Occupational Asthma: Classifying Chemical Allergens
Occupational Asthma and Challenges in Classifying Chemical Respiratory Allergens
Mark A Pemberton1, Stuart Hindle3, Andreas Flueckiger4 and Knut Kreuzer2
- Systox Associates, Macclesfield, UK;
- Röhm GmbH, Darmstadt, Germany;
- Dow Europe GmbH, Horgen, Switzerland;
- Occupational Toxicology and Containment for the Pharmaceutical Industry, Pratteln, Switzerland.
OPEN ACCESS
PUBLISHED 31 August 2025
CITATION Pemberton, MA., Hindle, S., et al., 2025. Occupational Asthma and Challenges in Classifying Chemical Respiratory Allergens. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6794
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6794
ISSN 2375-1924
ABSTRACT
Respiratory Sensitisation is a serious health condition that can have life-threatening consequences and that is regulated under the UN globally harmonised system (GHS) and the European system for classification, labelling and packaging of hazardous chemicals and mixtures (CLP). In the absence of widely accepted and validated approaches for the prospective identification of chemicals that induce respiratory sensitisation, classification relies on clinical evidence of causation of substance-specific respiratory hypersensitivity, typically seen as occupational asthma. Significant advances have been made in recent years in the phenotyping of both asthma and occupational asthma since the introduction of criteria and guidance for classification that are not reflected in the current criteria and guidance. Typically generated clinical data, including specific inhalation challenge tests, while sufficient for diagnosis and health management are often deficient for regulatory classification purposes as they are often not sufficiently documented to meet the required standards and fail to reliably distinguish between occupational asthma and non-specific irritant exacerbation of pre-existing asthma that is regulated under another classification endpoint. A multistakeholder forum is proposed to facilitate revision of the now outdated criteria and guidance. Differences between underlying mechanisms of respiratory sensitisation and occupational asthma jeopardise establishment of an Adverse Outcome Pathway for respiratory sensitisation and development of alternative methods, including non-animal methods.
Keywords: Respiratory Sensitisation; Occupational Asthma; Work Exacerbated Asthma; Clinical Data; Specific Inhalation Challenge (SIC) tests; Classification, Labelling and Packaging (CLP); Globally Harmonised System of Classification and Labelling of Chemicals (GHS).
1. Introduction
Respiratory Sensitisation (RS), also known as respiratory hypersensitivity, is an immune response to inhaled substances that can lead to allergic reactions, particularly asthma. It is one of the manifestations of type I allergy. This process, in common with contact (skin) sensitisers (a manifestation of type IV allergy), involves an initial exposure (sensitisation phase) followed by subsequent reactions upon re-exposure (elicitation phase). RS is identified as a classifiable health hazard under the United Nations Globally Harmonised System of Classification and Labelling of Chemicals (GHS) and as implemented within the EU under regulation 1272/2008 on the classification, labelling, and packaging of chemical substances and mixtures (CLP). On the basis that type I allergy can cause severe, potentially life-threatening reactions like anaphylactic shock it is recognised under Article 57(f) of the EU’s Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulation as being of equivalent concern to carcinogenicity, mutagenicity and reproductive toxicity and prioritised for regulatory action including restrictions, potentially limiting or banning substances of very high concern (SVHC) that pose unacceptable risks.
Although chemical-induced respiratory hypersensitivity has been known for decades, there are currently no widely accepted and validated approaches for the prospective identification of chemicals that induce respiratory sensitisation. Recent comprehensive reviews exist elsewhere. Consequently, GHS and CLP criteria for classification recognise that evidence that a substance can lead to specific respiratory hypersensitivity will normally be based on human experience and that this is normally seen as asthma, but other hypersensitivity reactions such as rhinitis/conjunctivitis and alveolitis are also considered. In this context, the criteria are referring to clinical evidence demonstrating the intrinsic property of a chemical to cause occupational asthma (OA) and the diagnosis consequently relies upon expert judgement of clinical and other evidence in a weight-of-evidence approach.
Clinical evidence confirming the presence of asthma and demonstrating a work-related pattern of disease indicative of causation of OA draws from a number of sources. Initial suspicions of the development of OA can be raised by general practitioners or occupational physicians, where they exist. Asthma diagnosis is typically confirmed through a combination of tests and assessments, including lung function tests like spirometry and peak flow tests, along with a review of symptoms and medical history. Additionally, referral to specialist centres for further evaluation of lung function and airway sensitivity using bronchodilator responsiveness tests and challenge tests may occur. Confirmation of a work-related association is typically done through interrogation of patient medical and work histories combined with robust characterisation of exposure profile by a proficient industrial hygienist, and may lead to the identification of possible causative/exacerbating agents.
Work-related asthma (WRA), i.e., asthma displaying a pattern of work-related clinical symptoms encompasses both asthma caused in the workplace (sensitiser induced (SIOA) and irritant induced (IIOA)) and asthma that is pre-existing and made worse by exposure to agents in the workplace (Work Exacerbated Asthma or WEA).
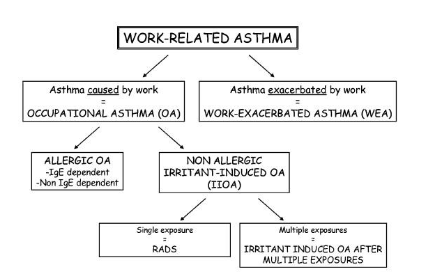
While it is generally accepted that it is important to distinguish between OA and non-specific WEA, it may be a moot point from a perspective whether there is a need to distinguish between SIOA and IIOA for occupational health management. As a result, the extent of the clinical evidence supporting a diagnosis may vary considerably. Indeed, more often than not, if there is exposure to a putative respiratory sensitiser within the workplace then both a work-related association and causation are assumed rather than identified by thorough workplace analysis. This impacts greatly on the utility of clinical data for regulatory classification purposes and the requirement to demonstrate causation, i.e., to demonstrate that a substance has led to specific respiratory hypersensitivity. This article discusses those aspects of clinical and related data procurement that are essential for effective health management and regulatory classification and labelling purposes.
2. Management of Occupational Asthma (OA)
It is important to recognise from the outset that asthma per se is characterised by a complex pathogenesis encompassing several well-defined phenotypes including: allergic, non-allergic, eosinophilic, neutrophilic, exercise-induced, and aspirin-induced. Asthma is a very prevalent disorder and it is within this setting that OA develops. Namely, a typical workforce will comprise both healthy individuals and asthmatics of varying degrees of sensitivity and susceptibility to WEA. This is reflected, but perhaps not sufficiently emphasised, in the generally accepted paradigm for WRA described by Moscato and coworkers in which there is clear distinction between asthma caused in the workplace (SIOA and IIOA) and pre-existing asthma made worse by exposure to agents in the workplace i.e., WEA.
In practice, however, the distinction between new asthma being caused, and pre-existing asthma being exacerbated, by agents in the workplace is extremely challenging. This is in no small part due to the inevitable retrospective nature of the events leading up to the development of the disease, and difficulties in distinguishing between IIOA and non-specific irritant WEA.
Approximately 16% of new asthma diagnoses in adults is thought to be work-related (WRA). OA, or more correctly speaking WRA, is recognised as an important health issue due to its extremely distressing and potentially life-threatening nature, high prevalence and social and economic burden. It has been reported that although the incidence of life-threatening asthmatic events is extremely low, individuals with WRA tend to express more severe symptoms when compared with their non-WRA counterparts. In this regard, the incidence of severe asthma has been estimated to be between 16.2% and 18%. The extent to which selection bias impacts on these estimates is unknown. It is only natural, therefore, that the primary objective of health management professionals is identifying and minimising exposure to workplace triggers, using medications to control symptoms, and implementing strategies to prevent future attacks. In this regard, early diagnosis and removal from the triggering substance are crucial for better outcomes. Some health professionals may even hold the viewpoint that it is unnecessary to determine the underlying mechanism by which causation occurred, or even if it was exacerbation of a pre-existing condition rather than causation. Occupational physicians and industrial hygienists, however, are likely to hold a very different viewpoint as the management of OA risk in an occupational setting is very different. Depending upon whether the underlying mechanism is true respiratory sensitisation or irritant induced asthma, as opposed to exacerbation by non-specific stressor agents including general air pollution such as dusts, irritant chemicals, molds, smoke or fumes and even physical stressors like temperature and humidity, different risk management measures apply.
In the absence of comprehensive and validated approaches for the prospective identification of chemicals that induce respiratory sensitisation it remains beholden upon respiratory physicians to make timely diagnostic decisions, often based upon incomplete data. Nevertheless, there are unavoidable consequences of not sufficiently distinguishing between SIOA, IIOA and WEA that go beyond the immediate objective of optimising patient well-being and healthcare outcomes. One likely consequence is that lists of respiratory sensitisers in addition to including will also contain positive that cause WEA as opposed to SIOA or IIOA. Proliferation of such lists based upon insufficiently defined or heterogeneous criteria encourage further a priori misdiagnosis that weakens then not only the basis of evidence-based medicine, but also research into approaches for the prospective identification of chemicals that induce respiratory sensitisation including new approach methodologies (NAMs).
3. Clinical diagnosis of Occupational Asthma (OA)
Clinical guidelines for the diagnosis of OA have been developed and revised in recent years. These guidelines converge in recommending investigation and comprehensive documentation of occupational and patient/familial clinical histories, and clinical data including a combination of physical examination, pulmonary function tests, allergy testing and bronchial provocation challenge tests. The scope and limitations of these investigations have recently been reviewed elsewhere.
Occupational history can provide valuable information on the job history, workplace activities and, if supported by occupational hygiene data, exposure to chemicals that are recognised, or suspected, to be linked to WRA. Patient and familial clinical history, especially of allergies, eczema, or early respiratory infections can indicate not only pre-existence of asthma, but also inherited genetic predisposition and the susceptibility to developing asthma. Additionally, understanding past exposures (like smoking or air pollution) and other respiratory health conditions can help identify potential confounding factors that might influence susceptibility, asthma development and severity.
Clinical investigations can confirm the diagnosis of asthma and cross-shift pulmonary function tests like spirometry and peak expiratory flow (PEF) monitoring can confirm that symptoms are work or even task related. Additionally, marked Non-Specific Bronchial Hyperreactivity (NSBH) in a Methacholine challenge test can confirm the diagnosis of asthma and indicate increased susceptibility to non-specific irritation and WEA. While allergy testing has proven to be of value in identifying specific hypersensitivity to high molecular weight allergens, such as proteins, this has proven unreliable for most low molecular weight chemicals suspected of causing OA.
A positive bronchial provocation inhalation challenge tests conducted with specific substances, also known as the Specific Inhalation Challenge or SIC test, if conducted according to recognised guidelines, has been claimed to be the gold standard for a diagnosis of OA. Indeed, the current criteria and guidance for classification and labelling (GHS and CLP) state that the results of positive bronchial challenge tests are considered to provide sufficient evidence for classification on their own. However, it has to be recognised that the SIC test only demonstrates that the specific substance under the exposure conditions used in the SIC test is capable of provoking a bronchial response in the subject. As such the test does not distinguish between OA and WEA.
The do include recommendations to mimic typical workplace conditions and to monitor exposure levels during the SIC test so as to maintain these below the occupational exposure limit (OEL), thereby assuming that non-specific irritation will be avoided. This is an over simplification of what is often a very complex aspect of occupational health management. Firstly, this approach assumes that the OEL is protective not only for healthy personnel but also for asthmatic individuals that are being subjected to SIC testing. However, this may not necessarily be the case since the OELs are primarily designed to protect the health of the majority of healthy adult workers, they are not always sufficient to protect individuals with pre-existing conditions like asthma, especially those with work-related asthma. While OELs aim to prevent adverse health effects from chemical exposures, they often don’t account for the increased sensitivity of some individuals or the potential for substances to trigger or exacerbate asthma. In this regard, asthmatics have been reported to have up to three-fold greater bronchial sensitivity than their healthy counterparts. Secondly, the profile of workplace exposures is typically complex with workers experiencing varying levels of exposure depending on the tasks they perform. These exposures often include background levels that are relatively consistent, overlaid with brief, but potentially high-intensity, “peak exposures” associated with specific tasks or operations. An exposure profile can be further determined by the availability of engineering control measures, such as local exhaust ventilation, and the use of respiratory protective equipment. Only a comprehensive survey by a trained industrial hygienists will provide a reliable insight into the exposure conditions that are required to be mimicked in a SIC test.
Furthermore, since it has been recognised that in practice, appropriate techniques for measuring the wide variety of agents causing occupational asthma are seldom available, exposure monitoring is rarely done with sufficient rigor. Consequently, the good intentioned use of excessively high exposure conditions in SIC tests so as to maximise test sensitivity can result in non-specific irritation and the triggering of false positive responses. Some respiratory physicians are of the view that observation of a Late Asthmatic Response (LAR) in a positive SIC test, whether present in isolated or dual reactions (DAR), reliably indicate involvement of immune mechanisms thereby confirming OA. However, this viewpoint is not supported by the available science as reviewed elsewhere. Furthermore, LAR are observed during exercise-induced asthma so the observation of LAR in positive SIC tests does not necessarily indicate the mechanism by which the challenge substance is acting and substance-specific hypersensitivity cannot be assumed.
It remains the case that thorough interrogation of patient history combined with robust characterisation of exposure profile by a proficient industrial hygienist is still the most reliable means of identifying possible causative/exacerbating agents for OA.
4. Classification and Labelling of Respiratory Sensitisers
With regards to Respiratory Sensitisation, criteria and Guidance on the Application of the CLP Criteria still reflect closely GHS Revision 1 published in 2003, and unlike information on other hazards that has been revised to reflect developments in toxicological hazard assessment, lag the current state of knowledge on the identification of RS and clinical diagnosis of OA. For example, current criteria state under §3.4.2.1.2.1 that Evidence that a substance can lead to specific respiratory hypersensitivity will normally be based on human experience. In this context, the use of the term lead conveys the requirement that the substance must have the intrinsic property to cause hypersensitivity to happen or exist as opposed to aggravating a pre-existing condition (WEA). This is consistent with the paradigm of work-related asthma (WRA) described by Moscato and coworkers and the differentiation between OA induced by agents in the workplace (SIOA and IIOA) as opposed to asthma that is exacerbated (WEA). However, the criteria go on to state that in this context, hypersensitivity is normally seen as asthma, but other hypersensitivity reactions such as rhinitis/conjunctivitis and alveolitis are also considered. The condition will have the clinical character of an allergic reaction. However, immunological mechanisms do not have to be demonstrated. This relief from having to demonstrate immune involvement was originally introduced to address uncertainties in the precise nature of the immune response(s) that drive the acquisition of sensitisation and inability to demonstrate substance-specific IgE antibodies. This is acknowledged in guidance where it states that the mechanisms by which substances induce symptoms of asthma are not yet fully known. For preventative measures, these substances are considered respiratory sensitisers. This broadening of the criteria ensured that irritant induced OA (IIOA) fell under the scope of Respiratory Sensitisation classification, but placed greater pressure on the need to distinguish between irritant IIOA and non-specific irritant exacerbation of pre-existing asthma, both in the workplace (WEA) and in SIC tests (false positive results). The criteria and guidance make a single reference to this critical aspect under Annex I: §3.4.2.1.3. of ECHA guidance, albeit in a section referring to animal studies, where it states under footnote: The mechanisms by which substances induce symptoms of asthma are not yet fully known. For preventative measures, these substances are considered respiratory sensitisers. However, if on the basis of the evidence, it can be demonstrated that these substances induce symptoms of asthma by irritation only in people with bronchial hyper reactivity, they should not be considered respiratory sensitisers.
It may therefore be justified in claiming that the criteria and guidance do not give sufficiently clear and precise requirements and limitations on the use of clinical data for classification purposes. Revision of the criteria and guidance is warranted to avoid contentious interpretation of clinical data and proliferation of scientifically unsound classifications, and to align clinical and regulatory understanding. This revision must recognise and balance the constraints imposed by medical confidentiality with the adequacy of clinical data in satisfying data quality, reliability and transparency criteria under CLP. A particular focus must be given to the role of the SIC test in decision making. There is growing evidence from recent EU harmonised classification and labelling (CLH) activities that the current requirement for tests to meet Annex I §3.4.2.1.2.3. (b) is too broad to be of any reliable value. In this regard, detailed guidance is required to highlight the key features, reporting standards and data requirements that are essential for the correct interpretation of SIC tests and the determination of substance-specific hypersensitivity, and compliance with CLP criteria relating to reliable and good quality.
In taking this forward, it should be recognised by all parties that the priorities of clinicians, toxicologists and regulators are in some aspects necessarily different and that the misalignment of GHS/CLP criteria and guidance and the state of knowledge underpinning acquisition and expression of OA inevitably leads to tension over the use and interpretation of human and non-human data. A multistakeholder forum is therefore essential to broker a common understanding of the state of the science and how best to revise the criteria and guidance.
It is also anticipated that this broader awareness by all stakeholder groups of the practical differences between RS and OA, the paradigm of WRA, and the contributions of sensitiser induced OA, IIOA and WEA, will contribute to the refinement of the Adverse Outcome Pathway (AOP) for respiratory sensitisation to recognise the involvement of non-immune mechanisms and future development of NAMs.
Conflict of Interest Statement:
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Mark Alan Pemberton reports financial support was provided by Cefic Methacrylate Sector Group. Knut Kreuzer reports financial support was provided by Röhm GmbH, Darmstadt, Germany. Stuart Hindle reports financial support was provided by Dow Europe GmbH, Horgen, Switzerland. Mark Alan Pemberton reports a relationship with CEFIC Methacrylate Sector Group that includes: consulting or advisory. Knut Kreuzer reports a relationship with Röhm GmbH, Darmstadt, Germany that includes: employment. Stuart Hindle reports a relationship with Dow Europe GmbH, Horgen, Switzerland that includes: employment. Andreas Flueckiger declares that he has no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding Statement:
The CEFIC Methacrylate Sector Group (the sponsors) provided financial support to the corresponding author for the preparation of the article. The sponsors had no input into the collection, analysis and interpretation of publications, in the writing of the paper or the decision to submit the article for publication.
Acknowledgements:
The authors gratefully acknowledge the expert views and comments of the Editor and of the anonymous peer-reviewers.
References:
- Hargitai R, Parráková L, Szatmári T, Monfort-Lanzas P, Galbiati V, Audouze K, Jornod F, Staal YCM, Burla S, Chary A, Gutleb AC, Lumniczky K, Vandebriel RJ, Gostner JM. Chemical respiratory sensitization-Current status of mechanistic understanding, knowledge gaps and possible identification methods of sensitizers. Front Toxicol. 2024 Jul 29;6:1331803. doi: 10.3389/ftox.2024.1331803. PMID: 39135743; PMCID: PMC11317441.
- UN. Globally Harmonized System of Classification and Labelling of Chemicals. 2023. GHS Rev. 10. The United Nations Economic Commission for Europe, Geneva, Switzerland. Available at https://unece.org/transport/dangerous-goods/ghs-rev10-2023. Accessed on 16th May 2025.
- EC. Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006 (Text with EEA relevance). 2008. Available at http://data.europa.eu/eli/reg/2008/1272/oj
- McLendon K, Sternard BT. Anaphylaxis. [Updated 2023 Jan 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482124/
- EC. Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), establishing a European Chemicals Agency, amending Directive 1999/45/EC and repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. 2006. Available at https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A02006R1907-20241010
- ECHA. Identification of substances as SVHCs due to equivalent level of concern to CMRs (Article 57(f)) sensitisers as an example. 2012. Available at: https://echa.europa.eu/documents/10162/13657/svhc_art_57f_sensitisers_en.pdf/a50728cc-6514-486c-9108-193a88b4bc9e
- Pemberton MA, Arts JH, Kimber I. Identification of true chemical respiratory allergens: Current status, limitations and recommendations. Regul Toxicol Pharmacol. 2024 Feb;147:105568. doi: 10.1016/j.yrtph.2024.105568. Epub 2024 Jan 14. PMID: 38228280.
- Tarlo SM, Lemiere C. Occupational asthma. N Engl J Med. 2014 Feb 13;370(7):640-9. doi: 10.1056/NEJMra1301758. PMID: 24521110.
- Vandenplas O, Wiszniewska M, Raulf M, de Blay F, Gerth van Wijk R, Moscato G, Nemery B, Pala G, Quirce S, Sastre J, Schlünssen V, Sigsgaard T, Siracusa A, Tarlo SM, van Kampen V, Zock JP, Walusiak-Skorupa J; European Academy of Allergy and Clinical Immunology. EAACI position paper: irritant-induced asthma. Allergy. 2014 Sep;69(9):1141-53. doi: 10.1111/all.12448. Epub 2014 Jul 5. PMID: 24854136.
- Vandenplas O, Suojalehto H, Cullinan P. Diagnosing occupational asthma. Clin Exp Allergy. 2017 Jan;47(1):6-18. doi: 10.1111/cea.12858. PMID: 27883240.
- Malo JL. The case for confirming occupational asthma: why, how much, how far? J Allergy Clin Immunol. 1993 May;91(5):967-70. doi: 10.1016/0091-6749(93)90207-v. PMID: 8491945.
- Tarlo SM, Balmes J, Balkissoon R, Beach J, Beckett W, Bernstein D, Blanc PD, Brooks SM, Cowl CT, Daroowalla F, Harber P, Lemiere C, Liss GM, Pacheco KA, Redlich CA, Rowe B, Heitzer J. Diagnosis and management of work-related asthma: American College Of Chest Physicians Consensus Statement. Chest. 2008 Sep;134(3 Suppl):1S-41S. doi: 10.1378/chest.08-0201. Erratum in: Chest. 2008 Oct;134(4):892. PMID: 18779187.
- Cartier A, Sastre J. Clinical assessment of occupational asthma and its differential diagnosis. Immunol Allergy Clin North Am. 2011 Nov;31(4):717-28, vi. doi: 10.1016/j.iac.2011.07.005. PMID: 21978853.
- Tiotiu AI, Novakova S, Labor M, Emelyanov A, Mihaicuta S, Novakova P, Nedeva D. Progress in Occupational Asthma. Int J Environ Res Public Health. 2020 Jun 24;17(12):4553. doi: 10.3390/ijerph17124553. PMID: 32599814; PMCID: PMC7345155.
- Moscato G, Pala G, Barnig C, De Blay F, Del Giacco SR, Folletti I, Heffler E, Maestrelli P, Pauli G, Perfetti L, Quirce S, Sastre J, Siracusa A, Walusiak-Skorupa J, van Wjik RG; European Academy of Allergy and Clinical Immunology. EAACI consensus statement for investigation of work-related asthma in non-specialized centres. Allergy. 2012 Apr;67(4):491-501. doi: 10.1111/j.1398-9995.2011.02784.x. Epub 2012 Jan 19. PMID: 22257175.
- Pemberton MA, Kimber I. Accurate regulatory classification of chemical respiratory allergens: The case for robust characterisation of causation. Regul Toxicol Pharmacol. 2025 Mar;157:105785. doi: 10.1016/j.yrtph.2025.105785. Epub 2025 Feb 12. PMID: 39952546.
- Jarjour NN, Kelly EA. Pathogenesis of asthma. Med Clin North Am. 2002 Sep;86(5):925-36. doi: 10.1016/s0025-7125(02)00087-1. PMID: 12428538.
- Holgate ST. Pathogenesis of asthma. Clin Exp Allergy. 2008 Jun;38(6):872-97. doi: 10.1111/j.1365-2222.2008.02971.x. PMID: 18498538.
- Carr TF, Sharma S. Characterizing severe asthma phenotypes. UpToDate.com Literature review current through: Dec 2024. This topic last updated: Jan 10, 2025. Accessed February 2 2025. Available at https://www.uptodate.com/contents/characterizing-severe-asthma-phenotypes
- Schatz M, Rosenwasser L. The allergic asthma phenotype. J Allergy Clin Immunol Pract. 2014 Nov-Dec;2(6):645-8; quiz 649. doi: 10.1016/j.jaip.2014.09.004. Epub 2014 Nov 6. PMID: 25439351.
- Pembrey L, Barreto ML, Douwes J, Cooper P, Henderson J, Mpairwe H, Ardura-Garcia C, Chico M, Brooks C, Cruz AA, Elliott AM, Figueiredo CA, Langan SM, Nassanga B, Ring S, Rodrigues L, Pearce N. Understanding asthma phenotypes: the World Asthma Phenotypes (WASP) international collaboration. ERJ Open Res. 2018 Aug 22;4(3):00013-2018. doi: 10.1183/23120541.00013-2018. PMID: 30151371; PMCID: PMC6104297.
- Kuruvilla ME, Lee FE, Lee GB. Understanding Asthma Phenotypes, Endotypes, and Mechanisms of Disease. Clin Rev Allergy Immunol. 2019 Apr;56(2):219-233. doi: 10.1007/s12016-018-8712-1. PMID: 30206782; PMCID: PMC6411459.
- Bourdin A, Brusselle G, Couillard S, Fajt ML, Heaney LG, Israel E, McDowell PJ, Menzies-Gow A, Martin N, Mitchell PD, Petousi N, Quirce S, Schleich F, Pavord ID. Phenotyping of Severe Asthma in the Era of Broad-Acting Anti-Asthma Biologics. J Allergy Clin Immunol Pract. 2024 Apr;12(4):809-823. doi: 10.1016/j.jaip.2024.01.023. Epub 2024 Jan 25. PMID: 38280454.
- AAAAI. The American Academy of Allergy, Asthma & Immunology. 2024. 555 East Wells Street, Suite 1100, Milwaukee, WI 53202-3823, USA. Available at https://www.aaaai.org/. Accessed 1 Jan 2025.
- WHO. World Health Organisation factsheet on asthma: global, regional and national incidences, prevalence and years lived with disability for 328 diseases and injuries for 195 countries 190-2016: a systematic analysis for the Global Burden of Disease Study. Lancet, 2016; 390, 1211-1259.
- Mattiuzzi C, Lippi G. Worldwide asthma epidemiology: insights from the Global Health Data Exchange database. Int Forum Allergy Rhinol. 2020 Jan;10(1):75-80. doi: 10.1002/alr.22464. Epub 2019 Oct 23. PMID: 31645084.
- Balmes J, Becklake M, Blanc P, Henneberger P, Kreiss K, Mapp C, Milton D, Schwartz D, Toren K, Viegi G; Environmental and Occupational Health Assembly, American Thoracic Society. American Thoracic Society Statement: Occupational contribution to the burden of airway disease. Am J Respir Crit Care Med. 2003 Mar 1;167(5):787-97. doi: 10.1164/rccm.167.5.787. PMID: 12598220.
- Torén K, Blanc PD. Asthma caused by occupational exposures is common – a systematic analysis of estimates of the population-attributable fraction. BMC Pulm Med. 2009 Jan 29;9:7. doi: 10.1186/1471-2466-9-7. PMID: 19178702; PMCID: PMC2642762.
- Blanc PD, Annesi-Maesano I, Balmes JR, Cummings KJ, Fishwick D, Miedinger D, Murgia N, Naidoo RN, Reynolds CJ, Sigsgaard T, Torén K, Vinnikov D, Redlich CA. The Occupational Burden of Nonmalignant Respiratory Diseases. An Official American Thoracic Society and European Respiratory Society Statement. Am J Respir Crit Care Med. 2019 Jun 1;199(11):1312-1334. doi: 10.1164/rccm.201904-0717ST. PMID: 31149852; PMCID: PMC6543721.
- Siracusa A, Folletti I, Gerth van Wijk R, Jeebhay MF, Moscato G, Quirce S, Raulf M, Ruëff F, Walusiak-Skorupa J, Whitaker P, Tarlo SM. Occupational anaphylaxis–an EAACI task force consensus statement. Allergy. 2015 Feb;70(2):141-52. doi: 10.1111/all.12541. PMID: 25369880.
- Vandenplas O, Toren K, Blanc PD. Health and socioeconomic impact of work-related asthma. Eur Respir J. 2003 Oct;22(4):689-97. doi: 10.1183/09031936.03.00053203. PMID: 14582924.
- Bakerly ND, Moore VC, Vellore AD, Jaakkola MS, Robertson AS, Burge PS. Fifteen-year trends in occupational asthma: data from the Shield surveillance scheme. Occup Med (Lond). 2008 May;58(3):169-74. doi: 10.1093/occmed/kqn007. Epub 2008 Feb 27. PMID: 18308695.
- Kenyon NJ, Morrisey BM, Schivo M, Albertson TE. Occupational asthma. Clin. Rev. Allergy Immunol. 2012;43, 3-13. DOI: 10.1007/s12016-011-8272-0.
- Lemière C, Boulet LP, Chaboillez S, Forget A, Chiry S, Villeneuve H, Prince P, Maghni K, Kennedy WA, Blais L. Work-exacerbated asthma and occupational asthma: do they really differ? J Allergy Clin Immunol. 2013 Mar;131(3):704-10. doi: 10.1016/j.jaci.2012.08.024. Epub 2012 Oct 8. PMID: 23058644.
- Mapp CE, Boschetto P, Maestrelli P, Fabbri LM. Occupational asthma. Am. J. Respir. Crit. Care Med. 2005;172, 280-305. DOI: 10.1164/rccm.200311-1575SO.
- Feary J, Pinnock H, Cullinan P. Occupational asthma. Br. Med. J. 2016;353, i2658. DOI: 10.1136/bmj.i2658.
- Friedman-Jimenez G, Harrison D, Luo H. Occupational asthma and work-exacerbated asthma. Seminars in respiratory and critical care medicine. 2015;36(3), 388-407. DOI: 10.1055/s-0035-1550157.
- Vandenplas O, Godet J, Hurdubaea L, Rifflart C, Suojalehto H, Walusiak-Skorupa J, Munoz X, Sastre J, Klusackova P, Moore V, Merget R, Talini D, Kirkeleit J, Mason P, Folletti I, Cullinan P, Moscato G, Quirce S, Hoyle J, Sherson D, Kauppi P, Preisser A, Meyer N, de Blay F; European network for the PHenotyping of OCcupational ASthma (E-PHOCAS) investigators. Severe Occupational Asthma: Insights From a Multicenter European Cohort. J Allergy Clin Immunol Pract. 2019 Sep-Oct;7(7):2309-2318.e4. doi: 10.1016/j.jaip.2019.03.017. Epub 2019 Mar 23. PMID: 30914232.
- Alif SM, Benke G, Kromhout H, Abramson MJ, Kogevinas M, Jarvis D, Le Moual N, Dharmage S, Schlünssen V, Torén K, Norback D, Lytras T, Carsin AE, Svanes C, Olivieri M, Dorado-Arenas S, Urrutia I, Pascual Erquicia S, Acke S, Bentouhami H, Wieslander G, Murgia N, Martínez-Moratalla J, Leynaert B, Radon K, Gerlich J, Nowak D, Villani S, Holm M, Verlato G, D’Errico A, Bakke P, Skorge TD, Storaas T, Dahlman-Höglund A, Hellgren J, Miedinger D, Sigsgaard T, Blanc PD, Zock JP. Occupational exposures and incidence of asthma over two decades in the European Community Respiratory Health Survey. Thorax. 2025 Apr 29:thorax-2024-222307. doi: 10.1136/thorax-2024-222307. Epub ahead of print. PMID: 40306950.
- Lemiere C, Chaboillez S, Welman M, Maghni K. Outcome of occupational asthma after removal from exposure: A follow-up study. Can Respir J. 2010 Mar-Apr;17(2):61-6. doi: 10.1155/2010/509807. PMID: 20422061; PMCID: PMC2866225.
- Holsapple MP, Jones D, Kawabata TT, Kimber I, Sarlo K, Selgrade MK, Shah J, Woolhiser MR. Assessing the potential to induce respiratory hypersensitivity. Toxicol Sci. 2006 May;91(1):4-13. doi: 10.1093/toxsci/kfj074. Epub 2005 Dec 9. PMID: 16339788.
- Kimber I, Agius R, Basketter DA, Corsini E, Cullinan P, Dearman RJ, Gimenez-Arnau E, Greenwell L, Hartung T, Kuper F, Maestrelli P, Roggen E, Rovida C; European Centre for the Validation of Alternative Methods. Chemical respiratory allergy: opportunities for hazard identification and characterisation. The report and recommendations of ECVAM workshop 60. Altern Lab Anim. 2007 May;35(2):243-65. doi: 10.1177/0261192907035000212. PMID: 17559314.
- Boverhof DR, Billington R, Gollapudi BB, Hotchkiss JA, Krieger SM, Poole A, Wiescinski CM, Woolhiser MR. Respiratory sensitization and allergy: current research approaches and needs. Toxicol Appl Pharmacol. 2008 Jan 1;226(1):1-13. doi: 10.1016/j.taap.2007.10.008. Epub 2007 Oct 22. PMID: 18023833.
- North CM, Ezendam J, Hotchkiss JA, Maier C, Aoyama K, Enoch S, Goetz A, Graham C, Kimber I, Karjalainen A, Pauluhn J, Roggen EL, Selgrade M, Tarlo SM, Chen CL. Developing a framework for assessing chemical respiratory sensitization: A workshop report. Regul Toxicol Pharmacol. 2016 Oct;80:295-309. doi: 10.1016/j.yrtph.2016.06.006. Epub 2016 Jul 7. PMID: 27396307.
- Chary A, Hennen J, Klein SG, Serchi T, Gutleb AC, Blömeke B. Respiratory sensitization: toxicological point of view on the available assays. Arch Toxicol. 2018 Feb;92(2):803-822. doi: 10.1007/s00204-017-2088-5. Epub 2017 Oct 16. PMID: 29038838.
- Arts J. How to assess respiratory sensitization of low molecular weight chemicals? Int J Hyg Environ Health. 2020 Apr;225:113469. doi: 10.1016/j.ijheh.2020.113469. Epub 2020 Feb 12. PMID: 32058937.
- Scheel J, Krutz NL, Rajagopal R, Sadekar N, Hindle S, Hickey C, Campbell C, Botham P. Use and limitations of clinical data in the identification and classification of low molecular weight chemicals (LMWCs) as respiratory sensitizers: recommendations for improvement. Crit Rev Toxicol. 2025 Jan;55(1):27-54. doi: 10.1080/10408444.2024.2433222. Epub 2025 Jan 9. PMID: 39785825.
- Tarlo SM, Liss GM. Occupational asthma: an approach to diagnosis and management. CMAJ. 2003 Apr 1;168(7):867-71. Erratum in: CMAJ. 2003 Apr 15;168(8):966. PMID: 12668547; PMCID: PMC151995.
- Nicholson PJ, Cullinan P, Taylor AJ, Burge PS, Boyle C. Evidence based guidelines for the prevention, identification, and management of occupational asthma. Occup Environ Med. 2005 May;62(5):290-9. doi: 10.1136/oem.2004.016287. PMID: 15837849; PMCID: PMC1741012.
- Aasen TB, Burge PS, Henneberger PK, Schlünssen V, Baur X; ERS Task Force on the Management of Work-related Asthma; EOM Society. Diagnostic approach in cases with suspected work-related asthma. J Occup Med Toxicol. 2013 Jun 14;8(1):17. doi: 10.1186/1745-6673-8-17. PMID: 23768266; PMCID: PMC3716794.
- Tarlo SM. The role and interpretation of specific inhalation challenges in the diagnosis of occupational asthma. Can Respir J. 2015 Nov-Dec;22(6):322-3. doi: 10.1155/2015/591010. PMID: 26641806; PMCID: PMC4676394.
- Preisser, A,M, Koschelm, D, Merget, R, Nowak, D, Raulf, M, Heidrich, J. Workplace-related inhalation test – Specific inhalation challenge: S2k Guideline of the German Society for Occupational and Environmental Medicine e.V. (DGAUM), the German Society for Pneumology and Respiratory Medicine e.V. (DGP) and the German Society for Allergology and Clinical Immunology e.V. (DGAKI). Allergol Select. 2021;315-334. doi: 10.5414/ALX02280E. PMID: 34646977; PMCID: PMC8507414.
- Baur X, Sigsgaard T, Aasen TB, Burge PS, Heederik D, Henneberger P, Maestrelli P, Rooyackers J, Schlünssen V, Vandenplas O, Wilken D; ERS Task Force on the Management of Work-related Asthma. Guidelines for the management of work-related asthma. Eur Respir J. 2012 Mar;39(3):529-45. doi: 10.1183/09031936.00096111. Erratum in: Eur Respir J. 2012 Jun;39(6):1553. PMID: 22379148.
- Johanson G. Are asthmatics more sensitive to irritants? Int J Hyg Environ Health. 2020 May;22(6):113488. doi: 10.1016/j.ijheh.2020.113488. Epub 2020 Feb 20. PMID: 32088597.
- Pemberton MA, Schmitt B, Hindle S, Flueckiger A, Kreuzer K. The role of exposure determinations in the exclusion of non-specific irritation from Specific Inhalation Challenge Tests. 2025. Submitted to Regulatory Toxicology and Pharmacology.
- Pemberton MA, Kimber I. Classification of chemicals as respiratory allergens based on human data: Requirements and practical considerations. Regul Toxicol Pharmacol. 2021 Jul;123:104925. doi: 10.1016/j.yrtph.2021.104925. Epub 2021 Apr 5. PMID: 33831493.
- Chhabra SK, Ojha UC. Late asthmatic response in exercise-induced asthma. Ann Allergy Asthma Immunol. 1998 Apr;80(4):323-7. doi: 10.1016/S1081-1206(10)62977-8. PMID: 9564982.
- Koh YY, Jeong JH, Jin SM, Kim CK, Min KU. The occurrence of late asthmatic response to exercise after allergen challenge. Ann Allergy Asthma Immunol. 1998 Oct;81(4):366-72. doi: 10.1016/S1081-1206(10)63130-4. PMID: 9809502.
- ECHA. Guidance on the Application of the CLP Criteria. Part 3: Health Hazards Guidance to Regulation (EC) No 1272/2008 on classification, labelling and packaging (CLP) of substances and mixtures Version 5.0. 2024. Available at https://echa.europa.eu/documents/10162/2324906/clp_part3_en.pdf/42e0397a-73f2-0583-958f-3830928e1604?t=1730718832043
- ECHA (European Chemicals Agency), Guidance on the application of the CLP Criteria Guidance to Regulation (EC) No 1272/2008 on classification, labelling and packaging (CLP) of substances and mixtures. Part 3, Health hazards, European Chemicals Agency, 2024, Available at https://data.europa.eu/doi/10.2823/9350054
- Pemberton MA, Kimber I. Methyl methacrylate and respiratory sensitisation: a comprehensive review. Crit Rev Toxicol. 2022 Feb;52(2):139-166. doi: 10.1080/10408444.2022.2064267. Epub 2022 May 24. PMID: 35607993.
- Pemberton MA, Kreuzer K, Kimber I. Challenges in the classification of chemical respiratory allergens based on human data: Case studies of 2-hydroxyethylmethacrylate (HEMA) and 2-hydroxypropylmethacrylate (HPMA). Regul Toxicol Pharmacol. 2023 Jun;141:105404. doi: 10.1016/j.yrtph.2023.105404. Epub 2023 Apr 26. PMID: 37105297.




