HRCT Lung Findings in Connective Tissue Diseases
A Scoping Literature Review of Lung HRCT in Connective Tissue Disease: Descriptions and Prediction of Mortality
Olga Morgan, Thasia Woodworth¹, Raymond Paria, Harsh Agrawal², Aishwarya Bollampally, Kevin Kafaja³, Adla B Hassan⁴, Nihal Fathi⁵, Daniel E Furst⁶
- Leading Edge Clinical Research
- Division of Cardiovascular Medicine, Department of Medicine, University of California, San Francisco
- Student in Organized Health Care Education/Training Program, Pomona, CA
- Internal Medicine Department, Arabian Gulf University Consultant Rheumatologist King Abdullah Medical City Bahrain
- Head Faculty of Medicine, Assiut University
- Prof. of Rheumatology (emeritus) at University of California Los Angeles, Prof. of Rheumatology at the University of Washington, Seattle Washington, Prof. of Rheumatology (research) at the University of Florence, Florence Italy
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION: Morgan, O., Woodworth, T., et al., 2025. A Scoping Literature Review of lung HRCT in Connective Tissue Disease. Descriptions and Prediction of Mortality. Medical Research Archives, [online] 13(5).
https://doi.org/10.18103/mra.v13i5.6602
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i5.6602
ISSN 2375-1924
Abstract
Background and Objectives: This scoping literature review is intended to describe the characteristic HRCT lung findings related to interstitial lung disease (ILD) which occur in: dermatomyositis/polymyositis (DM/PM), rheumatoid arthritis (RA), Sjogren’s Syndrome (SjS), systemic lupus erythematosus (SLE) and systemic sclerosis (SSc). In addition, we examined whether HRCT features in these diseases could predict mortality.
Methods: We reviewed the medical literature from PubMed, EMBASE and Cochran reviews from 1993 to 2021, in DM/PM, RA, SJS, SLE and SSc. The articles were extracted according to defined inclusion/exclusion criteria using standardized forms with double extraction of 20% of the articles. Analysis included descriptive statistics plus tabulations for comparisons of frequency, prevalence, and prediction of mortality.
Results: Of 478 publications, 110 studies were included. HRCT features consistent with ILD were reported with variable prevalence (often depending on study design) in RA (15-43%), SjS (~50%), DM/PM (~60%), SSc (~53%) and SLE (53%) patients. Usual interstitial pneumonia (UIP) frequently predicted mortality as did, less commonly, honeycombing and increasing fibrosis. Other non-HRCT factors such as decreased FVC, DLCO, increased age, smoking and male gender were predictors of mortality in RA and SSc. Additionally, predictors of mortality were: UIP in RA; increased skin involvement in SSC, diffuse alveolar hemorrhage in SLE (although rare) and MDA5 in DM, when available.
Conclusion: ILD and increased fibrosis, by HRCT, is relatively common in these connective tissue diseases and plays a significant role in mortality. Specific occurrences such as UIP (RA), increased skin involvement (SSC), diffuse alveolar hemorrhage (SLE) and MDA 5 (DM) also affected mortality.
Keywords
HRCT, Lung, RA, DM/PM, Sjogren’s, SLE, SSc, description, prognosis, scoping review
Introduction
Collagen vascular diseases (CVD) that show features of interstitial lung disease (ILD) include rheumatoid arthritis (RA), systemic sclerosis (SSc) Sjogren’s syndrome (SjS), systemic lupus erythematosus (SLE), polymyositis and dermatomyositis (DM/PM). There can be changes in pulmonary function, quality of life, function, survival, and imaging when considering the lung in these diseases.
We examined the use of High-Resolution Computerized Tomography (HRCT) imaging for diagnosis/screening and monitoring of CVD-ILD as it is a reasonable reflection of lung anatomy, and it is both noninvasive and sensitive. There was previously hesitation with respect to using this modality because a significant amount of radiation was necessary for imaging. However, lower radiation protocols have alleviated this concern to a large degree. While HRCT was the sole imaging modality used in this review, other modalities were also considered.
MRI was considered in connective tissue diseases because it does not expose patients to radiation. MRI correlates well with HRCT of the lungs (r=0.85) in small numbers of patients while extent of involvement on MRI is less than HRCT. Agreement between the two methods in SSc is good (kappa=0.73) 1. Some authors in fact, feel that MRI is more sensitive than HRCT, although it is less specific. In a 24 patient SSc study, sensitivity of MRI vs. HRCT was 100%, while specificity was 60%. However, the expense of MRI makes it much less feasible.
Ultrasound of the lungs, too, may be a measurement modality in the future but it is not yet sufficiently validated for use, as its sensitivity, specificity, accuracy, ability to examine change and minimally important clinical difference are not yet fully delineated.
As lung damage, logically correlated with mortality, the influence of HRCT and mortality was also examined. HRCT of the lungs was associated with mortality in DMS/PMS, rheumatoid arthritis, Sjogren’s syndrome and systemic sclerosis. However, subsets of HRCT damage such as UIP and other factors such as age and disease activity affected mortality.
In this review, the HRCT findings in DM/PM, RA, SJS, SLE and SSc were described and the contribution of HRCT of the lungs to mortality in these diseases was examined.
Methods
This was a scoping literature review of medical literature from PubMed, EMBASE and Cochrane Reviews (1993 to 2021) analyzing HRCT changes in the lung in patients with DM/PM, RA, SJS, SLE, and SSc. A case report form was developed and used to facilitate data extraction consistency.
Inclusion criteria were the following: reports of studies in English, conducted in patients >=18 years of age, containing pulmonary HRCT data, explicitly in at least one of DM/PM, RA, SjS, SLE or SSc, data separable for extraction if more than one disease was included. Exclusion criteria included non-English, exclusively animal or in vitro data and lack of separability into the diseases of interest. We accepted the diagnoses as reported, and explicit description of the HRCT technique was not required. Reviews were not included although the bibliographies were examined for articles not found in the initial literature review.
To ensure consistency, 20% of the articles were independently extracted by 2 individuals. 80% consistency was found. We used the EPHPP to rate articles. HRCT-ILD patterns of interest included usual interstitial pneumonitis (UIP), nonspecific interstitial pneumonia (NSIP) and their components. Predominant findings of UIP were characterized by patchy, ground glass appearance, reticulation, fibrosis and bronchiectasis plus honeycombing with basal preponderance. NSIP was defined by more uniform ground glass opacity, reticulation, fibrosis, +/- bronchiectasis but without honeycombing. It should be noted that these definitions were inconsistent in the articles and often missing, with only UIP and NSIP reported. Lymphocytic interstitial pneumonia (LIP) is defined by centrilobular nodules, ground-glass attenuation, septal and bronchovascular thickening and thin-walled cysts. Organizing pneumonia (OP) findings included patchy bilateral consolidation, ground-glass opacification, perilobular opacity, nodules or masses, bronchial wall thickening, bronchial dilatation, mediastinal lymphadenopathy and pleural effusion.
Our study additionally examined whether HRCT ILD lung changes were associated with patient morbidity/mortality. Because there was marked heterogeneity, we tabulated results for descriptive statistics and visually compared results.
Results
A total of 478 publications were identified, of which 104 met the selection criteria. HRCT clinical data were extracted for risk factors for ILD development, and/or prediction of mortality.
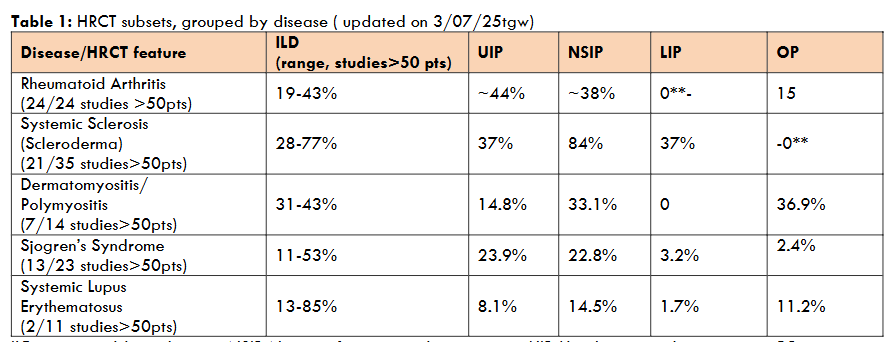
| Disease/HRCT feature | ILD (range, studies>50 pts) | UIP | NSIP | LIP | OP |
|---|---|---|---|---|---|
| Rheumatoid Arthritis (24/24 studies >50pts) | 19-43% | ~44% | ~38% | 0 | 15 |
| Systemic Sclerosis (Scleroderma) (21/35 studies>50pts) | 28-77% | 37% | 84% | 37% | -0 |
| Dermatomyositis/Polymyositis (7/14 studies>50pts) | 31-43% | 14.8% | 33.1% | 0 | 36.9% |
| Sjogren’s Syndrome (13/23 studies>50pts) | 11-53% | 23.9% | 22.8% | 3.2% | 2.4% |
| Systemic Lupus Erythematosus (2/11 studies>50pts) | 13-85% | 8.1% | 14.5% | 1.7% | 11.2% |
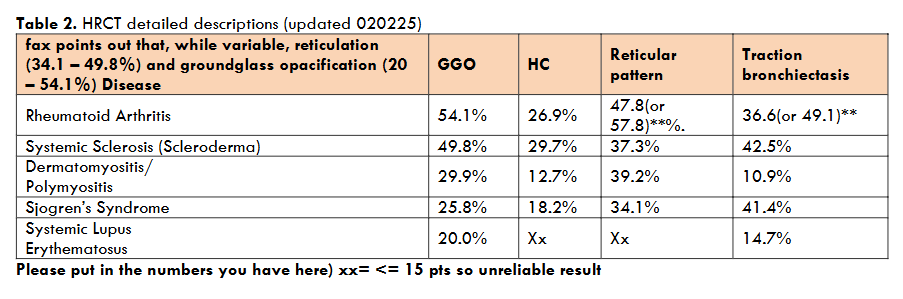
| Disease | GGO | HC | Reticular pattern | Traction bronchiectasis |
|---|---|---|---|---|
| Rheumatoid Arthritis | 54.1% | 26.9% | 47.8(or 57.8)**% | 36.6(or 49.1)** |
| Systemic Sclerosis (Scleroderma) | 49.8% | 29.7% | 37.3% | 42.5% |
| Dermatomyositis/Polymyositis | 29.9% | 12.7% | 39.2% | 10.9% |
| Sjogren’s Syndrome | 25.8% | 18.2% | 34.1% | 41.4% |
| Systemic Lupus Erythematosus | 20.0% | Xx | Xx | 14.7% |
| Factors affecting mortality | PM/DM | RA | SJS | SLE | SSc |
|---|---|---|---|---|---|
| ILD | + | + | |||
| NSIP | + | + | |||
| UIP | ++ | + | + | ||
| DAH | + | ||||
| GGO | + | ||||
| Fibrosis / reticulation extensive | + | + | + | ||
| HC | + | ||||
| Lower Lobe Consolidation | + | ||||
| Traction Bronchiectasis | + |

Dermatomyositis/polymyositis (DM/PM) 24 (out of 88) studies with DM/PM patients were analyzed in our review, including 1357 patients. ILD was found in approximately 31-43% (across 7 of 14 studies with>50 pts), Kameda and Takeuchi’s 30-50% prevalence. Only four studies looked at specific ILD patterns, finding NSIP (33.1%), organizing pneumonia (OP) (36.9%), then UIP (14.8%). Also, presence of ground glass opacity (29.9%), reticular pattern (39.2%) and fibrosis (36%) were common findings. A low prevalence of honeycombing was observed (12.7%). Among 4 studies concentrating on the HRCT among DM/PM patients, irregular linear patterns, consolidation and ground glass seemed the predominant patterns. Two of 30 patients in one study had lung cancer, introducing the need for caution when examining the HRCT.
A study by Kotani et al. found that ground glass opacities (GGO) ≥ 25% of the right middle lobe was the best cut-off value for a poor prognosis (sensitivity: 85.7%, specificity: 85.7%), and the survival rate after 24 weeks was much lower in these patients (survival rate: 0.0%) than in those with < 25%GG (92.9%) (p < 0.0001). Four studies examined predictors of survival. Two studies highlighted HRCT (ground glass, consolidation, UIP), and 2 studies also included function tests in the survival prediction while 4 studies included a relatively large range of blood tests (anti-MDA 5, anti-CADM110, decreased C3/4/8 T cells, increased CD19 cells, our YKL – 40, anti-RO 52, CEA, Ferritin), out of which only anti-MDA 5 was found in more than one study. Most prognostic and predictive studies combined serologies or laboratory tests with HRCT. Anti-CADM-140/MDA5 autoantibody titer predicted worse disease outcome in patients with dermatomyositis. Several studies with clinically amyopathic dermatomyositis showed that an HRCT score, when associated with extensive interstitial lung disease, significantly increased mortality. Additionally, lower lobe consolidation/ground glass attenuation on HRCT may predict worse survival in these patients.
For example, Tanizawa et al. showed that, in clinically amyopathic dermatomyositis, lower lobe consolidation/GG pattern (odds ratio, 23.1; P < 0.02) and the presence of MDA5 (odds ratio, 14.1; P < 0.03) were independent predictors of 90-day mortality. This was supported by Hallowell et al., for MDA5, as well as anti-JO-1 and anti-Ro52, especially in the presence of CCP, RF, SSA, and SSB antibodies.
Summary: In studies of HRCT lung findings in DM/PM, the most common findings were irregular linear patterns, ground glass, and consolidation. The HRCT plus serological markers, particularly anti-MDA5, predict poorer survival in these patients.
Rheumatoid Arthritis (RA)
Twenty-four (of 96) studies in RA patients were analyzed to examine the role of HRCT to diagnose and assess prevalence of ILD, as well as to examine ILD impact on survival. All studies described features (ground glass opacities, honeycombing, reticular pattern, traction bronchiectasis, or/and patterns of ILD on HRCT (UIP > NSIP > OP). Twelve studies (>50 patients) provided prevalence data, using retrospective chart reviews with a prevalence of 19-43%. Thirteen studies examined only HRCT-ILD features relative to clinical characteristics including morbidity/mortality, in retrospective ILD patient studies of various types. Specific HRCT patterns and/or features were described variously, either as UIP (~44%), NSIP (~35%), or less commonly as organizing pneumonia (OP) in 15%. In some studies, neither UIP nor NSIP were noted although the components were: ground glass opacity (GGO) (54.1%), bronchial wall thickening (50.3%), reticular pattern (57.8%), honeycombing (26.9%) and traction bronchiectasis (38.6%).
The presence of UIP was significantly associated with poor prognosis and survival in 8 studies. For example, during the 10.4 (95%CI:8.2-12.9) year follow-up period in a study by Solomon et al. in 108 UIP RA and 29 NSIP RA patients, 44% of the RA-UIP patients died vs. 24% who had RA-NSIP (Chi-squared 3.59, p=0.05) (median survival 10.18 years versus 13.62 years; log rank p=0.02).
Three studies demonstrated that the extent of lung disease or fibrosis on HRCT was associated with mortality, and two studies found that honeycombing, and traction bronchiectasis (features of UIP) were associated with patient mortality. Further, Nurmi et al. described HRCT findings in 60 RA-ILD patients and showed that the extent of reticulation (hazard ratio 1.14), traction bronchiectasis (HR: 1.18), and architectural distortion (HR 1.09) – all associated with UIP – predicted decreased survival. Yunt et al. showed decreased survival in the 23 of 158 RA-ILD patients with UIP.
Additional significant predictors of mortality on multivariate analyses in other studies were presence of emphysema, a decrease of FVC (% predicted) from baseline by >=10%, male gender, and smoking history.
Summary: Across all studies, ILD was identified in ~31% (range 19-43%) of RA patients. In most studies, UIP, characterized by ground glass opacity, bronchial wall thickening, reticular pattern, often with traction bronchiectasis and honeycombing, was more common than NSIP. UIP was significantly associated with poor prognosis and lowered survival. Additional significant predictors of mortality on multivariate analysis were decreased FVC from baseline by >=10% predicted, age >60, male gender, and smoking history.
Sjogren’s syndrome (SJS)
Thirteen studies with Sjogren’s syndrome (SJS) patients were analyzed, including 2810 patients. ILD was found in 49.8%. In 617 patients NSIP was demonstrated in 22.8% (mean), and UIP in a mean of 23.9% studies. An outlier with only 8 patients had 44.4% UIP. In the group of 12 studies, components were as follows: traction bronchiectasis (41.4%), reticular pattern (34.1%), honeycombing (18.2%) and ground glass opacity (25.8%). Lymphocytic Interstitial pneumonia (LIP), while very uncommon in our review (3.2%), is more common in SJS than the other reviewed connective tissue diseases (0-1.7%). It is characterized by extensive ground glass opacities, inter-lobular septal thickening and scattered thin-walled cysts. Although useful to increase the suspicion of Sjogren’s, this description also applies to other connective tissue diseases, and non-connective tissue diseases such as HIV and drug reactions. Separately and independently, diffuse alveolar hemorrhage was found in 3.2%.
Extent of HRCT lung involvement, presence of UIP patterns, and presence of honeycombing were significant factors in predicting mortality in Sjogren’s syndrome. Ming-Han et al. found that a high, semi-subjective, HRCT score (range 0-18, high/severe>=13) was significantly and independently associated with higher mortality, as was “low” FEV1 and FVC<60%. Kim et al found that UIP, increased age and increased ESR predicted mortality.
In one study examining the prognostic significance of HRCT findings in Sjogren’s Syndrome lung disease, verified by histology, a low PaO2 and microscopic honeycombing were independently associated with poor survival.
Summary: NSIP (22.8%) or UIP (23.9%) or LIP (3.2%) patterns were seen in this disease in the available 12 subset-analyzed studies. Although uncommon, lymphocytic interstitial pneumonia was more common in SJS than in the other connective tissue diseases, as were thin-walled cysts. LIP and thin-walled cysts should make one consider Sjogren’s Syndrome.
Systemic Lupus Erythematosus (SLE)
Sixteen (out of 39) studies with SLE patients, a total of 269 patients, met our inclusion criteria and were analyzed. SLE-ILD was found in 53%. Only two studies meeting our inclusion criteria included ILD patterns. NSIP was found in 14.5%, UIP in 8.1% and obliterating pneumonia in 11.2%. Pleural effusions (26.4%) were found relatively more frequently in SLE than other connective tissue diseases. In some cases, the number of patients was so small (e.g. 10 patients with honeycombing – “47.6%.”) that percentage results, while included here for completeness, should be viewed with suspicion.
Sant et al. concluded that HRCT had excellent ability to detect lung changes in patients despite absence of symptoms, and normal pulmonary function tests. In Fenlon et al, HRCT abnormalities were identified in 24 patients (70%), while pulmonary function abnormalities were present in only 14 patients (41%), and the plain chest radiograph was abnormal in only 8 patients (24%). It should be pointed out that diseases other than SLE, such as infection or renal disease, could also have explained the HRCT appearance of ILD.
A necropsy series of 90 SLE patients showed that interstitial lung disease was always secondary to other causes (e.g. tuberculosis, infection, renal disease). At necropsy, as expected, there were more pathological findings then would be seen on HRCT. Pleuropulmonary involvement was seen in 97.8% of 90 autopsies, pleuritis in 77.8%, bacterial infections in 57.8%, alveolar hemorrhage in 25.6%. Writing and pulmonary arterial hypertension in 4%.
Clinically significant alveolar hemorrhage by HRCT is most commonly found in SLE and vasculitis among the rheumatic diseases but is rare. The combination of bilateral diffuse, dependent ground glass opacity, airspace bronchograms and the superposition of ground glass and reticulation (called “crazy paving”) are highly suggestive of diffuse hemorrhage on HRCT. Mortality in diffuse alveolar hemorrhage was 28.6% and 61.9% in two studies.
The only study found which attempted to examine predictors of survival was by Enomoto et al in 55 patients. They identified that NSIP, NSIP+OP, and presence of fibrosis on HRCT were significant predictors of poor survival. Other non-HRCT findings that increased mortality were history of smoking, thrombocytopenia, positive Anti-ds DNA antibody, and Serum KL-6 antigen.
Summary: It appears that HRCT abnormalities are found frequently (55.7%) while symptoms, chest radiographs and pulmonary function tests are abnormal in far fewer SLE patients. Pleural effusions and infiltrates/consolidation were the most common HRCT findings. Rare diffuse alveolar hemorrhage is the most important predictor of mortality.
Systemic Sclerosis (SSc)
Thirty-two (out of 170) studies with SSc patients from 1993 to 2021 were reviewed and ILD was specifically examined in 13 of those studies (2534 patients). ILD was found in a mean of 53% of these SSc patients. Only five studies looked at specific ILD patterns. They showed a significant predominance of NSIP in 102 of 121 ILD patients. Other common chest CT characteristics of SSc-ILD included ground glass opacity (49.8%), reticular pattern (37.3%), honeycombing (29.7%) and traction bronchiectasis (42.5%). Studies that looked at prognosis showed that increased lung disease or baseline lung fibrosis, UIP, and honeycombing were associated with greater mortality.
A 160 SSc patient study showed that mortality correlated with the extent of baseline lung fibrosis on HRCT and low baseline FVC and FVC decline. A study by Sánchez-Cano et al found that SSc-associated mortality and ILD-related mortality were highest in diffuse cutaneous SSc (dcSSc) patients but that it was actually higher than non-SSc in all three SSc subgroups than controls.
Quantitation of the lung HRCT is a newer technique which predicts progression and outcome. For example, a 172-patient study demonstrated that the presence of >20% fibrosis, ground glass attenuation, reticulation and/or honeycombing was associated with a 3-fold increased risk of deterioration or death in SSc-ILD. Similar findings were noted by Goh et al. A different, operator-independent computerized quantitation of ILD predicted mortality in the study by Ariani et al and yet another system review showed response to treatment but did not address mortality. The most reliable, sensitive, and easiest to use algorithm still needs to be decided but quantitative lung HRCT may turn out to be an improved method by which to use lung HRCT for predicting mortality.
Summary: ILD by HRCT occurs in more than half of SSc patients. Both NSIP and UIP occur in SSC interstitial lung disease, although NSIP predominates. Fibrosis and ILD by HRCT predicted mortality in multiple studies, although other factors (e.g. male sex, smoking and low FVC) contribute significantly as well. Quantitative lung HRCT represents a new potentially useful application of HRCT.
Discussion
Our literature review of HRCT of the lungs included dermatomyositis/polymyositis, rheumatoid arthritis, Sjogren’s syndrome, SLE, and SSc. It allowed cross-disease qualitative comparisons of HRCT findings and examination of prognostic factors which included HRCT. While HRCT was the sole imaging modality used in this review, other modalities were also considered. As noted above, MRI correlates well with HRCT of the lungs. Further while MRI is more sensitive than HRCT, it is less specific. In one SSc study, sensitivity of MRI vs. HRCT was 100%, while specificity was 60%. Also, MRI is more expensive than MRI’s.
Also, as noted above, ultrasound of the lungs, while being increasingly used, needs validation in the lungs before it goes into common use. Further, it remains operator dependent, another source of variability.
A Taiwanese nationwide cohort study examined the incidence risk of ILD in those newly diagnosed with systemic autoimmune rheumatic diseases and found that the incidence risk of ILD was greatest among patients with SSc, followed by DM, PM, RA, and SLE. This differed somewhat from our study where the prevalence of ILD was highly variable. Multivariable analyses in the Taiwanese study showed that the risk of ILD was increased among patients with SSc, DM, PM, SLE, Sjogren’s, or RA.
A study by Kim et al supported these results, with NSIP more prevalent in SSc and PM/DM, and less so in Sjogren’s or SLE (NSIP was still more prevalent). Fang et al. and Flaherty et al., in their studies, addressed the controversy between NSIP and UIP. They pointed out that UIP is not always an accurate diagnosis when NSIP and UIP coexist, and NSIP can have regions of UIP. Therefore, a more standardized approach such as quantitative HRCT might give more definitive results.
We also examined the descriptions of HRCT features in detail, independent of the disease setting. Table 2 points out that groundglass opacification and reticulation, while somewhat variable are common enough across the diseases so they cannot be used to differentiate among these connective tissue diseases. At the same time, table 2 demonstrates that some patterns, such as traction bronchiectasis seem to be most common in rheumatoid arthritis, Sjogren’s syndrome, and systemic sclerosis. These detailed descriptions also pointed out some radiographic findings that were relatively specific, although sometimes rare, among these connective tissue diseases. A finding lymphocytic interstitial pneumonia, for example, should make one suspect the possibility that the patient has Sjogren’s syndrome or SLE. The presence of pleuritis might make one consider SLE. Organizing pneumonia was by far the most common in dermatomyositis/polymyositis. Diffuse alveolar hemorrhage was found predominantly in Sjogren’s and SLE among the diseases we considered and should make one consider these diseases.
We examined whether HRCT features of the lung could be applied to support predictions of mortality. In 107 patients with dermatomyositis/polymyositis, UIP was one component of predicting mortality; as mortality was also associated with older age, decreased FVC and DLCO ILD. Among 156 rheumatoid arthritis patients followed over 10.2 years, 25.6 % died and mortality was predicted by UIP as well as pleural effusions. In SSc, mortality was predicted by age, CRP, FVC and UIP (hazard ratio for UIP: 4.58) with a 5-year survival of 81.1%. UIP prognosticates mortality in RA, Sjogren’s syndrome and SSc, although often also associated with other factors, gender. Oftentimes, PFTs can be used as an additional tool to better evaluate lung involvement and assist in prognosis. In SSc, the combination of fibrosis on HRCT plus decreasing FVC is a logical and prognostic finding. In SLE, NSIP associates with poor prognosis and, alveolar hemorrhage alone also signals a very poor prognosis.
The lower half of table 3 indicates the details of the HRCT which were associated with mortality. On careful examination, however, these details generally supported the subsetting of the upper half of the table. For example, UIP correlated with mortality in RA as did honeycombing, extensive fibrosis and bronchiectasis; these latter three details are all elements of UIP. As another example in SLE, details correlated ground glass opacities and extensive fibrosis with mortality, and these are elements of NSIP.
Winstone et al. conducted a systematic review of predictors of mortality in scleroderma ILD. That study supported ours, although our study included more studies and more patients. HRCT (specifically the extent of fibrosis and presence of honeycombing) predicted mortality, while age, low FVC, low DLCO and male gender were also predictors. A review by Walsh et al. concluded traction bronchiectasis and the extent of honeycombing (associated with UIP), together with DLCO<45%, in CTD-ILD, were associated with mortality.
A strength of our review is that it examined the HRCT of the lung across five connective tissue diseases and sought to go beyond a description of HRCTs of the lung to use them for prognosis. Examining across these five connective tissue diseases also allowed some qualitative comparisons.
There are also limitations. We chose to examine the literature from 1993 on as there had been SLRs and meta-analyses prior to that date. This scoping literature review faced challenges typical of such an approach. Including a limited number of relevant studies. Also, diagnoses lacked uniformity, HRCT instruments varied widely, and HRCT methods and definitions inconsistent, often poorly described, and changed over time. Clearly, more standardization and quantitation of the HRCT of the chest are needed. Also, given the highly variable data, a meta-analysis was not possible or appropriate. In the future, when the HRCT quantitative methods are standardized, a meta-analysis may yet be possible. Finally, we examined mortality rather than worsening ILD in our predictive examination as we thought that mortality was both more important and a more solid endpoint.
Conclusion
ILD/increased fibrosis, by HRCT, is relatively common in DM/PM, RA, SJS, SLE, and SSc and plays a significant role in mortality. Specific occurrences such as UIP (RA), increased skin involvement (SSC), diffuse alveolar hemorrhage (SLE) and MDA5 (DM) also affected mortality.
Conflict of interest:
None
Acknowledgement:
Thanks to Claudia Real and Gabriel Valdivia for their invaluable help in completing this Manuscript.
Daniel E Furst – University of Washington, Seattle, Washington; University of Florence, Florence, Italy; University of California Los Angeles, Los Angeles, California
Degree(s) Daniel E Furst – MD, FACR, MACR
References
- Castelino FV, Varga J. Interstitial lung disease in connective tissue diseases: evolving concepts of pathogenesis and management. Arthritis Res Ther. 2010;12(4):213. doi:10.1186/ar3097
- Raghu G, Remy-Jardin M, Myers JL, et al. Diagnosis of Idiopathic Pulmonary Fibrosis. An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am J Respir Crit Care Med. Sep 1 2018;198(5):e44-e68. doi:10.1164/rccm.201807-1255ST
- Hallowell RW, Ascherman DP, Danoff SK. Pulmonary manifestations of polymyositis/dermatomyositis. Semin Respir Crit Care Med. Apr 2014;35(2):239-48. doi:10.1055/s-0034-1371528
- Fathi M, Vikgren J, Boijsen M, et al. Interstitial lung disease in polymyositis and dermatomyositis: longitudinal evaluation by pulmonary function and radiology. Arthritis Rheum. May 15 2008;59(5):677-85. doi:10.1002/art.23571
- Kowal-Bielecka O, Landewe R, Avouac J, et al. EULAR recommendations for the treatment of systemic sclerosis: a report from the EULAR Scleroderma Trials and Research group (EUSTAR). Ann Rheum Dis. May 2009;68(5):620-8. doi:10.1136/ard.2008.096677
- Matcham F, Scott IC, Rayner L, et al. The impact of rheumatoid arthritis on quality-of-life assessed using the SF-36: a systematic review and meta-analysis. Semin Arthritis Rheum. Oct 2014;44(2):123-30. doi:10.1016/j.semarthrit.2014.05.001
- Ohno Y, Koyama H, Yoshikawa T, Seki S. State-of-the-Art Imaging of the Lung for Connective Tissue Disease (CTD). Curr Rheumatol Rep. Dec 2015;17(12):69. doi:10.1007/s11926-015-0546-8
- Winklehner A, Berger N, Maurer B, Distler O, Alkadhi H, Frauenfelder T. Screening for interstitial lung disease in systemic sclerosis: the diagnostic accuracy of HRCT image series with high increment and reduced number of slices. Ann Rheum Dis. Apr 2012;71(4):549-52. doi:10.1136/annrheumdis-2011-200564
- Prior JA, Jordan KP, Kadam UT. Variations in patient-reported physical health between cardiac and musculoskeletal diseases: systematic review and meta-analysis of population-based studies. Health Qual Life Outcomes. May 30 2015;13:71. doi:10.1186/s12955-015-0265-x
- Gobi K, Arunachalam VK, Varatharajaperumal RK, Cherian M, Periaswamy G, Rajesh S. The role of ultra-low-dose computed tomography in the detection of pulmonary pathologies: a prospective observational study. Pol J Radiol. 2022;87:e597-e605. doi:10.5114/pjr.2022.121433
- Nguyen-Kim TDL, Maurer B, Suliman YA, Morsbach F, Distler O, Frauenfelder T. The impact of slice-reduced computed tomography on histogram-based densitometry assessment of lung fibrosis in patients with systemic sclerosis. J Thorac Dis. Apr 2018;10(4):2142-2152. doi:10.21037/jtd.2018.04.39
- Bastos AL, Correa RA, Ferreira GA. Tomography patterns of lung disease in systemic sclerosis. Radiol Bras. Sep-Oct 2016;49(5):316-321. doi:10.1590/0100-3984.2015.0116
- Milisenda JC, Selva-O’Callaghan A, Grau JM. The diagnosis and classification of polymyositis. J Autoimmun. Feb-Mar 2014;48-49:118-21. doi:10.1016/j.jaut.2014.01.025
- Sasaki H, Kohsaka H. Current diagnosis and treatment of polymyositis and dermatomyositis. Mod Rheumatol. Nov 2018;28(6):913-921. doi:10.1080/14397595.2018.1467257
- Findlay AR, Goyal NA, Mozaffar T. An overview of polymyositis and dermatomyositis. Muscle Nerve. May 2015;51(5):638-56. doi:10.1002/mus.24566
- Li H, Xiong Z, Liu J, Li Y, Zhou B. [Manifestations of the connective tissue associated interstitial lung disease under high resolution computed tomography]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. Aug 28 2017;42(8):934-939. doi:10.11817/j.issn.1672-7347.2017.08.010
- Materna O, Koubsky K, Padr R, Janousek J. Major left ventricular thrombi in an adolescent with COVID-19-associated inflammatory syndrome. Eur Heart J. Aug 31 2021;42(33):3207. doi:10.1093/eurheartj/ehab165
- Yamanaka Y, Baba T, Hagiwara E, et al. Radiological images of interstitial pneumonia in mixed connective tissue disease compared with scleroderma and polymyositis/dermatomyositis. Eur J Radiol. Oct 2018;107:26-32. doi:10.1016/j.ejrad.2018.08.005
- Young A, Vummidi D, Visovatti S, et al. Prevalence, Treatment, and Outcomes of Coexistent Pulmonary Hypertension and Interstitial Lung Disease in Systemic Sclerosis. Arthritis Rheumatol. Aug 2019;71(8):1339-1349. doi:10.1002/art.40862
- Moore OA, Proudman SM, Goh N, et al. Quantifying change in pulmonary function as a prognostic marker in systemic sclerosis-related interstitial lung disease. Clin Exp Rheumatol. Jul-Aug 2015;33(4 Suppl 91):S111-6.
- Marie I, Hatron PY, Dominique S, Cherin P, Mouthon L, Menard JF. Short-term and long-term outcomes of interstitial lung disease in polymyositis and dermatomyositis: a series of 107 patients. Arthritis Rheum. Nov 2011;63(11):3439-47. doi:10.1002/art.30513
- Zamora AC, Hoskote SS, Abascal-Bolado B, et al. Clinical features and outcomes of interstitial lung disease in anti-Jo-1 positive antisynthetase syndrome. Respir Med. Sep 2016;118:39-45. doi:10.1016/j.rmed.2016.07.009
- Tanizawa K, Handa T, Nakashima R, et al. The prognostic value of HRCT in myositis-associated interstitial lung disease. Respir Med. May 2013;107(5):745-52. doi:10.1016/j.rmed.2013.01.014
- Zhang PL, Yang HX, Zhang LN, et al. [Value of serum YKL-40 in the diagnosis of anti-MDA5-positive patients with dermatomyositis complicated with severe pulmonary injury]. Beijing Da Xue Xue Bao Yi Xue Ban. Dec 18 2021;53(6):1055-1060. doi:10.19723/j.issn.1671-167X.2021.06.008
- Vojinovic T, Cavazzana I, Ceruti P, et al. Predictive Features and Clinical Presentation of Interstitial Lung Disease in Inflammatory Myositis. Clin Rev Allergy Immunol. Feb 2021;60(1):87-94. doi:10.1007/s12016-020-08814-5
- Ariani A, Silva M, Seletti V, et al. Quantitative chest computed tomography is associated with two prediction models of mortality in interstitial lung disease related to systemic sclerosis. Rheumatology (Oxford). Jun 1 2017;56(6):922-927. doi:10.1093/rheumatology/kew480
- Kotani T, Takeuchi T, Yoshimatsu Y, et al. Initial limited three-level thin-section computed tomography scorings predict the prognosis of acute/subacute interstitial pneumonia in patients with dermatomyositis. Mod Rheumatol. Sep 2016;26(5):738-43. doi:10.3109/14397595.2015.1134392
- Wolfe F, Michaud K, Pincus T, Furst D, Keystone E. The disease activity score is not suitable as the sole criterion for initiation and evaluation of anti-tumor necrosis factor therapy in the clinic: discordance between assessment measures and limitations in questionnaire use for regulatory purposes. Arthritis Rheum. Dec 2005;52(12):3873-9. doi:10.1002/art.21494
- Reith PE, Freeman JB, Granner DK. Sputum cytology: a valuable addition to the investigation of hypercalcemia. Am Surg. Aug 1977;43(8):538-9.
- Faraguna MC, Musto F, Crescitelli V, et al. Mucopolysaccharidosis-Plus Syndrome, a Rapidly Progressive Disease: Favorable Impact of a Very Prolonged Steroid Treatment on the Clinical Course in a Child. Genes (Basel). Feb 28 2022;13(3) doi:10.3390/genes13030442
- Freitas S, Westram AM, Schwander T, et al. Parthenogenesis in Darevskia lizards: A rare outcome of common hybridization, not a common outcome of rare hybridization. Evolution. May 2022;76(5):899-914. doi:10.1111/evo.14462
- Kerr SM, Stark GR, Kerr IM. Excess antisense RNA from infectious recombinant SV40 fails to inhibit expression of a transfected, interferon-inducible gene. Eur J Biochem. Jul 15 1988;175(1):65-73. doi:10.1111/j.1432-1033.1988.tb14167.x
- Legras A, Tallet A, Didelot A, et al. Clinical and molecular characteristics of unicentric mediastinal Castleman disease. J Thorac Dis. Apr 2018;10(4):2079-2088. doi:10.21037/jtd.2018.03.159
- Khanna D, Distler O, Cottin V, et al. Diagnosis and monitoring of systemic sclerosis-associated interstitial lung disease using high-resolution computed tomography. J Scleroderma Relat Disord. Oct 2022;7(3):168-178. doi:10.1177/23971983211064463
- Uddin S, Munir MZ, Gull S, et al. Transcriptome Profiling Reveals Role of MicroRNAs and Their Targeted Genes during Adventitious Root Formation in Dark-Pretreated Micro-Shoot Cuttings of Tetraploid Robinia pseudoacacia L. Genes (Basel). Feb 27 2022;13(3) doi:10.3390/genes13030441
- Schiopu E, Phillips K, MacDonald PM, Crofford LJ, Somers EC. Predictors of survival in a cohort of patients with polymyositis and dermatomyositis: effect of corticosteroids, methotrexate and azathioprine. Arthritis Res Ther. Jan 27 2012;14(1):R22. doi:10.1186/ar3704
- Winstone TA, Assayag D, Wilcox PG, et al. Predictors of mortality and progression in scleroderma-associated interstitial lung disease: a systematic review. Chest. Aug 2014;146(2):422-436. doi:10.1378/chest.13-2626
- Yin Y, Liang D, Zhao L, et al. Anti-cyclic citrullinated Peptide antibody is associated with interstitial lung disease in patients with rheumatoid arthritis. PLoS One. 2014;9(4):e92449. doi:10.1371/journal.pone.0092449
- Chen J, Shi Y, Wang X, Huang H, Ascherman D. Asymptomatic preclinical rheumatoid arthritis-associated interstitial lung disease. Clin Dev Immunol. 2013;2013:406927. doi:10.1155/2013/406927
- Wang JX, Du CG. A retrospective study of clinical characteristics of interstitial lung disease associated with rheumatoid arthritis in Chinese patients. Med Sci Monit. Mar 7 2015;21:708-15. doi:10.12659/MSM.890880
- Zou YQ, Li YS, Ding XN, Ying ZH. The clinical significance of HRCT in evaluation of patients with rheumatoid arthritis-associated interstitial lung disease: a report from China. Rheumatol Int. Mar 2012;32(3):669-73. doi:10.1007/s00296-010-1665-1
- Zhang Y, Li H, Wu N, Dong X, Zheng Y. Retrospective study of the clinical characteristics and risk factors of rheumatoid arthritis-associated interstitial lung disease. Clin Rheumatol. Apr 2017;36(4):817-823. doi:10.1007/s10067-017-3561-5
- Li L, Liu R, Zhang Y, et al. A retrospective study on the predictive implications of clinical characteristics and therapeutic management in patients with rheumatoid arthritis-associated interstitial lung disease. Clin Rheumatol. May 2020;39(5):1457-1470. doi:10.1007/s10067-019-04846-1
- Salaffi F, Carotti M, Di Carlo M, Tardella M, Giovagnoni A. High-resolution computed tomography of the lung in patients with rheumatoid arthritis: Prevalence of interstitial lung disease involvement and determinants of abnormalities. Medicine (Baltimore). Sep 2019;98(38):e17088. doi:10.1097/MD.0000000000017088
- Gautam M, Masood MJ, Arooj S, Mahmud ME, Mukhtar MU. Rheumatoid Arthritis Related Interstitial Lung Disease: Patterns of High-resolution Computed Tomography. Cureus. Feb 4 2020;12(2):e6875. doi:10.7759/cureus.6875
- Li L, Gao S, Fu Q, et al. A preliminary study of lung abnormalities on HRCT in patients of rheumatoid arthritis-associated interstitial lung disease with progressive fibrosis. Clin Rheumatol. Nov 2019;38(11):3169-3178. doi:10.1007/s10067-019-04673-4
- Avouac J, Cauvet A, Steelandt A, et al. Improving risk-stratification of rheumatoid arthritis patients for interstitial lung disease. PLoS One. 2020;15(5):e0232978. doi:10.1371/journal.pone.0232978
- Zrour SH, Touzi M, Bejia I, et al. Correlations between high-resolution computed tomography of the chest and clinical function in patients with rheumatoid arthritis. Prospective study in 75 patients. Joint Bone Spine. Jan 2005;72(1):41-7. doi:10.1016/j.jbspin.2004.02.001
- Wilsher M, Voight L, Milne D, et al. Prevalence of airway and parenchymal abnormalities in newly diagnosed rheumatoid arthritis. Respir Med. Oct 2012;106(10):1441-6. doi:10.1016/j.rmed.2012.06.020
- Hanaka T, Kido T, Ishimoto H, et al. Radiological patterns of pulmonary involvement may predict treatment response in rheumatoid arthritis: A retrospective study. Respir Investig. Mar 2019;57(2):172-182. doi:10.1016/j.resinv.2018.12.006
- Kim EJ, Elicker BM, Maldonado F, et al. Usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease. Eur Respir J. Jun 2010;35(6):1322-8. doi:10.1183/09031936.00092309
- Ekici M, Baytar Y, Kardas RC, et al. Predictors of mortality in rheumatoid arthritis-associated lung disease: A retrospective study on ten years. Joint Bone Spine. May 2021;88(3):105133. doi:10.1016/j.jbspin.2021.105133
- Kim HC, Lee JS, Lee EY, et al. Risk prediction model in rheumatoid arthritis-associated interstitial lung disease. Respirology. Dec 2020;25(12):1257-1264. doi:10.1111/resp.13848
- Ito Y, Arita M, Kumagai S, et al. Radiological fibrosis score is strongly associated with worse survival in rheumatoid arthritis-related interstitial lung disease. Mod Rheumatol. Jan 2019;29(1):98-104. doi:10.1080/14397595.2018.1442170
- Yunt ZX, Chung JH, Hobbs S, et al. High resolution computed tomography pattern of usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease: Relationship to survival. Respir Med. May 2017;126:100-104. doi:10.1016/j.rmed.2017.03.027
- Nurmi HM, Kettunen HP, Suoranta SK, et al. Several high-resolution computed tomography findings associate with survival and clinical features in rheumatoid arthritis-associated interstitial lung disease. Respir Med. Jan 2018;134:24-30. doi:10.1016/j.rmed.2017.11.013
- Yamakawa H, Sato S, Tsumiyama E, et al. Predictive factors of mortality in rheumatoid arthritis-associated interstitial lung disease analysed by modified HRCT classification of idiopathic pulmonary fibrosis according to the 2018 ATS/ERS/JRS/ALAT criteria. J Thorac Dis. Dec 2019;11(12):5247-5257. doi:10.21037/jtd.2019.11.73
- Biederer J, Schnabel A, Muhle C, Gross WL, Heller M, Reuter M. Correlation between HRCT findings, pulmonary function tests and bronchoalveolar lavage cytology in interstitial lung disease associated with rheumatoid arthritis. Eur Radiol. Feb 2004;14(2):272-80. doi:10.1007/s00330-003-2026-1
- Yang JA, Lee JS, Park JK, Lee EB, Song YW, Lee EY. Clinical characteristics associated with occurrence and poor prognosis of interstitial lung disease in rheumatoid arthritis. Korean J Intern Med. Mar 2019;34(2):434-441. doi:10.3904/kjim.2016.349
- Fu Q, Wang L, Li L, Li Y, Liu R, Zheng Y. Risk factors for progression and prognosis of rheumatoid arthritis-associated interstitial lung disease: single center study with a large sample of Chinese population. Clin Rheumatol. Apr 2019;38(4):1109-1116. doi:10.1007/s10067-018-4382-x
- Solomon JJ, Chung JH, Cosgrove GP, et al. Predictors of mortality in rheumatoid arthritis-associated interstitial lung disease. Eur Respir J. Feb 2016;47(2):588-96. doi:10.1183/13993003.00357-2015
- Kelly CA, Saravanan V, Nisar M, et al. Rheumatoid arthritis-related interstitial lung disease: associations, prognostic factors and physiological and radiological characteristics–a large multicentre UK study. Rheumatology (Oxford). Sep 2014;53(9):1676-82. doi:10.1093/rheumatology/keu165
- Suleman Y, Clark KEN, Cole AR, Ong VH, Denton CP. Real-world experience of tocilizumab in systemic sclerosis: potential benefit on lung function for anti-topoisomerase-positive patients. Rheumatology (Oxford). Aug 2 2021;60(8):3945-3946. doi:10.1093/rheumatology/keab273
- Kotani T, Takeuchi T, Makino S, et al. Combination with corticosteroids and cyclosporin-A improves pulmonary function test results and chest HRCT findings in dermatomyositis patients with acute/subacute interstitial pneumonia. Clin Rheumatol. Aug 2011;30(8):1021-8. doi:10.1007/s10067-011-1713-6
- Go DJ, Lee EY, Lee EB, Song YW, Konig MF, Park JK. Elevated Erythrocyte Sedimentation Rate Is Predictive of Interstitial Lung Disease and Mortality in Dermatomyositis: a Korean Retrospective Cohort Study. J Korean Med Sci. Mar 2016;31(3):389-96. doi:10.3346/jkms.2016.31.3.389
- Douglas WW, Tazelaar HD, Hartman TE, et al. Polymyositis-dermatomyositis-associated interstitial lung disease. Am J Respir Crit Care Med. Oct 1 2001;164(7):1182-5. doi:10.1164/ajrccm.164.7.2103110
- Ikeda S, Arita M, Misaki K, et al. Incidence and impact of interstitial lung disease and malignancy in patients with polymyositis, dermatomyositis, and clinically amyopathic dermatomyositis: a retrospective cohort study. Springerplus. 2015;4:240. doi:10.1186/s40064-015-1013-8
- Begleiter A, Robotham E, Lacey G, Leith MK. Increased sensitivity of quinone resistant cells to mitomycin C. Cancer Lett. Jun 1989;45(3):173-6. doi:10.1016/0304-3835(89)90073-6
- Hill CL, Zhang Y, Sigurgeirsson B, et al. Frequency of specific cancer types in dermatomyositis and polymyositis: a population-based study. Lancet. Jan 13 2001;357(9250):96-100. doi:10.1016/S0140-6736(00)03540-6
- Prideaux N, van den Berg M, Drummond C, Barr C. Augmented Performance Feedback during Robotic Gait Therapy Results in Moderate Intensity Cardiovascular Exercise in Subacute Stroke. J Stroke Cerebrovasc Dis. Jun 2020;29(6):104758. doi:10.1016/j.jstrokecerebrovasdis.2020.104758
- Kerr SM, Stark GR, Kerr IM. Excess antisense RNA from infectious recombinant SV40 fails to inhibit expression of a transfected, interferon-inducible gene. Eur J Biochem. Jul 15 1988;175(1):65-73. doi:10.1111/j.1432-1033.1988.tb14167.x
- Legras A, Tallet A, Didelot A, et al. Clinical and molecular characteristics of unicentric mediastinal Castleman disease. J Thorac Dis. Apr 2018;10(4):2079-2088. doi:10.21037/jtd.2018.03.159
- Khanna D, Distler O, Cottin V, et al. Diagnosis and monitoring of systemic sclerosis-associated interstitial lung disease using high-resolution computed tomography. J Scleroderma Relat Disord. Oct 2022;7(3):168-178. doi:10.1177/23971983211064463
- Uddin S, Munir MZ, Gull S, et al. Transcriptome Profiling Reveals Role of MicroRNAs and Their Targeted Genes during Adventitious Root Formation in Dark-Pretreated Micro-Shoot Cuttings of Tetraploid Robinia pseudoacacia L. Genes (Basel). Feb 27 2022;13(3) doi:10.3390/genes13030441
- Schiopu E, Phillips K, MacDonald PM, Crofford LJ, Somers EC. Predictors of survival in a cohort of patients with polymyositis and dermatomyositis: effect of corticosteroids, methotrexate and azathioprine. Arthritis Res Ther. Jan 27 2012;14(1):R22. doi:10.1186/ar3704
- Winstone TA, Assayag D, Wilcox PG, et al. Predictors of mortality and progression in scleroderma-associated interstitial lung disease: a systematic review. Chest. Aug 2014;146(2):422-436. doi:10.1378/chest.13-2626
- Yin Y, Liang D, Zhao L, et al. Anti-cyclic citrullinated Peptide antibody is associated with interstitial lung disease in patients with rheumatoid arthritis. PLoS One. 2014;9(4):e92449. doi:10.1371/journal.pone.0092449
- Chen J, Shi Y, Wang X, Huang H, Ascherman D. Asymptomatic preclinical rheumatoid arthritis-associated interstitial lung disease. Clin Dev Immunol. 2013;2013:406927. doi:10.1155/2013/406927
- Wang JX, Du CG. A retrospective study of clinical characteristics of interstitial lung disease associated with rheumatoid arthritis in Chinese patients. Med Sci Monit. Mar 7 2015;21:708-15. doi:10.12659/MSM.890880
- Zou YQ, Li YS, Ding XN, Ying ZH. The clinical significance of HRCT in evaluation of patients with rheumatoid arthritis-associated interstitial lung disease: a report from China. Rheumatol Int. Mar 2012;32(3):669-73. doi:10.1007/s00296-010-1665-1
- Zhang Y, Li H, Wu N, Dong X, Zheng Y. Retrospective study of the clinical characteristics and risk factors of rheumatoid arthritis-associated interstitial lung disease. Clin Rheumatol. Apr 2017;36(4):817-823. doi:10.1007/s10067-017-3561-5
- Li L, Liu R, Zhang Y, et al. A retrospective study on the predictive implications of clinical characteristics and therapeutic management in patients with rheumatoid arthritis-associated interstitial lung disease. Clin Rheumatol. May 2020;39(5):1457-1470. doi:10.1007/s10067-019-04846-1
- Salaffi F, Carotti M, Di Carlo M, Tardella M, Giovagnoni A. High-resolution computed tomography of the lung in patients with rheumatoid arthritis: Prevalence of interstitial lung disease involvement and determinants of abnormalities. Medicine (Baltimore). Sep 2019;98(38):e17088. doi:10.1097/MD.0000000000017088
- Gautam M, Masood MJ, Arooj S, Mahmud ME, Mukhtar MU. Rheumatoid Arthritis Related Interstitial Lung Disease: Patterns of High-resolution Computed Tomography. Cureus. Feb 4 2020;12(2):e6875. doi:10.7759/cureus.6875
- Li L, Gao S, Fu Q, et al. A preliminary study of lung abnormalities on HRCT in patients of rheumatoid arthritis-associated interstitial lung disease with progressive fibrosis. Clin Rheumatol. Nov 2019;38(11):3169-3178. doi:10.1007/s10067-019-04673-4
- Avouac J, Cauvet A, Steelandt A, et al. Improving risk-stratification of rheumatoid arthritis patients for interstitial lung disease. PLoS One. 2020;15(5):e0232978. doi:10.1371/journal.pone.0232978
- Zrour SH, Touzi M, Bejia I, et al. Correlations between high-resolution computed tomography of the chest and clinical function in patients with rheumatoid arthritis. Prospective study in 75 patients. Joint Bone Spine. Jan 2005;72(1):41-7. doi:10.1016/j.jbspin.2004.02.001
- Wilsher M, Voight L, Milne D, et al. Prevalence of airway and parenchymal abnormalities in newly diagnosed rheumatoid arthritis. Respir Med. Oct 2012;106(10):1441-6. doi:10.1016/j.rmed.2012.06.020
- Hanaka T, Kido T, Ishimoto H, et al. Radiological patterns of pulmonary involvement may predict treatment response in rheumatoid arthritis: A retrospective study. Respir Investig. Mar 2019;57(2):172-182. doi:10.1016/j.resinv.2018.12.006
- Kim EJ, Elicker BM, Maldonado F, et al. Usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease. Eur Respir J. Jun 2010;35(6):1322-8. doi:10.1183/09031936.00092309
- Ekici M, Baytar Y, Kardas RC, et al. Predictors of mortality in rheumatoid arthritis-associated lung disease: A retrospective study on ten years. Joint Bone Spine. May 2021;88(3):105133. doi:10.1016/j.jbspin.2021.105133
- Kim HC, Lee JS, Lee EY, et al. Risk prediction model in rheumatoid arthritis-associated interstitial lung disease. Respirology. Dec 2020;25(12):1257-1264. doi:10.1111/resp.13848
- Ito Y, Arita M, Kumagai S, et al. Radiological fibrosis score is strongly associated with worse survival in rheumatoid arthritis-related interstitial lung disease. Mod Rheumatol. Jan 2019;29(1):98-104. doi:10.1080/14397595.2018.1442170
- Yunt ZX, Chung JH, Hobbs S, et al. High resolution computed tomography pattern of usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease: Relationship to survival. Respir Med. May 2017;126:100-104. doi:10.1016/j.rmed.2017.03.027
- Nurmi HM, Kettunen HP, Suoranta SK, et al. Several high-resolution computed tomography findings associate with survival and clinical features in rheumatoid arthritis-associated interstitial lung disease. Respir Med. Jan 2018;134:24-30. doi:10.1016/j.rmed.2017.11.013
- Yamakawa H, Sato S, Tsumiyama E, et al. Predictive factors of mortality in rheumatoid arthritis-associated interstitial lung disease analysed by modified HRCT classification of idiopathic pulmonary fibrosis according to the 2018 ATS/ERS/JRS/ALAT criteria. J Thorac Dis. Dec 2019;11(12):5247-5257. doi:10.21037/jtd.2019.11.73
- Biederer J, Schnabel A, Muhle C, Gross WL, Heller M, Reuter M. Correlation between HRCT findings, pulmonary function tests and bronchoalveolar lavage cytology in interstitial lung disease associated with rheumatoid arthritis. Eur Radiol. Feb 2004;14(2):272-80. doi:10.1007/s00330-003-2026-1
- Yang JA, Lee JS, Park JK, Lee EB, Song YW, Lee EY. Clinical characteristics associated with occurrence and poor prognosis of interstitial lung disease in rheumatoid arthritis. Korean J Intern Med. Mar 2019;34(2):434-441. doi:10.3904/kjim.2016.349
- Fu Q, Wang L, Li L, Li Y, Liu R, Zheng Y. Risk factors for progression and prognosis of rheumatoid arthritis-associated interstitial lung disease: single center study with a large sample of Chinese population. Clin Rheumatol. Apr 2019;38(4):1109-1116. doi:10.1007/s10067-018-4382-x
- Solomon JJ, Chung JH, Cosgrove GP, et al. Predictors of mortality in rheumatoid arthritis-associated interstitial lung disease. Eur Respir J. Feb 2016;47(2):588-96. doi:10.1183/13993003.00357-2015
- Kelly CA, Saravanan V, Nisar M, et al. Rheumatoid arthritis-related interstitial lung disease: associations, prognostic factors and physiological and radiological characteristics–a large multicentre UK study. Rheumatology (Oxford). Sep 2014;53(9):1676-82. doi:10.1093/rheumatology/keu165
- Suleman Y, Clark KEN, Cole AR, Ong VH, Denton CP. Real-world experience of tocilizumab in systemic sclerosis: potential benefit on lung function for anti-topoisomerase-positive patients. Rheumatology (Oxford). Aug 2 2021;60(8):3945-3946. doi:10.1093/rheumatology/keab273
- Kotani T, Takeuchi T, Makino S, et al. Combination with corticosteroids and cyclosporin-A improves pulmonary function test results and chest HRCT findings in dermatomyositis patients with acute/subacute interstitial pneumonia. Clin Rheumatol. Aug 2011;30(8):1021-8. doi:10.1007/s10067-011-1713-6
- Go DJ, Lee EY, Lee EB, Song YW, Konig MF, Park JK. Elevated Erythrocyte Sedimentation Rate Is Predictive of Interstitial Lung Disease and Mortality in Dermatomyositis: a Korean Retrospective Cohort Study. J Korean Med Sci. Mar 2016;31(3):389-96. doi:10.3346/jkms.2016.31.3.389
- Douglas WW, Tazelaar HD, Hartman TE, et al. Polymyositis-dermatomyositis-associated interstitial lung disease. Am J Respir Crit Care Med. Oct 1 2001;164(7):1182-5. doi:10.1164/ajrccm.164.7.2103110
- Ikeda S, Arita M, Misaki K, et al. Incidence and impact of interstitial lung disease and malignancy in patients with polymyositis, dermatomyositis, and clinically amyopathic dermatomyositis: a retrospective cohort study. Springerplus. 2015;4:240. doi:10.1186/s40064-015-1013-8
- Begleiter A,

