Distinguishing Non-Neoplastic Pancreatic Masses
Diagnostic strategy to distinguish non-neoplastic masses of the pancreas from pancreatic cancer
Sahar Mack¹, Suzanne Chartier², Jean-Louis Frossard¹
- Department of Medical Specialties, Division of Gastroenterology, University Hospital of Geneva, Switzerland
- Department of Pathology, University Hospital of Geneva, Switzerland University Hospital of Geneva, Switzerland
OPEN ACCESS
PUBLISHED:30 October 2024
CITATION: Mack, S., et al., 2024. Diagnostic strategy to distinguish non-neoplastic masses of the pancreas from pancreatic cancer. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5925
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i10.5925
ISSN: 2375-1924
ABSTRACT
Background:
Pancreatic cancer presents a significant challenge in oncology due to its aggressive nature and poor prognosis. Obtaining an accurate diagnosis can be difficult because of the presence of mimicking masses, which may correspond to chronic pancreatitis, pancreatic neuroendocrine tumors, pancreatic pseudocysts, autoimmune pancreatitis, or primary pancreatic lymphomas. The aim of this paper is to understand the distinctive features of these diseases in order to guide appropriate management and improve patient outcomes.
Methods:
This article reviews the clinical presentation, diagnostic modalities, and differential diagnosis of pancreatic cancer and its mimicking masses.
Results:
Pancreatic cancer often presents with nonspecific symptoms such as abdominal pain, weight loss, and jaundice. It requires a multidisciplinary diagnostic approach that includes clinical evaluation, imaging, laboratory tests, and, when possible, histological samplings. Mimicking masses such as chronic pancreatitis exhibit overlapping symptoms and imaging features with pancreatic cancer, making histopathological evaluations essential for differentiation. Pancreatic pseudocysts may mimic cancer on imaging but lack malignant potential, often resolving spontaneously or requiring a minimally invasive drainage procedure. Pancreatic neuroendocrine tumors, characterized by hormone secretion and distinct histological patterns, require specialized imaging modalities and tissue biopsy for accurate diagnosis. Autoimmune pancreatitis and primary pancreatic lymphomas present diagnostic challenges due to their autoimmune and lymphoproliferative symptoms, respectively, necessitating a combination of serological, histopathological, radiological and molecular analyses to obtain a definitive diagnosis.
Conclusion:
Multidisciplinary approach integrating clinical expertise, advanced imaging techniques, and molecular diagnostics are crucial to diagnose pancreatic cancer and its mimicking masses. A good understanding of distinctive features can help for an accurate diagnosis and guide optimal management strategies. Continued research efforts are warranted to enhance diagnostic precision, identify novel biomarkers, and improve therapeutic outcomes for patients affected by these challenging conditions.
Keywords:
pancreatic cancer, chronic pancreatitis, pancreatic pseudocysts, pancreatic neuroendocrine tumors, IgG4-related disease, autoimmune pancreatitis, pancreatic lymphoma.
Introduction
Pancreatic cancer poses a significant challenge in the field of oncology¹. Due to its aggressive nature, late presentation, and limited treatment options, it is one of the deadliest forms of cancer.
Despite advances in medical imaging and molecular diagnostics, differentiating pancreatic cancer from other pancreatic and peri-pancreatic masses remains a complex diagnostic challenge²˒³. Furthermore, the number of pancreatic masses identified has increased over the past decade, and probably because of the expanded use of radiological imaging and the higher incidence of pancreatic cancer, this number will continue to increase.
Benign mass-like lesions of the pancreas represent a heterogenous group of diseases, including chronic pancreatitis (CP), pancreatic pseudocysts (PPs), pancreatic neuroendocrine tumors (PNETs), autoimmune pancreatitis (AIP), or primary pancreatic lymphomas (PPL). These diseases can present with a variety of clinical signs and symptoms that often mimic those of pancreatic cancer, particularly pancreatic ductal adenocarcinoma (PDAC). The overlap in clinical and radiological presentation, can make the distinction between benign pancreatic disease and malignancy challenging.
According to several guidelines, core biopsy investigations are required to exclude pancreatic cancer whenever feasible⁴. It is critical to distinguish pancreatic cancer from other benign pancreatic diseases, as it directly influences treatment decisions and patient outcomes and precludes any surgery if a benign disease is diagnosed.
Table 1 provides a summary of the distinguishing features between PDAC and its differential diagnoses. This article aims to elucidate the differential diagnoses of pancreatic cancer by describing both malignant and benign entities that mimic its presentation.
PANCREATIC CANCER: SYMPTOMS AND DIAGNOSIS
Pancreatic ductal adenocarcinoma, which constitutes approximately 90% of all pancreatic malignancies, originates most commonly in the exocrine component of the pancreas¹. According to recent statistics, pancreatic cancer has a 5-year survival rate of around 10% due to its silent progression, often remaining asymptomatic until the advances stages⁵˒⁶. About 40% of patients with pancreatic cancer are smokers. Other risk factors include aging, obesity, male sex, type 2 diabetes, and previous exposure to pesticides⁷.
Pancreatic cancer often presents insidiously with nonspecific symptoms such as abdominal pain, weight loss, jaundice, and gastrointestinal disturbances. These symptoms arise from the tumors’ effects on pancreatic function, local tissue invasion, and systemic alterations in metabolism and hormone regulation. However, the overlapping of symptoms with other pancreatic diseases complicates the diagnostic process.
Advanced imaging modalities play a crucial role in detecting and characterizing pancreatic lesions. Computed tomography (CT), scan, magnetic resonance imaging (MRI), and endoscopic ultrasound (EUS) offer detailed anatomical information and contribute to assessing tumor size, vascular involvement, and distant metastases⁸. Additionally, functional imaging techniques such as positron emission tomography (PET) can provide valuable insights into tumor metabolism and extent².
Laboratory tests, including serum biomarkers such as carbohydrate antigen 19-9 (CA 19-9), are used in complement to imaging studies⁹˒¹⁰. However, this marker has poor sensitivity and specificity¹¹. Other biomarkers under investigation, including circulating tumor DNA and exosomes, hold promise for improving early detection and monitoring treatment response¹⁰˒¹².
Histological confirmation of the diagnosis remains highly recommended. Microscopically, PDAC most often exhibits a marked desmoplastic stroma. Well- and moderately differentiated carcinomas exhibit a variable number of duct-like structures with mild to moderate atypia, whereas poorly differentiated carcinomas show marked atypia and limited gland forming pattern¹³.
Distinct characteristics criteria between pancreatic ductal adenocarcinoma and differential diagnoses.
| Criteria | PDAC | CP | PP | NET | AIP |
|---|---|---|---|---|---|
| Clinical presentation | Weight loss, jaundice, abdominal pain, fatigue | Chronic abdominal pain, diabetes, weight loss | Elevated amylase, palpable mass | Elevated chromogranin A, synaptophysin | Elevated LDH, variable amylase |
| Biological | Elevated CA 19-9, elevated bilirubin, elevated glucose | Slightly elevated amylase, diabetes, palpable mass | Elevated amylase, diabetes, palpable mass | Elevated chromogranin A, variable pancreatic function tests | Elevated IgG4 levels, palpable mass |
| Radiological | Hypodense mass CT: hypodense mass after contrast, biliary ductal obstruction | Calcifications, dilated pancreatic ducts, glandular atrophy | Fluid collections, cystic lesions | Well-defined mass with enhancement | Diffuse enlargement, perivascular hypodensity |
| Histological | Cellularity, atypical cells, desmoplastic stroma | Chronic inflammation, fibrosis | Fibrosis, chronic inflammation | Neuroendocrine features | IgG4-positive plasma cells, storiform fibrosis |
Abbreviations: PDAC: pancreatic ductal adenocarcinoma, CP: chronic pancreatitis, PP: pancreatic pseudocysts, PNET: pancreatic neuroendocrine tumors, AIP: autoimmune pancreatitis, PPL: primary pancreatic lymphoma, CA 19-9: carbohydrate antigen 19-9, IgG4: immunoglobulin G4, LDH: lactate dehydrogenase, US: ultrasonography, CT: computed tomography, CD: cluster of differentiation.
DIFFERENTIAL DIAGNOSIS: MIMICKING MASSES
Chronic pancreatitis
Chronic pancreatitis is characterized by recurrent episodes of pancreatic inflammation, leading to fibrotic changes, ductal strictures, and parenchymal atrophy¹⁴. These structural alterations can mimic the radiographic appearance of pancreatic malignancy, thus posing diagnostic challenges, while imaging features such as pancreatic calcifications, ductal dilatation, and parenchymal heterogeneity can be suggestive of CP, although they can also be observed in PDAC¹⁵.
Patients developing CP seem to be younger and usually have a long disease history characterized by painful attacks of acute pancreatitis and the development of pseudocysts. Risk factors for CP are well established and include excessive alcohol consumption as well as smcking, which are also shared by patients with PDAC. Patients with PDAC tend to be older and often present with painless jaundice¹⁶. The exception to this rule, however, is patients with autoimmune pancreatitis (see below).
From a biological perspective, the distinction between CP and PDAC is difficult. Elevated CA 19-9 and CEA levels might suggest malignancy in the absence of cholestasis, although elevated levels have also been observed in benign conditions. Furthermore, their absence does not exclude PDAC.
Distinguishing CP from PDAC can often prove difficult, since these two entities can coexist. In the case of pancreatic parenchymal nodule or clinical doubt, histological evaluation remains the gold standard for distinguishing between these two entities. Tissue sampling and ductal brushing using EUS and endoscopic retrograde cholangiopancreatography (ERCP) may be necessary⁴˒¹⁷. ERCP findings include a malignant-looking pancreatic duct, distal bile duct stricture, and upstream duct dilatation in the case of PDAC, although these can also be seen in CP¹⁷. Furthermore, EUS is helpful for the diagnosis, staging, and treatment of both diseases (Figure 1). A core biopsy or fine needle aspiration has a sensitivity of up to 90% in the case of pancreatic parenchymal anomalies or peripancreatic lymph node¹⁸˒¹⁹. However, distinguishing CP from PDAC can be a challenge. These entities share overlapping histological features such as fibrosis and the presence of distorted ducts. A combination of features is often necessary to discriminate between CP and PDAC. Retention of the lobular pattern, no or mild atypia, and regular ductal contours favor CP. Perineural or lymphovascular invasion are diagnostic of PDAC¹⁵˒²⁰.
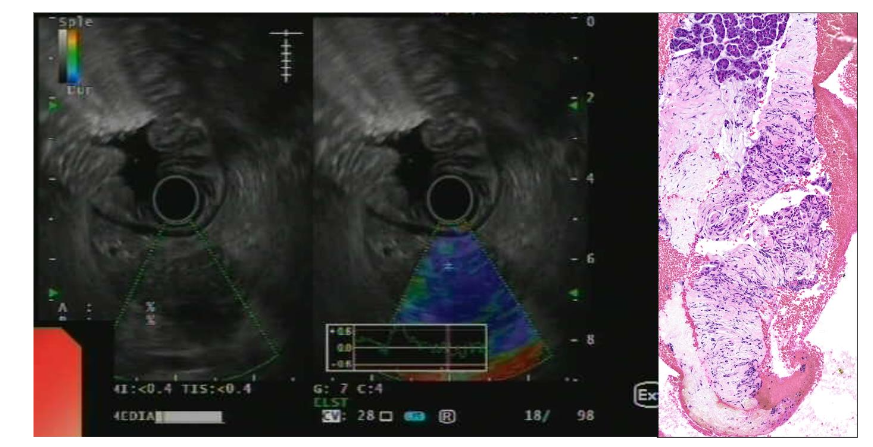
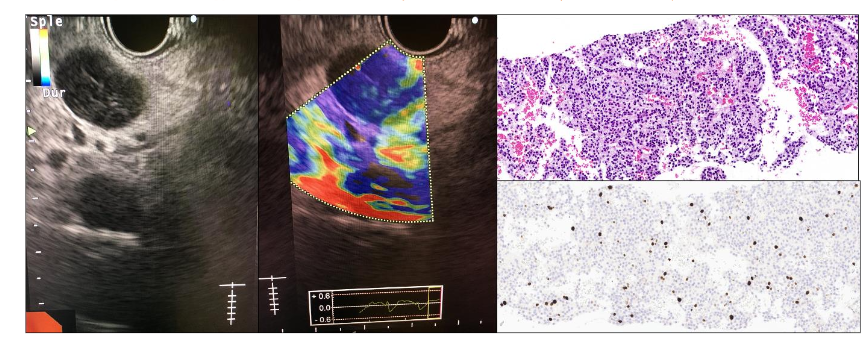
Figure 1 EUS imaging showing a pancreatic neuroendocrine tumor with a low stroma ratio. Biopsy reveals monomorphic neoplastic cells with minimal atypia in trabecular pattern. The stroma is minimal, rich in vascular with fine, capillary-sized vessels (H&E, 200x). Neoplastic cells express chromogranin A and synaptophysin with a slightly elevated Ki67 proliferation index (Ki67 immunostaining, 200x).
Pancreatic pseudocysts
Pancreatic pseudocysts are encapsulated collections of fluid that develop as a complication of acute, chronic pancreatitis or pancreatic trauma, thus posing diagnostic challenges akin to pancreatic malignancies²¹. Discriminating between these entities is based on the patients’ clinical history, imaging, and cytological analyses.
Unlike true cystic neoplasms, which originate from epithelial cells, PPs lack an epithelial lining and instead arise from the inflammatory response to a pancreatic injury. These fluid collections typically develop in the context of pancreatic duct disruption or parenchymal necrosis, leading to the extravasation of pancreatic enzymes and inflammatory mediators into the surrounding tissues.
The clinical presentation of pancreatic pseudocyst varies depending on their size, location, and complications²². Small asymptomatic PPs may be detected incidentally on imaging studies performed for other indications, whereas larger cysts may cause abdominal pain, fullness, or obstructive symptoms.
secondary to the mass effect. Indeed, larger PPs can be misdiagnosed as PDAC. In rare cases, PPs may become infected or hemorrhage, leading to life-threatening complications that require an urgent medical intervention.
Imaging such as CT or MRI are instrumental in diagnosing PPs and assessing their size, content, and relationship to the adjacent structures²³. Pancreatic pseudocysts typically appear as well-defined, round, or oval fluid collections with thin walls and homogeneous fluid attenuation or signal intensity on imaging. Contrast-enhanced studies may demonstrate peripheral enhancement or internal septations, particularly in the setting of chronic PPs with organized debris or hemorrhage²³.
Endoscopic ultrasound offers a better examination and visualization of PPs, thus facilitating the accurate assessment of their morphology and their proximity to adjacent structures. Moreover, endoscopic ultrasound-guided fine-needle aspiration (FNA) can provide additional diagnostic information by sampling the cyst fluid for analysis, including amylase levels, cytology, exosomes, proteomics, and microbiological cultures¹²˒²⁴˒²⁵. However, as FNA carries the risk of infection or hemorrhage, it should only be reserved for cases where the diagnosis is uncertain, or complications are suspected.
Not all PPs require intervention. Indeed, spontaneous resolution is reported in 40% to 50% of patients²⁴. For this reason, only symptomatic patients should be treated using a step-up approach, first endoscopic or percutaneous and then surgically if the first approach fails²⁶.
Pancreatic neuroendocrine tumors
Pancreatic neuroendocrine tumors arise from neuroendocrine cells scattered throughout the pancreas. Differential diagnosis involves discerning their distinct histological features, secretory profiles, and molecular signatures.
Pancreatic neuroendocrine tumors encompass a heterogenous group of neoplasms with variable clinical presentations and biological behaviors²⁷˒²⁸. Although the majority of PNETs are sporadic, a subset may occur in the context of genetic syndromes such as multiple endocrine neoplasia type 1 (MEN1)²⁹ or von Hippel-Lindau (VHL) disease³⁰. These tumors can secrete a variety of hormones, leading to a diverse range of clinical syndromes, including insulinoma, gastrinoma, glucagonoma, and VIPoma²⁷.
Imaging studies play a crucial role in localizing and characterizing PNETs. Contrast-enhanced CT and MRI can define the size, location, and vascularity of the tumor, which can help with therapeutic management and prognosis. MRI is similar to CT in detecting PNETs; they appear as round hypointense lesions on T1 and hyperintense compared with pancreas on T2-weighted images¹⁶. Functional imaging with somatostatin receptor scintigraphy (SRS) or 68Ga-DOTATATE PET can identify tumors expressing somatostatin receptors, thus facilitating targeted treatment options with somatostatin analogs. Finally, EUS contributes to the histological confirmation of the diagnosis using elastometry studies (Figure 2) and provides histological material to assess the histological grade and stage of the PNETs²⁷˒²⁹˒³³.
Microscopically, PNETs are well-differentiated tumors. They exhibit characteristic architectural patterns, including nested, trabecular, or glandular growth, and express neuroendocrine markers such as chromogranin A and synaptophysin. Based on their proliferative activity, PNETs are graded from G1 to G3³⁴. Unlike their pulmonary counterparts, neuroendocrine carcinomas (NECs) of the pancreas are rare³⁵.
Treatment options for PNETs are based on their size, location, and aggressiveness. Different options exist, ranging from surveillance to locoregional therapy such as endoscopic or percutaneous ablation, surgery, or systemic treatment including somatostatin analogs, targeted therapy, and peptide receptor radionuclide therapy³⁶. As such, treatment plan is tailored to the individual patient’s condition and the specific characteristics of their tumor, it should be discussed at a multidisciplinary team meeting.
Figure 2. EUS imaging showing a pancreatic neuroendocrine tumor with a low strain ratio. Biopsy reveals monomorphic neoplastic cells with minimal atypia in trabecular pattern. The stroma is minimal, richly vascular with fine, capillary-sized vessels (H&E, 200x). Neoplastic cells express chromogranin A and synaptophysin with a slightly elevated Ki67 proliferation index (Ki67 immunostaining, 200x).
Autoimmune pancreatitis
Autoimmune pancreatitis is a distinct form of chronic pancreatitis characterized by the autoimmune-mediated inflammation of the pancreatic parenchyma and ducts³⁷˒³⁹. Two subtypes of AIP exist: Type 1, which is associated with elevated serum immunoglobulin G4 (IgG4) levels and the systemic involvement of other organs, and type 2, which lacks these features and is often referred to as idiopathic duct-centric pancreatitis characterized by granulocytic epithelial lesions³⁹˒⁴¹. Furthermore, type 1 AIP is now recognized as the pancreatic manifestation of the IgG4-related disease (IgG4-RD)⁴²˒⁴⁴. The latter can affect almost all organs with a variable clinical presentation, although the most common manifestations are autoimmune pancreatitis, salivary gland disease, orbital/lacrimal gland disease, and retroperitoneal fibrosis⁴⁵.
While the exact pathogenesis of AIP is poorly understood, multiple immunological pathways have been proposed to contribute to the development of this entity. Autoimmune pancreatitis³⁹ and IgG4-RD⁴³˒⁴⁴ have multiple diagnostic criteria with a sequential methodology based on the integration of clinical, radiological, biological, and histological features that weight the diagnosis.
The clinical presentation of AIP can mimic that of pancreatic cancer with patients presenting with abdominal pain, jaundice, weight loss, and pancreatic mass lesions on imaging studies⁴⁶˒⁵⁰. However, several key features can help distinguish AIP from malignancy.
Clinically, AIP patients usually present with mild abdominal pain such as discomfort, while PDAC patients present severe, persistent, and progressive abdominal pain with possible weight loss and progressive jaundice in the advance stages. Extraparenchymal manifestations, particularly when accompanied by other autoimmune manifestations such as sclerosing cholangitis, salivary or lacrimal gland involvement, or retroperitoneal fibrosis, are more frequent in AIP, whereas PDAC is more localized in the pancreatic gland and induces lower bile duct stenosis, presenting metastatic lesions and direct invasions in some cases. Fluorodeoxyglucose (FDG)-positron emission tomography (PET)-CT scans can help to determine other organ involvement. Immunoglobulin G4 is elevated in AIP patients, mainly type 1 AIP (around 60–70%) although elevated levels have also been reported in a few cases of PDAC⁴⁶˒⁵¹. By contrast, elevated CA 19-9 is uncommon in AIP. Radiologically, smooth margins and capsule-like rims in the body and tail region that represent
severe fibrotic changes are observed in CT and MRI imaging of patients with AIP⁵². Duct dilatation should raise the suspicion of PDAC. Using contrast-enhanced CT, AIP is characterized by homogenous delayed enhancement of the gland, which indicates the diffuse loss of parenchymal volume and severe fibrosis, whereas heterogenous enhancement that represents necrosis or bleeding in the tumor can be seen in PDAC. EUS for AIP reveals a duct-penetrating sign, a diffuse homogenous hypoechoic pattern, and linear or reticular hyperechoic inclusions, reflecting interlobular fibrosis⁵³. In PDAC, EUS findings include a localized hypoechoic mass and double-duct sign, often with lymph node enlargement or vascular invasion⁵⁴.
Characteristic histological findings of AIP include diffuse lymphoplasmacytic infiltration with abundant IgG4 positive plasma cells, storiform fibrosis, and obliterative venulitis³⁸˒⁵⁰ (Figure 3). Not all these criteria are always present on biopsy due to the small size of the sample.
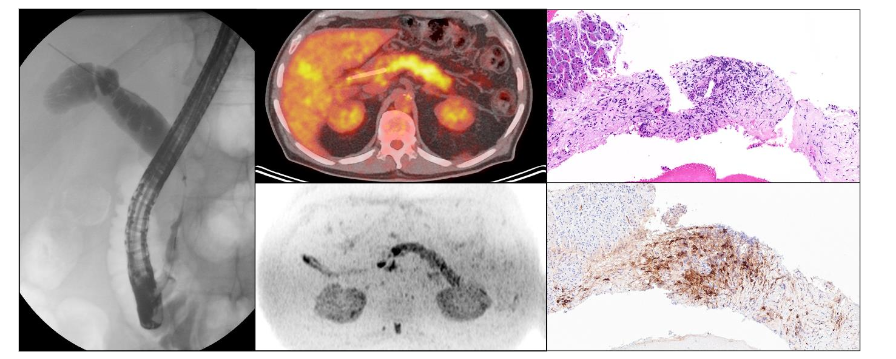
Figure 3. Focal common bile duct stricture with smooth, concentric thickening, suggestive of IgG4-related cholangiopathy. Increased FDG uptake in the pancreas on PET-CT, indicating hypermetabolic activity. Biopsy reveals fibrosis and lymphoplasmocytic inflammation (H&E, 200x). IgG4 immunostaining shows abundant IgG4-positive plasma cells. Response to corticosteroid therapy is another hallmark of AIP, with patients typically experiencing a good response within 2–4 weeks with an improvement in symptoms as well as biological and radiographic abnormalities following treatment initiation⁵⁵˒⁵⁷. This contrasts with the lack of response observed in PDAC, where chemotherapy and surgical resection are the mainstays of therapy. However, it is important to note that AIP can coexist with pancreatic cancer, thus requiring careful evaluation and follow-up to ensure accurate diagnosis and appropriate management⁵⁸.
Primary pancreatic lymphomas
Primary pancreatic lymphomas are a rare malignancy accounting for less than 1% of pancreatic tumors⁵⁹. Nevertheless, 25% of non-Hodgkin lymphomas originate from extralymphatic organs, with approximately 30% involving the pancreas⁵⁹. Primary pancreatic lymphomas arise from lymphoid tissue within the pancreas and can present as a focal mass or diffuse infiltrative lesion mimicking PDAC⁶⁰. The clinical presentation of pancreatic lymphomas is nonspecific, with patients typically presenting with abdominal pain, weight loss, and constitutional symptoms such as fever, night sweats, and fatigue (B symptoms). Primary pancreatic lymphomas can involve any part of the pancreas, with a predominance in the head, and may manifest as a solitary mass or multiple nodules. Imaging studies such as CT, MRI, and PET-CT can help delineate the extent of the disease and guide biopsy planning. Primary pancreatic lymphomas
with well-preserved peripancreatic fat planes⁶¹˒⁶² and rarely cause pancreatic duct obstruction unlike PDAC, which is often observed as a smaller irregular mass with ductal dilatation and obstruction due to its invasive nature. Although elevated levels of lactate dehydrogenase and beta-2 microglobulin have been observed and can contribute to the diagnosis, these markers remain nonspecific. Carbohydrate antigen 19-9 levels will remain normal in PPL unless biliary involvement is present. However, the definitive diagnosis of pancreatic lymphoma requires histopathological confirmation, as imaging findings alone are insufficient to distinguish it from other pancreatic malignancies. Fine-needle aspiration or core needle biopsy of the pancreatic lesion can often provide tissue samples for histological examination and immunophenotypic analysis.
Histologically, pancreatic lymphomas are subtyped following the classification used in lymph nodes and other extranodal sites. The main types of lymphomas involving the pancreas, with the most frequent being diffuse large B-cell lymphoma, extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue, and follicular lymphoma⁵²˒⁵³. Lymphomas form aggregates or sheets of neoplastic lymphoid cells that are distinct from the glandular duct-like structures formed by the malignant epithelial cells of PDAC. Immunohistological staining for lymphoid markers such as CD20, CD3, and CD5 help subtype lymphomas and guide treatment decisions. Molecular studies, including fluorescence in situ hybridization or polymerase chain reaction analysis for clonal immunoglobulin gene rearrangements, may also be performed to confirm the diagnosis and assess for genetic aberration associated with aggressive disease behavior.
The treatment of PPL depends on the type of lymphoma and most often consists of aggressive chemotherapy, sometimes combined with immunotherapy agents; radiation therapy can also be used for local tumor control or symptom relief. Surgical resection is not common but can be considered for managing specific complications like obstructive jaundice.
Conclusion
Accurately diagnosing pancreatic lesions is crucial for determining the appropriate treatment path and significantly improving patient outcomes. Continued advancements in diagnostic technologies and a better understanding of pancreatic disease pathology are likely to enhance diagnostic accuracy, thus offering hope for better management and improved survival rates for patients with pancreatic disease.
Few biological markers can help distinguish these entities¹⁰ for instance CA 19-9 which has a sensitivity of approximately 70–80% and a specificity of about 80–90% for PDAC in symptomatic patients⁶⁴. Carbohydrate antigen 19-9 levels >200U/ml are associated with a high likelihood of malignancy, particularly in non-jaundiced patient. However, approximately 5% to 10% of individuals cannot synthesize CA 19-9 antigens and are therefore regarded as Lewis-a b-negative⁶⁵. Hence, negative CA 19-9 test results do not necessarily exclude PDAC, thus necessitating a comprehensive diagnostic approach based on clinical findings, imaging results, and other biomarkers to ensure an accurate diagnosis and appropriate treatment planning. Although surgical resection should not be delayed in the case of PDAC, surgical restraint is crucial in other cases to avoid morbi-mortality caused by unnecessary pancreatic surgery, since surgical pancreatectomy is associated with substantial morbidity (30–50%) and mortality (3–5%). The management of patients harboring a pancreatic mass should always be discussed and coordinated by a multidisciplinary team specializing in oncology, visceral surgery, radiology, and gastroenterology to tailor treatment strategies to individual patient needs, thus improving overall patient outcomes.
Conflicts of interests:
The authors have no conflicts of interest to declare.
Acknowledgements:
None
References
1. McGuigan A, Kelly P, Turkington RC, Jones C, Coleman HG, McCain RS. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes. World J Gastroenterol. 2018;24 (43):4846-4861. doi:10.3748/wjg.v24.i43.4846
2. Kobi M, Veillette G, Narurkar R, et al. Imaging and Management of Pancreatic Cancer. Semin Ultrasound CT MR. 2020;41(2):139-151. doi:10.1053/j.sult.2019.12.003
3. Centeno BA, Thomas SC. Non-Neoplastic Masses of the Pancreas. Monogr Clin Cytol. 2020; 26:42-52. doi:10.1159/000455734
4. ASGE Standards of Practice Committee, Eloubeidi MA, Decker GA, et al. The role of endoscopy in the evaluation and management of patients with solid pancreatic neoplasia. Gastrointest Endosc. 2016;83(1):17-28.
doi:10.1016/j.gie.2015.09.009
5. Huang J, Lok V, Ngai CH, et al. Worldwide Burden of, Risk Factors for, and Trends in Pancreatic Cancer. Gastroenterology. 2021;160(3):744-754. doi:10.1053/j.gastro.2020.10.007
6. Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3): 209-249. doi:10.3322/caac.21660
7. Brugel M, Carlier C, Reyes-Castellanos G, Callon S, Carrier A, Bouché O. Pesticides and pancreatic adenocarcinoma: A transversal epidemiological, environmental and mechanistic narrative review. Dig Liver Dis Off J Ital Soc Gastroenterol Ital Assoc Study Liver. 2022;54 (12):1605-1613. doi:10.1016/j.dld.2022.08.023
8. Chu LC, Goggins MG, Fishman EK. Diagnosis and Detection of Pancreatic Cancer. Cancer J Sudbury Mass. 2017;23(6):333-342. doi:10.1097/PPO.0000000000000290
9. Takaori K, Bassi C, Biankin A, et al. International Association of Pancreatology (IAP)/ European Pancreatic Club (EPC) consensus review of guidelines for the treatment of pancreatic cancer. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2016;16(1):14-27. doi:10.1016/j.pan.2015.10.013
10. O’Neill RS, Stoita A. Biomarkers in the diagnosis of pancreatic cancer: Are we closer to finding the golden ticket? World J Gastroenterol. 2021;27(26):4045-4087. doi:10.3748/wjg.v27.i26.4045
11. Gui JC, Yan WL, Liu XD. CA19-9 and CA242 as tumor markers for the diagnosis of pancreatic cancer: a meta-analysis. Clin Exp Med. 2014;14(2) :225-233. doi:10.1007/s10238-013-0234-9
12. Farina A, Dumonceau JM, Frossard JL, Hadengue A, Hochstrasser DF, Lescuyer P. Proteomic analysis of human bile from malignant biliary stenosis induced by pancreatic cancer. J Proteome Res. 2009;8(1):159-169.
doi:10.1021/pr8004925
13. Hruban RH, Adsay NV, Esposito I. Pancreatic ductal adenocarcinoma. In: Digestive System Tumours WHO Classification of Tumours, 5th Edition, Volume 1. Vol 1. 5th ed. International Agency for Research on Cancer, Lyon, 2019; 2019.
14. Beyer G, Habtezion A, Werner J, Lerch MM, Mayerle J. Chronic pancreatitis. Lancet Lond Engl. 2020;396(10249):499-512. doi:10.1016/S0140-6736(20)31318-0
15. Klöppel G, Adsay NV. Chronic pancreatitis and the differential diagnosis versus pancreatic cancer. Arch Pathol Lab Med. 2009;133(3):382-387. doi:10.5858/133.3.382
16. Younan G. Pancreas Solid Tumors. Surg Clin North Am. 2020;100(3):565-580. doi:10.1016/j.suc.2020.02.008
17. Hanada K, Minami T, Shimizu A, et al. Roles of ERCP in the Early Diagnosis of Pancreatic Cancer. Diagn Basel Switz. 2019;9(1):30. doi:10.3390/diagnostics9010030
18. Syed A, Babich O, Rao B, et al. Endoscopic ultrasound guided fine-needle aspiration vs core needle biopsy for solid pancreatic lesions: Comparison of diagnostic accuracy and procedural efficiency. Diagn Cytopathol. 2019;47(11):1138-1144. doi:10.1002/dc.24277
19. Selvaggi SM. The value of concurrent endoscopic ultrasound-guided fine needle aspirates and needle core biopsies in the diagnosis of pancreatic neoplasms. Diagn Cytopathol. 2022;50(10):459-462. doi:10.1002/dc.25016
20. Klöppel G. Chronic pancreatitis, pseudotumors and other tumor-like lesions. Mod Pathol Off J U S Can Acad Pathol Inc. 2007;20 Suppl 1:S113-131. doi:10.1038/modpathol.3800690
21. Sand J, Nordback I. The differentiation between pancreatic neoplastic cysts and pancreatic pseudocyst. Scand J Surg SJS Off Organ Finn Surg Soc Scand Surg Soc. 2005;94(2):161-164. doi:10.1177/145749690509400213
22. Habashi S, Draganov PV. Pancreatic pseudocyst. World J Gastroenterol. 2009;15(1):38-47. doi:10.3748/wjg.15.38
23. Hookey LC, Debroux S, Delhaye M, Arvanitakis M, Le Moine O, Devière J. Endoscopic drainage of pancreatic-fluid collections in 116 patients: a comparison of etiologies, drainage techniques, and outcomes. Gastrointest Endosc. 2006;63(4): 635-643. doi:10.1016/j.gie.2005.06.028
24. Gumaste VV, Aron J. Pseudocyst management: endoscopic drainage and other emerging techniques. J Clin Gastroenterol. 2010; 44(5):326-331. doi:10.1097/MCG.0b013e3181cd9d2f
25. Baron TH. Treatment of pancreatic pseudocysts, pancreatic necrosis, and pancreatic duct leaks. Gastrointest Endosc Clin N Am. 2007; 17(3):559-579, vii. doi:10.1016/j.giec.2007.05.013
26. Melman L, Azar R, Beddow K, et al. Primary and overall success rates for clinical outcomes after laparoscopic, endoscopic, and open pancreatic cystgastrostomy for pancreatic pseudocysts. Surg Endosc. 2009;23(2):267-271. doi:10.1007/s00464-008-0196-2
27. Ma ZY, Gong YF, Zhuang HK, et al. Pancreatic neuroendocrine tumors: A review of serum biomarkers, staging, and management. World J Gastroenterol. 2020;26(19):2305-2322. doi:10.3748/wjg.v26.i19.2305
28. Ro C, Chai W, Yu VE, Yu R. Pancreatic neuroendocrine tumors: biology, diagnosis, and treatment. Chin J Cancer. 2013;32(6):312-324. doi:10.5732/cjc.012.10295
29. Effraimidis G, Knigge U, Rossing M, Oturai P, Rasmussen ÅK, Feldt-Rasmussen U. Multiple endocrine neoplasia type 1 (MEN-1) and neuroendocrine neoplasms (NENs). Semin Cancer Biol. 2022;79:141-162.
doi:10.1016/j.semcancer.2021.04.011
30. Laks S, van Leeuwaarde R, Patel D, et al. Management recommendations for pancreatic manifestations of von Hippel-Lindau disease. Cancer. 2022;128(3):435-446. doi:10.1002/cncr.33978
31. Lewis RB, Lattin GE, Paal E. Pancreatic endocrine tumors: radiologic-clinicopathologic correlation. Radiogr Rev Publ Radiol Soc N Am Inc. 2010;30(6):1445-1464. doi:10.1148/rg.306105523
32. Franchellucci G, Andreozzi M, Carrara S, et al. Contrast Enhanced EUS for Predicting Solid Pancreatic Neuroendocrine Tumor Grade and Aggressiveness. Diagn Basel Switz. 2023;13(2):239. doi:10.3390/diagnostics13020239
33. Crinò SF, Ammendola S, Meneghetti A, et al. Comparison between EUS-guided fine-needle aspiration cytology and EUS-guided fine-needle biopsy histology for the evaluation of pancreatic neuroendocrine tumors. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2021;21(2):443-450. doi:10.1016/j.pan.2020.12.015
34. Kloeppel G, Klimstra DS, Singhi AD. Pancreatic neuroendocrine neoplasms: Introduction. In: Digestive System Tumours WHO Classification of Tumours, 5th Edition, Volume 1. Vol 1. 5th ed. International Agency for Research on Cancer, Lyon, 2019; 2019.
35. Adsay NV, Singhi AD, Perren A. Pancreatic neuroendocrine carcinoma. In: Digestive System Tumours WHO Classification of Tumours, 5th Edition, Volume 1. Vol 1. 5th ed. International Agency for Research on Cancer, Lyon, 2019; 2019.
36. So H, Ko SW, Shin SH, et al. Comparison of EUS-guided ablation and surgical resection for nonfunctioning small pancreatic neuroendocrine tumors: a propensity score-matching study. Gastrointest Endosc. 2023;97(4):741-751.e1. doi:10.1016/j.gie.2022.11.004
37. Notohara K, Burgart LJ, Yadav D, Chari S, Smyrk TC. Idiopathic chronic pancreatitis with periductal lymphoplasmacytic infiltration: clinicopathologic features of 35 cases. Am J Surg Pathol. 2003;27(8):1119-1127.
doi:10.1097/00000478-200308000-00009
38. Zamboni G, Lüttges J, Capelli P, et al. Histopathological features of diagnostic and clinical relevance in autoimmune pancreatitis: a study on 53 resection specimens and 9 biopsy specimens. Virchows Arch Int J Pathol. 2004;445(6):552-563. doi:10.1007/s00428-004-1140-z
39. Shimosegawa T, Chari ST, Frulloni L, et al. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas. 2011;40(3):352-358.
doi:10.1097/MPA.0b013e3182142fd2
40. Vonlaufen A, Frossard JL. [Autoimmune pancreatitis]. Rev Med Suisse. 2010;6(261):1662-1666.
41. Mack S, Flattet Y, Bichard P, Frossard JL. Recent advances in the management of autoimmune pancreatitis in the era of artificial intelligence. World J Gastroenterol. 2022;28(48) :6867-6874. doi:10.3748/wjg.v28.i48.6867
42. Kamisawa T, Funata N, Hayashi Y, et al. A new clinicopathological entity of IgG4-related autoimmune disease. J Gastroenterol. 2003;38 (10):982-984. doi:10.1007/s00535-003-1175-y
43. Wallace ZS, Naden RP, Chari S, et al. The 2019 American College of Rheumatology/European League Against Rheumatism classification criteria for IgG4-related disease. Ann Rheum Dis. 2020; 79(1):77-87. doi:10.1136/annrheumdis-2019-216561
44. Umehara H, Okazaki K, Kawa S, et al. The 2020 revised comprehensive diagnostic (RCD) criteria for IgG4-RD. Mod Rheumatol. 2021;31(3): 529-533. doi:10.1080/14397595.2020.1859710
45. Kamisawa T, Zen Y, Pillai S, Stone JH. IgG4-related disease. Lancet Lond Engl. 2015;385(9976): 1460-1471. doi:10.1016/S0140-6736(14)60720-0
46. Hedfi M, Charfi M, Nejib FZ, et al. Focal Mass-Forming Autoimmune Pancreatitis Mimicking Pancreatic Cancer: Which strategy? Tunis Med. 2019;97(5):731-735.
47. Hoshimoto S, Aiura K, Tanaka M, Shito M, Kakefuda T, Sugiura H. Mass-forming type 1 autoimmune pancreatitis mimicking pancreatic cancer. J Dig Dis. 2016;17(3):202-209. doi:10.1111/1751-2980.12316
48. Hsu WL, Chang SM, Wu PY, Chang CC. Localized autoimmune pancreatitis mimicking pancreatic cancer: Case report and literature review. J Int Med Res. 2018;46(4):1657-1665. doi:10.1177/0300060517742303
49. Khanna K B J, Mesenas SJ, Loh TJ, Chin YK. Autoimmune pancreatitis masquerading as pancreatic cancer. VideoGIE Off Video J Am Soc Gastrointest Endosc. 2021;6(12):546-548. doi:10.1016/j.vgie.2021.09.001
50. Chari ST, Takahashi N, Levy MJ, et al. A Diagnostic Strategy to Distinguish Autoimmune Pancreatitis From Pancreatic Cancer. Clin Gastroenterol Hepatol. 2009;7(10):1097-1103. doi:10.1016/j.cgh.2009.04.020
51. Ghazale A, Chari ST, Smyrk TC, et al. Value of serum IgG4 in the diagnosis of autoimmune pancreatitis and in distinguishing it from pancreatic cancer. Am J Gastroenterol. 2007;102(8):1646-1653. doi:10.1111/j.1572-0241.2007.01264.x
52. Schima W, Böhm G, Rösch CS, Klaus A, Függer R, Kopf H. Mass-forming pancreatitis versus pancreatic ductal adenocarcinoma: CT and MR imaging for differentiation. Cancer Imaging Off Publ Int Cancer Imaging Soc. 2020;20(1):52. doi:10.1186/s40644-020-00324-z
53. Farrell JJ, Garber J, Sahani D, Brugge WR. EUS findings in patients with autoimmune pancreatitis. Gastrointest Endosc. 2004;60(6):927-936. doi:10.1016/s0016-5107(04)02230-8
54. Kitano M, Yoshida T, Itonaga M, Tamura T, Hatamaru K, Yamashita Y. Impact of endoscopic ultrasonography on diagnosis of pancreatic cancer. J Gastroenterol. 2019;54(1):19-32. doi:10.1007/s00535-018-1519-2
55. Kamisawa T, Egawa N, Nakajima H, Tsuruta K, Okamoto A. Morphological changes after steroid therapy in autoimmune pancreatitis. Scand J Gastroenterol. 2004;39(11):1154-1158. doi:10.1080/00365520410008033
56. Bi Y, Hart PA, Law R, et al. Obstructive jaundice in autoimmune pancreatitis can be safely treated with corticosteroids alone without biliary stenting. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2016;16(3):391-396.
doi:10.1016/j.pan.2016.03.017
57. Okazaki K, Chari ST, Frulloni L, et al. International consensus for the treatment of autoimmune pancreatitis. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2017;17(1):1-6. doi:10.1016/j.pan.2016.12.003
58. Macinga P, Bajer L, Del Chiaro M, et al. Pancreatic cancer in patients with autoimmune pancreatitis: A scoping review. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2021;21(5):928-937. doi:10.1016/j.pan.2021.03.007
59. Behrns KE, Sarr MG, Strickler JG. Pancreatic lymphoma: is it a surgical disease? Pancreas. 1994;9(5):662-667.
60. Kato N, Yamaguchi A, Sugata S, et al. Clinical features and images of malignant lymphoma localized in the pancreatic head to differentiate from pancreatic ductal adenocarcinoma: a case series study. BMC Gastroenterol. 2023;23(1):138. doi:10.1186/s12876-023-02779-3
61. Merkle EM, Bender GN, Brambs HJ. Imaging findings in pancreatic lymphoma: differential aspects. AJR Am J Roentgenol. 2000;174(3):671-675. doi:10.2214/ajr.174.3.1740671
62. Li Z, Zhang S, Vasdani N, Castillo E. Clues for diagnosing primary pancreatic lymphoma. Case Rep Gastroenterol. 2012;6(2):438-445. doi:10.1159/000339968
63. Alexander RE, Nakeeb A, Sandrasegaran K, et al. Primary pancreatic follicle center-derived lymphoma masquerading as carcinoma. Gastroenterol Hepatol. 2011;7(12):834-838.
64. Ballehaninna UK, Chamberlain RS. The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: An evidence based appraisal. J Gastrointest Oncol. 2012;3(2):105-119. doi:10.3978/j.issn.2078-6891.2011.021
65. Scarà S, Bottoni P, Scatena R. CA 19-9: Biochemical and Clinical Aspects. Adv Exp Med Biol. 2015;867:247-260. doi:10.1007/978-94-017-7215-0_15