Transgenerational Dioxin Risks in Children: Half-Life Insights
Transgenerational Implications of Age-Dependent Half-Life of Dioxins in Children
Brent D. Kerger1 and Anne E. Loccisano2
- Exponent Inc., Tampa, FL USA
- Exponent Inc., Alexandria, VA USA
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION: Kerger, BD. and Loccisano, AE., 2025. Transgenerational Implications of Age-Dependent Half-Life of Dioxins in Children. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6902
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6902
ISSN 2375-1924
ABSTRACT
Transgenerational health risks of dioxin-like compounds (dioxins) have been a prominent concern due to environmental persistence and food chain bioaccumulation related to combustion processes and widespread historical usage of certain commercial compounds like 2,4,5-trichlorophenoxyacetate and polychlorinated biphenyls. Earlier research showed that human adults excreted dioxins extremely slowly, e.g., with a plasma clearance half-life of perhaps 7 to 11 years, which indicated potential transfer of elevated maternal body burden via breast-feeding. These concerns led to the development of regulatory strategies targeting the reduction of environmental releases of dioxins to help reduce childhood and maternal body burdens and associated health risks. Fortunately, these regulatory strategies resulted in substantially reduced dioxin body burdens and further research revealed that infants, adolescents, and young adults exhibit an age-dependent half-life, e.g., breast-fed infants showed dioxin half-life of ~0.4 years with only gradual increase with each subsequent year (e.g., 0.12 years per annum). These findings equate with reduced health concerns to children and mothers today and for future generations. Available research of highly exposed populations also demonstrates a concentration-dependent dioxin half-life reduction relating to liver enzyme induction triggered by aryl hydrocarbon receptor activation starting at relatively high blood dioxin concentrations. Epidemiological studies of the highest dioxin exposures demonstrate this liver enzyme induction and chloracne, but other potential effects were clinically subtle or not coherently linked to the aryl hydrocarbon receptor-driven toxicological impacts. These highly exposed populations have not demonstrated strong and consistent evidence of increased cause-specific cancer rates, and proposed epidemiology-based dose-response factors for total cancer and other specific diseases may be overstated due to the influences of age- and concentration-dependent dioxin half-lives on body burden estimates. Thus, while continued research of potential dioxin health hazards may be warranted, researchers must carefully consider influences of age- and concentration-dependent pharmacokinetics, diminishing background exposures, and aryl hydrocarbon receptor-based mechanism of action that likely limit transgenerational health impacts to young children and mothers going forward.
Keywords: pharmacokinetics, human, exposure, internal dose modeling.
1.0 Introduction
Exposures to infants and young children have been a prominent concern for human health risk assessment of dioxin-like compounds or DLCs. These highly lipophilic and persistent compounds are transferred from mother to infant during breast-feeding and are known to be present in the fats of dairy products, meats, and other foods commonly consumed by young children. The higher fat intake per body weight in younger children (e.g., ages 0-2 years) reinforces concerns that they receive considerably higher intake of DLCs per body weight compared to adults. Moreover, the greater hand-to-mouth contact rates among younger children may lead to intake of DLCs present in environmental sources such as contaminated soils. Accordingly, the combined impact of these higher potential exposures to dietary and environmental sources of DLCs in young children and the associated health risks must be properly characterized and understood.
It is commonly assumed that children may be more sensitive to potential toxic effects of chemicals in general based on conservative choices for childhood exposure parameters in risk assessment. Enhanced concerns may apply to chemicals with potential neurodevelopmental effects like heavy metals (e.g., mercury and lead) and DLCs that are known to bioaccumulate through the food chain and in relevant human tissues like the brain. Bioaccumulation leads to potential hazards that are not well predicted by daily dose, but rather must be examined with respect to body burden or target organ/tissue concentration accumulated over time. While animal toxicology studies have identified a wide range of adverse effects of DLCs in species shown to be sensitive to aryl hydrocarbon receptor (AhR) driven high dose toxicity, long-term medical monitoring of highly exposed human populations have failed to demonstrate strong, specific, consistent and coherent evidence of chronic disease causation other than chloracne and liver enzyme induction. Chloracne is a skin disease involving acne-like lesions and hyperpigmentation that can occur within days after relatively high doses of 2,3,7,8-tetrachlorodibenzo-p-dioxin (dioxin) via inhalation, ingestion, or dermal contact. Dioxin is thought to accumulate in sebaceous gland oils and eventually cause inflammation and blockage of these glands in skin. Differing density of sebaceous glands leads to a characteristic pattern of occurrence of chloracne lesions that allows this condition to be differentiated from adolescent acne (acne vulgaris). Like acne vulgaris, chloracne can occur as clusters of dark-colored keratin plugs (blackheads) or comedones (bumps) on more oily regions of the face. However, chloracne is more prominent and persistent in the malar crescent region around the eyes, behind the ears, and on the genitals. With higher exposures the chloracne may involve all skin regions except the nose, which is somehow spared. In more severe cases, the chloracne-related lesions can be present for decades and are resilient to common acne treatments.
Dioxin is unique in its relatively high potency to cause adverse effects on reproduction, growth, development, certain organ dysfunctions, and cancers in certain experimental animal species. However, major species differences in susceptibility to dioxin have been demonstrated, and this broad scope of toxicity and high potency is not confirmed in the numerous clinical and epidemiological studies of human populations with high occupational or environmental exposure. Recent findings suggest that these human versus animal dose-response discrepancies may relate to species differences in functionality of the aryl hydrocarbon receptor or AhR. Binding to AhR is linked to enzyme induction response in humans at high dioxin doses (e.g., over 1000 ppt in adult serum lipid) and to induction plus several other adverse responses in experimental animals at much lower doses. Other than the clearly demonstrated causal connection between dioxin and human chloracne and liver enzyme induction, the noncancer human health consequences are often described as or lacking in clinical importance. The mechanistic and epidemiologic studies of dioxin and related compounds are important topics of continued research and scientific debate on the public health importance of these compounds.
The carcinogenic potential of dioxin and related compounds in human populations is also an important topic of continued scientific debate. Starting in the 1990s, epidemiological studies were able to distinguish groups with highest dioxin exposures, and some have identified elevated total cancer rates among the most exposed groups with more than 20 years latency from first exposure. However, unlike other recognized occupational carcinogens such as benzene and vinyl chloride, the available studies do not identify strong, coherent, and consistent associations of dioxin exposures and specific cancer types. The idea that dioxin could promote diverse types of human cancer is not generally accepted and continues to be debated. Fortunately, regulatory efforts to limit sources of dioxin have demonstrably reduced background exposures.
The prominent concern among public health officials following the Seveso trichlorophenol reactor explosion in 1976 was based on available animal studies that suggested possible risks of spontaneous abortions and birth defects from dioxin exposures. As reviewed by Kerger et al., in the years after the accident extensive studies were conducted to assess Seveso reproductive health, clinical monitoring of exposed children and adults for dermatological, liver, and neurological outcomes, and more long-term studies centered on potential cancer incidence and cause-specific mortality. The surveillance program investigators were supervised and periodically reviewed by an International Steering Committee, which completed its findings in 1984. For the period of 1976 to 1984, the committee concluded that chloracne represented the only health outcome clearly attributable to the accidental dioxin exposure. While researchers have continued to follow-up on a variety of health outcomes among Seveso residents, on balance these studies have not identified convincing causal associations for disease endpoints other than chloracne. However, the foresight of the Seveso local investigators to collect, store, and later analyze hundreds of blood samples from highly-exposed children, adolescents and young adults led to the discovery of age- and concentration-dependent dioxin pharmacokinetics discussed below.
The aim of this review is to evaluate the transgenerational implications of three key discoveries pertaining to potential health impacts of DLCs in human populations: 1) documentation of age-dependent and concentration-dependent half-life for these congeners; 2) documentation of reduced body burdens of these congeners over time in the general population since initial concerns were raised in the 1970s; and 3) documentation of the threshold-dependent aryl hydrocarbon receptor-mediated mechanism of action for DLCs and the more limited hazard potential in humans compared to sensitive animal models.
2.0 Dioxin-like compound environmental fate and chemistry relating to childhood dioxin exposures and health risks
In the environment, humans are often exposed to a mixture of three closely related families of compounds that include dioxin-like congeners: Polychlorinated Dibenzo-p-dioxins (PCDDs), Polychlorinated Dibenzofurans (PCDFs), and Polychlorinated Biphenyls (PCBs).
The effects of DLCs share a common mechanism of action in that they are mediated through the AhR, resulting in similar outcomes after binding in selected in vitro and in vivo test systems. Although they share these quantifiable endpoints, potencies of DLCs vary with base molecule and chlorine substitution pattern, with 2,3,7,8-substituted PCDDs and PCDFs being considerably more potent than PCBs (see
| Compound | WHO 1998 TEF | WHO 2005 TEF | WHO 2022 TEF |
|---|---|---|---|
| 2,3,7,8-TCDD | 1 | 1 | 1 |
| 1,2,3,7,8-PeCDD | 1 | 1 | 0.4 |
| 1,2,3,4,7.8-HxCDD | 0.1 | 0.1 | 0.09 |
| 1,2,3,6,7,8-HxCDD | 0.1 | 0.1 | 0.07 |
| 1,2,3,7,8,9-HxCDD | 0.1 | 0.1 | 0.05 |
below). In order to provide an estimate of the toxic potency of mixtures of DLCs while accounting for the potency differences among them, the dioxin toxicity equivalence factor (TEF) approach was developed where the relative potency of individual DLC congeners for producing AhR-mediated effects is estimated relative to that of the reference compound, 2,3,7,8-tetrachlorodibenzo-p-dioxin (TEF = 1). An expert panel organized by the World Health Organization (WHO) initially developed TEFs for all DLCs in 1993; subsequent WHO panels then updated the TEFs in 1998, 2005, and 2022.
| Compound | WHO 1998 TEF | WHO 2005 TEF | WHO 2022 TEF |
|---|---|---|---|
| 2,3,7,8-TCDD | 1 | 1 | 1 |
| 1,2,3,7,8-PeCDD | 1 | 1 | 0.4 |
| 1,2,3,4,7.8-HxCDD | 0.1 | 0.1 | 0.09 |
| 1,2,3,6,7,8-HxCDD | 0.1 | 0.1 | 0.07 |
| 1,2,3,7,8,9-HxCDD | 0.1 | 0.1 | 0.05 |
The dominant properties that influence environmental distribution of DLCs are water solubility, vapor pressure, and the organic carbon partition coefficient (Koc). Although the molecular weight of DLCs will vary depending on the specific compound and degree of chlorination, molecular weights of these compounds are generally high (e.g., dioxin @ 322 g/mol). In addition, DLCs have low volatility with vapor pressures of 10-8 to 10-12 mm Hg. The Koc quantifies the tendency of organic compounds to adsorb to organic matter in soil/sediment relative to aqueous phase. The high Koc values of DLCs and their strong affinity for organic matter leads to accumulation in soils, sediments, and biota with limited presence in water and air. These compounds bioaccumulate up the food chain, resulting in higher concentrations in certain livestock, carnivorous wildlife, and fish. The high lipophilicity of DLCs leads to their concentration in fatty tissues including milk.
According to ATSDR, the partitioning ratio of 2,3,7,8-TCDD between adipose tissue lipids and serum lipids is approximately 1 and remains near unity for 1000-fold above background levels. Accordingly, serum lipid DLC concentrations are commonly used for body burden dosimetry. According to the ATSDR, food consumption (including human breastmilk), particularly animal products and fish/seafood, is the most important pathway for background exposure to PCDDs and PCDFs in the general population; food represents >90% of total daily intake.
Fortunately, the general population body burdens of DLCs in developed countries have decreased over the past two decades. Indeed, general population body burden estimates for PCDD/Fs expressed in World Health Organization (WHO) dioxin toxic equivalents (TEQ) were lowered more than an order of magnitude between 1980 and 2000. This downward trend was associated with the recognition of dioxin hazards in the 1970s and interventions to limit emissions and exposure through research that characterized the prominent sources, and through regulatory actions and application of risk assessment principles. This trend is also important to consider in the assessment of total exposures and health risks of DLCs in young children since the body burdens present in reproductive-age women contribute to baseline body burden in their offspring via in utero transfer and breast-feeding. Further, it is expected that background dietary exposures to DLCs have been lowered concurrently with the observed decrease in general population body burdens.
The intake from these other pathways is estimated to be approximately 2% of daily DLC intake. The USEPA 2004 Dioxin Reassessment characterized background exposures to DLCs, including estimates of average background intake dose and average background body burden. These estimates were derived from data from the mid-1990s and then updated with data from studies conducted in the mid-2000s. The average background intake in the mid-1990s was 61.0 pg TEQ/day; using the updated data from the mid-2000s, the average background intake was 40.6 pg TEQ/day. More recent reviews have indicated continuing decreases of DLC body burdens and dietary levels. Arisawa reviewed the median concentrations of DLC body burdens in various countries as summarized in
| Country | Population | N | Matrix | Mean or Median | Year | |
|---|---|---|---|---|---|---|
| Germany | General population | 95 | blood | 42.67 (PCDD/Fs) | 1991 | |
| 157 | blood | 38.12 (PCDD/Fs) | 1992 | |||
| 17 | blood | 29.05 (PCDD/Fs) | 1993 | |||
| 74 | blood | 29.14 (PCDD/Fs) | 1994 | |||
| 69 | blood | 24.06 (PCDD/Fs) | 1995 | |||
| 95 | blood | 20.74 (PCDD/Fs) | 1996 | |||
| United States | NHATS | 1980-1981 | 57 | serum | 55 (PCDD/Fs) | 1980-1981 |
| NHANES | 2001-2002 | 408 | serum | 12.9 (PCDD/Fs) | 2001-2002 | |
| Japan | General population | 259 | blood | 26.7 (PCDD/Fs; DL-PCBs) | 2002 | |
| 272 | blood | 23.6 (PCDD/Fs; DL-PCBs) | 2003 | |||
| 264 | blood | 23.7 (PCDD/Fs; DL-PCBs) | 2004 | |||
| 288 | blood | 25.6 (PCDD/Fs; DL-PCBs) | 2005 | |||
| 291 | blood | 21 (PCDD/Fs; DL-PCBs) | 2006 | |||
| Russia | Women living in vicinity of chemical plant | 8 | serum | 36 | 2000 | |
| serum | 25 | 2009 | ||||
| Korea | Residents, workers | 954 | serum | 8.15 (PCDD/Fs) | 2001-2011 | |
| Residents, workers | 539 | serum | 4.15 (DL-PCBs) | 2001-2011 | ||
| Japan | General population | 2264 | blood | 19 | 2002-2010 | |
| General population | 490 | blood | 11 | 2011-2016 |
Bichteler et al. analyzed DLC body burdens from 2001 to 2008 in the US National Health and Nutrition Examination Survey by race/ethnicity and age. Temporal trends observed by Bichteler et al. indicate that TEQ body burdens have continued to decrease over time. When stratified by race/ethnicity, Mexican Americans, Non-Hispanic Blacks, and Non-Hispanic Whites all had approximately a 50% decrease in TEQ (pg/g lipid). The 20-39 age group had the greatest decrease in TEQ (~64%) and the 60+ age group had the smallest decrease (~43%). Bichteler et al. reported generally decreasing trends across racial groups with some indications of flattened DLC body burdens between 2006 and 2008.
Methods for conducting human health risk assessments for DLCs have evolved significantly since the CDC developed their risk assessment for dioxin in residential soils. Since the 1 ppb guideline for dioxin TEQ in soils was developed by Kimbrough et al., many toxicology, epidemiology, exposure, and fate and transport studies have been conducted for the DLCs. Paustenbach et al. attempted to integrate these additional data into a probabilistic analysis to identify safe concentrations dioxin TEQ in residential soils. These authors considered updated cancer and non-cancer toxicity criteria as well as updated data distributions of child soil ingestion/dermal uptake, bioavailability of dioxin in soils, and residential exposure duration. This probabilistic risk assessment approach has several advantages to the point-estimate approach used in past risk assessments for dioxin, including: 1) eliminates debate over the point estimates for exposure parameters; 2) provides more realistic estimates of upper bound exposures; 3) provides information on how risks are distributed based on population exposure; 4) gives the risk manager a more complete picture regarding the factors that drive the assessment as well as the degree of uncertainty in those factors; and 5) provides the information useful to performing sensitivity and uncertainty analyses.
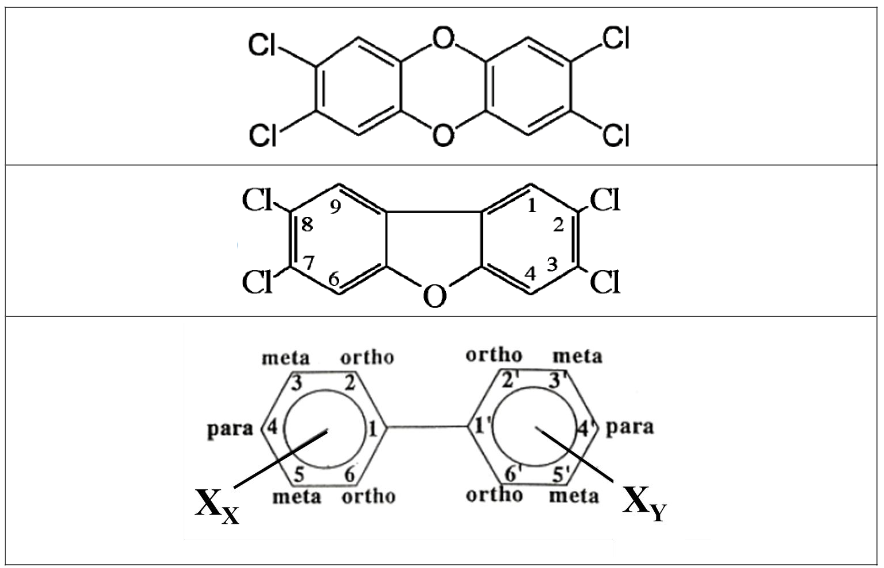
Another important development which changed risk assessment approaches for DLCs and concerns regarding child exposures and breast milk transfer was the discovery of age-dependent half-lives. Kerger et al. developed a childhood body burden model (birth to 7 years) for assessing serum lipid DLCs from dietary (breast milk, formula, other foods) and environmental exposure pathways (soil ingestion, dermal contact). This model incorporated empirical data on model parameters (including DLC congener-specific infant half-lives and rates of increase in half-lives with age) and allows for user input of site-specific DLC congener data in environmental media, foods, and breastmilk. Model output is a profile of blood lipid TEQ concentration versus age and allows for calculation of the TWA child internal dose from age 0-7 years. Exemplary output of the childhood PCDD/F body burden model is shown in
.
3.0 Pharmacokinetics and childhood dioxin exposures
Measurements of adipose and liver concentrations of dioxin in infants have suggested that the relatively high daily doses associated with breast-feeding may not lead to the high body burdens predicted by toxicokinetic models using a constant half-life for all ages. Using a simple model that accommodated an age-dependent adipose tissue volume, Kreuzer et al. estimated that the half-life of dioxin in infants was only about 0.4 years, much shorter than the 7 to 11 years estimated for adults. Lorber and Phillips reported a modeling exercise of PCDD/F body burdens as TEQ during breast-feeding of infants using a similar one-compartment model. Leung et al. verified the relatively short half-life of dioxin in infants and identified similar short half-lives for other PCDD/F DLC congeners (e.g., 0.2-0.5 years). The reasons for the shorter half-lives in young children are not fully understood, but are likely due to a combination of factors, including body burden dilution from rapid growth of the adipose mass, higher fecal lipid excretion relative to adults, and increased metabolism. Recent observations that children and adolescents (under age 18 years) reduced their total DLC body burdens more readily than adults exposed during the Seveso incident, beyond those accounted for by growth-related dilution, suggest that excretion probably plays an important role.
| Congener | Half-life (yr) by age–child (0-12 years) | TCDD | TCDF | PeCDD | 4-PeCDF | HxCDD/F | HpCDD/F | OCDD/F |
|---|---|---|---|---|---|---|---|---|
| 0-12 months | 0.4 | 0.4 | 0.32 | 0.27 | 0.39 | 0.32 | 0.46 | |
| 12-24 months | 0.52 | 0.52 | 0.44 | 0.39 | 0.51 | 0.44 | 0.58 | |
| 24-36 months | 0.64 | 0.64 | 0.56 | 0.51 | 0.63 | 0.56 | 0.70 | |
| 36-48 months | 0.76 | 0.76 | 0.68 | 0.63 | 0.75 | 0.68 | 0.82 | |
| 48-60 months | 0.88 | 0.88 | 0.80 | 0.75 | 0.87 | 0.80 | 0.94 | |
| 60-72 months | 1.00 | 1.00 | 0.92 | 0.87 | 0.99 | 0.92 | 1.06 | |
| 72-84 months | 1.12 | 1.12 | 1.04 | 0.99 | 1.11 | 1.04 | 1.18 | |
| 84-96 months | 1.24 | 1.24 | 1.16 | 1.11 | 1.23 | 1.16 | 1.30 | |
| 96-108 months | 1.36 | 1.36 | 1.28 | 1.23 | 1.35 | 1.28 | 1.42 | |
| 108-120 months | 1.48 | 1.48 | 1.40 | 1.35 | 1.47 | 1.40 | 1.54 | |
| 120-132 months | 1.60 | 1.60 | 1.52 | 1.47 | 1.59 | 1.52 | 1.66 | |
| 132-144 months | 1.72 | 1.72 | 1.64 | 1.59 | 1.71 | 1.64 | 1.78 |
| Half-life (yr) by age–adolescent (age 13-18) | TCDD | TCDF | PeCDD | 4-PeCDF | HxCDD/F | HpCDD/F | OCDD/F |
|---|---|---|---|---|---|---|---|
| 144-156 months | 1.8 | 1.8 | 1.8 | 1.7 | 1.8 | 1.8 | 1.9 |
| 156-168 months | 2.0 | 2.0 | 1.9 | 1.8 | 1.9 | 1.9 | 2.0 |
| 168-180 months | 2.1 | 2.1 | 2.0 | 2.0 | 2.1 | 2.0 | 2.1 |
| 180-192 months | 2.2 | 2.2 | 2.1 | 2.1 | 2.2 | 2.1 | 2.3 |
| 192-204 months | 2.3 | 2.3 | 2.2 | 2.2 | 2.3 | 2.2 | 2.4 |
| 204-216 months | 2.4 | 2.4 | 2.4 | 2.3 | 2.4 | 2.4 | 2.5 |
| Half-life (yr) by age–adult (age 20-50) | TCDD | TCDF | PeCDD | 4-PeCDF | HxCDD/Fs | HpCDD/Fs | OCDD/F |
|---|---|---|---|---|---|---|---|
| Age 20 | 2.7 | 2.7 | 2.6 | 2.6 | 2.7 | 2.6 | 2.7 |
| Age 30 | 3.9 | 3.9 | 3.8 | 3.8 | 3.9 | 3.8 | 3.9 |
| Age 40 | 5.1 | 5.1 | 5.0 | 5.0 | 5.1 | 5.0 | 5.1 |
| Age 50 | 6.3 | 6.3 | 6.2 | 6.2 | 6.3 | 6.2 | 6.3 |
The PCB/PCDF rice oil poisoning incidents (Yusho in Japan and Yucheng in Taiwan) showed that dioxin-like compounds are excreted through sebaceous glands and accumulate in gingival tissues and skin at high doses. Pharmacokinetic studies of DDE also demonstrate partitioning to skin as a depot for distribution and storage/excretion in mammals. The skin and other epidermal tissues involve competing biochemical processes for partitioning, sequestering, and sloughing of skin cells most prone to DLC accumulation. Sebaceous glands secrete DLCs to stratum corneum and not likely reabsorbed, while subcutaneous fat concentrations equilibrate with the growing mass of dermis and epidermis. Through continued growth and exfoliation of the skin, average daily excretion of unchanged DLCs may exceed the loss via feces. Similar processes probably impact the epidermal lining of the gastrointestinal tract and influence fecal excretion. These mechanisms plausibly influence body burden dilution and more rapid DLC losses during rapid growth stages, e.g., fetal, neonatal and early childhood.
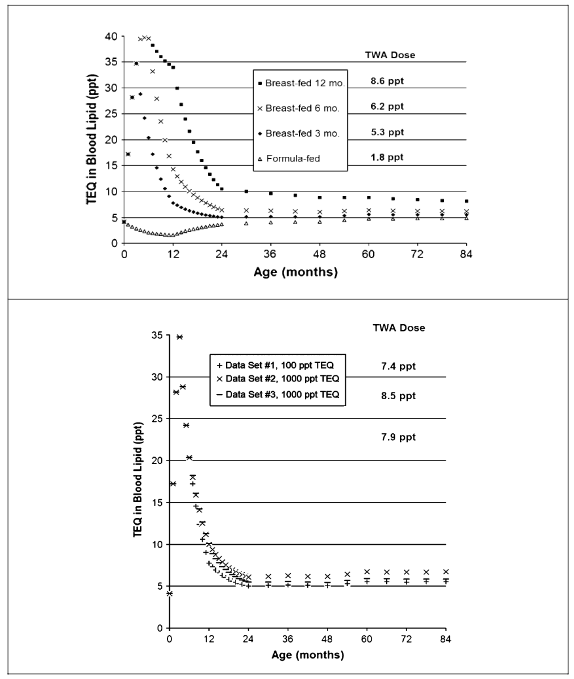
Kerger et al. reported an adaptable internal dose model for risk assessment of dietary and soil PCDD/F exposures in young children that included consideration of breast-feeding and varied patterns of dioxin-like compounds in foods and soils. These modeling exercises illustrate that the much shorter half-life of dioxins in young children (e.g., 0.27 to 0.46 years in children ages 0 to 6 years) are capable of rapidly moderating internal doses from background dietary and environmental intakes, translating into average internal doses from age 0 to 7 that are well below those for adults who exhibit a much longer half-life (e.g., 7 to 11 years). An illustration of the childhood DLC body burden model presented by Kerger et al. for PCDD/F TEQ is shown in

.
4.0 Aryl hydrocarbon receptor mechanistic implications
Due to the ubiquitous presence of dioxins in foods, they are present in all reproductive age women at some level. Thus, everyone is exposed to dioxin-like compounds from the point of conception forward. However, potential health impacts of AhR binding are dependent upon the dose and potency of DLCs absorbed over time, and the duration of sustained tissue concentrations above a certain threshold level leading to AhR-mediated injury or disease in specific target organs. All receptor-mediated events are believed to follow a threshold-type dose-response relationship. Thus, conclusions about correlations between DLC body burdens and AhR-mediated consequences must consider that there are doses at which there is no organ injury and disease potentially linked to AhR activation or other receptor mediated effects. The AhR binding and associated gene expression changes are inherently reversible and require sustained stimulation to cause organ injury or disease. Little is known regarding the magnitude and duration of target tissue receptor stimulation that equates with definable early signs of organ injury or disease in humans. However, it is well recognized that there are no-effect levels for AhR-mediated effects in both test animals and in humans. Thus, one must understand the threshold conditions for tissue-specific sustained AhR activation that may or may not lead to clinically definable organ injury or disease in order to conclude whether or not a given body dose of dioxin-like compounds will cause organ injury or disease in a given individual.
Test animals uniformly exhibit increased liver enzyme metabolism in response to excessive intake of TCDD. This liver enzyme induction is a response related to specific binding of dioxin to a cellular protein known as the AhR in liver cells. The AhR protein is a gene transcription factor that is found in most mammalian tissues, including humans. Through a series of direct and indirect events occurring within liver cells, the AhR activates dioxin-responsive genes that lead to production of additional cellular capacity to metabolize and excrete aryl hydrocarbon compounds such as dioxin. The AhR with respect to dioxin has a main function of facilitating excretion of dioxin and other aryl hydrocarbon chemicals. The AhR upregulates phase I metabolism enzymes, particularly CYP1A1 and CYP1A2, which add oxygen molecules to aryl hydrocarbon compounds such as dioxin and thereby facilitate their removal from the body. Such induction of liver enzymes is an early response to excessive dioxin intake and is uniformly observed as the AhR-mediated effect occurring at the lowest dose in animal studies. Importantly, all receptor-mediated adverse effects of chemicals or drugs follow a threshold-type dose-response relationship whereby low doses may activate biological receptors without any adverse consequences, and a sustained high tissue concentration is required to trigger more severe adverse effects. This is an important observation because if one can determine that TCDD intake in animals or humans is below the lowest observed effect level (LOEL) for clinically important CYP1A1 or CYP1A2 liver enzyme induction, then it is reasonable to conclude that other adverse effects or disease endpoints from dioxin will not occur. Dioxin has been used as a research tool to study dose-related activation of the AhR and its health consequences in test animals. Dioxin binds to the AhR with exceptionally high affinity, and it has been used as a yardstick for characterizing binding affinity and potency of other DLCs. The AhR is relatively nonselective and can be occupied by many different compounds including TCDD, other aryl hydrocarbons, and a variety of dissimilar endogenous and exogenous compounds, e.g., amino acids, hormones, lipids, foods, and certain drugs. The AhR activation by a particular ligand does not necessarily result in injury or disease; some ligands bind to the AhR and have various beneficial actions which have led to recent research focused on utilizing AhR agonist drugs to treat various human diseases. Some of these molecules, like flavones that are present in teas, wines, and citrus products bind to the AhR but apparently do not activate transcription of genes.
In receptor theory, agents that bind the receptor but cause relatively limited effects are called partial agonists and agents that bind the receptor and cause no effects are called antagonists. Okey notes, receptor theory predicts that compounds which compete for the same receptor site will antagonize each other if the mixture contains partial agonists. Receptor theory and numerous empirical precedents predict that responses should be sublinear at low doses and that a practical threshold should exist. Responses mediated by interaction of a ligand with its receptor are reversible when the ligand is withdrawn because response depends on the continued presence of the ligand-receptor complex. Accordingly, sustained high tissue concentrations of AhR agonists are required to significantly influence potential responses like cancer, a disease process that involves multiple steps that may or may not be propagated by AhR-mediated events alone. The timing between sufficient receptor activation and the associated potential clinical effect (e.g., latency between sufficient exposure and onset of chloracne) generally can be shortened with increased dose, leading to longer duration of sustained high tissue concentrations; this is the basis for determining efficacy of all receptor-active drugs. Thus, in order to predict the potential health risks from DLCs related to a particular exposure source or from a measured serum lipid DLC concentration, one must consider whether or not the associated tissue dose of AhR activators is sufficient to exceed the empirical dose threshold for a sufficiently long period to result in the clinically defined injury or disease of concern.
The study of biological receptor activation is considered the fundamental element of pharmacology because it has led to the identification of the neurotransmitters, hormones, and cytokines that are at the core of development of many therapeutic agents. However, receptor activation and associated gene expression are transient events that cannot be considered an early stage of organ injury or disease. In receptor pharmacology, the occurrence of a threshold-dependent dose-response relationship is also considered a fundamental concept that is deeply rooted in empiricism. Poland notes, since the dawn of studying receptors nearly 100 years ago I am unaware of a single instance of a drug (i.e., any biologically active substance) acting on a receptor which upon sufficient decrease in concentration or dose does not display a no-effect level, i.e., a threshold. In other words, there is always a threshold below which no effects are observed.
The natural role of the AhR is apparently tied to regulation of growth and certain cellular maintenance processes across several different organs in mammals. Recent studies have created AhR knockout strains of mice where the genes coding for the Ah receptor are omitted; these mice exhibit abnormal development of liver and vascular tissues, abnormal immune function, birth defects, cancer and lethality. Thus, AhR function in many ways is necessary for normal development, but the intertwined roles it may play in growth regulation and cellular maintenance are complex and have not been fully elucidated.
There is strong and consistent evidence indicating that, at the cellular level, humans are less sensitive than rodents to AhR-mediated effects of dioxin. For example, Silkworth et al. showed that AhR-mediated responses to dioxin in human liver cells were 10- to 1000-fold less sensitive than the responses observed in AhR-sensitive Sprague-Dawley rats and Rhesus monkeys, not accounting for toxicokinetic and anatomical differences between the test species and humans. These species-related differences in AhR sensitivity are best explained by differences in TCDD kinetics and receptor numbers, receptor structure, and/or in the cascade of responses triggered. The AhR receptor activation by various agonists occurs continuously in completely healthy individuals and is necessary for normal development and bodily functions in humans and animals. Connor et al. reported that global activation of the AhR pathway (from all potential AhR agonists) in human blood samples was about 1000-fold higher than that from dioxin or dioxin-like compounds. At low tissue concentrations, there is greater competition with other endogenous or exogenous molecules for occupying the AhR, which reduces triggering of AhR-mediated events by dioxin. Relatively high dioxin concentrations in humans are required to successfully compete with other AhR agonists for receptor occupancy. Sufficiently high tissue dioxin concentrations must be present to induce sustained high levels of receptor activation in order to cause organ injury or disease. In the absence of sufficient intensity and duration of AhR activation in relevant tissues, the downstream effects of receptor activation by dioxin may not occur and/or may be completely reversible. This may explain why the broad range of adverse health effects of dioxin in sensitive animal species has not been demonstrated in human studies.
Total body doses of DLCs cannot be directly transposed between in vitro findings and studies of intact animals or humans for several reasons: 1. Human cells are less responsive to DLCs compared to sensitive animal species likely due to differences in AhR structure and/or functions. 2. Humans have a considerably larger adipose tissue depot compared to sensitive animal species, allowing for greater dilution and sequestration of DLCs and other persistent organic compounds that partition into body fats. 3. Human metabolism of DLCs is much slower relative to sensitive animal species because the lowest observed effect of liver enzyme induction (CYP1A) is not observed until much higher internal doses, e.g., at serum lipid dioxin concentrations exceeding 1000 ppt. 4. Upregulation of CYP1A metabolism is associated with a variety of serious DLC effects in sensitive animal species but only a limited set of health effects in humans. Importantly, this CYP1A upregulation in both animals and humans through AhR-mediated events leads to more rapid DLC excretion, reducing tissue levels and concurrently reducing the duration and severity of potential AhR-mediated adverse effects. 5. Plausible exposure pathways for DLC uptake in humans (i.e., typically oral and dermal absorption) involve prompt partitioning to lipid carriers and adipose tissues. This process moderates the delivery rate and concentration of DLCs occurring at key target organs like the liver in sensitive animal species. These primary factors collectively indicate that any inferences that may be gleaned from in vitro studies require validation in more detailed studies in the intact test animal or in humans.
Due to lower AhR responsiveness and distinctly different pharmacokinetic factors, humans exhibit CYP1A enzyme induction at relatively high dioxin concentrations (e.g., 1,000 ppt in serum lipid). This lowest observable effect level or LOEL for CYP1A induction in humans is 15-fold greater than that for Sprague-Dawley rats; the difference reflects the lower AhR responsiveness in humans. Dioxin distributes evenly in serum lipids and body fat in humans and the higher concentration required to induce CYP1A is observed on a cellular level and a whole organism level. Among the most highly exposed human populations studied to date, only a small fraction have ever attained serum lipid dioxin concentrations substantially above the LOEL of 1,000 ppt. As shown in
| Reference | Study population (n) | Range | Mean |
|---|---|---|---|
| Centers for Disease Control | Vietnam ground combat troops with service in heavily sprayed areas (646) | ND-45 | 4.2 |
| Non-Vietnam veterans (97) | ND-15 | 4.1 | |
| Air Force Health Study | Ranch Hand veterans (866) | 0-617.8 | – |
| Comparisons (804) | 0-54.8 | – | |
| Mocarelli et al. | Zone A with Chloracne (10) | 828-56,000 | 19,144 |
| Zone A without Chloracne (9) | 1770-10,400 | 5240 | |
| Non-ABR zone (10)* | ND–137 | – | |
| Fingerhut et al. | Exposed workers (253) | 2-3400 | – |
| Unexposed workers (79) | <20 | 7 | |
| Workers with yr exposure (119) | – | 418 | |
| Ott et al. | Workers involved in clean-up after reactor accident (138) | <1-553.0 | 15.4 |
| External referents (102) | 0.6-9.1 | 3.0 | |
| Kogevinas et al. | Australian sprayers (37) | 2-34 | – |
| Austrian production workers (9) | 98-659 | 389 | |
| Dutch production workers (31) | 1.9-194 | 53 | |
| New Zealand sprayers (9) | 3.0-131 | 53.3 | |
| Swedish production workers (5) | 9-37 | 17 | |
| German production workers (19) | 1.3-6.49 | 3.2 | |
| German production workers (190) | 3-2252 | 141 | |
| German production workers (20) | 23-1935 | 401.7 |
.
5.0 Conclusions
The age- and concentration-dependent kinetics of DLCs in the human body have potentially important public health implications, particularly with regard to exposures during childhood including breast-feeding, i.e., transgenerational impacts. A much shorter elimination half-life of DLCs in young children and adolescents is important because it limits accumulation of body burdens during development and puberty (e.g., birth to age 18) and is reasonably expected to limit potential health impacts on females of typical reproductive age (ages 18 to 35 years). Observations of age- and concentration-dependent elimination of DLCs have important risk assessment implications with respect to evaluating accumulated body burdens and dose-response trends in humans. This includes consideration of transgenerational impacts of DLCs for several reasons. First, background DLC dietary exposures and human body burdens have been reduced dramatically over the past 4 decades which reduces the likelihood of high DLC dosages to current and future generations. Second, the age-dependent half-life of DLCs predominantly reduces body burdens in younger persons (e.g., children and reproductive age females) who comprise the primary population at risk for potential body burden transfer from mother to child during breast-feeding. Third, the AhR-mediated adverse effects of DLCs follow a threshold-dependent dose-response relationship and complex interactions with many other binding ligands that likely ameliorate associated human health risks particularly at low general population body burdens. Fourth, despite the continued research concerns and scientific debate about potential health risks of dietary DLCs on children and reproductive age adults, there is a strong base of information suggesting limited evidence for human health impacts based on the highest exposed populations, some of which have been studied for more than 40 years. Finally, shorter concentration-dependent DLC half-lives at higher tissue concentrations suggest that earlier attempts to back-calculate cumulative body burdens in highly exposed workers based on an assumption of uniform half-life (e.g., 7 to 11 years half-life for dioxin in adults) may lead to overstated potency estimates.
Conflict of Interest Statement: The authors are employed by an engineering and health sciences consulting firm with diverse professional staff providing assistance to governmental and private sector clients including litigation support and expert witness testimony. The authors declare no conflict of interest based on client work or expert testimony pertaining to dioxin-like compounds in the past 10 years.
Funding Statement: None.
Acknowledgements: None.
6. References:
- Greene JF, Hays S, Paustenbach D. Basis for a proposed reference dose (RfD) for dioxin of 1-10 pg/kg-day: a weight of evidence evaluation of the human and animal studies. J Toxicol Environ Health B Crit Rev. 2003 Mar-Apr;6(2):115-59. doi: 10.1080/10937400306470. PMID: 12554432
- USEPA (United States Environmental Protection Agency). 2003. Exposure and Human Health Reassessment of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) and Related Compounds. Office of Research and Development, National Center for Environmental Assessment. NAS Review Draft. December 2003
- ATSDR (Agency for Toxic Substances and Disease Registry). 2024. Toxicological Profile for Chlorinated Dibenzo-p-Dioxins. Draft for Public Comment. October 2024. https://www.atsdr.cdc.gov/toxprofiles/tp104.pdf
- Paustenbach DJ, Fehling K, Scott P, Harris M, Kerger BD. Identifying soil cleanup criteria for dioxins in urban residential soils: how have 20 years of research and risk assessment experience affected the analysis? J Toxicol Environ Health B Crit Rev. 2006 Mar-Apr;9(2):87-145. doi: 10.1080/10937400500538482. PMID: 16613806
- USEPA (United States Environmental Protection Agency). 2002a. US Environmental Protection Agency Child-Specific Exposure Factor Handbook. Pp. 3-59. NCEA, Washington DC. EPA-600-P-00-002B
- USEPA (United States Environmental Protection Agency). 1989. Risk Assessment Guidance for Superfund Vol 1. Human Health Evaluation Manual. Office of Emergency and Remedial Response. EPA/540/1-89/002
- Williams, PRD; Holicky, KC; Paustenbach DJ exposures for use in health risk assessment. J Child Health 1, 41-98
- USEPA (United States Environmental Protection Agency). 2002b. Short Sheet: Overview of the IEUBK Model for Lead in Children. EPA #PB 99-965-8, OSWER #9285-7-31. Office of Solid Waste and Emergency Response, USEPA, Washington DC
- Kerger BD; Gerthoux PM; Mocarelli P. 2011. 1976 Trichlorophenol reactor explosion at Seveso, Italy. Encyclopedia of Environmental Health. Amsterdam; Elsevier. Editor-in-chief: Jerome O. Nriagu
- ATSDR (Agency for Toxic Substances and Disease Registry). 2023. Toxicological Profile for Chlorodibenzofurans (CDFs). April 2023. https://www.atsdr.cdc.gov/toxprofiles/tp32.pdf
- Aylward LL, Hays SM. Temporal trends in human TCDD body burden: decreases over three decades and implications for exposure levels. J Expo Anal Environ Epidemiol. 2002 Sep;12(5):319-28. doi: 10.1038/sj.jea.7500233. PMID: 12198580
- Hays SM, Aylward LL. Dioxin risks in perspective: past, present, and future. Regul Toxicol Pharmacol. 2003 Apr;37(2):202-17. doi: 10.1016/s0273-2300(02)00044-2. PMID: 12726754
- Lorber M. A pharmacokinetic model for estimating exposure of Americans to dioxin-like compounds in the past, present, and future. Sci Total Environ. 2002 Apr 8;288(1-2):81-95. doi: 10.1016/s0048-9697(01)01119-6. PMID: 12013550
- Richter RO; Kerger BD; Leung HW; Paustenbach DJ. 2006. Implications of age-dependent half-lives of dioxins on assessment of breast milk dose and body burden. Toxicol Sci 89, 177 (Abstract 1561)
- Lorber M, Patterson D, Huwe J, Kahn H. Evaluation of background exposures of Americans to dioxin-like compounds in the 1990s and the 2000s. Chemosphere. 2009 Oct;77(5):640-51. doi: 10.1016/j.chemosphere.2009.08.016. Epub 2009 Sep 4. PMID: 19733382
- Arisawa K. Recent decreasing trends of exposure to PCDDs/PCDFs/dioxinlike PCBs in general populations, and associations with diabetes, metabolic syndrome, and gout/ hyperuricemia. J Med Invest. 2018;65(3.4):151-161. doi: 10.2152/jmi.65.151. PMID: 30282853
- Bichteler A, Wikoff DS, Loko F, Harris MA. Estimating serum concentrations of dioxin-like compounds in the U.S. population effective 2005-2006 and 2007-2008: A multiple imputation and trending approach incorporating NHANES pooled sample data. Environ Int. 2017 Aug;105:112-125. doi: 10.1016/j.envint.2017.05.003. Epub 2017 May 18. PMID: 28527750
- Kimbrough RD, Falk H, Stehr P, Fries G. Health implications of 2,3,7,8-tetrachlorodibenzo-dioxin (TCDD) contamination of residential soil. J Toxicol Environ Health. 1984;14(1):47-93. doi: 10.1080/15287398409530562. PMID: 6389894
- Kreuzer PE, Csanády GA, Baur C, Kessler W, Päpke O, Greim H, Filser JG. 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) and congeners in infants. A toxicokinetic model of human lifetime body burden by TCDD with special emphasis on its uptake by nutrition. Arch Toxicol. 1997;71(6):383-400. doi: 10.1007/s002040050402. PMID: 9195020
- Leung HW, Kerger BD, Paustenbach DJ. Elimination half-lives of selected polychlorinated dibenzodioxins and dibenzofurans in breast-fed human infants. J Toxicol Environ Health A. 2006 Mar;69(6):437-43. doi: 10.1080/15287390500246886. PMID: 16574620
- Lorber M, Phillips L. Infant exposure to dioxin-like compounds in breast milk. Environ Health Perspect. 2002 Jun;110(6):A325-32. doi: 10.1289/ehp.021100325. PMID: 12055063; PMCID: PMC1240886
- Kerger BD, Leung HW, Scott P, Paustenbach DJ, Needham LL, Patterson DG Jr, Gerthoux PM, Mocarelli P. Age- and concentration-dependent elimination half-life of 2,3,7,8-tetrachlorodibenzo-p-dioxin in Seveso children. Environ Health Perspect. 2006 Oct;114(10):1596-602. doi: 10.1289/ehp.8884. PMID: 17035149; PMCID: PMC1626409
- Kerger BD, Leung HW, Scott PK, Paustenbach DJ. An adaptable internal dose model for risk assessment of dietary and soil dioxin exposures in young children. Toxicol Sci. 2007 Nov;100(1):224-37. doi: 10.1093/toxsci/kfm199. Epub 2007 Aug 6. PMID: 17682006
- Abraham K, Knoll A, Ende M, Päpke O, Helge H. Intake, fecal excretion, and body burden of polychlorinated dibenzo-p-dioxins and dibenzofurans in breast-fed and formula-fed infants. Pediatr Res. 1996 Nov;40(5):671-9. doi: 10.1203/00006450-199611000-00005. PMID: 8910931
- Abraham K, Päpke O, Gross A, Kordonouri O, Wiegand S, Wahn U, Helge H. Time course of PCDD/PCDF/PCB concentrations in breast-feeding mothers and their infants. Chemosphere. 1998 Oct-Nov;37(9-12):1731-41. doi: 10.1016/s0045-6535(98)00238-0. PMID: 9828301
- Leung, H-W, Loccisano, A.E., Kerger, B. 2017. Burdens of Polychlorinated Dioxins and Dioxin-like Compounds in Humans: A Review of the Literature. Environmental Sciences 2017; 156(1): 348. Abstract #2476
- Milbrath MO, Wenger Y, Chang CW, Emond C, Garabrant D, Gillespie BW, Jolliet O. Apparent half-lives of dioxins, furans, and polychlorinated biphenyls as a function of age, body fat, smoking status, and breast-feeding. Environ Health Perspect. 2009 Mar;117(3):417-25. doi: 10.1289/ehp.11781. Epub 2008 Oct 3. PMID: 19337517; PMCID: PMC2661912
- Grandjean P, Budtz-Jørgensen E, Barr DB, Needham LL, Weihe P, Heinzow B. Elimination half-lives of polychlorinated biphenyl congeners in children. Environ Sci Technol. 2008 Sep 15;42(18):6991-6. doi: 10.1021/es800778q. PMID: 18853821; PMCID: PMC2600453
- Paustenbach DJ, Kerger BD. The University of Michigan Dioxin Exposure Study: estimating residential soil and house dust exposures to young children. Chemosphere. 2013 Apr;91(2):200-4. doi: 10.1016/j.chemosphere.2012.12.047. Epub 2013 Jan 22. PMID: 23351485
- Demond A, Adriaens P, Towey T, Chang SC, Hong B, Chen Q, Chang CW, Franzblau A, Garabrant D, Gillespie B, Hedgeman E, Knutson K, Lee CY, Lepkowski J, Olson K, Ward B, Zwica L, Luksemburg W, Maier M. Statistical comparison of residential soil concentrations of PCDDs, PCDFs, and PCBs from two communities in Michigan. Environ Sci Technol. 2008 Aug 1;42(15):5441-8. doi: 10.1021/es702554g. PMID: 18754458.
- Demond A, Franzblau A, Garabrant D, Jiang X, Adriaens P, Chen Q, Gillespie B, Hao W, Hong B, Jolliet O, Lepkowski J. Human exposure from dioxins in soil. Environ Sci Technol. 2012 Feb 7;46(3):1296-302. doi: 10.1021/es2022363. Epub 2011 Dec 22. PMID: 22136605.
- Garabrant DH, Franzblau A, Lepkowski J, Gillespie BW, Adriaens P, Demond A, Hedgeman E, Knutson K, Zwica L, Olson K, Towey T, Chen Q, Hong B, Chang CW, Lee SY, Ward B, Ladronka K, Luksemburg W, Maier M. The University of Michigan Dioxin Exposure Study: predictors of human serum dioxin concentrations in Midland and Saginaw, Michigan. Environ Health Perspect. 2009 May;117(5):818-24. doi: 10.1289/ehp.11779. Epub 2008 Dec 22. PMID: 19479027; PMCID: PMC2685847.
- Okey AB. 2007. An aryl hydrocarbon receptor odyssey to the shores of toxicology: the Deichmann Lecture, International Congress of Toxicology-XI. Toxicol Sci 98:5-38
- Sorg O. 2014. AhR signalling and dioxin toxicity. Toxicol Lett 230:225-33
- Kohle C, Bock KW. 2007. Coordinate regulation of Phase I and II xenobiotic metabolisms by the Ah receptor and Nrf2. Biochem Pharmacol 73:1853-62
- Abel J, Haarmann-Stemmann T. 2010. An introduction to the molecular basics of aryl hydrocarbon receptor biology. Biol Chem 391:1235-48
- Guyot E, Chevallier A, Barouki R, Coumoul X. 2013. The AhR twist: ligand-dependent AhR signaling and pharmaco-toxicological implications. Drug Discov Today 18:479-86
- Hahn ME. 2002. Aryl hydrocarbon receptors: diversity and evolution. Chem Biol Interact 141:131-60
- Poland A. 1997. Reflections on risk assessment of receptor-acting xenobiotics. Regul Toxicol Pharmacol 26:41-3
- Chun OK, Chung SJ, Song WO. 2007. Estimated dietary flavonoid intake and major food sources of U.S. adults. J Nutr 137:1244-52
- Quintana FJ, Sherr DH. 2013. Aryl hydrocarbon receptor control of adaptive immunity. Pharmacol Rev 65:1148-61
- O’Donnell EF, Koch DC, Bisson WH, Jang HS, Kolluri SK. 2014. The aryl hydrocarbon receptor mediates raloxifene-induced apoptosis in estrogen receptor-negative hepatoma and breast cancer cells. Cell Death Dis 5:e1038
- Wahlang B, Falkner KC, Clair HB et al. 2014. Human receptor activation by aroclor 1260, a polychlorinated biphenyl mixture. Toxicol Sci 140:283-97
- Forgacs, A. L., Dere, E., Angrish, M. M., & Zacharewski, T. R. (2013). Comparative Analysis of Temporal and Dose-Dependent TCDD-Elicited Gene Expression in Human, Mouse, and Rat Primary Hepatocytes. Toxicol Sci, 133(1):54 66
- Nault R, Forgacs AL, Dere E, Zacharewski TR. 2013. Comparisons of differential gene expression elicited by TCDD, PCB126, betaNF, or ICZ in mouse hepatoma Hepa1c1c7 cells and C57BL/6 mouse liver. Toxicol Lett 223:52-9
- Black MB, Budinsky RA, Dombkowski A, Cukovic D, LeCluyse EL, et al. 2012. Cross-species comparisons of transcriptomic alterations in human and rat primary hepatocytes exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Toxicol Sci 127:199-215
- Budinsky RA, LeCluyse EL, Ferguson SS, Rowlands JC, Simon T. 2010. Human and rat primary hepatocyte CYP1A1 and 1A2 induction with 2,3,7,8-tetrachlorodibenzo-p-dioxin, 2,3,7,8-tetrachlorodibenzofuran, and 2,3,4,7,8-pentachlorodibenzofuran. Toxicol Sci 118:224-35
- Budinsky RA, Schrenk D, Simon T, et al. 2014. Mode of action and dose-response framework analysis for receptor-mediated toxicity: the aryl hydrocarbon receptor as a case study. Crit Rev Toxicology 44(1):83-119
- Carlson EA, McCulloch C, Koganti A, Goodwin SB, Sutter TR, Silkworth JB. 2009. Divergent transcriptomic responses to aryl hydrocarbon receptor agonists between rat and human primary hepatocytes. Toxicol Sci 112:257-72
- Connor KT, Aylward LL. 2006. Human response to dioxin: aryl hydrocarbon receptor (AhR) molecular structure, function, and dose-response data for enzyme induction indicate an impaired human AhR. J Toxicol Environ Health B 9:147-71
- Peters AK, van Londen K, Bergman A et al. 2004. Effects of polybrominated diphenyl ethers on basal and TCDD-induced ethoxyresorufin activity and cytochrome P450-1A1 expression in MCF-7, HepG2, and H4IIE cells. Toxicol Sci 82:488-96
- Zeiger M, Haag R, Hockel J, Schrenk D, Schmitz HJ. 2001. Inducing effects of dioxin-like polychlorinated biphenyls (PCBs) on CYP1A in the human hepatoblastoma cell line HepG2, the rat hepatoma cell line H4IIE, and rat primary hepat

