphatidylinositol 3′-kinase (PI3K)-protein kinase B (PI3K-Akt) and the mitogen-activated protein kinase (MAPK) and JAK/STAT signaling pathways in several cellular types. These pathways are also involved in adipogenesis in WAT. However, whether CO supplementation can stimulate these mechanisms in WAT is unknown.
It is well recognized that visceral WAT depot expansion preferentially by hypertrophy. Thus, at least in female MSG-obese, CO supplementation appears to raise the proliferation capacity of visceral WAT event, which would explain the reduction in triglyceride levels found in female MSG-obese. The female mice presented higher adipogenesis capability on the subcutaneous WAT when fed with a high-fat diet. Although subcutaneous and visceral WAT depots utilize different cellular and molecular mechanisms to regulate adipogenesis; our results suggest that visceral WAT in females is more responsive to adipogenesis induced by CO supplementation than in male obese rats. Sex differences in adipogenesis control have been demonstrated previously.
To our knowledge, no study explores adipogenesis or lipogenesis on WAT of rodents supplemented with CO, and the probable mechanisms of CO in this tissue are unknown. Molecular studies with CO in other cellular types can offer some clues. Studies in neuronal cells indicate that cannabinoid receptors subtype 2 (CB2) mediated the effects of CO on the pI3K/Akt/mTOR signaling pathway. Endocannabinoid systems are represented in the WAT, which regulates critical homeostatic processes, including adipogenesis and lipogenesis, expressing the cannabinoid receptors CB1 and CB2. Interestingly, the CB2 receptor stimulates proliferation on WAT depot. Then, we speculated that oral CO supplementation would modulate this receptor pathway in the visceral WAT of MSG-obese females. It has been shown that endocannabinoid activation on the hypothalamus differs in male and female obese mice.
Finally, we cannot exclude the possibility that CO supplementation would affect metabolism via CNS and from the intestine, activating vagal afferent pathways. Unfortunately, none of these mechanisms are investigated. Still, it is important to consider that MSG-obese shows hypervagal dysfunctions and hypothalamic lesions, conditions that may explain the reduced anti-adiposity effects of CO supplementation on this obesity model.
Our study presents some important limitations among those we emphasized. First, the commercially traded CO, as used here, is often a mixture of oleoresin from many trees and species, and it is challenging to recognize the principal compound responsible for its effects. However, the primary compounds are generally very similar between different analyses performed. For example, terpenes (approximately 60 types) are frequent in CO, with β-caryophyllene the dominant terpene, representing approximately 51% of the total terpenes in CO. Second, CO exerts differential effects on the molecular signaling pathways in different cell type. Moreover, the opposite effects of isolated β-caryophyllene compound and CO signaling pathways indicate that complex interactions of its constituents are important to determine its functional effects.
CONCLUSION
In conclusion, chronic and oral CO supplementation could not avoid obesity development and visceral WAT expansion in the MSG-treated rats. Sexes influence CO supplementation effects on WAT and metabolism. Thus, MSG-obese female CO-supplemented showed a slight reduction in adiposity with increased adipocyte proliferation and improved fasting triglycerides plasma levels. Further studies are necessary to clarify eventual molecular CO mechanisms in WAT from MSG-obese females.
Conflicts of Interest Statement
The authors have no conflicts of interest to declare.
Funding Statement
Programa de Apoio à Pós-Graduação (PROAP), Fundação Coordenação de Aperfeiçoamento de Pessoal de Nível Superior CAPES, Grant nº 88887.695630/2022-00. Doctorate scholarships.
Acknowledgments
We thank Amazon Oil for donating Copaiba’s oil used in this experiment.
References
- Kershaw EE, Flier JS. Adipose Tissue as an Endocrine Organ. J Clin Endocrinol Metab. 2004;89(6):2548-2556. doi:10.1210/jc.2004-0395
- Wellen KE, Hotamisligil GS. Inflammation, stress, and diabetes. Journal of Clinical Investigation. 2005;115(5):1111-1119. doi:10.1172/JCI200525102
- Kawai T, Autieri M V., Scalia R. Adipose tissue inflammation and metabolic dysfunction in obesity. American Journal of Physiology-Cell Physiology. 2021;320(3):C375-C391. doi:10.1152/ajpcell.00379.2020
- Alberti KGM, Zimmet P, Shaw J. The metabolic syndrome—a new worldwide definition. The Lancet. 2005;366(9491):1059-1062. doi:10.1016/S0140-6736(05)67402-8
- Chait A, den Hartigh LJ. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front Cardiovasc Med. 2020;7. doi:10.3389/fcvm.2020.00022
- Gray SL, Vidal-Puig AJ. Adipose Tissue Expandability in the Maintenance of Metabolic Homeostasis. Nutr Rev. 2007;65(SUPPL.1):7-12. doi:10.1111/j.1753-4887.2007.tb00331.x
- Suzuki M, Shinohara Y, Ohsaki Y, Fujimoto T. Lipid droplets: size matters. Microscopy. 2011;60(suppl 1):S101-S116. doi:10.1093/jmicro/dfr016
- Crandall DL, Hausman GJ, Kral JG. A Review of the Microcirculation of Adipose Tissue: Anatomic, Metabolic, and Angiogenic Perspectives. Microcirculation. 1997;4(2):211-232. doi:10.3109/10739689709146786
- Nookaew I, Svensson PA, Jacobson P, et al. Adipose Tissue Resting Energy Expenditure and Expression of Genes Involved in Mitochondrial Function Are Higher in Women than in Men. J Clin Endocrinol Metab. 2013;98(2):E370-E378. doi:10.1210/jc.2012-2764
- Mauvais-Jarvis F. Sex differences in metabolic homeostasis, diabetes, and obesity. Biol Sex Differ. 2015;6(1):14. doi:10.1186/s13293-015-0033-y
- Li J, Ruggiero-Ruff RE, He Y, et al. Sexual dimorphism in obesity is governed by RELMα regulation of adipose macrophages and eosinophils. Elife. 2023;12. doi:10.7554/eLife.86001
- Mauvais-Jarvis F. Sex differences in energy metabolism: natural selection, mechanisms and consequences. Nat Rev Nephrol. 2024;20(1):56-69. doi:10.1038/s41581-023-00781-2
- Santos-Marcos JA, Mora-Ortiz M, Tena-Sempere M, Lopez-Miranda J, Camargo A. Interaction between gut microbiota and sex hormones and their relation to sexual dimorphism in metabolic diseases. Biol Sex Differ. 2023;14(1):4. doi:10.1186/s13293-023-00490-2
- Steiner BM, Berry DC. The Regulation of Adipose Tissue Health by Estrogens. Front Endocrinol (Lausanne). 2022;13. doi:10.3389/fendo.2022.889923
- Li S ying, Zhao Y ling, Yang Y fan, et al. Metabolic Effects of Testosterone Replacement Therapy in Patients with Type 2 Diabetes Mellitus or Metabolic Syndrome: A Meta-Analysis. Int J Endocrinol. 2020:1-12. doi:10.1155/2020/4732021
- Groti K, Žuran I, Antonič B, Foršnarič L, Pfeifer M. The impact of testosterone replacement therapy on glycemic control, vascular function, and components of the metabolic syndrome in obese hypogonadal men with type 2 diabetes. The Aging Male. 2018;21(3):158-169. doi:10.1080/13685538.2018.1468429
- World Health Organization. World Health Statistics 2023: Monitoring Health for the SDGs, Sustainable Development Goals.; 2023.
- Ornellas F, Mello VS, Mandarim-de-Lacerda CA, Aguila MB. Sexual dimorphism in fat distribution and metabolic profile in mice offspring from diet-induced obese mothers. Life Sci. 2013;93(12-14):454-463. doi:10.1016/j.lfs.2013.08.005
- Dhanraj P, van Heerden MB, Pepper MS, Ambele MA. Sexual Dimorphism in Changes That Occur in Tissues, Organs and Plasma during the Early Stages of Obesity Development. Biology (Basel). 2021;10(8):717. doi:10.3390/biology10080717
- Shanley P, Margaret C, Murilo S, Gabriel M. Fruit trees and useful plants in Amazonian life. In: Food and Agriculture Organization of the United Nations, the Center for International Forestry Res; 2011.
- Telles LO, Silva BS da, Paulino AMB, et al. Copaiba oleoresin presents anti-obesogenic effect and mitigates inflammation and redox imbalance in adipose tissue. Acta Amazon. 2022;52(4):331-338. doi:10.1590/1809-4392202201411
- De Paula MG, Rocha LA, Telles LO, et al. Óleo-resina de Copaíba atenua ganho de peso e não altera marcadores inflamatórios e do sistema redox no tecido adiposo de animais saudáveis. Scientific Electronic Archives. 2023;16(4). doi:10.36560/16420231694
- Veiga VF, Rosas EC, Carvalho MV, Henriques MGMO, Pinto AC. Chemical composition and anti-inflammatory activity of copaiba oils from Copaifera cearensis Huber ex Ducke, Copaifera reticulata Ducke and Copaifera multijuga Hayne—A comparative study. J Ethnopharmacol. 2007;112(2):248-254. doi:10.1016/j.jep.2007.03.005
- Dias D, Fontes L, Crotti A, et al. Copaiba Oil Suppresses Inflammatory Cytokines in Splenocytes of C57Bl/6 Mice Induced with Experimental Autoimmune Encephalomyelitis (EAE). Molecules. 2014;19(8):12814-12826. doi:10.3390/molecules190812814
- Santos AO dos, Ueda-Nakamura T, Dias Filho BP, Veiga Junior VF, Pinto AC, Nakamura CV. Antimicrobial activity of Brazilian copaiba oils obtained from different species of the Copaifera genus. Mem Inst Oswaldo Cruz. 2008;103(3):277-281. doi:10.1590/S0074-02762008005000015
- Santos AO, Ueda-Nakamura T, Dias Filho BP, Veiga Junior VF, Pinto AC, Nakamura CV. Effect of Brazilian copaiba oils on Leishmania amazonensis. J Ethnopharmacol. 2008;120(2):204-208. doi:10.1016/j.jep.2008.08.007
- Kian D, Lancheros CAC, Assolini JP, et al. Trypanocidal activity of copaiba oil and kaurenoic acid does not depend on macrophage killing machinery. Biomedicine & Pharmacotherapy. 2018;103:1294-1301. doi:10.1016/j.biopha.2018.04.164
- Urasaki Y, Beaumont C, Talbot JN, Hill DK, Le TT. Akt3 Regulates the Tissue-Specific Response to Copaiba Essential Oil. Int J Mol Sci. 2020;21(8):2851. doi:10.3390/ijms21082851
- Caputo LS, Campos MIC, Dias HJ, et al. Copaiba oil suppresses inflammation in asthmatic lungs of BALB/c mice induced with ovalbumin. Int Immunopharmacol. 2020;80:106177. doi:10.1016/j.intimp.2019.106177
- Lopes LN, Santos FAF, Oliveira LCM, Percário S, Barros CAV de, Brito MVH. Copaiba oil effect on induced fecal peritonitis in rats. Acta Cir Bras. 2015;30(8):568-573. doi:10.1590/S0102-865020150080000008
- Urasaki Y, Beaumont C, Workman M, Talbot JN, Hill DK, Le TT. Fast-Acting and Receptor-Mediated Regulation of Neuronal Signaling Pathways by Copaiba Essential Oil. Int J Mol Sci. 2020;21(7):2259. doi:10.3390/ijms21072259
- Kuchler JC, Siqueira BS, Ceglarek VM, et al. The Vagus Nerve and Spleen: Influence on White Adipose Mass and Histology of Obese and Non-obese Rats. Front Physiol. 2021;12. doi:10.3389/fphys.2021.672027
- Balbo SL, Grassiolli S, Ribeiro RA, et al. Fat storage is partially dependent on vagal activity and insulin secretion of hypothalamic obese rat. Endocrine. 2007;31(2):142-148. doi:10.1007/s12020-007-0021-z
- Olney JW. Brain Lesions, Obesity, and Other Disturbances in Mice Treated with Monosodium Glutamate. Science (1979). 1969;164(3880):719-721. doi:10.1126/science.164.3880.719
- Lubaczeuski C, Balbo SL, Ribeiro RA, et al. Vagotomy ameliorates islet morphofunction and body metabolic homeostasis in MSG-obese rats. Brazilian Journal of Medical and Biological Research. 2015;48(5):447-457. doi:10.1590/1414-431X20144340
- Galúcio C de S, Benites CI, Rodrigues RAF, Maciel MRW. Sesquiterpenes Recovery Of Copaiba Oil-Resin From Molecular Distillation. Quim Nova. Published online 2016. doi:10.5935/0100-4042.20160096
- Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord. 2008;6(4):299-304. doi:10.1089/met.2008.0034
- Waterson MJ, Horvath TL. Neuronal Regulation of Energy Homeostasis: Beyond the Hypothalamus and Feeding. Cell Metab. 2015;22(6):962-970. doi:10.1016/j.cmet.2015.09.026
- Meister B, Ceccatelli S, Håkfelt T, Andén NE, Andén M, Theodorsson E. Neurotransmitters, neuropeptides and binding sites in the rat mediobasal hypothalamus: effects of monosodium glutamate (MSG) lesions. Exp Brain Res. 1989;76(2). doi:10.1007/BF00247894
- Scallet AC, Olney JW. Components of hypothalamic obesity: bipiperidyl-mustard lesions add hyperphagia to monosodium glutamate-induced hyperinsulinemia. Brain Res. 1986;374(2):380-384. doi:10.1016/0006-8993(86)90434-8
- Balbo SL, Gravena C, Bonfleur ML, de Freitas Mathias PC. Insulin Secretion and Acetylcholinesterase Activity in Monosodium L-Glutamate-InducedObese Mice. Horm Res Paediatr. 2000;54(4):186-191. doi:10.1159/000053257
- Hernández Bautista RJ, Mahmoud AM, Königsberg M, López Díaz Guerrero NE. Obesity: Pathophysiology, monosodium glutamate-induced model and anti-obesity medicinal plants. Biomedicine & Pharmacotherapy. 2019;111:503-516. doi:10.1016/j.biopha.2018.12.108
- Hirata AE, Andrade IS, Vaskevicius P, Dolnikoff MS. Monosodium glutamate (MSG)-obese rats develop glucose intolerance and insulin resistance to peripheral glucose uptake. Brazilian Journal of Medical and Biological Research. 1997;30(5):671-67. doi:10.1590/S0100-879X1997000500016
- Kopchick JJ, Berryman DE, Puri V, Lee KY, Jorgensen JOL. The effects of growth hormone on adipose tissue: old observations, new mechanisms. Nat Rev Endocrinol. 2020;16(3):135-146. doi:10.1038/s41574-019-0280-9
- Miranda RA, Torrezan R, de Oliveira JC, et al. HPA axis and vagus nervous function are involved in impaired insulin secretion of MSG-obese rats. Journal of Endocrinology. 2016;230(1):27-38. doi:10.1530/JOE-15-0467
- Scomparin DX, Gomes RM, Grassiolli S, et al. Autonomic activity and glycemic homeostasis are maintained by precocious and low intensity training exercises in MSG-programmed obese mice. Endocrine. 2009;36(3):510-517. doi:10.1007/s12020-009-9263-2
- Roth CL, Zenno A. Treatment of hypothalamic obesity in people with hypothalamic injury: new drugs are on the horizon. Front Endocrinol (Lausanne). 2023;14. doi:10.3389/fendo.2023.1256514
- Sara Cristina Sagae, Sabrina Grassiolli, Charlis Raineki, Sandra Lucinei Balbo, Ana Carla Marques da Silva. Sex differences in brain cholinergic activity in MSG-obese rats submitted to exercise. Can J Physiol Pharmacol. 2011;89(11):845-853.
- Pimenta F da S, Tose H, Jr ÉW, et al. Lipectomy associated to obesity produces greater fat accumulation in the visceral white adipose tissue of female compared to male rats. Lipids Health Dis. 2019;18(1):44. doi:10.1186/s12944-019-0988-5
- Kong M, Xu M, Zhou Y, et al. Assessing Visceral Obesity and Abdominal Adipose Tissue Distribution in Healthy Populations Based on Computed Tomography: A Large Multicenter Cross-Sectional Study. Front Nutr. 2022;9. doi:10.3389/fnut.2022.871697
- Allan CA, McLachlan RI. Androgens and obesity. Curr Opin Endocrinol Diabetes Obes. 2010;17(3):224-232. doi:10.1097/MED.0b013e3283398ee2
- Kelly DM, Jones TH. Testosterone and obesity. Obesity Reviews. 2015;16(7):581-606. doi:10.1111/obr.12282
- Kayode OT, Rotimi DE, Kayode AAA, Olaolu TD, Adeyemi OS. Monosodium Glutamate (MSG)-Induced Male Reproductive Dysfunction: A Mini Review. Toxics. 2020;8(1):7. doi:10.3390/toxics8010007
- Muscogiuri G, Verde L, Vetrani C, Barrea L, Savastano S, Colao A. Obesity: a gender-view. J Endocrinol Invest. 2023;47(2):299-306. doi:10.1007/s40618-023-02196-z
- D’Archivio M, Coppola L, Masella R, Tammaro A, La Rocca C. Sex and Gender Differences on the Impact of Metabolism-Disrupting Chemicals on Obesity: A Systematic Review. Nutrients. 2024;16(2):181. doi:10.3390/nu16020181
- Paula MG de, Rocha LA, Silva IL, et al. Óleo-resina de copaíba diminui o índice de adiposidade e melhora o sistema redox, os níveis de IL-10 e função renal de ratos submetidos à dieta rica em sacarose. Brazilian Journal of Health Review. 2024;7(3):e70425. doi:10.34119/bjhrv7n3-332
- Guareschi ZM, Ceglarek VM, Rodrigues PF, et al. Exercise and Vitamin D Supplementation Modify Spleen Morphology in Lean, but not, in Monosodium-Glutamate-Obese Rats. Journal of Spleen and Liver Research. 2019;1(3):1-14. doi:10.14302/issn.2578-2371.jslr-19-2819
- Andreazzi AE, Scomparin DX, Mesquita FP, et al. Swimming exercise at weaning improves glycemic control and inhibits the onset of monosodium l-glutamate-obesity in mice. Journal of Endocrinology. 2009;201(3):351-359. doi:10.1677/JOE-08-0312
- Soares GM, Cantelli KR, Balbo SL, et al. Liver steatosis in hypothalamic obese rats improves after duodeno-jejunal bypass by reduction in de novo lipogenesis pathway. Life Sci. 2017;188:68-75. doi:10.1016/j.lfs.2017.08.035
- Nardelli TR, Ribeiro RA, Balbo SL, et al. Taurine prevents fat deposition and ameliorates plasma lipid profile in monosodium glutamate-obese rats. Amino Acids. 2011;41(4):901-908. doi:10.1007/s00726-010-0789-7
- Guareschi ZM, Valcanaia AC, Ceglarek VM, et al. The effect of chronic oral vitamin D supplementation on adiposity and insulin secretion in hypothalamic obese rats. British Journal of Nutrition. 2019;121(12):1334-1344. doi:10.1017/S0007114519000667
- Willett RA, Tryndyak VP, Hughes Hanks JM, et al. A preclinical model of severe NASH-like liver injury by chronic administration of a high-fat and high-sucrose diet in mice. Toxicol Appl Pharmacol. 2024;491:117046. doi:10.1016/j.taap.2024.117046
- Castro Ghizoni C V., Arssufi Ames AP, Lameira OA, et al. Anti‐Inflammatory and Antioxidant Actions of Copaiba Oil Are Related to Liver Cell Modifications in Arthritic Rats. J Cell Biochem. 2017;118(10):3409-3423. doi:10.1002/jcb.25998
- Torrezan R, Malta A, de Souza Rodrigues W do N, et al. Monosodium l-glutamate-obesity onset is associated with disruption of central control of the hypothalamic-pituitary-adrenal axis and autonomic nervous system. J Neuroendocrinol. 2019;31(6). doi:10.1111/jne.12717
- Khan T, Muise ES, Iyengar P, et al. Metabolic Dysregulation and Adipose Tissue Fibrosis: Role of Collagen VI. Mol Cell Biol. 2009;29(6):1575-1591. doi:10.1128/MCB.01300-08
- Dong H, Sun W, Shen Y, et al. Identification of a regulatory pathway inhibiting adipogenesis via RSPO2. Nat Metab. 2022;4(1):90-105. doi:10.1038/s42255-021-00509-1
- Shao M, Vishvanath L, Busbuso NC, et al. De novo adipocyte differentiation from Pdgfrβ+ preadipocytes protects against pathologic visceral adipose expansion in obesity. Nat Commun. 2018;9(1):890. doi:10.1038/s41467-018-03196-x
- Shao M, Hepler C, Zhang Q, et al. Pathologic HIF1α signaling drives adipose progenitor dysfunction in obesity. Cell Stem Cell. 2021;28(4):685-701.e7. doi:10.1016/j.stem.2020.12.008
- Saavedra-Peña R del M, Taylor N, Flannery C, Rodeheffer MS. Estradiol cycling drives female obesogenic adipocyte hyperplasia. Cell Rep. 2023;42(4):112390. doi:10.1016/j.celrep.2023.112390
- Shan B, Barker CS, Shao M, Zhang Q, Gupta RK, Wu Y. Multilayered omics reveal sex- and depot-dependent adipose progenitor cell heterogeneity. Cell Metab. 2022;34(5):783-799.e7. doi:10.1016/j.cmet.2022.03.012
- Jung KM, Lin L, Piomelli D. The endocannabinoid system in the adipose organ. Rev Endocr Metab Disord. 2022;23(1):51-60. doi:10.1007/s11154-020-09623-z
- Ruhl T, Karthaus N, Kim BS, Beier JP. The endocannabinoid receptors CB1 and CB2 affect the regenerative potential of adipose tissue MSCs. Exp Cell Res. 2020;389(1):111881. doi:10.1016/j.yexcr.2020.111881
- Miralpeix C, Fosch A, Casas J, et al. Hypothalamic endocannabinoids inversely correlate with the development of diet-induced obesity in male and female mice. J Lipid Res. 2019;60(7):1260-1269. doi:10.1194/jlr.M092742
- Hawwal MF, Ali Z, Wang M, et al. (E)-2,6,10-Trimethyldodec-8-en-2-ol: An Undescribed Sesquiterpenoid from Copaiba Oil. Molecules. 2021;26(15):4456. doi:10.3390/molecules26154456

