Urine Output as a Key Indicator in Cardiac Surgery
URINE OUTPUT – A CRITICAL VITAL SIGN AND THERAPEUTIC TARGET IN CARDIAC SURGERY
Richard Solomon MD1, FASN
- Division of Nephrology and Hypertension, University of Vermont Larner College of Medicine Burlington, VT, USA 05401
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION: Solomon, R., URINE OUTPUT – A CRITICAL VITAL SIGN AND THERAPEUTIC TARGET IN CARDIAC SURGERY. Medical Research Archives, [online] 13(8).
https://doi.org/10.18103/mra.v13i8.6858
COPYRIGHT: © 2025 European Society of Medicine.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i8.6858
ISSN 2375-1924
ABSTRACT
Cardiac surgery associated acute kidney injury is one of the most serious adverse events occurring in this population. To date, there are no widely accepted therapies for treating cardiac surgery associated acute kidney injury once it occurs; therefore, prevention is most important. Multiple therapies have been proposed for prevention of cardiac surgery associated acute kidney injury but none has been successful and uniformly adopted by anesthesia and surgical societies. In the review that follows, I propose a new approach to prevention of cardiac surgery associated acute kidney injury based upon a readily available marker of kidney function, urine flow rate. The relationship of urine flow rate to cardiac surgery associated acute kidney injury and the effects of raising urine flow rate with diuretics is discussed. The physiology behind the beneficial effects of high urine flow rate is applicable to other causes of acute kidney injury.
Keywords
- Urine output
- Acute kidney injury
- Cardiac surgery
- Diuretics
Introduction
Urine flow rate (UR) is routinely available in patients undergoing general anesthesia and like other vital signs informs the operating team of the hemodynamic status of the patient. It is considered a marker of kidney perfusion and the overall hydration state of the patient. A widely accepted definition of oliguria is <0.5 ml/kg/h. However, there are no clear guidelines on how to interpret UR during the surgical period. Oliguria may be considered physiologic; a result of anesthesia induced ADH release. For this reason, ‘permissive oliguria’ has been proposed to minimize fluid administration during surgery. However, recent large randomized controlled trials in general surgical patients have found using a lower UR (<0.3 vs <0.5) to trigger fluid administration was associated with a higher incidence of post-operative acute kidney injury (AKI). Thus, there remains uncertainty about how to interpret intraoperative UR. In this review, the importance of UR and specific UR targets in the context of cardiac surgery are presented. Methods for achieving specific UR targets in cardiac surgery are frequently used and will be presented using the existing evidence from observational and randomized controlled trials.
Non-Cardiac Surgery
A 2023 review of management of intraoperative oliguria found that a UR <0.5 ml/kg/h was associated with post-operative acute kidney injury. A meta-analysis of 18,743 non-cardiac surgery patients found that oliguria defined as either <0.3 ml/kg/h or <0.5 ml/kg/h was associated with post op AKI. For the <0.3 ml/kg/h threshold definition, the OR was 2.64 while for the <0.5 ml/kg/h threshold, the OR was 1.72 (both p<.00001). The RELIEF trial randomized 2444 non-cardiac surgery patients to a liberal versus a restrictive intraoperative fluid management protocol. No diuretics were used. Intraoperative oliguria (<0.5 ml/kg/h for at least 1 hour) occurred in 889 patients (36%). A total of 21% of the total group developed post-operative AKI. The presence of oliguria conveyed an OR for post-operative AKI after adjustment for liberal vs restricted fluid management of 1.38 (p <.001). As the duration of oliguria increased there is a parallel increase in the incidence of post op AKI. This association of oliguria with AKI however is weak as a predictor of AKI. A review of post-operative AKI in 2015 did not find oliguria (<0.5 mg/kg/h) to be useful for fluid management and suggested avoiding diuretics to increase UR. A 2021 meta-analysis of 7 studies of 3862 patient undergoing thoracic surgery (non-cardiac) also did not find oliguria predictive. Most post-operative AKI occurs in subjects who were not oliguric. Furthermore, many physiologic mechanisms can result in oliguria and only some of those mechanisms might lead to AKI. Two other earlier (2016) meta-analyses of non-cardiac surgery patients suggested that targeting oliguria with either triggered fluid management or goal directed hemodynamics did not alter the incidence of AKI.
Cardiac surgery
Cardiac surgery patients may be a more informative cohort to look for a relationship between UR and AKI. First, this is a cohort with a high incidence of post-operative AKI (up to 30%) or cardiac surgery associated acute kidney injury (CSA-AKI). It is a more homogenous group of patients where there are identified risk factors for CSA-AKI that can be used for adjusting CSA-AKI rates between different levels of UR or treatments. Interestingly, a 2025 review of CSA-AKI doesn’t mention intraoperative UR as a risk factor. Nevertheless, there have been many observational studies suggesting that a low intraoperative UR, well above the traditional definition of oliguria, is a predictor of CSA-AKI. Hori in 2016 reported on CSA-AKI in a retrospective analysis of patients undergoing cerebral perfusion monitoring with transcranial doppler. A total of 579 patients were analyzed for predictors of CSA-AKI. CSA-AKI occurred in 29.5% of patients and 98% received mannitol in the cardiopulmonary perfusate. UR was influenced by MAP and pump flow rates. After adjusting for a number of risk covariates (age, diabetes, baseline renal function, use of diuretics, intraoperative MAP, pump flow rate), a UR <1.5 ml/kg/h was significantly associated with CSA-AKI. The intraoperative use of diuretics was also associated with a higher incidence of CSA-AKI. In the same year, Yilmaz studied 200 patients with baseline normal kidney. CSA-AKI occurred in 7%. All patients received mannitol in the perfusate. The relative risk of CSA-AKI began to increase at UR < 3.70 ml/kg/h. Song studied 696 patients with a CSA-AKI rate of 37%. Mannitol was used in all patients. That study found that UR <4.0 ml/kg/h was associated with a higher incidence of CSA-AKI. UR was also lower in the first 24h in those with CSA-AKI. In a study of 1184 children undergoing cardiac surgery for congenital heart disease by BIE, no relationship with intraoperative UR was found. CSA-AKI occurred in 23.1% of the children. Of note, 78% of the patients received intraoperative diuretics (see discussion below). In these observational trials, a target UR was not prescribed. It is unclear what percentage of patients were above threshold levels found to be associated with lowest incidence of CSA-AKI. Data extracted from one trial suggested only 20% were above the threshold. To confound the situation further, routine use of mannitol in the perfusion solution was the standard of care and many patients received intraoperative diuretics as well. While MAP and CO were found to be correlated with UR there was no differences in either hemodynamic parameter between those who developed CSA-AKI and those who didn’t. This collection of retrospective observational studies supports the importance of intraoperative UR as a predictor of CSA-AKI. This association remains an independent risk factor after adjustment for multiple additional preoperative and intraoperative variables. Different thresholds, below which the incidence of CSA-AKI increases, are reflected in the different cohort characteristics from which the data are derived such as elective surgery, kidney function, presence of diabetes, use of diuretics and transfusions. These studies at best support the concept that low UR during the time of maximum hemodynamic insult may play a role either as a predictor of subsequent kidney injury or as part of the pathophysiologic mechanism of injury itself. What this data doesn’t tell us is whether interventions to increase UR would decrease CSA-AKI.
Randomized controlled trials increasing intraoperative UR with diuretics
Mannitol: As noted above, many of the trials finding an association between UR and CSA-AKI add mannitol to the perfusate during cardiopulmonary bypass. This use is based in part upon mannitol’s pharmacodynamics to increase serum osmolality, reduce in oxidative stress, enhance renal perfusion and enhance urine output resulting from osmotic diuresis. The expectation is that these effects will reduce the incidence of CSA-AKI. A reduction in CSA-AKI with addition of mannitol to the CPB circuit has been found in some but not all randomized clinical trials. The early trials tended to be small and performed primarily in low-risk patients. However, recently an increasing number of RCT’s found no benefit of adding mannitol to the perfusate, including in patients with baseline renal insufficiency. A 2019 meta-analysis of 22 studies including 7 RCT’s in cardiac surgery also found no benefit despite increases in UR. These studies in cardiac surgery patients included patients with chronic kidney disease at baseline. They are, however, small trials (usually <100 patients). The most recent society guidelines in 2023 do not recommend the use of mannitol. Mannitol, acting as an osmotic diuretic in the proximal tubule, delivers more sodium to distal sites, including the loop of Henle. This results in increased Na uptake at those distal sites resulting in increased oxygen consumption. Depending upon renal blood flow, this could actually enhance the risk of AKI. Interestingly, Redfors showed that this increased oxygen consumption could be blocked by the addition of furosemide.
Loop diuretics: Diuretics, given in the presurgical period do not appear to offer an advantage for preventing CSA-AKI. Indeed, some authors have identified pre-operative use of diuretics as a risk factor for CSA-AKI. Likewise, a number of retrospective analyses of cardiac surgery patients have identified use of intraoperative furosemide as a risk factor for CSA-AKI. Moreira et al analyzed 329 cardiac surgery patients with pre-operative normal kidney function. CSA-AKI occurred in 19%. Decreased UR and use of furosemide were independently associated with CSA-AKI. Parolari analyzed 3219 cardiac surgery patients. CSA-AKI occurred in 8.9% and again low UR and use of furosemide were independently associated with CSA-AKI. Maruniak found no benefit of furosemide in 120 patients with a CSA-AKI rate of 26%. A small number of RCT’s of furosemide use during cardiac surgery have also found no benefit in preventing CSA-AKI. Lassnigg randomized 126 patients with normal kidney function to either dopamine, furosemide (0.5 ug/kg/min=~ 2-3 mg/h), or placebo for up to 48h post-surgery. Patients could get an additional bolus of 20 mg of furosemide if UO fell below 0.5 ml/kg/h. 38% and 50% of the dopamine and placebo group developed oliguria and required the additional furosemide bolus compared to 20% of the furosemide group. As expected, the furosemide group had the largest UR during surgery and for the initial 12 hours in the ICU. Urine output was replaced throughout the study. Seven patients developed CSA-AKI (not specifically defined). The furosemide group had the highest rate of CSA-AKI with 6 of the 7 cases. Fakhari randomized 81 patients having elective cardiac surgery to a constant infusion of furosemide at 2 mg/h for up to 12 h post-surgery vs 0.9% saline. UO was similar during surgery but higher for the first 24 hour period in the furosemide group (2.25 ml/kg/h vs 1.89 ml/kg/h respectively). The incidence of CSA-AKI (~27%) was similar in both groups. There was no difference in the use of inotropes, blood products or mannitol. Mahesh randomized 42 high risk patients with heart failure, diabetes, renal insufficiency to intraoperative furosemide infusion (4 mg /h) for up to 12 hours post-operatively vs placebo (saline 2 ml/h). While furosemide increased UR (3.4 ml/kg/h vs 1.2 ml/kg/h) there was no difference in CSA-AKI rates (43% furosemide vs 38% placebo). Kumada also found furosemide deleterious in off-pump cardiac surgery.
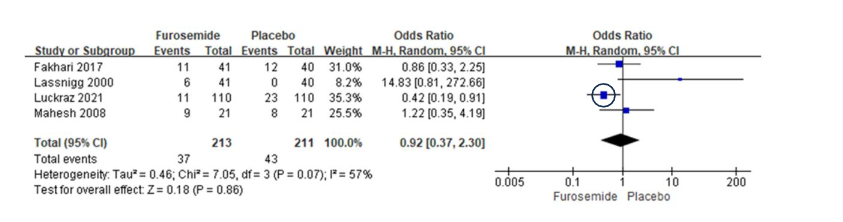
A recent meta-analysis involving 4 RCT trials found no benefit of intraoperative furosemide on CSA-AKI, but a reduction in days on mechanical ventilation and days in the ICU. There was no advantage on 90d mortality.
Why the discordance between the data indicating that lower intraoperative UR is associated with an increase in CSA-AKI yet administration of diuretics such as furosemide to enhance intraoperative UR do not show any benefit? The answer may lay in the fluid shifts induced by the diuretic. These shifts that can result in decreases in intravascular and extracellular volume, stimulate counter regulatory mechanisms (renin-angiotensin system, sympathetic system, and vasopressin) which maintain systemic hemodynamics at the expense of decreasing renal blood flow and glomerular filtration rate. These decreases in renal perfusion and oxygen delivery may mask the beneficial effects of a high UR. It is interesting that there is one trial (circled) in the Figure 1 meta-analysis that showed a benefit of furosemide. This was more specifically a trial to replace urine output in real time, ml for ml, so as not to allow any fluid shifts. Increasing UR without fluid shifts led to renal protection in that trial, a statistically significant 50% reduction in CSA-AKI. This supports the important role that fluid shifts and the counter regulatory mechanisms that they induce play in determining the outcome of kidney function.
Matching fluid administration with diuretic enhanced UR:
As noted above, the one positive study in the meta-analysis by Xie was that of Luckraz. This study was not designed as a trial of furosemide but rather of a device that matched intravenous fluid administration with urine output in real time. By this approach, fluid shifts were prevented and presumably the compensatory hormonal effects that reduce renal blood flow were also prevented. This is not the first use of this approach. What has been referred to a ‘forced matched diuresis’ has been used in the clinical arena for many years to treat nephrotoxin exposure (rhabdomyolysis, tumor lysis syndrome, contrast media), drug overdoses (barbiturates), and chemotherapeutic exposure (methotrexate). The underlying concept is that by having a high UR, not only is the nephrotoxin diluted in the urine but the contact time of the nephrotoxin with the renal epithelium is reduced thus mitigating injury. This approach has been used in the cardiac catheterization laboratory to reduce the incidence of contrast associated acute kidney injury (CA-AKI). Multiple RCT’s have shown the benefit of this approach in preventing CA-AKI and recent meta-analyses of these outcomes have been published. However, this is not an easy therapy to administer manually. When attempted manually it requires measuring urine output every 5 minutes during surgery and adjusting the iv fluid rate accordingly. A device to automatically monitor UR and simultaneously control an adjustable perfusion pump to match output with input is much simpler and more accurate.
Mechanisms of renal protection induced by high UR.
The mechanism of action of such high urine output is still unclear, although as discussed above, elimination of nephrotoxin is one possibility. For example, under the right conditions of dose and time of exposure, contrast is directly toxic the renal tubule cells. The incidence of CA-AKI and biomarkers of kidney injury are directly related to the density of nephrograms obtained immediately post the cardiac procedure. Forced matched diuresis is known to reduce the density of nephrograms post contrast exposure. This supports the idea that dilution of contrast and wash out from the nephron may be important for the beneficial effects of high urine outputs. However, this would not easily explain the benefit on CSA-AKI unless another toxin such as free hemoglobin is a major mechanism of injury. Alternatively, a high urine output may have some other benefit. Observations in man indicate that ingestion of water sufficient to increase urine output in the subsequent few hours alters kidney hemodynamics. For example, ingestion of 20 mL/kg of water over 15 minutes leads to an increase in medullary oxygen content as detected by BOLD MRI. The increase in medullary oxygen levels involves enhanced perfusion of the vasa recti in the medulla mediated by enhanced prostaglandins and nitric oxide. It can be inhibited by NSAIDs and restored with NO donors. Oral intake of water of this amount reduces the incidence of CA-AKI in STEMI patients undergoing primary PCI. Whether the clinical benefit of increase UO in cardiac surgery is mediated by similar mechanisms is an intriguing hypothesis.
Conclusion:
Monitoring urine output during cardiac surgery is already standard of care. What is being presented here is the rationale for purposefully increasing UO during the intraoperative and early post-operative period. This rationale is supported by the observations that higher intraoperative UO is associated with a lower incidence of CSA-AKI. Use of diuretics, such as mannitol or furosemide, alone doesn’t improve CSA-AKI perhaps because the associated fluid shifts offset the benefits of the higher UO. Preventing the fluid shifts by matching fluid intake with UO in real time seems to obviate those fluid shifts and allows the benefits of high UO to be revealed. While the mechanism(s) of benefit is still speculative, achieving a high UO is within the scope of routine practice in the OR and ICU with the help of a device to automate the process.
Conflict of Interest:
None.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Tallarico RT, McCoy IE, Depret F, Legrand M. Meaning and Management of Perioperative Oliguria. Anesthesiology. Feb 1 2024;140(2):304-312. doi:10.1097/ALN.0000000000004746
- Kunst G, Ostermann M. Intraoperative permissive oliguria – how much is too much? Br J Anaesth. Dec 1 2017;119(6):1075-1077. doi:10.1093/bja/aex387
- Mizota T, Yamamoto Y, Hamada M, Matsukawa S, Shimizu S, Kai S. Intraoperative oliguria predicts acute kidney injury after major abdominal surgery. Br J Anaesth. Dec 1 2017;119(6):1127-1134. doi:10.1093/bja/aex255
- Myles PS, Bellomo R, Corcoran T, et al. Restrictive versus Liberal Fluid Therapy for Major Abdominal Surgery. N Engl J Med. Jun 14 2018;378(24):2263-2274. doi:10.1056/NEJMoa1801601
- Ostermann M, Shaw AD, Joannidis M. Management of oliguria. Intensive Care Med. Jan 2023;49(1):103-106. doi:10.1007/s00134-022-06909-5
- Pang Z, Liang S, Xing M, Zhou N, Guo Q, Zou W. The correlation of intraoperative oliguria with acute kidney injury after noncardiac surgery: a systematic review and meta-analysis. Int J Surg. Mar 1 2023;109(3):449-457. doi:10.1097/JS9.0000000000000284
- Myles PS, McIlroy DR, Bellomo R, Wallace S. Importance of intraoperative oliguria during major abdominal surgery: findings of the Restrictive versus Liberal Fluid Therapy in Major Abdominal Surgery trial. Br J Anaesth. Jun 2019;122(6):726-733. doi:10.1016/j.bja.2019.01.010
- Shiba A, Uchino S, Fujii T, Takinami M, Uezono S. Association Between Intraoperative Oliguria and Acute Kidney Injury After Major Noncardiac Surgery. Anesth Analg. Nov 2018;127(5):1229-1235. doi:10.1213/ANE.0000000000003576
- du Toit L, Biccard BM. The relationship between intraoperative oliguria and acute kidney injury. Br J Anaesth. Jun 2019;122(6):707-710. doi:10.1016/j.bja.2019.03.008
- Goren O, Matot I. Perioperative acute kidney injury. Br J Anaesth. Dec 2015;115 Suppl 2:ii3-14. doi:10.1093/bja/aev380
- Zhao BC, Lei SH, Yang X, et al. Assessment of prognostic value of intraoperative oliguria for postoperative acute kidney injury: a retrospective cohort study. Br J Anaesth. Apr 2021;126(4):799-807. doi:10.1016/j.bja.2020.11.018
- Egal M, deGeusHRH, vanBommelJ, Groeneveld ABJ. Targeting oliguria reversal in perioperative restrictive fluid management does not influence the occurrence of renal dysfunction: A systematic review and meta-analysis. European Journal of Anaesthesiology EJA. 2016;33(6):425-435. doi:10.1097/eja.0000000000000416
- Egal M, Erler NS, de Geus HRH, van Bommel J, Groeneveld ABJ. Targeting Oliguria Reversal in Goal-Directed Hemodynamic Management Does Not Reduce Renal Dysfunction in Perioperative and Critically Ill Patients: A Systematic Review and Meta-Analysis. Anesthesia & Analgesia. 2016;122(1):173-185. doi:10.1213/ane.000000000000102
- Kamla CE, Meersch-Dini M, Palma LMP. Kidney Injury Following Cardiac Surgery: A Review of Our Current Understanding. Am J Cardiovasc Drugs. May 2025;25(3):337-348. doi:10.1007/s40256-024-00715-8
- Hori D, Katz NM, Fine DM, et al. Defining oliguria during cardiopulmonary bypass and its relationship with cardiac surgery-associated acute kidney injury. Br J Anaesth. Dec 2016;117(6):733-740. doi:10.1093/bja/aew340
- Yilmaz M, Aksoy R, Kilic Yilmaz V, Balci C, Duzyol C, Tekeli Kunt A. Urine Output during Cardiopulmonary Bypass Predicts Acute Kidney Injury after Coronary Artery Bypass Grafting. Heart Surg Forum. Dec 16 2016;19(6):E289-E293. doi:10.1532/hsf.1495
- Song Y, Kim DW, Kwak YL, et al. Urine Output During Cardiopulmonary Bypass Predicts Acute Kidney Injury After Cardiac Surgery: A Single-Center Retrospective Analysis. Medicine (Baltimore). May 2016;95(22):e3757. doi:10.1097/MD.0000000000003757
- Bie D, Li Y, Wang H, et al. Relationship between intra-operative urine output and postoperative acute kidney injury in paediatric cardiac surgery: A retrospective observational study. Eur J Anaesthesiol. Dec 1 2024;41(12):881-888. doi:10.1097/EJA.0000000000002044
- Yallop KG, Sheppard SV, Smith DC. The effect of mannitol on renal function following cardio-pulmonary bypass in patients with normal pre-operative creatinine. Anaesthesia. Jun 2008;63(6):576-82. doi:10.1111/j.1365-2044.2008.05540.x
- Smith MN, Best D, Sheppard SV, Smith DC. The effect of mannitol on renal function after cardiopulmonary bypass in patients with established renal dysfunction. Anaesthesia. Jul 2008;63(7):701-4. doi:10.1111/j.1365-2044.2007.05408.x
- Sköld A, Dardashti A, Lindstedt S, Hyllén S. No benefit of adding mannitol to cardiopulmonary bypass priming solution assessing cystatin C. A randomized clinical trial. Perfusion. May 24 2025:2676591251344857. doi:10.1177/02676591251344857
- Saleem M, Awan AH, Mushtaq A. Effect of Mannitol on Electrolytes and Its Relation with Kidney Injury in Cardiopulmonary Bypass. Biological and Clinical Sciences Research Journal. 2023;2023(1)doi:10.54112/bcsrj.v2023i1.535
- Hemmati Maslakpak M, Bilejani E, Negargar S, Khalili A, Alinejad V, Faravan A. The effect of mannitol on postoperative renal function in patients undergoing coronary artery bypass surgery: A double-blinded randomized controlled trial. J Cardiovasc Thorac Res. 2024;16(3):146-151. doi:10.34172/jcvtr.32992
- Svenmarker S, Claesson Lingehall H, Malmqvist G, Appelblad M. Plasma hyperosmolality during cardiopulmonary bypass is a risk factor for postoperative acute kidney injury: Results from double blind randomised controlled trial. Perfusion. Mar 2025;40(2):466-474. doi:10.1177/02676591241240726
- Sköld A, Dardashti A, Lindstedt S, Hyllén S. No benefit of adding mannitol to cardiopulmonary bypass priming solution assessing cystatin C. A randomized clinical trial. Perfusion. 0(0):02676591251344857. doi:10.1177/02676591251344857
- Waskowski J, Pfortmueller CA, Erdoes G, et al. Mannitol for the Prevention of Peri-Operative Acute Kidney Injury: A Systematic Review. Eur J Vasc Endovasc Surg. Jul 2019;58(1):130-140. doi:10.1016/j.ejvs.2019.02.003
- Brown JR, Baker RA, Shore-Lesserson L, et al. The Society of Thoracic Surgeons/Society of Cardiovascular Anesthesiologists/American Society of Extracorporeal Technology Clinical Practice Guidelines for the Prevention of Adult Cardiac Surgery-Associated Acute Kidney Injury. Ann Thorac Surg. Jan 2023;115(1):34-42. doi:10.1016/j.athoracsur.2022.06.054
- Redfors B, Swärd K, Sellgren J, Ricksten SE. Effects of mannitol alone and mannitol plus furosemide on renal oxygen consumption, blood flow and glomerular filtration after cardiac surgery. Intensive Care Med. Jan 2009;35(1):115-22. doi:10.1007/s00134-008-1206-5
- Kumada Y, Yoshitani K, Shimabara Y, Ohnishi Y. Perioperative risk factors for acute kidney injury after off-pump coronary artery bypass grafting: a retrospective study. JA Clin Rep. 2017;3(1):55. doi:10.1186/s40981-017-0125-2
- Moreira R, Jacinto T, Neves P, Vouga L, Baeta C. Predictors of Acute Kidney Injury Associated with Cardiopulmonary Bypass. Rev Port Cir Cardiotorac Vasc. Apr-Jun 2019;26(2):109-115.
- Parolari A, Pesce LL, Pacini D, et al. Risk factors for perioperative acute kidney injury after adult cardiac surgery: role of perioperative management. Ann Thorac Surg. Feb 2012;93(2):584-91. doi:10.1016/j.athoracsur.2011.09.073
- Maruniak S, Loskutov O, Swol J, Todurov B. Factors associated with acute kidney injury after on-pump coronary artery bypass grafting. J Cardiothorac Surg. Oct 8 2024;19(1):598. doi:10.1186/s13019-024-03103-0
- Lassnigg A, Donner E, Grubhofer G, Presterl E, Druml W, Hiesmayr M. Lack of renoprotective effects of dopamine and furosemide during cardiac surgery. J Am Soc Nephrol. Jan 2000;11(1):97-104. doi:10.1681/asn.V11197
- Fakhari S, Bavil FM, Bilehjani E, Abolhasani S, Mirinazhad M, Naghipour B. Prophylactic furosemide infusion decreasing early major postoperative renal dysfunction in on-pump adult cardiac surgery: a randomized clinical trial. Res Rep Urol. 2017;9:5-13. doi:10.2147/rru.S126134
- Mahesh B, Yim B, Robson D, Pillai R, Ratnatunga C, Pigott D. Does furosemide prevent renal dysfunction in high-risk cardiac surgical patients? Results of a double-blinded prospective randomised trial. Eur J Cardiothorac Surg. Mar 2008;33(3):370-6. doi:10.1016/j.ejcts.2007.12.030
- Xie CM, Yao YT, Yang K, et al. Furosemide does not reduce the incidence of postoperative acute kidney injury in adult patients undergoing cardiac surgery: A PRISMA-compliant systematic review and meta-analysis. J Card Surg. Dec 2022;37(12):4850-4860. doi:10.1111/jocs.17120
- Luckraz H, Giri R, Wrigley B, et al. Reduction in acute kidney injury post cardiac surgery using balanced forced diuresis: a randomized, controlled trial. Eur J Cardiothorac Surg. Apr 13 2021;59(3):562-569. doi:10.1093/ejcts/ezaa395
- Putzu A, Boscolo Berto M, Belletti A, et al. Prevention of Contrast-Induced Acute Kidney Injury by Furosemide With Matched Hydration in Patients Undergoing Interventional Procedures: A Systematic Review and Meta-Analysis of Randomized Trials. JACC Cardiovasc Interv. Feb 27 2017;10(4):355-363. doi:10.1016/j.jcin.2016.11.006
- Zhang W, Zhang J, Yang B, et al. Effectiveness of oral hydration in preventing contrast-induced acute kidney injury in patients undergoing coronary angiography or intervention: a pairwise and network meta-analysis. Coronary artery disease. Jun 2018;29(4):286-293. doi:10.1097/MCA.0000000000000607
- Romano G, Briguori C, Quintavalle C, et al. Contrast agents and renal cell apoptosis. Eur Heart J. Oct 2008;29(20):2569-76. doi:10.1093/eurheartj/ehn197
- Chu LL, Katzberg RW, Solomon R, et al. Clinical Significance of Persistent Global and Focal Computed Tomography Nephrograms After Cardiac Catheterization and Their Relationships to Urinary Biomarkers of Kidney Damage and Procedural Factors: Pilot Study. Invest Radiol. Dec 2016;51(12):797-803. doi:10.1097/RLI.0000000000000268
- Ben-Haim Y, Chorin E, Hochstadt A, et al. Forced Diuresis with Matched Isotonic Intravenous Hydration Prevents Renal Contrast Media Accumulation. J Clin Med. Feb 8 2022;11(3)doi:10.3390/jcm11030885
- Leaf DE, Rajapurkar M, Lele SS, et al. Increased plasma catalytic iron in patients may mediate acute kidney injury and death following cardiac surgery. Kidney Int. May 2015;87(5):1046-54. doi:10.1038/ki.2014.374
- Prasad PV, Epstein FH. Changes in renal medullary pO2 during water diuresis as evaluated by blood oxygenation level-dependent magnetic resonance imaging: effects of aging and cyclooxygenase inhibition. Kidney Int. Jan 1999;55(1):294-8. doi:10.1046/j.1523-1755.1999.00237.
- Prasad PV, Priatna A, Spokes K, Epsein F. Changes in intrarenal oxygenation as evaluated by BOLD MRI in a rat kidney model for radiocontrast nephropathy. J Magn Reson Imaging. 2011;13:744-47.
- Xie W, Zhou Y, Liao Z, Lin B. Effect of Oral Hydration on Contrast-Induced Acute Kidney Injury among Patients after Primary Percutaneous Coronary Intervention. Cardiorenal medicine. 2021;11(5-6):243-251. doi:10.1159/000520088

