Wharton’s Jelly Stem Cells in Retinitis Pigmentosa: Safety & Outcomes
Injection of Wharton’s Jelly-Derived Mesenchymal Stem Cells into the Subtenon Space in a Retinitis Pigmentosa Patient: A Case Report on Safety and Visual Outcomes
Qi Zhe Ngoo, MD, MMED(Ophthalmology)1, Nor Shahidah Khairullah, FRCPath2, Angelina Tiah, MD(MedSci)2*
- Department of Ophthalmology and Visual Science, School of Medical Sciences, University Sains Malaysia, Kota Bharu, Kelantan, Malaysia
- Century International Life Science Centre, Seri Kembangan, Selangor, Malaysia
OPEN ACCESS
PUBLISHED: 31 December 2025
CITATION: Ngoo, QZ., Khairullah, NS., Tiah, A., 2025. Injection of Wharton’s Jelly-Derived Mesenchymal Stem Cells into the Subtenon Space in a Retinitis Pigmentosa Patient: A Case Report on Safety and Visual Outcomes. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.7171
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i12.7171
ISSN 2375-1924
ABSTRACT
Introduction: Retinitis pigmentosa (RP) is an inherited retinal dystrophy which involves the progressive degeneration and dysfunction of the retina. It affects 1 in 4000 individuals worldwide. The clinical manifestation of retinitis pigmentosa often begins with night blindness, constriction of peripheral visual field with variable visual acuity and eventually total blindness. Despite the significant impact on the patient’s quality of life, there is no definitive cure for retinitis pigmentosa currently. Hence, it is necessary to explore innovative treatment that can preserve and slow down the programmed cell death of photoreceptors to delay visual deterioration and improve functional status of retinitis pigmentosa patients. This case report describes a 36 years old Chinese gentleman was diagnosed with retinitis pigmentosa for more than 25 years. He has no other medical illnesses or family history of retinitis pigmentosa and suffered from bilateral severe vision loss starting the age of 4 that has severely impacted his daily living activities.
Methods: The patient has received a total of 4 sessions of Wharton’s Jelly-derived Mesenchymal Stem Cells implanted in the subtenon space. The injection interval was 2 months for the first three injections and 5 months for the fourth injection.
Results: Following four subtenon injections of Wharton’s Jelly-Derived Mesenchymal Stem Cells (WJ-MSCs) over 10 months, the patient demonstrated improved visual function from light perception to hand motion with better light projection and shape recognition. OPTICAL COHERENCE TOMOGRAPHY showed increased central foveal thickness, partial reappearance of the ellipsoid zone, and preservation of macular architecture, while fundus photographs revealed stabilization of retinal changes without further pigment migration or vascular attenuation. No treatment-related complications were observed throughout the follow-up period.
Conclusion: Subtenon Wharton’s Jelly-Derived Mesenchymal Stem Cells therapy was safe and associated with structural stabilization and functional improvement in advanced retinitis pigmentosa. These findings support its potential as a regenerative and neuroprotective therapeutic option, warranting further controlled studies.
Keywords
Wharton’s Jelly-Derived Mesenchymal Stem Cells (WJ-MSCs), retinitis pigmentosa, subtenon implantation, ophthalmology.
INTRODUCTION
Retinitis pigmentosa (RP) comprises a heterogeneous group of inherited retinal dystrophies characterized by progressive degeneration of photoreceptors and the retinal pigment epithelium, ultimately leading to severe visual impairment and, in advanced stages, blindness. The mode of inheritance includes autosomal recessive (50–60%), autosomal dominant (30–40%), X-linked (5–15%), and sporadic forms with no identifiable pattern. A substantial proportion of patients progress to legal blindness by approximately 40 years of age, depending on genotype.
Although RP is fundamentally a genetically driven disorder, increasing evidence suggests that secondary inflammatory mechanisms contribute to disease progression. Apoptotic photoreceptors release pro-inflammatory cytokines and damage-associated molecular patterns, triggering activation of microglia and Müller cells and amplifying retinal degeneration. Supportive treatments—such as omega-3 fatty acids, taurine, and vitamin A—offer limited benefit. Gene therapy with voretigene neparvovec (Luxturna) has shown meaningful clinical outcomes but applies only to a small subset of patients with RPE65 mutations and remains cost-prohibitive for many.
Mesenchymal stem cells (MSCs), particularly those derived from Wharton’s jelly, have emerged as a potential therapeutic option because of their regenerative, immunomodulatory, and neurotrophic properties. Through paracrine signaling and extracellular vesicle release, MSCs may slow or halt photoreceptor apoptosis, stabilize the retinal microenvironment, and enhance cellular survival in RP. Here, we present a case exploring the safety and possible functional effects of subtenon Wharton’s Jelly-derived Mesenchymal Stem Cell injection in advanced retinitis pigmentosa.
CASE PRESENTATION
This case report describes a 36 years old Chinese gentleman who was diagnosed with retinitis pigmentosa for more than 25 years. He has no other medical illnesses or family history of retinitis pigmentosa and suffered from bilateral severe vision loss starting the age of 4 that has severely impacted his daily living activities. He has not received any prior mesenchymal stem cell or related therapy prior to enrolling into this study. However, he did take lutein supplements but there was no improvement in helping his eyesight. The diagnosis of retinitis pigmentosa was confirmed with ophthalmologist by history and clinical examination. Objective assessment such as optical coherence tomography (OCT) scan of the macula showed macula involvement of retinitis pigmentosa with significant thinning of retinal layers and disruption of the ellipsoid zone. Humphrey visual field showed almost total visual field lost especially at the periphery.
PREPARATION OF UMBILICAL CORD WHARTON’S JELLY-DERIVED MESENCHYMAL CELL
Preparation of Wharton’s jelly-derived mesenchymal stem cell used in this study were isolated from a single donated umbilical cord Wharton’s jelly. The mother’s consent was taken according to good clinical practice and the Helsinki declaration. It is then treated as followed:
The umbilical cord is washed with 0.9% sodium chloride injection and disinfected in filtered 70/75% medical grade ethanol after being immersed in cord preservative solution. The length of the umbilical cord is clamped with haemostats at both ends and measured before being cut into 2-3 cm small pieces. The Wharton’s jelly was processed by weighing, cutting, washing and centrifuging before being seeded in a complete culture media, incubated in a CO2 chamber under controlled conditions: 37°C, 5.0% CO2, and 95% relative humidity. The culture expanded cells were cryopreserved at passage 4 (P4) following standard cryopreservation protocols and stored until further use.
All cell preparation and cultivation procedures were performed at Beike 23 Century International Stem Cell Laboratory, an MOH-accredited cGMP/cGTP facility and underwent characterization via flow cytometric analysis in compliance with FDA and ISCT guidelines to confirm the expression of positive surface markers (CD90, CD73, CD105, CD29) and the absence of negative markers (CD45, CD34, CD79a, CD14, HLA-DR). Quality control testing included: mycoplasma analysis according to Ph.Eur 2.6.7; endotoxin analysis according to Ph.Eur 2.6.14. Microbial limit testing according to US-6.1, Sterility testing according to USP 7.1.
INJECTION OF WHARTON’S JELLY-DERIVED MESENCHYMAL STEM CELLS
He has received a total of 4 transplantations of Wharton’s Jelly-Derived Mesenchymal Stem Cells (WJ-MSCs) injected deep into the subtenon space ranging from 2 months apart and last injection with a 5 months gap. The first injection was given in October 2024 and the last injection was given on August 2025. Patient was then followed up closely with an optical coherence tomography test. Each course of subtenon injection consists of 10 to 12 million of WJ-MSCs suspended in 1.5 ml solution for each eye. He underwent four sessions of WJ-MSCs subtenon injection, which was carried out under aseptic technique.
PRE INJECTION FINDINGS
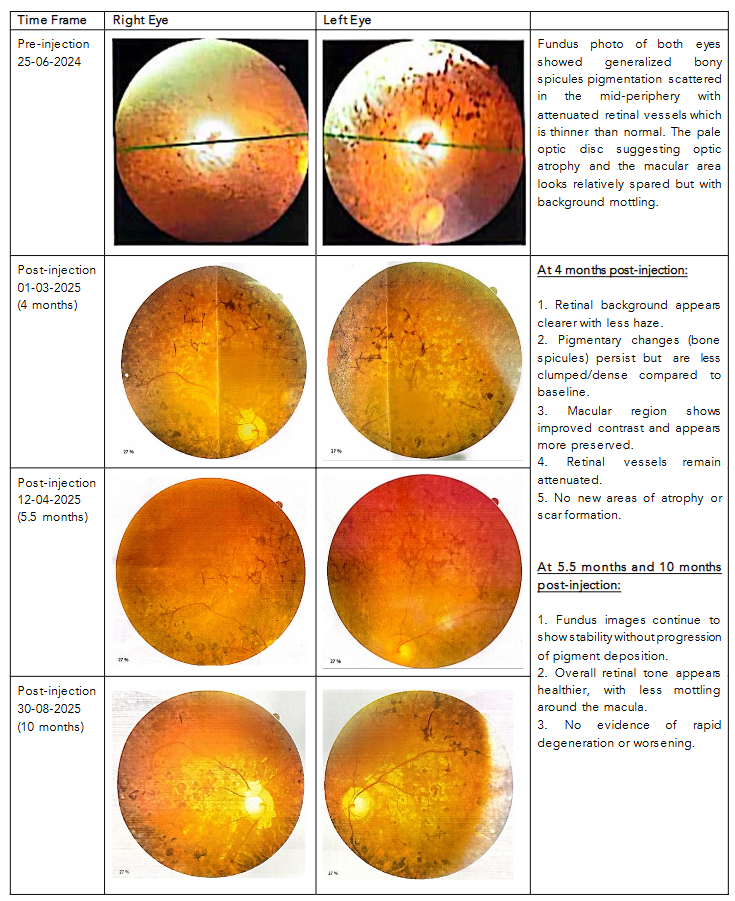
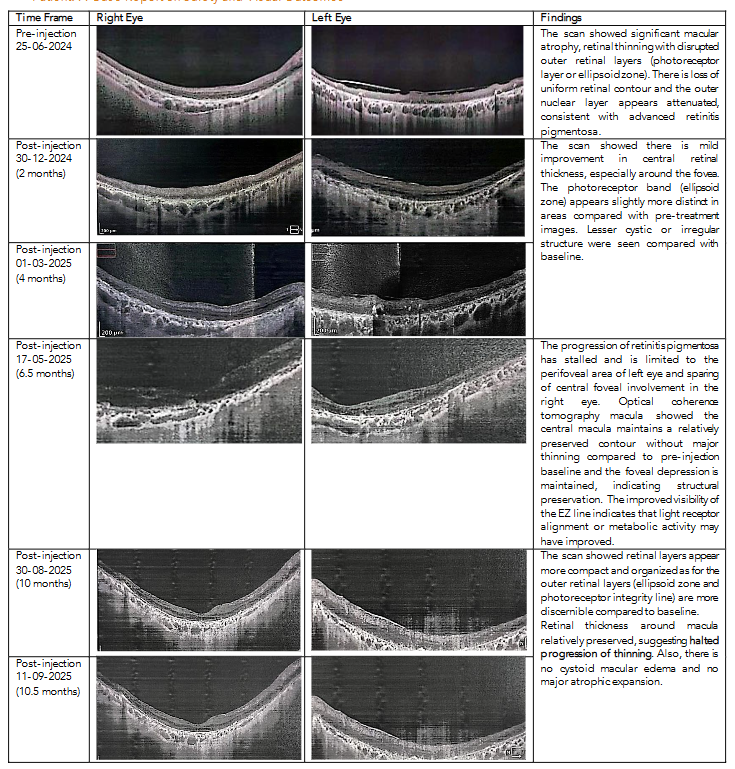
Patient had horizontal jerk nystagmus and vision was light perception with poor light projection bilaterally. Optical coherence tomography (OCT) test reveals significant macular thinning which signifies macular atrophy associated with retinitis pigmentosa. The ellipsoid zone (EZ) line was poorly defined, indicating photoreceptor degeneration and the outer nuclear layer (ONL) was attenuated. From retina fundus photo, we can see classic retinitis pigmentosa features are evident such as the marked bone spicule pigmentation scattered in the mid-periphery, attenuated retinal vessels and pale optic disc suggesting optic atrophy.
POST INJECTION FINDINGS
After 3 injections of subtenon WJ-MSCs, nystagmus improved. Vision has improved to hand motion with good light projection. Subjectively, patient claimed he could appreciate shapes and colours with moderate contrast sensitivity.
The central fundus images suggest stabilization of retinitis pigmentosa changes after WJ-MSCs injection starting 4 months post-injection. There are no signs of acute worsening (such as increased pigment migration, vessel loss, or macular scarring). Subtle improvements in retinal clarity and reduced pigment clumping may indicate some degree of metabolic or supportive benefit. The macular zone demonstrated better preservation and contrast. From the mid to late follow-up images, we can see that the fundus photographs showed stable retinal appearance without further pigment migration or vessel narrowing. The macula continued to appear preserved, with no evidence of atrophic expansion or scarring. Asides, there is no treatment-related complications (such as neovascularization, edema) were observed post treatment.
On optical coherence tomography macula, the central foveal thickness increases which signifies regeneration of the inner retinal layers. During the early post treatment phase, we can see improvement in the definition of the outer retinal architecture and the ellipsoid zone (EZ) became more distinct in the central macula with some central retinal thickness compared to baseline. After 6 months from the first injection, optical coherence tomography confirmed preserved macular contour and ONL showed less attenuation with improved structural density compared to baseline. Partial reappearance of the EZ line was noted, suggesting photoreceptor structural support. During the late follow-up phase (10 months after first injection), the retinal layers appeared more compact and organized with EZ and photoreceptor integrity were more visible than baseline. Overall optical coherence tomography demonstrated preservation of macular thickness, improved definition of the photoreceptor zone (EZ), and stabilization of retinal architecture over 10 months post-injection.
FUNDUS PHOTOGRAPH FINDINGS
| Time Frame | Right Eye | Left Eye |
|---|---|---|
| Pre-injection 25-06-2024 | Fundus photo of both eyes showed generalized bony spicules pigmentation scattered in the mid-periphery with attenuated retinal vessels which is thinner than normal. The pale optic disc suggesting optic atrophy and the macular area looks relatively spared but with background mottling. | Fundus photo of both eyes showed generalized bony spicules pigmentation scattered in the mid-periphery with attenuated retinal vessels which is thinner than normal. The pale optic disc suggesting optic atrophy and the macular area looks relatively spared but with background mottling. |
| Post-injection 01-03-2025 (4 months) |
|
|
| Post-injection 12-04-2025 (5.5 months) | Post-injection findings continue to show stability without progression of pigment deposition. | Post-injection findings continue to show stability without progression of pigment deposition. |
| Post-injection 30-08-2025 (10 months) | Overall retinal tone appears healthier, with less mottling around the macula. | Overall retinal tone appears healthier, with less mottling around the macula. |
OPTICAL COHERENCE TOMOGRAPHY FINDINGS
| Time Frame | Right Eye | Left Eye | Findings |
|---|---|---|---|
| Pre-injection 25-06-2024 | The scan showed significant macular atrophy, retinal thinning with disrupted outer retinal layers (photoreceptor layer or ellipsoid zone). There is loss of uniform retinal contour and the outer nuclear layer appears attenuated, consistent with advanced retinitis pigmentosa. | The scan showed significant macular atrophy, retinal thinning with disrupted outer retinal layers (photoreceptor layer or ellipsoid zone). There is loss of uniform retinal contour and the outer nuclear layer appears attenuated, consistent with advanced retinitis pigmentosa. | The scan showed significant macular atrophy, retinal thinning with disrupted outer retinal layers (photoreceptor layer or ellipsoid zone). There is loss of uniform retinal contour and the outer nuclear layer appears attenuated, consistent with advanced retinitis pigmentosa. |
| Post-injection 30-12-2024 (2 months) | The scan showed there is mild improvement in central retinal thickness, especially around the fovea. The photoreceptor band (ellipsoid zone) appears slightly more distinct in areas compared with pre-treatment images. Lesser cystic or irregular structure were seen compared with baseline. | The scan showed there is mild improvement in central retinal thickness, especially around the fovea. The photoreceptor band (ellipsoid zone) appears slightly more distinct in areas compared with pre-treatment images. Lesser cystic or irregular structure were seen compared with baseline. | The scan showed there is mild improvement in central retinal thickness, especially around the fovea. The photoreceptor band (ellipsoid zone) appears slightly more distinct in areas compared with pre-treatment images. Lesser cystic or irregular structure were seen compared with baseline. |
| Post-injection 01-03-2025 (4 months) | The progression of retinitis pigmentosa has stalled and is limited to the perifoveal area of left eye and sparing of central foveal involvement in the right eye. Optical coherence tomography macula showed the central macula maintains a relatively preserved contour without major thinning compared to pre-injection baseline and the foveal depression is maintained, indicating structural preservation. The improved visibility of the EZ line indicates that light receptor alignment or metabolic activity may have improved. | The progression of retinitis pigmentosa has stalled and is limited to the perifoveal area of left eye and sparing of central foveal involvement in the right eye. Optical coherence tomography macula showed the central macula maintains a relatively preserved contour without major thinning compared to pre-injection baseline and the foveal depression is maintained, indicating structural preservation. The improved visibility of the EZ line indicates that light receptor alignment or metabolic activity may have improved. | The progression of retinitis pigmentosa has stalled and is limited to the perifoveal area of left eye and sparing of central foveal involvement in the right eye. Optical coherence tomography macula showed the central macula maintains a relatively preserved contour without major thinning compared to pre-injection baseline and the foveal depression is maintained, indicating structural preservation. The improved visibility of the EZ line indicates that light receptor alignment or metabolic activity may have improved. |
| Post-injection 30-08-2025 (10 months) | The scan showed retinal layers appear more compact and organized as for the outer retinal layers (ellipsoid zone and photoreceptor integrity line) are more discernible compared to baseline. Retinal thickness around macula relatively preserved, suggesting halted progression of thinning. Also, there is no cystoid macular edema and no major atrophic expansion. | The scan showed retinal layers appear more compact and organized as for the outer retinal layers (ellipsoid zone and photoreceptor integrity line) are more discernible compared to baseline. Retinal thickness around macula relatively preserved, suggesting halted progression of thinning. Also, there is no cystoid macular edema and no major atrophic expansion. | The scan showed retinal layers appear more compact and organized as for the outer retinal layers (ellipsoid zone and photoreceptor integrity line) are more discernible compared to baseline. Retinal thickness around macula relatively preserved, suggesting halted progression of thinning. Also, there is no cystoid macular edema and no major atrophic expansion. |
DISCUSSION
Mesenchymal stem cell (MSC) therapy represents an expanding frontier in regenerative medicine, offering a biologically plausible approach to degenerative retinal diseases such as retinitis pigmentosa (RP). MSCs possess anti-inflammatory, neuroprotective, angiogenic, and trophic properties, and have demonstrated promise in conditions characterized by progressive photoreceptor loss. The source of MSCs includes adipose tissue, umbilical cord, and bone marrow, with Wharton’s Jelly–derived MSCs (WJ-MSCs) showing particular advantages owing to their regenerating ability, immunomodulatory effect, trophic secretion, low tumorigenesis, higher proliferation capacity, paracrine potency, and low immunogenicity.
This has garnered significant scientific interest for treating degenerative and autoimmune diseases. Emerging evidences suggest that retinitis pigmentosa’s pathogenesis not only genetic defects but also secondary inflammatory mechanisms and microglial activation. The apoptotic photoreceptors release pro-inflammatory chemokines and cytokines (such as IL-6, TNF-α, IL-1β) and damage-associated molecular patterns (DAMPs) activating microglia and Müller cells, perpetuating and exacerbating retinal degeneration through a chronic low-grade inflammatory environment and amplifying oxidative stress. This provides a strong biological rationale for using MSCs, which secrete immunomodulatory cytokines such as TGF-β, IL-10, and PGE2 and thereby attenuate inflammation while stabilizing the degenerating microenvironment and improves retinal metabolism by promoting angiogenesis and enhancing retinal pigment epithelium function through VEGF (vascular endothelial growth factor) and IGF-1 (insulin-like growth factor 1).
In addition to cytokine-mediated immunomodulation, MSCs release extracellular vesicles (EVs) and exosomes enriched with neurotrophic molecules, microRNAs, and anti-apoptotic cargo. These vesicles are now recognized as primary mediators of retinal repair. Mead et al. demonstrated that MSC-derived exosomes significantly enhance retinal neuron survival and promote neurite outgrowth, supporting their ability to rescue photoreceptors even without direct cell integration. Similar studies show that EVs can ameliorate oxidative stress, modulate microglial activity, and promote photoreceptor stability in models of inherited retinal degeneration. This paracrine and EV-based mechanism could explain the structural stabilization seen in our patient even months after injections.
Neurotrophic factor delivery is another critical mechanism. MSCs secrete glial cell line–derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), and basic fibroblast growth factor (bFGF), all of which support photoreceptor survival and reduce apoptosis. These mechanisms collectively align with the structural improvements observed in this patient, including increased central foveal thickness, improved definition of the ellipsoid zone (EZ), and preservation of macular contour over serial OCT assessments. Beyond paracrine activity, some studies have shown that mesenchymal stem cells can differentiate into retinal progenitor cells which then give rise to all major retina cells including photoreceptors and inner retinal conducting neural cells. The visual improvement in our patient primarily stems from the paracrine signalling of the implanted cells. These grafted cells are able to functionally integrate into the retinal tissue, potentially replacing lost photoreceptors and exert neuroprotective and neurotrophic effects to retain recipient functional photoreceptors. These were further proven objectively with the increase of central foveal thickness and the partial return of the fovea contour at the end of the treatment. Although this remains controversial, animal models have shown partial integration and photoreceptor replacement potential.
CLINICAL EVIDENCE SUPPORTING WHARTON’S JELLY-DERIVED MESENCHYMAL STEM CELLS USE IN RETINITIS PIGMENTOSA
Multiple human studies have assessed MSC-based therapies for RP and other retinal degenerations, supporting their safety and potential efficacy. Jonas and colleagues reported safety of intravitreal bone marrow–derived cells in RP eyes without major adverse events. Park et al. further demonstrated safety in a phase I trial using intravitreal autologous MSCs, with some functional improvement. Subretinal MSC implantation has shown stabilization of retinal structure over months in early-phase clinical trials.
Wharton’s Jelly–derived MSCs, in particular, have shown encouraging results. Özmert and Arslan reported improved visual function and retinal stability following WJ-MSC implantation in RP over one year. A similar study in Malaysia by 23 Century with a similar injection technique reported visual improvement using the same WJ-MSC source and subtenon delivery demonstrated visual gains and no adverse reactions. These findings are consistent with our case, where repeated subtenon injections yielded functional improvement from light perception to hand motion, enhanced light projection, and subjective improvements in shape recognition.
RATIONALE FOR SUBTENON DELIVERY AND DOSING
The subtenon route offers a minimally invasive alternative to subretinal or intravitreal injection. It avoids intraocular complications and allows sustained local exposure of MSC-secreted factors to posterior segment tissues. Prior clinical experience indicates favorable safety, making it suitable for repeat dosing as performed in this case. Unlike intravitreal methods, subtenon administration reduces the risk of proliferative vitreoretinopathy, neovascularization, or vitreous opacities.
Regarding dosage, this patient received approximately 10–12 million Wharton’s Jelly-Derived Mesenchymal Stem Cells per eye in four sessions. While repeat dosing appears to have enhanced structural and functional stabilization, the optimal dose and interval remain to be determined.
SAFETY PROFILE
In this case, no complications—including intraocular inflammation, infection, neovascularization, retinal edema, or worsening of retinal degeneration—were observed over 10.5 months of follow-up. This aligns with previously published human studies showing a strong safety profile for MSC-based therapies when performed in controlled clinical settings. This highlights the favorable safety profile of repeated subtenon WJ-MSCs injection in retinitis pigmentosa.
LIMITATIONS
Limitations of this report include the single-patient sample, lack of electrophysiological studies (such as Electroretinography) to correlate structural recovery with retinal function, and the relatively short follow-up of 10 months, which may not fully capture the long-term trajectory of disease stabilization. Larger, controlled trials are needed to validate these findings.
CONCLUSION
In conclusion, this case showed the use of Wharton’s Jelly-Derived Mesenchymal Stem Cells (WJ-MSCs) therapy in retinitis pigmentosa patients is safe with no adverse retinal events observed. This therapy demonstrated inner retinal cellular regeneration with structural improvements in photoreceptor-related layers (EZ and ONL), correlating with fundus evidence of macular preservation and halted progression of retinitis pigmentosa related retinal degeneration seen on optical coherence tomography. The improvements in functional vision, including better light projection and shape appreciation, further support clinical benefit. These findings suggest that WJ-MSCs therapy may slow photoreceptor loss, support retinal metabolism, and preserve retinal structure in RP through immunomodulation, neurotrophic factor release, EV-mediated repair, and possible regenerative mechanisms.
This case supports repeat subtenon Wharton’s Jelly-Derived Mesenchymal Stem Cells injection as a feasible and safe strategy in advanced retinitis pigmentosa. WJ-MSCs therapy may provide a broader therapeutic option across multiple retinitis pigmentosa genotypes providing advantages over mutation-specific therapies such as gene therapy. Continued clinical development may position MSC therapy as a viable adjunct or standalone approach for managing advanced RP.
Therefore, mesenchymal stem cells therapy can spark a new treatment option for retinitis pigmentosa patients as it may slow down the disease process by preserving the remaining function of the retina and potentially reverse the course of disease with no significant side effects. However, further studies are required to determine the optimal dosing, treatment intervals, and long-term safety. Well-designed, larger-scale clinical trials are necessary to establish WJ-MSCs therapy as a standardized treatment option for retinitis pigmentosa patients.
ACKNOWLEDGEMENTS
We would like to thank the Director General of Health Malaysia for allowing the publication of this manuscript and the staff of Department of Ophthalmology and Visual Science, University Sains Malaysia for their contribution and support to this study. We also thank the Chief Executive Officer of 23 Century International Life Science Centre for her continuous support in this case study.
CONFLICT OF INTEREST STATEMENT
None.
FUNDING STATEMENT
None.
REFERENCES
- Verbakel SK, van Huet RAC, Boon CJF, den Hollander AI, Collin RWJ, Klaver CCW, et al. Non-syndromic retinitis pigmentosa. Prog Retin Eye Res. 2018;66:157-86.
- Khairullah SN, Fariza NN, Roslin AZ, Angelina T. Subtenon implantation of wharton’s jelly-derived mesenchymal stromal cells in retinitis pigmentosa. The Medical journal of Malaysia. 2022 Sep 1; 77(5):564–8.
- Zhao L, Zabel MK, Wang X, Ma W, Shah P, Fariss RN, et al. Microglial phagocytosis of living photoreceptors contributes to inherited retinal degeneration. EMBO Mol Med. 2015;7(9):1179-97.
- Lee H, Lotery A. Gene therapy for RPE65-mediated inherited retinal dystrophy completes phase 3. Lancet. 2017;390(10097):823-4.
- Zhao L, Zabel MK, Wang X, Ma W, Shah P, Fariss RN, et al. Microglial phagocytosis of living photoreceptors contributes to inherited retinal degeneration. EMBO Mol Med. 2015;7(9):1179-97.
- Yoshida N, Ikeda Y, Notomi S, Ishikawa K, Murakami Y, Hisatomi T, et al. Microglia-initiated photoreceptor degeneration via IL-1β in inherited retinal degeneration. PLoS One. 2013;8(12):e79107.
- Siqueira RC, Messias A, Voltarelli JC, Scott IU, Jorge R. Intravitreal injection of autologous bone marrow-derived mononuclear cells for hereditary retinal dystrophy: a phase I trial. Stem Cells Int. 2015;2015:673631.
- Tzameret A, Sher I, Belkin M, Treves AJ, Meir A, Nagler A, et al. Transplantation of human bone marrow mesenchymal stem cells as a thin subretinal layer ameliorates retinal degeneration in a rat model of retinal dystrophy. Stem Cells Dev. 2015;24(1):10-23.
- Harrell CR, Jovicic N, Djonov V, Arsenijevic N, Volarevic V. Mesenchymal stem cell-derived exosomes and other extracellular vesicles as new remedies in the therapy of inflammatory diseases. Cells. 2019;8(12):1605.
- Özmert E, Arslan U. Management of retinitis pigmentosa by Wharton’s jelly-derived mesenchymal stem cells: prospective analysis of 1-year results. Stem Cell Res Ther. 2020;11(1):353.
- Jonas JB, Witzens-Harig M. Intravitreal autologous bone marrow–derived mononuclear cell transplantation: feasibility and effects in retinitis pigmentosa. Acta Ophthalmol. 2014;92 (6):e495–e496.
- Park SS, Bauer G, Abedi M, et al. Intravitreal autologous bone marrow–derived mesenchymal stem cells in retinal degeneration: a safety study. Ophthalmology. 2015;122(5):1053–1060.
- Arslan U, Özmert E. Wharton’s jelly–derived mesenchymal stem cell implantation in patients with advanced retinitis pigmentosa: 6-month follow-up. Int J Ophthalmol. 2021;14(4):487–494.
- García-Benítez L, Aller Á, Arias L, et al. Stability of retinal structure after subretinal adipose-derived mesenchymal stem cell transplantation in a phase I clinical trial for retinitis pigmentosa. Stem Cells Transl Med. 2021;10(10):1438–1448.
- Mead B, Berry M, Logan A, Scott RA, Leadbeater W, Scheven BA. Mesenchymal stem cell–derived exosomes enhance survival of retinal ganglion cells and neurite regeneration. Mol Ther. 2017;25(4):957–971.
- Llonch S, Carido M, Ader M. Organoid technology for retinal repair. Dev Biol. 2018;433(2):132–143.
- Johnson TV, Bull ND, Hunt DPJ, Marina N, Tomarev SI, Martin KR. Neuroprotective effects of intravitreal mesenchymal stem cell transplantation in experimental glaucoma. Invest Ophthalmol Vis Sci. 2010;51(4):2051–2059.
- Tsai Y, Lu B, Bakondi B, et al. Human iPSC-derived neural progenitors preserve vision in an AMD-like mouse model. Sci Transl Med. 2015;7(296):296ra110.
- Shen J, Tsutsumi S, Maeda N, et al. Anti-inflammatory effects of mesenchymal stem cells promote photoreceptor survival in a rodent model of retinal degeneration. Invest Ophthalmol Vis Sci. 2016;57(13):5562–5573.
- Wang S, Lu B, Girman S, et al. Non-integrating lentiviral vectors: a promising strategy for retinal gene therapy. Int J Ophthalmol. 2013;6(6):766–773.
- Petrou PA, Gothelf Y, Argov Z, et al. Safety and clinical effects of mesenchymal stem cell intravenous infusion in patients with neurodegenerative disorders. JAMA Neurol. 2016;73(3):337–344.
- Chen J, Luo Y, Hui H, et al. Wharton’s jelly–derived mesenchymal stem cells: biological characteristics and therapeutic potential. Stem Cell Res Ther. 2021;12(1):1–14.
- Becker S, Jayaram H, Limb GA. Recent advances towards the clinical application of stem cells for retinal regeneration. Cells. 2020;9(6):1443.
- Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368(9549):1795–1809.

