High Velocity Therapy for Warming Acutely Injured Patients
High Velocity Therapy as A Warming Tool for Acutely Injured Patients
Megan G. Wooldridge1, JJ. Lago Valdez2, María Chacón Gómez3, W. Douglas Gregorie4, Jessica S. Whittle5
- Department of Medical Affairs, Vapotherm, Exeter NH. ORCID: 0009-2497-139X
- Respiratory Therapy Consultant, Clinical Education Specialist, Texas, United States
- Division of intensive care and critical medicine, Centro Nacional de Investigacion y Atencion a Quemados (CENIAQ), National Rehabilitation Institute, Mexico
- Department of Emergency Medicine, University of Tennessee Health Science Center, Chattanooga TN. ORCID 0000-0002-3886-5240
- Department of Emergency Medicine, University of Tennessee Health Science Center, Chattanooga TN. Chief Medical Officer Vapotherm, Exeter NH. ORCID 0000-0003-3177-8934
OPEN ACCESS
PUBLISHED 31 December 2025
CITATION Wooldridge, MG., Valdez, JJL., et al., 2025. High Velocity Therapy as A Warming Tool for Acutely Injured Patients. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.7037
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i12.7037
ISSN 2375-1924
ABSTRACT
Background: Hypothalamic thermoregulation allows humans to adjust and maintain core body temperature to support essential metabolic processes. When heat energy loss exceeds endogenous heat production, core body temperature decreases, slowing biochemical reactions. Without patient warming solutions, patients with injuries resulting in thermoregulatory disturbances are at risk of multiple organ failure and/or death.
Aim: To illustrate the potential thermoregulatory effects of heated, humidified high velocity therapy in broad clinical scenarios which traditionally require active temperature management.
Scope: In the two patient examples presented, clinicians utilized high velocity therapy to modulate gas and heat exchange in the lungs (140m2 surface area). These patient examples demonstrate an opportunity to deliver adjunctive thermoregulatory support alongside non-invasive oxygenation and ventilation.
Example 1: A 67-year-old female presented with accidental hypothermia due to environmental exposure, with hyperextension injury at C4-C5. The patient’s core body temperature was 30°C [86°F] upon discovery. Within 4 hours the patient’s temperature increased to 35°C [95°F] with the use of heated blankets and high velocity therapy. The underlying injury and pattern of recovery was consistent with central cord syndrome.
Example 2: A 5-month-old female sustained severe burns and was weaned from intubation using high velocity therapy. This patient was at high risk for mucosal airway dysfunction, temperature instability, and other poor outcomes, but remained stable and recovered.
Discussion: High velocity therapy delivers warmed humidified gas that oxygenates and ventilates simultaneously, reduces work of breathing, and preserves mucociliary clearance. By reducing the metabolic burden of heating and humidifying inspired air, it may contribute to temperature stabilization in patients with impaired thermoregulatory capacity. In the cases presented, the therapy performed within its accepted safety profile with no identified adverse events.
Conclusion: High velocity therapy may provide adjunctive warming benefit for patients with diminished temperature control who require respiratory support. Further investigation is needed to quantify warming effects and define patient populations most likely to benefit.
Keywords
High velocity therapy, thermoregulation, hypothermia, burns, respiratory support
1. Introduction
High velocity therapy (HVT) may potentially assist thermoregulatory stabilization in acutely injured patients that require respiratory support. Clinicians may not be aware that the lung surface area covers between 100-140 m2 which can be targeted for temperature modulation via airway warming and humidification.
In the United States (US) hypothermia accounts for between 700 to 1,500 deaths each year, with approximately 12,000 emergency department (ED) admissions annually. Common rewarming methods such as extracorporeal membrane oxygenation (ECMO), indicated in the most severe cases and cardiac arrest, can cost up to US$13,384 daily. In 2020, The American Burn Association (ABA) commissioned a report of burn related incidence in the US and found 698,555 burn-related interactions with healthcare systems, with 3,800 deaths attributed to fire and smoke inhalation. The median total cost of burn injuries was reported to be up to US$125,598, with in-hospital costs per day ranging from US$24.23 to US$4,125: the most severe burns significantly associated with higher expenditure. Early implementation of multiple concurrent non-invasive rewarming solutions which reduce the metabolic cost of humidifying and heating inspired air may prevent escalation to more costly, invasive techniques.
The notion of regulated body temperature was pioneered by James Currie in 1798, using thermometers to quantify body temperature change following cold water immersion experiments. The clinical adoption of thermometry followed the 1868 publication of ‘Manual of Medical Thermometry’ by Wunderlich, which defined normative temperature ranges across health and disease. By the 1960s, Carlsson and colleagues identified the hypothalamus as the neurological thermoregulator, marking a pivotal shift in understanding behavioral and physiological responses to temperature changes.
Subsequent advancements in molecular biology have illuminated the coordinated thermoregulatory pathways required for endothermic life. Humans (Homo sapiens) are considered endothermic, generating internal heat to maintain a stable body temperature of 37 ± 0.5°C [98.6 ± 0.9°F] for optimal enzymatic and metabolic activity. In states of disease and injury, thermoregulatory pathways may be impaired by central nervous system dysfunction or overwhelmed by environmental conditions. Disruption of the systemic ability to thermoregulate, such as in hypothermia and major burns, result in loss of essential biochemical reactions required for metabolism, leading to eventual organ failure, coma and/or death.
Respiratory interventions have historically been explored as adjunctive warming strategies. In 1975 Lloyd et al. identified that inhalation of a warmed inhaled gas allowed for a 3°C [5.4°F] temperature rise over 3 hours for patients who suffered accidental hypothermia. The European Resuscitation Council guidelines for rewarming in hypothermia advocate for minimally invasive rewarming and airway management in patients with moderate to severe hypothermia, specifying use of high flow nasal cannula (HFNC) in patients with concomitant hypoxia. Similarly, ABA identified pulmonary insufficiency as the primary cause of death in severe burns and recommend early implementation of HFNC to reduce mortality and improve outcomes for major burn and inhalation injuries. The associated ABA report demonstrated HFNC as a successful intervention for treatment of inhalation injury and burn-related acute respiratory distress syndrome (ARDS).
1.1 HYPOTHERMIA
Hypothermia is defined as a core body temperature of < 35°C [95°F]. Causes of hypothermia include environmental exposure, perioperative heat loss, iatrogenic interventions and impaired thermoregulation in vulnerable populations such as neonates and the elderly. The patient in example 1 presents with accidental hypothermia via environmental exposure (expanded upon below [2.1.1]). Hypothermia may also develop in highly controlled environments such as the operating room or intensive care unit. Incidence of inadvertent perioperative hypothermia has been reported to vary from 10 to 80% of patients who undergo surgery, despite the accessibility of warming devices. The above iatrogenic causes can amplify risk of coagulopathy, wound infection, or prolong recovery, but are preventable with temperature management.
1.1.1 Environmental Exposure
The most recognized pathway into hypothermia remains environmental exposure. Sudden immersion events and prolonged exposure to cold air or wind can overwhelm thermoregulatory processes, particularly when combined with inadequate or wet clothing. Medical and social comorbidities can compound hypothermia risk, even in relatively mild conditions. Alcohol and drug intoxication can impair both judgment and thermoregulatory responses, while advanced age reduces baseline metabolic heat production. Neurologic injuries such as stroke or traumatic brain injuries blunt the hypothalamic thermoregulatory centers, while systemic illnesses such as sepsis or endocrine disorders further compromise thermogenesis. Advanced age and low body mass index are among the top risk factors for hypothermia from any cause. Despite advancements in hospital care, mortality rates in patients with moderate to severe hypothermia still approach 50%.
1.2 BURNS
Burns are injuries to the skin and deeper surrounding tissues caused by exposure to thermal, electrical, chemical, radiation, and frictional energy. Burn severity is classified by the depth of damage (first to fourth), and percentage of total body surface area (TBSA) affected. Burns greater than 15% TBSA can lead to hypovolemic shock, organ dysfunction and death of patient. The skin is the largest organ in the human body (surface area of up to 2m2 in adults) and forms a physical and immunological barrier to external agents, regulates temperature and metabolic homeostasis. Its destruction compromises systemic physiology, exposing patients to infections and thermoregulatory disturbance.
1.2.1 Airway and Inhalation injury
Burn patients commonly sustain airway injuries alongside inhalation injuries, which can result in ARDS; chronic pulmonary inflammation due to pneumonia, sepsis or septic shock. Inhalation of smoke or toxins may result in supraglottic or intrapulmonary injury, which induce edema, narrowing of the airways, and impair gas exchange in respiratory tissues. The most extensive burns generate greater edema and hypothermia risk. Airway lesions can generate similar respiratory sequelae with decreased internal diameter, obstruction, and presence of respiratory distress which require repeated intubation, tracheostomy, or non-invasive support in the form of HFNC. Inhalation injury was found to be the second leading cause of death in the first week post-burn (16%), following burn shock (62%). Patients with full thickness burns may have non-compliant dead tissues which contribute to poor perfusion and inadequate ventilation and oxygenation of tissues. Invasive mechanical ventilation is often required to prevent hypoxia, however up to 40% of ventilated patients develop ARDS. High flow nasal cannula is increasingly used to reduce hypoxemia, avoid re-intubation and support recovery, particularly when the ROX index is employed to predict extubation success.
1.3 HIGH VELOCITY THERAPY SYSTEM
High Velocity Therapy represents a refined form of HFNC and has proven to be an alternative to non-invasive positive pressure ventilation (NiPPV). Compared to other high flow systems HVT utilizes small-bore symmetrical nasal cannulas to increase kinetic energy of delivered medical gases, enhancing dead space washout and providing ventilatory benefits at lower flow rates. High velocity devices can deliver FiO2 ranging from 0.21 to 1.0 at flow rates of 5-45 L/min, with approximately 5-6 cmH2O positive end-expiratory pressure (PEEP), heated humidification (33mg H2O/L) and can support nebulization. Temperature can be easily adjusted for patient comfort and condition, with guidelines recommending 34 – 37°C [93.2 – 98.6°F] as optimal. High Velocity Therapy has a well-established safety and tolerability profile across populations. Interface options include a double-pronged nasal cannula and a single-pronged cannula (“Unicorn™”), the latter having potential advantages in patients with facial burns or trauma, naris obstruction, or patients who need concomitant respiratory support and nasogastric nutrition.
2. Patient Case Examples
2.1. PATIENT 1: ACCIDENTAL HYPOTHERMIA FROM ENVIRONMENTAL EXPOSURE
67-year-old female presented as a trauma alert with quadriplegia after a fall while intoxicated (approximately 01:00). She was found the next morning (08:00), and was transported to the ED. Upon arrival, she was alert and oriented but exhibited an inability to move all four extremities with intact sensation, classic for central cord syndrome (CCS). She was severely hypothermic at 30°C [86°F] and bradycardic. Magnetic Resonance Imaging (MRI) confirmed a C4-C5 hyperextension injury featuring a cord contusion, ligamentous damage, and an anterior epidural hemorrhage, consistent with diagnosis.
Treatment focused on prompt rewarming to address life-threatening hypothermia and autonomic dysfunction. She was managed with a heated blanket (Bair Hugger™) and high-velocity therapy (Vapotherm, 30 L/min, 0.21 FiO2, 38°C), which utilized the lungs’ surface area for efficient heat exchange. This strategy successfully raised core temperature from 30°C [86°F] to 34.8°C [94.7°F] within approximately four hours of adjunctive therapy initiation, stabilizing her for surgical intervention.
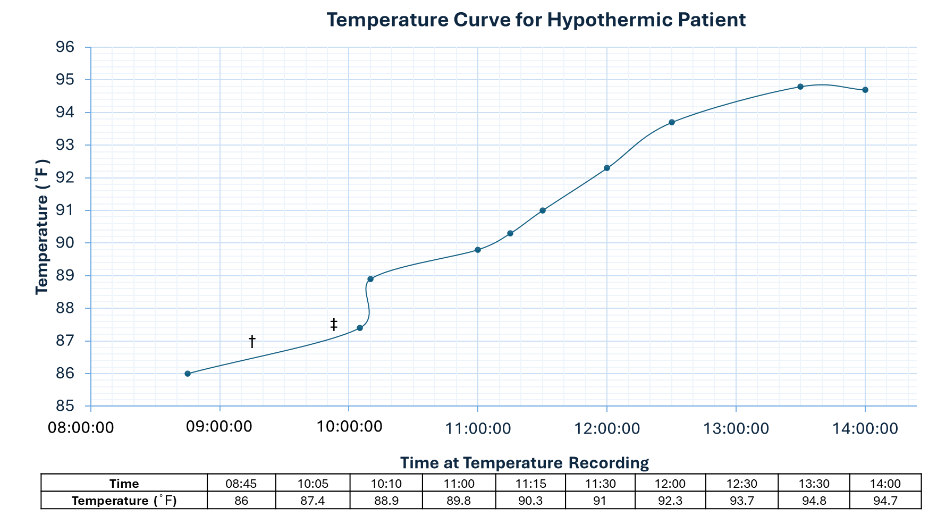
The patient was discharged to rehabilitation after eight days with moderate neurological improvement. While she regained shoulder strength and right toe flexion, deficits in hand grip and lower extremity mobility remained, a recovery pattern typical of CCS. This case illustrates the severe consequences of cervical hyperextension injuries and highlights the critical role of active airway rewarming as an effective adjunct therapy for rapidly correcting hypothermia in a trauma setting.
2.2. PATIENT 2: BURN PATIENT
A 5-month-old female suffered a fire burn at home in an enclosed space, with a burn extent of 14% TBSA. She was intubated at the accident site with a 3.5Fr cannula without a balloon. Carbonaceous debris was found, and she was nebulized with heparin. She underwent 24 hours of conventional mechanical ventilation. Due to respiratory deterioration, she progressed to high-frequency oscillatory ventilation for five days, weaned to conventional ventilation for another 48 hours, and extubated with an intravenous dexamethasone protocol at 0.25 mg/kg for four doses. She was extubated and presented with irritability, stridor, and wheezing audible at one meter. Nebulized adrenaline and steroids were administered on schedule. Subglottic stenosis was diagnosed via bronchoscopy, making her a candidate for tracheostomy. High-velocity therapy was initiated at 1 L/min/kg/dose (Vapotherm, 10 L/min, 0.4 FiO2, 34°C) and increased to 2 L/min/kg/dose (Vapotherm, 14 L/min, 0.45 FiO2 34°C), with clinical improvement.
| Time (Mins) | Clinical Data | Vital Signs Monitoring |
|---|---|---|
| 0 | + Irritability, + Wheezing, + Stridor | HR = 160, RR = 38, Sat (%) = 89, ROX = 5.8 |
| 30 | – Irritability, – Wheezing, – Stridor | HR = 130, RR = 17, Sat (%) = 97, ROX = 12.67 |
3. Discussion:
3.1 PATIENT CASE 1: HYPOTHERMIA
Physiological stabilization during rewarming was the primary goal for this patient as prompt rewarming enabled timely surgical management of cervical injury. The combined use of HVT and heated blanket achieved a mean rewarming rate of 1.0 ̊C /hr. [1.8 ̊F /hr.] (∆ 4.1 ̊C [∆ 7.3 ̊ F]) despite intermittent pauses in therapy. This rate is comparable with warmed IV fluids; 1.5 ± 1 ̊C /hr. (2.7 ± 1.8 ̊F /hr.), and heated blankets; 1.7 ̊C /hr. (range; 0.7-3.4 ̊C /hr. [3.1 ̊F /hr., range; 1.26-6.12 ̊F /hr.]). The observed temperature response supports the potential role of HVT as an adjunct to warming strategies, particularly when respiratory support is also indicated. While findings suggest an efficient and non-invasive means of internal rewarming, the contribution of HVT to overall temperature recovery cannot be isolated.
3.1.1 Treatment for Hypothermia
Immediate passive external rewarming involves removing wet clothing and covering the patient with dry, insulating layers while moving them to a warmer environment. Warm oral fluids may be administered to support internal heat generation. This approach is primarily indicated for mild hypothermia and may also be used in moderate hypothermia when more aggressive interventions are not immediately available. Passive rewarming supports compensatory heat production, while minimizing further heat loss. Passive methods are considered for initial interventions in hypothermic patients in both prehospital and clinical settings.
Active external rewarming methods elevate patients’ core temperature and are typically applied in clinical settings. Common methods include warming systems and heated blankets, as used in patient example 1. This approach is indicated for patients with moderate hypothermia or mild hypothermia where passive measures were insufficient. Evidence shows that heated blankets and warming systems effectively increase core temperature, especially when combined with insulation. These methods are widely recommended in perioperative and emergency medicine.
Severe hypothermia without cardiac arrest is treated with active internal rewarming. Active internal strategies such as ECMO and intravascular temperature management (IVTM) are invasive techniques that can achieve a rewarming rate of ≥ 5 ̊C /hr. (9 ̊F /hr.). Heated IV fluids should be warmed to 40-42°C [104-107.6°F] to avoid inadvertent cooling. Extracorporeal membrane oxygenation is indicated for patients in cardiac arrest or with cardiovascular instability.
3.1.2 Pathophysiology and Complications in Hypothermia
The combination of acidosis, hypothermia, and coagulopathy is coined as the ‘trauma triad of death’. It is important to carefully monitor trauma patients for signs associated with increased mortality. Early hypothermic responses include shivering, increased metabolic rate and elevated cardiac output which work to maintain core body temperature (Table 2: mild hypothermia). As energy reserves deplete, compensatory thermogenic mechanisms fail, and trigger progression to more severe hypothermia (Table 2: moderate-severe hypothermia). Major complications in grade II and above hypothermia, such as in patient case 1, include arrhythmia, low cardiac output states, coagulopathy, metabolic collapse, and cardiac arrest. As core temperatures drop below 30°C [86°F] there is an increased risk for malignant arrhythmia (ventricular fibrillation). Risk increases exponentially with moderate and severe hypothermia (< 30°C [86°F]) with profound hypothermia (< 28°C [82.4°F]) carrying the highest risk. Hypothermia depresses myocardial contractility, reduces heart rate and stroke volume. Gentle handling, and controlled rewarming are essential to reduce the risk of cardiac arrest and further heat loss. Afterdrop was not observed in patient case 1. Pulmonary complications can occur from hypothermia-induced disruption of the alveolar-capillary membrane. Cold stress and ischemia reperfusion during rewarming provoke cytokine release and free radical formation, which makes the pulmonary endothelium “leaky”. Furthermore, surfactant production and function become impaired which promotes alveolar collapse and pulmonary edema. Clinically, this mirrors the inflammatory microvascular injuries of ARDS which must be recognized early to prevent further complications.
| Classification (grade) | Core temperature range (°C [°F]) | Level of consciousness & Clinical Manifestations |
|---|---|---|
| Mild Hypothermia (I) | 32-35 [90-95] | Conscious, possibly altered, shivering GCS: 13-15 |
| Moderate Hypothermia (II) | 28-32 [82-90] | Impaired consciousness, with or without shivering GCS: 9-13 |
| Severe hypothermia (III, IV, V) | <28 [<82] | Unconsciousness, not shivering apparent death (<24°C), irreversible hypothermia (13.7°C) GCS: 3-9 |
3.2 PATIENT CASE 2: SEVERE BURNS WITH INHALATION INJURY
High velocity therapy was initiated immediately following extubation and titrated over 20 minutes from 10 L/min, 0.40 FiO2, to 14 L/min, 0.45 FiO2 in response to respiratory requirements. Within 30 minutes of HVT initiation, notable clinical improvements were observed. The patient’s irritability and audible signs of respiratory distress (inspiratory and expiratory wheeze and stridor) were markedly reduced. Similarly, vital sign parameters and monitoring observations also improved. Heart rate decreased from 160 bpm to 130 bpm, and respiratory rate declined from 38 to 17 breaths per minute within 30 minutes of therapy. Oxygen saturation (PaO2, Sat %) also increased from 89% to 97%. Corresponding improvements in the ROX index suggested enhanced respiratory stability and a reduced likelihood of weaning failure.
3.3 AIRWAY WARMING TO MAINTAIN TEMPERATURE
Airway warming with heated humidified oxygen can decrease insensible heat loss. The respiratory system, with an alveolar surface area of 100-140m2 represents a substantial yet overlooked conduit for heat exchange. High velocity therapy can support a patient’s oxygenation and ventilation requirements and offer an additional non-invasive active rewarming solution to stabilize patient temperature, without additional set-up or procedural disruptions. Adequate humidification is central to both respiratory support and thermal balance. Inhalation injuries result in direct cellular damage and disruption of mucociliary clearance (MCC), increase the risk of infections and may contribute to ARDS development. Following severe burn cases, fluid loss via plasma evaporation may exceed 4mL/kg/hr. It is imperative to retain as much fluid as possible to prevent hypovolemic shock, heat loss and maintain adequate tissue perfusion. Similarly, it is beneficial to provide early humidification in hypothermia, as inadequate humidity has been found to decrease the airway surface layer temperature by 5°C [9°F]. Adequately humidifying inspired air preserves MCC and respiratory physiology, reduces daily evaporative water by approximately 250 mL, and conserves metabolic heat by 350 kCal or 1500 kJ. In addition, adequate humidification has been shown to improve tolerance, with discomfort reported after only 10 minutes with inadequate humidification.
3.3.1 Use of High Flow Nasal Cannula in Hypothermia
Evidence for HFNC usage in hypothermia appears as small cohorts, case reports and animal studies, but remains promising. The use of HVT specifically has been reported to aid quicker time to normothermia when compared to current non-invasive active rewarming strategies. In a control matched study by Gilardi et al. patients with primary hypothermia (median temperature on admission 32.4°C [90.3°F]) were treated with HFNC or warm blankets as a control (n=8). Median time to sustained rewarming (>35°C [95 °F]) was 120 minutes in HFNC arm vs 345 minutes in the control arm, with patients rewarmed via HFNC having significantly higher body temperature at 5 hours compared to control. Similarly, Petersen et al compared the time to reach normothermia in patients with unintended perioperative hypothermia using heated blankets (n=15) versus HFNC (n=10). Patients warmed with HFNC regained normothermia 9.1 minutes faster than those in heated blanket group, although differences were not statistically significant. In the studies discussed above, no treatment adverse events were reported, which confirm the known safety spectrum and tolerability of HFNC devices. Importantly, no evidence was found to suggest the two modalities, HFNC and heated blankets, could not be used simultaneously.
3.3.2 Use of High Flow Nasal Cannula in Burns
High flow nasal cannula has been proven beneficial in acute respiratory failure from major burns, with reduced symptoms and lowered re-intubation rate. Marco et al. evaluated the safety and efficacy of HFNC for severely burned adult patients during enzymatic debridement procedures under deep sedation (n=14, mean 35.3% TBSA, second-third degree lesions, 0.5 FiO2). High flow nasal cannula was reported as invaluable to critically ill burn patients due to the ability to: deliver 1.0 FiO2, low PEEP, dead space washout, reduced risk of rebreathing, ability to precisely control the flow of heated and humidified inspired gas. Additionally, authors comment on the utility of a single pronged nasal cannula which blocks only one nostril for patients fed via NG tube but require concomitant nasal oxygen therapy. Marco et al. concluded HFNC should be considered for deep sedation for escharotomy procedures in severely burned patients. The effectiveness of high flow (>15 L/min, FiO2 0.45) vs conventional nasal oxygen therapy (<15 L/min) was evaluated in acute parenchymatous respiratory failure in burns without inhalation trauma. At 48 hours, HFNC patients had higher values of oxygenation (342 vs 305.5, p=0.02, n=74), faster normalization of blood gas composition, greater respiratory comfort (8.4 vs 5.3 Visual Analog Scale scores, p=0.03), and lower need for mechanical ventilation (4 vs 11, p=0.04), compared to control. Comparably, Akin et al reported HFNC to be a successful treatment for burn injury related to ARDS. After 7 days of HFNC treatment, ubiquitous improvements were seen in all symptoms of ARDS (n=3, 35-60% TBSA).
3.3.3 Using High Velocity Therapy for Weaning from Intubation
Whilst necessary for survival, prolonged endotracheal intubation (IMV) is associated with pneumonia, airway injury and increased risk of reintubation. Weaning from IMV represents the transition from total ventilatory support to spontaneous breathing and is an important milestone in recovery for patients requiring respiratory support. HFNC offers the opportunity to avoid IMV-associated complications and is increasingly used in extubation protocols. HFNC implementation during planned extubation protocols has been shown to reduce post-extubation respiratory failure, decrease respiratory rate, increase PaO2 and has further proven statistically non-inferior to NiPPV in multiple studies. Francesco et al. reported on 12 severe burn patients (7M, 5F, mean age 53 [38-70]) who were extubated and weaned to HFNC after a mean duration of 10.4 days on IMV (>30% TBSA, covered head and neck). Transition to HFNC improved PaO2: FiO2 ratios (Initiation vs 48 hours; 379.33 ± 31.56 vs 292.58 ± 41.25). No patient showed signs of respiratory distress or significant increase in lactate levels at 6 hours. Furthermore, the low contact nasal cannula interface was found to be more comfortable than NiPPV and allowed patients to practice respiratory physical therapy. The combined treatment expedited recovery compared to patients treated with only passive physical therapy. Similar benefits in a pediatric burn patient with inhalation injury and post-extubation stridor (12 – month- old female; 8% TBSA) were found by Byerly et al. The use of HVT decreased respiration rate immediately following initiation (63 breaths/min to 38 breaths/min), and a sustained decrease in heart rate. (175 beats/min to 144 beats/min).
4. Limitations
This concept paper is based on two illustrative patient examples selected to demonstrate potential thermoregulatory benefits of heated, humidified HVT in broad clinical contexts where respiratory support is already indicated. These examples do not represent the full spectrum of patients who may benefit from HVT. Observations are descriptive and inherently limited by absence of controlled conditions, small sample size, and short duration of therapy.
5. Call for more research
The relevant literature available for airway warming in hypothermia and burn patients is discussed in this paper, although the magnitude of thermoregulatory effect remains undefined. Larger prospective cohorts and controlled trials are needed to validate preliminary findings and should incorporate longer-term monitoring, standardized temperature metrics, and comparative modalities to quantify the adjunctive role HVT can offer.
6. Conclusion
High velocity therapy remains a clinically accepted alternative to NiPPV capable of reducing work of breathing, aiding extubation protocols, and improving patient comfort. The patient examples and discussed literature suggest HVT may also confer thermoregulatory advantages when applied to patients with temperature instability, however definitive evidence regarding thermal efficacy is limited. HVT can provide adequate non-invasive respiratory support to spontaneously breathing patients and may offer additional thermoregulatory advantages in line with existing guidance without procedural disruptions.
7. Conflicts of interest
Megan Wooldridge and Jessica Whittle are employees of Vapotherm. Maria Chacon and JJ. Lago Valdez are consultants for Vapotherm.
8. Acknowledgments
Authors would like to acknowledge Jeanne Pettinichi for her critical review and appraisal of this paper.
Funding Statement: Submission via membership – no funding received for this work.
9. References
- Petersson J, Glenny RW, Respir E. Gas exchange and ventilation-perfusion relationships in the lung REVIEW IN PRESS | CORRECTED PROOF ERJ Express. J (Basel). Published online 2014. doi:10.1183/09031936.00037014
- Paal P, Pasquier M, Darocha T, et al. Accidental Hypothermia: 2021 Update. Int J Environ Res Public Health. 2022;19(1):501. doi:10.3390/IJERPH19010501
- Teresiński G, Staśkiewicz G. Hypothermia. Forensic Imaging of Trauma. Published online January 19, 2024:141-167. doi:10.1007/978-3-031-48381-3_8
- Oude Lansink-Hartgring A, van Minnen O, Vermeulen KM, et al. Hospital Costs of Extracorporeal Membrane Oxygenation in Adults: A Systematic Review. Pharmacoecon Open. 2021;5(4):613. doi:10.1007/S41669-021-00272-9
- Fire loss in the United States | NFPA Research. Accessed October 14, 2025. https://www.nfpa.org/education-and-research/research/nfpa-research/fire-statistical-reports/fire-loss-in-the-united-states
- Ivanko A, Garbuzov AE, Schoen JE, et al. The Burden of Burns: An Analysis of Public Health Measures. Journal of Burn Care & Research. 2024;45(5):1095-1097. doi:10.1093/JBCR/IRAE053
- Lam NN, Hung NT, Khanh NN. Cost Analysis and Influencing Factors Amongst Severe Burn Patients. Ann Burns Fire Disasters. 2023;36(3):183. Accessed October 14, 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC11041904/
- Saavedra PAE, Leal JVDO, Areda CA, Galato D. The Costs of Burn Victim Hospital Care around the World: A Systematic Review. Iran J Public Health. 2021;50(5):866. doi:10.18502/IJPH.V50I5.6104
- Biffi S, Di Bella S, Scaravilli V, et al. Infections during extracorporeal membrane oxygenation: epidemiology, risk factors, pathogenesis and prevention. Int J Antimicrob Agents. 2017;50(1):9-16. doi:10.1016/j.ijantimicag.2017.02.025
- Yi N, Wang Z, Cui R. Warming with a composite warming strategy reduces intraoperative hypothermia in patients undergoing open hepatectomy for liver cancer: A randomized controlled study. Medicine. 2025;104(8):e41616. doi:10.1097/MD.0000000000041616
- Cooper KE. Some historical perspectives on thermoregulation. J Appl Physiol. 2002;92:1717-1724. doi:10.1152/japplphysiol.01051.2001.-In
- Oprita B, Olaru I, Botezatu L, Diaconu AE, Oprita R. Management of Severe Hypothermia: Challenges and Advanced Strategies. J Clin Med. 2025;14(5):1584. doi:10.3390/JCM14051584
- Paal P, Gordon L, Strapazzon G, et al. Accidental hypothermia-an update. Scand J Trauma Resusc Emerg Med. 2016;24(1):1-20. doi:10.1186/S13049-016-0303-7/FIGURES/2
- Lloyd EL, Senior FFARCS. 48 AIRWAY WARMING IN ACCIDENTAL HYPOTHERMIA.
- Lott C, Truhlář A, Alfonzo A, et al. European Resuscitation Council Guidelines 2021: Cardiac arrest in special circumstances. Resuscitation. 2021;161:152-219. doi:10.1016/J.RESUSCITATION.2021.02.011
- Akın M, Tuncer HB, Akgün AE, Erkılıç E. New Treatment Modality for Burn Injury-Related Acute Respiratory Distress Syndrome: High-Flow Nasal Oxygen Therapy in Major Burns. J Burn Care Res. 2024;45(4):1060-1065. doi:10.1093/JBCR/IRAE066
- Teległów A, Cicha I. Single immersion in cold water below 4 °C: A health hazard in young healthy men? PLoS One. 2025;20(5):e0324502. doi:10.1371/journal.pone.0324502
- Peixoto C de A, Ferreira MBG, dos Santos Felix MM, et al. Factors contributing to intraoperative hypothermia in patients undergoing elective surgery. Perioper Care Oper Room Manag. 2021;22:100150. doi:10.1016/J.PCORM.2020.100150
- Science P, Wang J feng, Deng X ming. Inadvertent hypothermia: a prevalent perioperative issue that remains to be improved. Anesthesiology and Perioperative Science 2023 1:3. 2023;1(3):1-13. doi:10.1007/S44254-023-00022-6
- Vacante M, Biondi A, Basile F, et al. Hypothyroidism as a Predictor of Surgical Outcomes in the Elderly. Front Endocrinol (Lausanne). 2019;10(APR). doi:10.3389/FENDO.2019.00258
- Yoo JH, Sung TY, Oh CS. Cold temperatures, hot risks: perioperative hypothermia in geriatric patients – a narrative review. Anesth Pain Med (Seoul). 2025;20(3):189-199. doi:10.17085/APM.25294
- Rauch S, Miller C, Bräuer A, Wallner B, Bock M, Paal P. Perioperative Hypothermia-A Narrative Review. Int J Environ Res Public Health. 2021;18(16). doi:10.3390/IJERPH18168749
- Gowda R, Jaffa M, Badjatia N. Thermoregulation in brain injury. Handb Clin Neurol. 2018;157:789-797. doi:10.1016/B978-0-444-64074-1.00049-5
- Cheshire WP. Thermoregulatory disorders and illness related to heat and cold stress. Autonomic Neuroscience. 2016;196:91-104. doi:10.1016/J.AUTNEU.2016.01.001
- Doman M, Thy M, Dessajan J, et al. Temperature control in sepsis. Front Med (Lausanne). 2023;10. doi:10.3389/FMED.2023.1292468
- Warby R, Maani C V. Burn Classification. StatPearls. Published online September 26, 2023. Accessed September 12, 2025. https://www.ncbi.nlm.nih.gov/books/NBK539773/
- Body surface area in children (image) | Medicines guidance | BNFC | NICE. Accessed September 11, 2025. https://bnfc.nice.org.uk/medicines-guidance/body-surface-area-in-children-image/
- McCann C, Watson A, Barnes D. Major burns: Part 1. Epidemiology, pathophysiology and initial management. BJA Educ. 2022;22(3):94-103. doi:10.1016/j.bjae.2021.10.001
- Driver J, Fielding A, Mullhi R, Chipp E, Torlinski T. Temperature management of adult burn patients in intensive care: findings from a retrospective cohort study in a tertiary centre in the United Kingdom. Anaesthesiol Intensive Ther. 2022;54(3):226. doi:10.5114/AIT.2022.119131
- Żwierełło W, Piorun K, Skórka-Majewicz M, Maruszewska A, Antoniewski J, Gutowska I. Burns: Classification, Pathophysiology, and Treatment: A Review. Int J Mol Sci. 2023;24(4):3749. doi:10.3390/IJMS24043749
- Alshammari RA, Qaisi HM, Zaben M, et al. Burns: Biochemical Aspects, Pathophysiology, Classification, Treatment, and Nursing Care. Journal of Medicinal and Chemical Sciences. 2024;7(12):1754-1770. doi:10.26655/JMCHEMSCI.2024.12.1
- Zimmermann L, Maiellare F, Veyckemans F, et al. Airway management in pediatrics: improving safety. J Anesth. 2024;39(1):123. doi:10.1007/S00540-024-03428-Z
- Byerly FL, Haithcock JA, Buchanan IB, Short KA, Cairns BA. Use of high flow nasal cannula on a pediatric burn patient with inhalation injury and post-extubation stridor. Burns. 2006;32(1):121-125. doi:10.1016/J.BURNS.2005.05.003
- Francesco C, Vincenzo P, Antonio T, et al. High-flow nasal cannula oxygen therapy in the weaning of severe burn patients: a preliminary report of data collection. Signa Vitae. 2023;19(4):58-62. doi:10.22514/sv.2023.009
- Petersen P, Liberio J, Dominguez S, Tojong J, Sanford A, Baldea A. 210 The Use of Humidified High Flow Nasal Cannula Post-Extubation to Decrease the Incidence of Reintubations in Adult and Pediatric Patients Treated in a Burn Center. Journal of Burn Care & Research. 2019;40(Supplement_1):S85-S85. doi:10.1093/JBCR/IRZ013.138
- Swanson JW, Otto AM, Gibran NS, et al. Trajectories to death in patients with burn injury. J Trauma Acute Care Surg. 2013;74(1):282-288. doi:10.1097/TA.0B013E3182788A1C
- Sjoberg F, Elmasry M, Abdelrahman I, et al. The impact and validity of the Berlin criteria on burn-induced ARDS: Examining mortality rates, and inhalation injury influences. A single center observational cohort study. Burns. 2024;50(6):1528-1535. doi:10.1016/J.BURNS.2024.05.005
- Bittner E, Sheridan R. Acute Respiratory Distress Syndrome, Mechanical Ventilation, and Inhalation Injury in Burn Patients. Surg Clin North Am. 2023;103(3):439. doi:10.1016/J.SUC.2023.01.006
- Hernández G, Vaquero C, González P, et al. Effect of Postextubation High-Flow Nasal Cannula vs Conventional Oxygen Therapy on Reintubation in Low-Risk Patients: A Randomized Clinical Trial. JAMA. 2016;315(13):1354-1361. doi:10.1001/JAMA.2016.2711
- Zhou X, Liu J, Pan J, Xu Z, Xu J. The ROX index as a predictor of high-flow nasal cannula outcome in pneumonia patients with acute hypoxemic respiratory failure: a systematic review and meta-analysis. BMC Pulm Med. 2022;22(1):121. doi:10.1186/S12890-022-01914-2
- Haywood ST, Whittle JS, Volakis LI, et al. HVNI vs NIPPV in the treatment of acute decompensated heart failure: Subgroup analysis of a multi-center trial in the ED. Am J Emerg Med. 2019;37(11):2084-2090. doi:10.1016/j.ajem.2019.03.002
- Yamane DP, Jones CW, Wilkerson RG, et al. High-velocity nasal insufflation versus noninvasive positive pressure ventilation for moderate acute exacerbation of chronic obstructive pulmonary disease in the emergency department: A randomized clinical trial. Academic Emergency Medicine. 2025;32(4):403-413. doi:10.1111/ACEM.15038
- Doshi P, Whittle JS, Bublewicz M, et al. High-Velocity Nasal Insufflation in the Treatment of Respiratory Failure: A Randomized Clinical Trial. Ann Emerg Med. 2018;72(1):73-83.e5. doi:10.1016/J.ANNEMERGMED.2017.12.006
- Milési C, Boubal M, Jacquot A, et al. High-flow nasal cannula: recommendations for daily practice in pediatrics. Ann Intensive Care. 2014;4(1):29. doi:10.1186/S13613-014-0029-5
- Smith AL, Kelly DP, Ruiz EA, et al. Optimizing High-Flow Nasal Cannula Weaning in Patients With Bronchiolitis. Hosp Pediatr. 2025;15(6):511-518. doi:10.1542/HPEDS.2024-008141
- Mcqueen M, Rojas J, Sun SC, et al. Safety and Long Term Outcomes with High Flow Nasal Cannula Therapy in Neonatology: A Large Retrospective Cohort Study. J Pulm Respir Med. 2014;4(6):216. doi:10.4172/2161-105X.1000216
- Marco DG. Use Of High Flow Nasal Cannula In Critical Burn Patient During Deep Sedation In Enzymatic Bromelain Debridement (Nexobrid ® ): A Single Center Brief Report Utilisation D’oxygène À Haut Débit Sur Lunettes Nasales Pendant La Sédation Profonde Nécessitée Par Le Débridement Enzymatique À La Bromélaïne (Nexobrid ® ) Chez Des Patients Graves: Une Courte Série Monocentrique. Ann Burns Fire Disasters. Published online 2024.
- UNICORN® Nasal Cannulas | Vapotherm. Accessed October 6, 2025. https://vapotherm.com/unicorn/
- Atwood C, Sethi J, Bergeski A, Dungan GC, Volakis LI, Whittle JS. Effect of Single-Prong Cannula Design With High Velocity Therapy: Comparable Efficacy at Lower Gas Flow Rates. Crit Care Explor. 2025;7(2):e1209. doi:10.1097/CCE.0000000000001209
- Kimberger O, Held C, Stadelmann K, et al. Resistive polymer versus forced-air warming: Comparable heat transfer and core rewarming rates in volunteers. Anesth Analg. 2008;107(5):1621-1626. doi:10.1213/ANE.0B013E3181845502
- Kornberger E, Schwarz B, Lindner KH, Mair P. Forced air surface rewarming in patients with severe accidental hypothermia. Resuscitation. 1999;41(2):105-111. doi:10.1016/S0300-9572(99)00069-6
- Levi Kitchen C. Joint Trauma System Clinical Practice Guideline (JTS CPG).
- Xu H, Xu G, Ren C, Liu L, Wei L. Effect of forced-air warming system in prevention of postoperative hypothermia in elderly patients: A Prospective controlled trial. Medicine. 2019;98(22). doi:10.1097/MD.0000000000015895
- Al-Dardery NM, Abdelwahab OA, El-Samahy M, Seif AM, Mouffokes A, Khaity A. Self-warming blankets versus active warming by forced-air devices for preventing hypothermia: A systematic review and meta-analysis. Medicine. 2023; 102(18):E33579. doi:10.1097/MD.0000000000033579
- Mccullough L, Arora S. Diagnosis and Treatment of Hypothermia. Am Fam Physician. 2004;70(12): 2325-2332. Accessed September 23, 2025. https://www.aafp.org/pubs/afp/issues/2004/1215/p2325.html

