Morphological Insights on Arteriolar Atherosclerosis in CAD
Morphological findings of arteriolar atherosclerosis and prognosis in patients undergoing artery bypass grafting in Armenia
Nona Kukurtchyan1, Gohar Karapetyan1, Arman Kocharyan1, Mher Voskanyan1, Mamicon Hovhannisyan1
- Best life Armenian-Japanise Medical Centre, Yerevan, RA
OPEN ACCESS
PUBLISHED 31 October 2025
CITATION Kukurtchyan, N., Karapetyan, G., et al., 2025. Morphological findings of arteriolar atherosclerosis and prognosis in patients undergoing artery bypass grafting in Armenia. Medical Research Archives, [online] 13(10). https://doi.org/10.18103/mra.v13i10.7045
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i10.7045
ISSN 2375-1924
Abstract
Cardiovascular disease remains the leading global cause of mortality and morbidity. Atherosclerotic alterations play a pivotal role not only in coronary vessels but also within the microcirculatory system, particularly affecting arterioles.
Right atrial appendage specimens from patients with coronary artery disease (CAD), obtained during cardiac surgical interventions, were processed using transmission electron microscopy (TEM), stained with Azure-2, and examined under a light microscope.
Morphological analysis revealed a marked increase in arteriolar formation via the intussusceptive angiogenesis mechanism. In certain patients, the number of de novo arterioles reached 4 points on a 5-point pathological vascular growth scale for intussusceptive angiogenesis. Coronary artery bypass grafting (CABG) is generally indicated for patients presenting with the most advanced forms of ischemic heart disease (IHD). Arteriolar atherosclerosis in CAD may represent one of the underlying mechanisms contributing to heart failure.
Early detection of such pathological changes may facilitate prognostication of disease progression, estimation of life expectancy, and the selection of optimal therapeutic strategies.
Keywords
CAD, atherosclerosis arteriole, intussusception growth, light optical microscopy.
Introduction
Ischemic heart disease (IHD) remains a leading global cause of mortality and morbidity. Myocardial perfusion is maintained through an extensive vascular network comprising large epicardial coronary arteries, smaller arteries, arterioles, and capillaries. The microvascular network, consisting of arterioles and capillaries, plays a critical role in myocardial oxygen delivery.
Atherosclerotic damage affects not only the major coronary vessels but also the primary microcirculatory elements—the arterioles. 11,9
Coronary artery disease (CAD), characterized by plaque accumulation in the coronary arterial walls, gradually narrows the vascular lumen and contributes to impaired myocardial perfusion.12 While CAD is often associated with large epicardial vessels, recent studies emphasize the crucial role of coronary microvascular dysfunction. 11,9
In IHD, coronary arterioles undergo morphological alterations due to reduced blood flow and oxygen supply. These resistance vessels, typically with diameters around 200 μm, are key regulators of myocardial reperfusion.
Heterogeneity in the myocardial vascular network affects structural and functional parameters such as vessel diameter, length, connectivity, flow patterns, velocity, wall shear stress, and oxidative stress. 22,21
Normally, arterioles have thin walls composed of endothelium, smooth muscle cells, and minimal connective tissue. Microvascular atherosclerosis leads to structural remodeling, including wall thickening, reduced elasticity, and impaired function. Coronary microcirculation disturbances are involved in multiple pathophysiological mechanisms of ischemia-reperfusion injury. 1 Knowledge of these mechanisms remains limited but is essential for understanding disease progression.22
Since atherosclerosis is a systemic process, its effects extend throughout the myocardium, including the right atrium. Cardiac surgical interventions, such as coronary artery bypass grafting (CABG), are employed to improve myocardial perfusion. Small fragments of the right atrial appendage, routinely obtained during cardiothoracic surgery, provide an opportunity to study myocardial arterioles using detailed histological methods.[18] These methods enable visualization of arteriolar morphology and assessment of the degree of atherosclerotic involvement.16,17 Morphological analyses have demonstrated pathological growth of arterioles by intussusception type, highlighting the need for further morphological investigation.
Materials and Methods
Small fragments of the right atrial appendage from 6 patients with CAD were obtained during routine cardiac surgical procedures. Biopsy specimens were immediately fixed in a cold mixture of paraformaldehyde and glutaraldehyde, followed by post-fixation in 1% OsO₄. The tissues were dehydrated in an ascending series of alcohols and acetone, then embedded in epoxy resin. Semithin epoxy slices with up to 1 micrometer thickness were prepared using an Ultracut Riechert. Sections were stained with 1% solution of Azur II by special method 18 and examined under a light microscope using a 40× objective lens combined with a 10× ocular lens.
All obtained images of arterioles were analyzed and evaluated by 5 score scale of pathological growth of microvessels by intussusceptive type 17.
All procedures involving human subjects were approved by the Institutional Review Board and Bioethical Committee of the Yerevan State Medical University, RRI, and conformed to the ethical guidelines and legal standards for research in accordance with the European Community Directives (2001/20/EC).
The aim of Study
To investigate new morphological findings of arteriolar atherosclerosis in CAD patients, focusing on intussusceptive arteriogenesis (de novo formation) and the direction of these changes, with the goal of predicting potential complications.
Results
On semithin epoxy sections stained with Azure-2, images of arterioles from the right atrial appendage were obtained. Six male patients with CAD, aged 48 to 65 years, were included in the study. To obtain a reliable representation of pathological changes in the arterioles, the entire biopsy material was utilized. Very small myocardial fragments of the atrial appendage were processed and embedded in epoxy resin, resulting in up to eight gelatin blocks.
The obtained images of arterioles were characteristic for each individual patient, showing both similarities and differences. The arterioles displayed variable lumen diameters, ranging from very large to small profiles. Wall thickness also varied, with some arterioles showing pronounced intussusceptive invaginations into the lumen. In certain cases, bridges connecting opposite arteriolar walls were observed, sometimes multiple in a single vessel.
Atherosclerotic changes were particularly notable in larger arterioles. These vessels exhibited elongated lumens, significant wall thickening, and abundant smooth muscle cells. Some large arterioles showed up to two convolutions at different ends of the lumen. Evidence of newly forming arterioles was observed, including residual fragments indicating separation. Notably, the newly separated arterioles differed markedly in lumen size compared to the parent vessel. In some separated segments, chambers were present, indicating the formation of a new convoluted vessel.
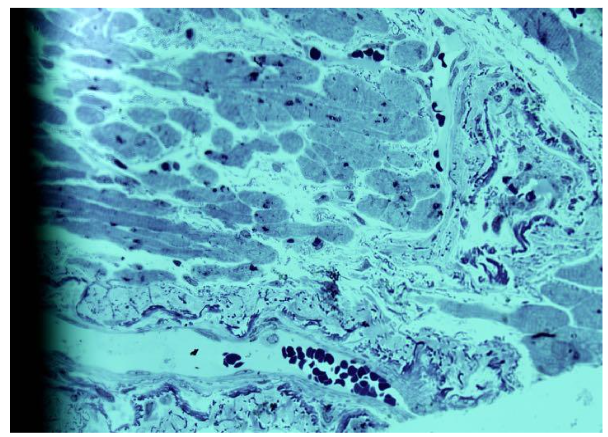
Frequently, arterioles with thickened walls consisted of multiple aligned lumens of different sizes, suggesting formation through bridging of opposite walls.
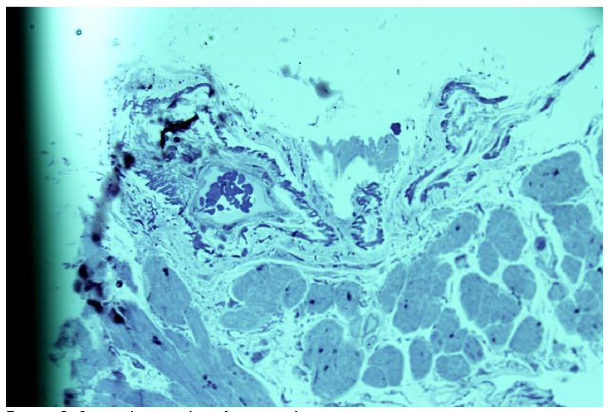
In cases of single de novo arteriole formation, the pathological vascular growth score reached up to 3 -4 points.17 In other patients, de novo arterioles exhibited variable lumen sizes, with multiple aligned or isolated lumens, sometimes associated with residual shared membranes.
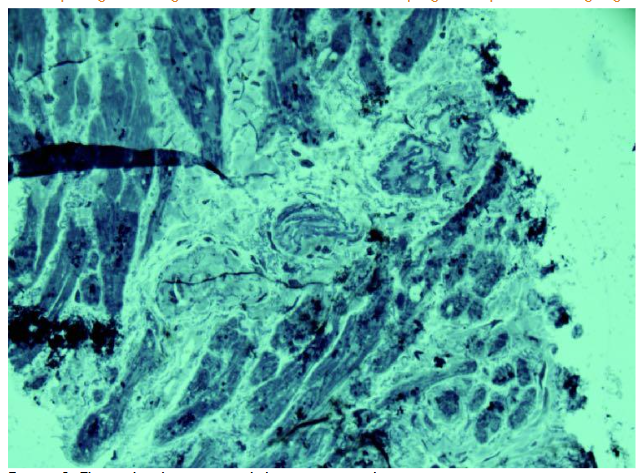
One arteriole showed chambers indicative of a separating convolution. Pathological arteriolar growth in this patient was also scored 4 due to increased de novo vessel formation.
The observed arteriolar growth sometimes resulted in highly irregular forms. Formation of pseudo-vessels occurred through protrusion of one arteriolar wall into the lumen, and growing walls occasionally created new vessels with dual lumens.

De novo formation from these vessels typically exhibited thickened walls and narrow lumen profiles.
Discussion
In IHD, arterioles undergo morphological changes due to reduced blood pressure and compromised oxygen supply. These changes contribute to structural remodeling of the vessel walls, including increased thickness, hypertrophy, and fibrosis. In the early stages of CAD, structural changes in arterioles are often compensatory and are manifested as arteriolar dilation, a response to pathological processes, as indicated by our findings.
Beyond varying degrees of wall thickening, significant alterations were observed in arterioles consistent with intussusceptive arteriogenesis.
It could lead to microvascular dysfunction.
In recent decades, new terms such as coronary vasospasm, microvascular spasm, and microvascular angina have emerged, though their identification and clinical relevance remain poorly understood in daily practice.
The observed arteriolar changes may also have clinical implications, contributing to manifestations of myocardial ischemia.
Microvascular dysfunction often undetected on routine coronary angiography, has emerged as a key determinant of prognosis acute and chronic coronary syndromes.
Patients with microvascular dysfunction are at higher risk of adverse cardiovascular events, such as recurred myocardial infarction, heart failure, hospitalization and mortality. A notable phenomenon is the pathological growth of arterioles via intussusception.
Morphological findings microvessels of patients with pulmonary artery valve stenosis occurred pathological growth of microvessels by intussusception type, which indicate that such phenomena could take place at other heart diseases as well.
Conclusion
Coronary artery bypass grafting (CABG) is indicated for patients with the most severe forms of ischemic heart disease (IHD), including those presenting emergently to clinical care. Arteriolar atherosclerosis in CAD may represent one of the underlying mechanisms contributing to heart failure. Timely detection of such microvascular changes can aid in predicting disease progression, estimating patient prognosis, and guiding appropriate pharmacological therapy.
References
- Alrfary A, Kabach M.J, Nieves J., Pino J., Chait R.,”Microvascular Coronary artery disease.’ USE/US Cardiology Review 2017.
- Burry P.H., Hlushchuk R., Djonov V., ‘Intussusceptive angiogenesis its significanse. Dev. Dyn. 2004, 231; 474-488. Doi:101002/dvdy.20184.
- Camiei P.G., Crea F. “Coronary microvascular disfunction’j. N. Egl. Med.2007, 356, 830-40. Doi:10.1056/NFJ Mra 061889
- Chen H., Kasseb Ch.S. ‘Microstructure- Bsaed Biochemical of Coronary Arteries in Health and Disease’ J. Biomech, 2016; 49(12); 2548-2559: Doi:10.1016/j biomech. 2016.03.023.
- Chilian W. M. ‘Coronary Microcirculation in Health and Disease. Summary of an NHLBI Workshop. Circulation 1997; 95, 522-528. Doi. Org:/10.1161/01.CIR 95.2.522.
- Del Buono M. G., Montone R.A., Camilli M. Et al. ‘Coronary microvascular dysfunction across the spectrum of cardiovascular disease. J AM. Call Cardiol. 2021, 78, 1352-1371.
- Djonov V, Baum O, Burry P.H. Vascular Remodeling by intussusceptive angiogenesis, j. Cell Tissue Res. 2003, 314(1); 107-117.Doi: 10.1007/S00441-003-0784-3.
- Egginton S., Zhow A.L., Brown M.D., Hudlicka D. ‘Unortodox angiogenesis in skeletal muscle’ cardiovascular. Res. 2001; 49; 634-646.
- Fonseka D.A., Antunes R.E., Doulce D., Cotrin D. The morphology, Physiology and Pathohystology of Coronary microcirculation. Open access pear-reviewed chapter oct. 2016. Doi: 10.5772/64537.
- Gianni-Barera, Tzani M, Fontanellar C, Hebery M, Djonov V, Hlushchuk R., Banfi A. ‘VEGF other expression in skeletal muscle induces angiogenesis by intussusception rather than sprouting’ Angiogenesis 2013, 16, 123-136. Doi: 10.1007/9 10456-012-93049
- Gianni-Barere R., Butsch K.A., Uccelli A., Gertelli A., Valente P., Bartolomeo M., Groppa E., Bargen M. G., Heberrer M, Schafor D., Gurske L, Djonov V, Vallmar B, Banfi A. “PDGF-BB regulating splitting angiogenesis in skeletal muscle by limiting VEGF-induced endothelial proliferation’. Angiogenesis, 2018; 21; 883-900.
- Jivorny R., Taqueti, Marculo F, Dicarli; Coronary microvascular disease. Patogenic mechanisms and Therapeutic Options. J. Acc-Stable-of-the Azt Review, J, Am. Call Cardiol, 2018, N27; 72(21); 2625-2641. Doi; 10.1016/I-iacc. 2018.09.042.
- John-Michael Azpino. ‘’Low-flow intussusceptive and metastable VEGFR, Sigma Link lanch angiogenesis ischemic muscle” Science advances. Nov.2021, Vol 7, issue 48. Doi:10.11.11.1126/Sciadv.abg9509
- Juan Carlos Kaskietal et al., Reappraised of Ischemic Heart Disease. Circulation 2018 Ana 138(4); 1463-1480. Doi; 10.1161/Circulation Ana, 118.03.1373
- Kukurtchyan N.S., Karapetyan G.R., Heart microvessels research at ischemic heart disease at open heart surgery’ EJBPT, 2021, 8(8): 71-75.
- Kukurtchyan N.S., Karapetyan G.R., Case report visualisation and Evaluation study of some patients with coronary artery bypass grafting 2024, European Society of Medicine. Medical research archives. 2024; 12/3. Doi org: /10.18103/mra.v.12
- Kukurtchyan N., Karapetyan.G., Kocharyan A., Voskanyan M., Hovhannisyan M., Farsyan N, Sahakyan I., Tumasyan N., ‘Microvessels Atheroscleroses study and Evaluation of some patients with coronary artery disease at open heart surgery in Armenia. The European Society of Medicine. Medical Research Archives, Volume 13 Issue 1, 2025.
- Kukurtchyan N.S., Karapetyan G.R., Patent 2844A. Rep Armenia. Staining method of histological material.
- L. Dear-Flosy, R. Cutiesver, S Cayso, Mp Gassia, M. Gonzales-Comes, L. Dear-Flores, R Sanchez, J.M. Carrasco, J. F. Madrid. ‘Intussusceptive angiogenesis and anterpart intussusceptive limphangiogenesis “ Histol. Histopathology 2022, oct., 35(10)1083-1103.
- Mentzez S.I., Konerding M.A. ‘Intussusception amuogenesis expansion and remodeling of microvascular network’ Angiogenesis 2014; 17, 499-509.
- Pries A. R., Regin B. ‘Coronary microcirculatory pathophysiology’ j. Eur. Heart 2017; 38; 478-488. Doi; 10.1093/eurheart j/ehv 760.
- Pries A.R, Coronary Microcirculation in Ischemic Heart Disease. PubMed Curry Pharm Des., 2018.
- Sorrob O., Van Der Wow, Chadler S., Ohanyan V., Tuni J.D., Chilian W.M., Mecus D., Bender S.B., Duncker D.J. ‘Experimental animal model of coronary microvascular disfunction’ Cardivascular research 2020; 116(4); 756-787. Doi.org:10.1093/CV2/cvas002
- Sucator V., Novos, Rubino N., Di Agestino A, Erolos Novo G., ‘Prognosis in patients with microvascular angina. A clinical follow up’ j. Cardiovascular. Med 2019, 20, 794-795. Doi: 10.2459/j.CM0000000000000842.
- Zhow A., Egginton S., Hudllicka O., Brown M.D. ‘Internal division of capillaries in rat skeletal muscle in response to chronic vasodilation treatment with alphas-antagonist pvarvosin’. Cell. Tissue Res 1992; 293; 293-303.

