Nanosecond Pulsed Field Ablation in Oncology: Advances and Efficacy
Nanosecond Pulsed Field Ablation in Oncology
Arbind Kumar Choudhary1
- Pulse Biosciences, Hayward, CA 94545
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION: Nuccitelli, R., 2025. Nanosecond Pulsed Field Ablation in Oncology. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i7.6875
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6875
ISSN 2375-1924
ABSTRACT
Cancer therapy has evolved significantly over the past three decades and this review focuses on some of the new ablation modalities that have been developed as well as the importance of combination therapies which enlist the immune system. During the late 1990s various immune system checkpoints were discovered, and inhibitors of those immune checkpoints were found to boost the immune system enough in some patients to eliminate metastases. This revealed the power of the immune system to attack and eliminate tumors. However, for most patients simply inhibiting the immune checkpoints was insufficient and additional therapies were required. Current approaches involve ablation of the primary tumor followed by stimulation of the immune system to pick up tumor antigens released by the ablation and generate cytotoxic CD8+ T-cells to eliminate metastases. Tumor ablation involves the targeted delivery of either thermal or nonthermal energy sufficient to kill the tumor cells. Thermal ablation modalities were the first to be used but recent data suggest that the immune system is more strongly recruited by nonthermal modalities which have the additional advantage of sparing acellular structures such as neighboring nerves and blood vessels. We reviewed two studies that compared the efficacy of thermal with that of nonthermal ablation and concentrate on our progress using nanosecond pulsed field ablation. Recent studies that combine ablation with the injection of immune stimulants have shown that combination therapies are the most effective. Combining nsPFA with the intratumoral injection of immune stimulants boosts the immune system sufficiently to eliminate untreated tumors or metastases. We review the immune stimulants currently being used in preclinical studies and summarize the two clinical studies conducted thus far using nanosecond pulsed field ablation (nsPFA).
Keywords
Nanosecond pulsed field ablation, oncology, cancer therapy, immune system, combination therapies
Introduction
Cancer therapy has improved dramatically over the past three decades in part due to improved surgical, radiation and ablation techniques, but more importantly due to advances in our understanding of the critical role of the immune system. This began with the discovery of immune checkpoint inhibitors in the late 1990s and their amazing ability to completely eliminate cancer in a small percentage of patients. This demonstrated the importance of recruiting the immune system as an important component of any successful therapy in order to lead in the attack of tumor metastases. Specifically, the combination therapy of ablating the primary tumor followed by stimulation of the immune response is becoming a popular approach. This review will briefly describe both the commonly used ablation therapies and the many ways that the immune system can be stimulated following ablation of the primary tumor. nsPFA is the newest ablation therapy and will be the most thoroughly discussed.
Methods Used
A search of PubMed.gov was conducted in July, 2025 using keywords tumor ablation and immune response to identify combination studies using ablation techniques as well as the stimulation the immune system.
Tumor Ablation Technologies
Tumor ablation technologies fall into two main categories: thermal and nonthermal. Thermal mechanisms have been used for decades and include radiofrequency ablation (RFA), cryoablation, high intensity focused ultrasound (HIFU), hyperthermia (HT), and photothermal therapy (PTT). All of these thermal modalities generate sufficient temperature transitions to disrupt protein structure and damage cell membranes. Not only does this kill the treated cells but the spreading heat can damage nearby structures such as neighboring nerves and blood vessels. Two reviews of these thermal ablation modalities have appeared recently so they will not be considered in detail here.
Nonthermal ablation mechanisms employ either pulsed electric fields to insert pores in cell membranes (electroporation) or short ultrasound bursts to mechanically disrupt cells (histotripsy). Pulsed electric fields can have a range of cellular effects depending on their pulse duration and amplitude. The first pulsed fields applied to cells were in the microsecond range with amplitudes of 1-2 kV/cm. When the pulse amplitude was below 2 kV/cm, the induced membrane pores and increased membrane permeability were transient and could be routinely used to load extracellular molecules into cells. However, when the pulse amplitude was increased to 2.5-3 kV/cm, the pores became permanent leading to the term, irreversible electroporation or IRE.
IRREVERSIBLE ELECTROPORATION (IRE)
This permanent electroporation ablates cells by necrosis and is available as the NanoKnife marketed by Angiodynamics. It has been approved by the FDA for ablating soft tissue and has been used in many clinical trials treating tumors. The initial therapy applied between 8 and 90 pulses 100 µs in duration which required the application of paralytics due to the strong muscle contraction stimulated. More recently, shorter, high frequency biphasic signals have been used to reduce the muscle contractions and avoid paralytics.
HIGH FREQUENCY IRREVERSIBLE ELECTROPORATION (HFIRE)
These high frequency, biphasic pulses are typically composed of many one microsecond pulse pairs (positive followed by negative) strung together one after the other into packets of hundreds of pulse pairs. The biphasic nature reduces the strength of the muscle contractions triggered by IRE pulses, but these pulses are often delivered during the refractory period of the heart to avoid interfering with the normal cardiac rhythm. While not considered HFIRE, one company using biphasic waveforms is the Aliya platform marketed by Galvanize Therapeutics.
NANOSECOND PULSED FIELD ABLATION (nsPFA)
The most recent type of pulsed field ablation uses much shorter pulses in the nanosecond range (nsPFA) with much higher amplitudes. This technology is available as the nPulseTM marketed by Pulse Biosciences, Inc. nsPFA ablates cells using a different ablation mechanism. Rather than generating permanent pores that kill cells by necrosis, these shorter pulses are able to penetrate into the cell and are strong enough to generate smaller, transient pores in both the plasma membrane and the smaller organelle membranes. This increases organelle permeability and disrupts the mitochondrial membrane potential, releasing cytochrome C and initiating the unique pathway of regulated cell death that is used by all cells when they reach the end of their useful life. nsPFA can therefore be thought of as a natural cell death pathway rather than introducing sufficient energy to introduce permanent pores in cell membranes.
Histotripsy
The third modality for nonthermal ablation is histotripsy. Histotripsy uses a nonthermal modification of HIFU which applies the ultrasound waves in short bursts that mechanically vibrate the target cells to liquify them without significant heating. It has been approved by the FDA for hepatic targets and the first clinical trial treating hepatocellular carcinoma in 44 patients with liver tumors smaller than 1.5 cm demonstrated a primary efficacy of 95.5% with minimal complications. This technology is being marketed by HistoSonics Corp. Preclinical studies treating murine melanoma indicated a CD8+-dependent inhibition of tumor growth along with intratumoral infiltration of CXCR3+/CD8+ T-cells. Studies using two tumors in murine models indicated that treatment of one tumor with histotripsy slowed the growth of the untreated abscopal tumor. There is a substantial literature comprised of over 400 papers describing the effectiveness of this nonthermal ablation modality which will not be covered any further in this review which will concentrate on the shorter electric pulses in the nanosecond domain and their use in oncology.
Studies comparing thermal ablation with nonthermal nsPFA
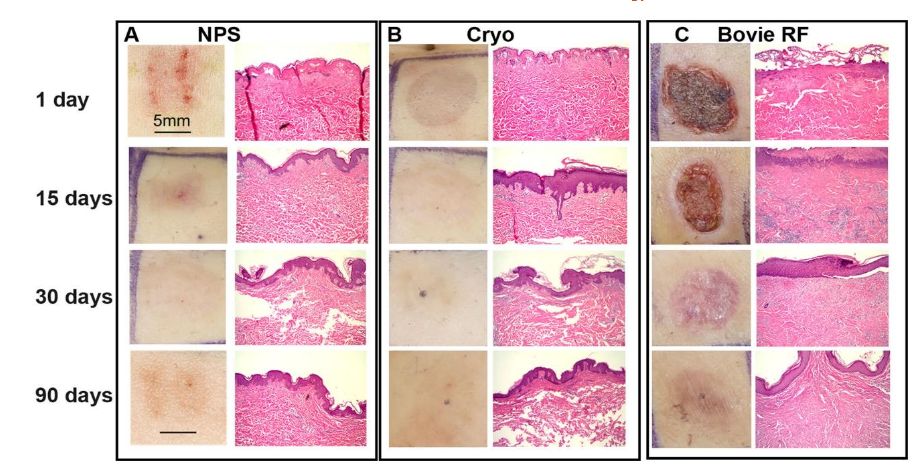
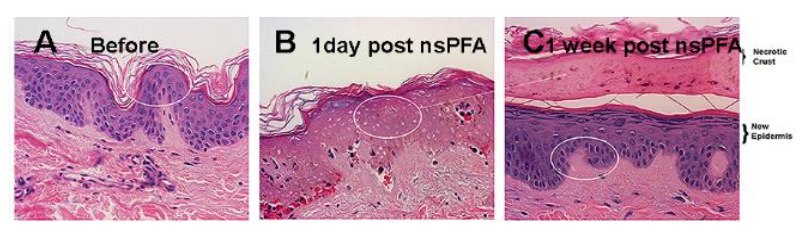
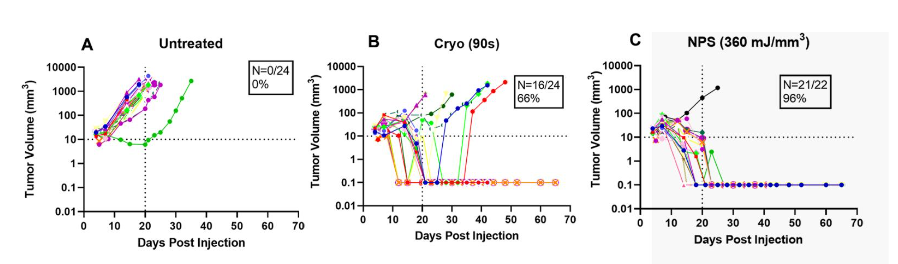
Two studies have been conducted to compare thermal ablation modalities with nonthermal nanosecond pulsed field ablation (nsPFA). The first of these compared the responses of human skin to nsPFA, radiofrequency ablation (RFA) and cryoablation and the second study compared the efficacies of nsPFA and cryoablation on eliminating murine melanoma tumors. nsPFA triggers regulated cell death as indicated by the appearance of activated Caspase-3 at 2 h post treatment along with the absence of nuclear staining 1 day post treatment. Epidermal regeneration follows without impacting the acellular dermis in contrast to both cryoablation and RFA. RFA was found to trigger necrosis and cause scarring, inflammation or permanent pigmentary changes. The main differences between nsPFA and the other ablation modalities are the level of fibrosis, amount of scarring, elastic fiber concentration and inflammation. An analysis of the skin thickness 30 days after the treatments indicated that nsPFA-treated skin was most similar to untreated skin while cryoablated and RF-ablated skin were 2- and 3.5-fold thicker, respectively, suggesting that they induce inflammatory wound-healing processes that result in a substantially altered dermal layer.
The second comparative study contrasted cryoablation with nsPFA in the treatment of murine B16-F10 melanoma tumors. nsPFA permanently eliminated up to 91% of all tumor lesions with a single treatment compared to cryoablation which eliminated only 66%. In addition, nsPFA exhibited much lower dermal fibrosis, underlying muscle atrophy and hair loss.
Unique Nature of Nanosecond Pulses
nsPFA has the unique ability to permeabilize intracellular organelle membranes because of the short duration and high amplitude of the fields applied. It is well known that biological lipid membranes can be permeabilized by applying a pulsed electric field of 400-500 mV/membrane. For a sub-micron organelle, that requires fields on the order of 1 volt across the diameter of 0.5 µm less which comes to a field strength of 20 kV/cm or greater. Applying such a large field for a duration of microseconds or milliseconds would heat the cell beyond hyperthermia levels. However, the much shorter delivery time of nsPFA avoids this heating and allows a nonthermal energy delivery that generates 1 nm-wide pores in both plasma and organelle membranes while leaving these membranes intact. This nsPFA thereby disrupts mitochondrial function and initiates regulated cell death (RCD). RCD is the death process used by all cells at the end of their useful life. This process is initiated by cellular signals indicating that something has gone wrong, such as a DNA mutation or membrane damage that the cell cannot repair. These cells signal the dendritic cells of the immune system to phagocytose them and incorporate all the tumor antigens. Sharing these antigens with T-cells in the lymph nodes leads to the activation of cytotoxic T-cells specific for cells displaying those tumor antigens.
Evidence that nsPFA initiates RCD
The RCD mechanism has several well-studied components that have been observed following nsPFA. The first is the release of signaling molecules called, DAMPs, which stands for danger-associated molecular patterns. These signaling molecules include ATP, HMGB1 and calreticulin and attract dendritic cells to the treated region to phagocytose the dying cells. nsPFA treatment has been shown to lead to the release of these DAMP molecules. In fact, a recent study of the DAMPs released by nanosecond, microsecond and millisecond pulse widths found that nsPFA was the only pulse width that released all three of the DAMP molecules. ATP and HMGB1 serve as chemo-attractants for dendritic cells such as macrophages. Calreticulin is a protein normally resident in the endoplasmic reticulum, but it is translocated to the plasma membrane during RCD. There it can bind to CD40L on dendritic cells and promote their maturation, initiating phagocytosis that incorporates tumor antigens. If any of the antigens are foreign, the immune system will generate cytotoxic CD8+ T-cells which will circulate in the body and attack any cells expressing those foreign antigens.
Evidence that nsPFA treatment can result in CD8+ cytotoxic T-cell production
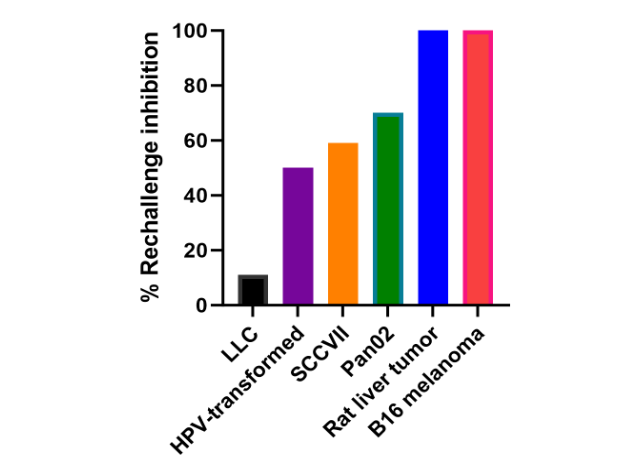
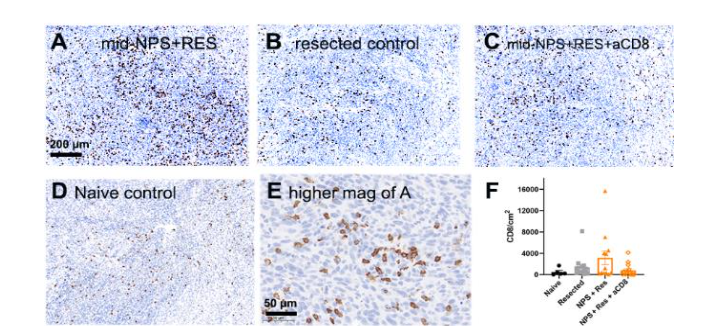
The earliest indication that nsPFA tumor treatment resulted in CD8+ T-cell production came from attempts to rechallenge mice several weeks after the primary tumor had been eliminated by nsPFA. The injection of the same line of some tumor cells failed to generate another tumor. Further studies on a variety of tumor types indicated that the strength of this blockage of rechallenge tumor growth varied with the tumor type suggesting that there are varying amounts of CD8+ T-cell production. Further evidence for the production of CD8+ T-cells came from immunohistochemistry labeling of the untreated, abscopal tumors. Labeling thin sections of untreated tumors in mice for which the primary tumor had been either ablated with nsPFA or resected indicated much more CD8+ infiltration in those untreated tumors in mice whose primary tumor had been ablated. In addition, this increase was reduced when CD8+ antibodies were introduced providing more evidence that these were indeed CD8+ T-cells.
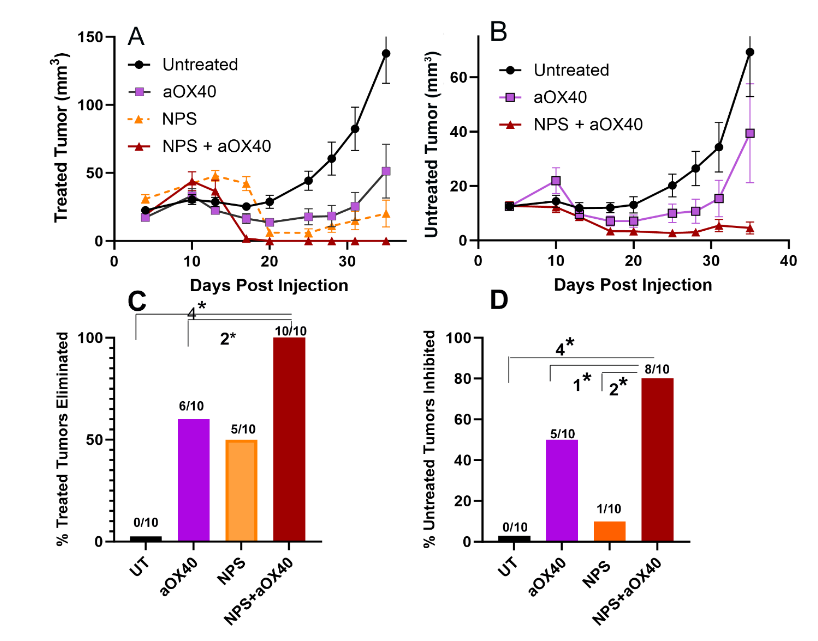
Perhaps the strongest data in support of tumor-specific CD8+ T-cell production comes from abscopal studies in which two tumors are injected into the same mouse and only one is ablated. By following the changes in the growth of the untreated tumor, one can determine if CD8+ T-cells are attacking it. We recently completed some abscopal studies of pancreatic tumors. In order to strengthen the immune response, we injected an immune stimulant, aOX40, into the nsPFA-treated tumor immediately after treatment. We observed that the combination of nsPFA treatment plus aOX40 injection completely eliminated the primary tumor and also eliminated the abscopal tumor in 80% of the mice. These data suggest that we had elicited an immune response that would eliminate metastases which is the ultimate goal. The use of immune stimulants is becoming more common as we find that ablation modalities that generate DAMPs do not normally sufficiently stimulate the immune response to completely eliminate metastases.
Immune Stimulants Used in Combination with Ablation Modalities
A survey of the literature identified several immune stimulants used in combination with various ablation modalities to enhance the immune system’s ability to eliminate untreated tumors:
- aOX40 and CpG: OX40, also known as CD134, is a co-stimulatory receptor on the surface of activated T-cells that belongs to the TNF receptor superfamily. It plays a role in T-cell activation, proliferation and survival. OX40 agonists, such as ivuxolimab or aOX40, can enhance the function of tumor-reactive T-cells and promote the anti-tumor immune response. In the work cited above, we injected aOX40 intratumorally after nsPFA to eliminate untreated pancreatic abscopal tumors. One of the pioneering studies using intratumoral injection of aOX40 combined with the synthetic toll-like receptor TLR9 ligand, CpG, eradicated untreated sites of murine colon carcinoma and breast cancer. Unfortunately this therapy was not as successful when used in a clinical trial.
- Imiquimod or Resiquimod: stimulates the innate immune system by activating toll-like receptor 7 (TLR7), commonly involved in pathogen recognition. Cells activated by imiquimod secrete cytokines such as interferon-α, interleukin-6 and tumor necrosis factor. Intratumoral injection of a gel form of imiquimod combined with aPD-1 rescued mice after colon carcinoma treatment with cryoablation.
- Granulocyte-Macrophage Colony Stimulating Factor (GM-CSF) and Bacillus Calmette-Guerin (BCG): GM-CSF is a cytokine that plays a crucial role in the development and function of white blood cells, particularly granulocytes and macrophages. BCG is a weakened form of the bacterium Mycobacterium bovis that is primarily used as a vaccine against tuberculosis and also in the treatment of recurrent bladder cancer. The combination of RFA on the primary tumor and GM-CSF/BCG injection completely eliminated distant mouse liver tumors. Similarly, injection of GM-CSF and purified protein derivatives (from BCG) eliminated the abscopal tumors in a murine colon carcinoma model.
- CD40: a transmembrane protein found on antigen-presenting cells that is required for their activation. When CD154 binds to CD40, it activates the antigen-presenting cells. Addition of the CD40 agonist, aCD40, to IRE-treated murine pancreatic tumors improved dendritic cell activation and generated a strong systemic antitumor T-cell response that inhibited metastatic disease progression.
- OK432: a freeze-dried product of streptococcus pyrogenes plus penicillin, also known as Picibanil, induces an inflammatory response. When the injection of OK-432 followed RFA treatment of osteosarcoma, distant untreated tumors shrink in size.
Ablating Murine Tumors with nsPFA
We have developed a very simple method for generating tumors in mice and treating them with nsPFA. When tumor cells are injected intradermally, the tumors will develop within the mouse skin which can be stretched over a light post for easy imaging of the tumor outline. We use a row of electrodes on either side of the tumor to expose all the tumor cells to our energy and the holes formed by the electrode penetration indicate the boundary of the applied field. Using this approach we have treated a wide range of murine tumor types with several field strengths, including B16 melanoma, 4T1 breast tumors, LLC lung carcinomas, SCCVII squamous cell carcinomas, and Pan02 pancreatic tumors. We found that the nsPFA energy required to eliminate these tumors varies with tumor type.
| Murine Tumor Type | Energy for 90% Ablation (mJ/mm3) |
|---|---|
| Pancreatic carcinoma | 240 |
| Lung carcinoma | 240 |
| Breast carcinoma | 360 |
| Squamous cell carcinoma | 360 |
| Melanoma | 480 |
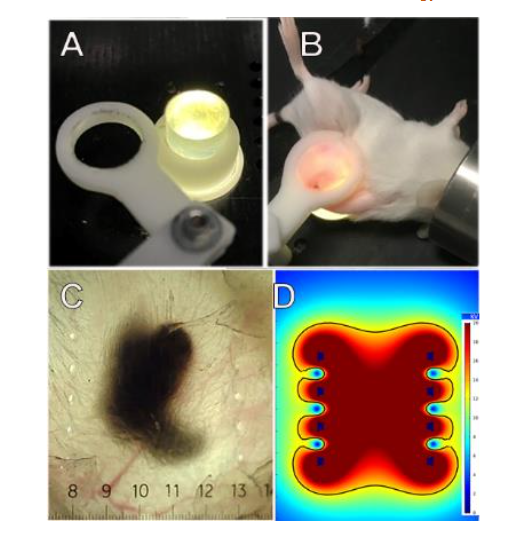
Figure 7. Method for treating the entire murine tumor. A. Photo of lighted silicone post over which the skin encapsulating the tumor is stretched. The white ring is placed over the skin to hold it once the tumor is in position. B. Photo of mouse with skin stretched over post for transillumination; C. Image of a melanoma tumor after treatment with two rows of bipolar needle electrodes. Holes indicate position of the two rows of electrodes; D. COMSOL Multiphysics model of the electric field distribution between the two rows of needle electrodes (black dots) indicating the uniformity of the applied electric field. (Reprinted with permission28)”
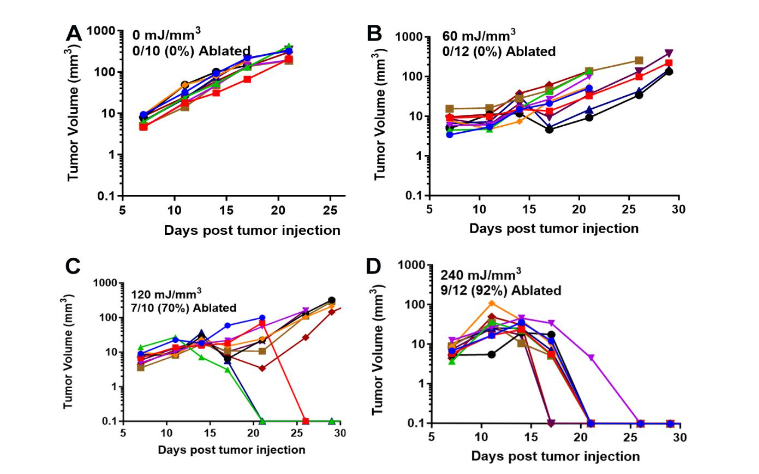
Figure 8. Growth of Lewis Lung carcinomas injected on Day 0 and treated on Day 5 with the indicated NPS energy. A. In the Sham control, none of the 10 treated tumors were ablated; B. 60 mJ/mm³ treatment ablates none of the 12 treated tumors; C. 120 mJ/mm³ ablates three of the 10 tumors treated; D. 240 mJ/mm³ treatment ablated all 10 of the 10 treated tumors. (reprinted with permission28)”
Clinical Studies
Despite the large number of studies demonstrating success in using nsPFA to treat murine tumors, there have been very few clinical trials applying nsPFA to human tumors. Up to now, only two human oncology clinical trials have been published, basal cell carcinoma and hepatocellular carcinoma.
BASAL CELL CARCINOMA
The first oncology clinical trial using NPS therapy treated 10 basal cell carcinomas (BCC) on only three subjects. Seven of the treated lesions were completely free of basaloid cells when biopsied, but 2 of the 7 exhibited seborrheic keratosis in the absence of basaloid cells. No scar was visible on the healed sites of any of the successfully ablated lesions. More recently, a larger trial treated 37 BCC lesions on 30 subjects, and 92% showed complete histological clearance of BCC. Histological analysis of the three cases where residual BCC was noted indicated that full energy coverage was not achieved. This study concluded that these nanosecond pulses were safe and effective for clearing both nodular and superficial BCC lesions. This success treating the deeper, nodular BCCs is noteworthy because they are resistant to treatment with other modalities.
HEPATOCELLULAR CARCINOMA
The first clinical study to evaluate the efficacy and safety of NPS for the treatment of hepatocellular carcinoma (HCC) began in 2020 in five hospitals in China and treated 192 patients with HCC. These HCC tumors were in high-risk locations where thermal ablation was not advised due to nearby blood vessels or biliary ducts. After contrast-enhanced magnetic resonance imaging, three-dimensional tumor measurements were used to determine the required electrode number and configuration. Two-six 19-gauge unipolar nsPEF electrodes were inserted percutaneously in a parallel manner with ultrasound or CD guidance. They had an interelectrode distance of 10-25 mm and active tip length of 20 mm. 800 pulses 300ns long and 25-30 kV/cm in amplitude were applied. The complete ablation rate at 1 month was 86% (90% for tumors smaller than 2 cm and 72% for tumors larger than 2 cm) with a low incidence of high-grade AEs.
Conclusion
We have concentrated here on the nsPFA nonthermal ablation modality due to its enhanced safety and cellular-specific targeting which avoids damaging acellular structures. Pulsed electric fields vary mainly by pulse width and amplitude as well as the type of cell death that they initiate. It is only the ultrashort nanosecond pulses that can be applied at amplitudes large enough to permeabilize the small intracellular organelles and initiate RCD rather than necrosis. Initiating RCD has the advantage of recruiting the immune system to generate cytotoxic CD8+ T-cells to attack any cells expressing tumor antigens. While the immune response triggered by nsPEF alone is not usually sufficient to eliminate untreated tumor cells, the addition of immune system stimulants such as aOX40 can strengthen the immune response sufficiently to eliminate untreated tumor cells, raising the hope that metastases might be eliminated when nsPFA is combined with immune stimulants. While the clinical work applying nsPFA is in its infancy, the preliminary results are quite encouraging and suggest that high efficacy should be possible. The application of nonthermal nsPFA along with immune stimulants will be a major focus for oncology research in the coming years.
Conflict of Interest Statement:
R. Nuccitelli is employed by Pulse Biosciences, Inc.
Funding Statement:
None
Acknowledgements:
None
References:
- Krummel MF, Allison JP. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. The Journal of experimental medicine 1995;182:459-465.
- Mc Neil V, Lee SW. Advancing Cancer Treatment: A Review of Immune Checkpoint Inhibitors and Combination Strategies. Cancers 2025;17.
- Engelen Y, Demuynck R, Ramon J, et al. Immunogenic cell death as interplay between physical anticancer modalities and immunotherapy. J Control Release 2025;384:113721.
- Ma J, Wei Z, Ye X. Interventional oncology and immunotherapy: current status and future perspectives. Front Immunol 2025;16:1541105.
- Xu Z, Hall TL, Vlaisavljevich E, Lee FT, Jr. Histotripsy: the first noninvasive, non-ionizing, non-thermal ablation technique based on ultrasound. Int J Hyperthermia 2021;38:561-575.
- Xu Z, Khokhlova TD, Cho CS, Khokhlova VA. Histotripsy: A Method for Mechanical Tissue Ablation with Ultrasound. Annu Rev Biomed Eng 2024;26:141-167.
- Neumann E, Rosenheck K. Permeability changes induced by electric impulses in vesicular membranes. J Membr Biol 1972;10:279-290.
- Davalos RV, Mir IL, Rubinsky B. Tissue ablation with irreversible electroporation. Ann Biomed Eng 2005;33:223-231.
- George AK, Miocinovic R, Patel AR, et al. A Description and Safety Overview of Irreversible Electroporation for Prostate Tissue Ablation in Intermediate-Risk Prostate Cancer Patients: Preliminary Results from the PRESERVE Trial. Cancers (Basel) 2024;16.
- Narayanan G, Bilimoria MM, Hosein PJ, Su Z, Mortimer KM, Martin RCG, 2nd. Multicenter randomized controlled trial and registry study to assess the safety and efficacy of the NanoKnife® system for the ablation of stage 3 pancreatic adenocarcinoma: overview of study protocols. BMC Cancer 2021;21:785.
- Yilmaz M, Karaaslan M, Şirin ME, et al. Salvage irreversible electroporation for locally recurrent prostate cancer after definitive radiotherapy: a systematic review. Prostate Cancer Prostatic Dis 2024.
- Partridge BR, O’Brien TJ, Lorenzo MF, et al. High-Frequency Irreversible Electroporation for Treatment of Primary Liver Cancer: A Proof-of-Principle Study in Canine Hepatocellular Carcinoma. J Vasc Interv Radiol 2020;31:482-491.e484.
- O’Brien TJ, Passeri M, Lorenzo MF, et al. Experimental High-Frequency Irreversible Electroporation Using a Single-Needle Delivery Approach for Nonthermal Pancreatic Ablation In Vivo. J Vasc Interv Radiol 2019;30:854-862.e857.
- Pakhomov AG, Shevin R, White JA, et al. Membrane permeabilization and cell damage by ultrashort electric field shocks. Arch Biochem Biophys 2007;465:109-118.
- Vernier PT. Mitochondrial membrane permeabilization with nanosecond electric pulses. Conf Proc IEEE Eng Med Biol Soc 2011;2011:743-5.:743-745.
- Ren W, Sain NM, Beebe SJ. Nanosecond pulsed electric fields (nsPEFs) activate intrinsic caspase-dependent and caspase-independent cell death in Jurkat cells. Biochem Biophys Res Commun 2012;421:808-812.
- …