Evolving Pediatric Vesicoureteral Reflux Management Strategies
Evolving Approaches to Pediatric Vesicoureteral Reflux: Diagnostic Innovations, Risk Stratification, and Treatment Trends
Benjamin Press¹,², Rachael Germany³, Andrew Kirsch¹,²,³
- Pediatric Urology Department Emory University School of Medicine
- Pediatric Urology Department, Children’s Healthcare of Atlanta
- Georgia Urology Pediatrics
OPEN ACCESS
PUBLISHED 31 July 2025
CITATION Press, B., Germany, R., Kirsch, A., 2025. Evolving Approaches to Pediatric Vesicoureteral Reflux: Diagnostic Innovations, Risk Stratification, and Treatment Trends. Medical Research Archives, [online] 13(7). https://doi.org/10.18103/mra.v13i7.6718
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6718
ISSN 2375-1924
Abstract
Vesicoureteral reflux (VUR) is a common pediatric condition historically managed with a goal of preventing renal injury from recurrent urinary tract infections (UTIs). However, contemporary understanding recognizes that not all reflux confers the same risk. This review highlights evolving diagnostic strategies, risk stratification tools, and treatment trends in VUR management. The natural history of VUR is influenced by multiple factors, including grade, timing on voiding cystourethrogram (VCUG) presence of bladder and bowel dysfunction (BBD), and anatomical anomalies. While many cases of low-grade reflux resolve spontaneously, high-grade or early-filling VUR, especially with BBD, poses a higher risk of persistent disease and recurrent febrile UTIs. Diagnosis has shifted toward minimizing invasive testing and radiation exposure. Contrast-enhanced voiding urosonography (ceVUS) offers a non-radiating alternative to VCUG with excellent diagnostic accuracy. Adjunctive tools such as the ureteral diameter ratio (UDR) and Vesicoureteral Reflux Index (VURx) enhance risk stratification and predict likelihood of resolution or breakthrough infections. Treatment has similarly evolved. Observation and selective use of continuous antibiotic prophylaxis (CAP) are now standard for low-risk patients, reflecting concerns about resistance and microbiome disruption. Endoscopic injection using the Double HIT method provides high success rates with minimal morbidity and is now widely favored over open surgery in most cases. Robotic ureteral reimplantation is increasingly utilized, though its cost and learning curve remain barriers to widespread adoption. Emerging research in artificial intelligence, urinary biomarkers, and microbiome preservation offers promise for even more personalized care. Revised guidelines and multicenter registries are fostering data-driven, risk-based approaches. VUR management has transitioned from a uniform treatment model to a nuanced, individualized strategy. Innovations in diagnostics, quantitative risk tools, and minimally invasive therapies now support safe, effective, and tailored care. The focus continues to shift toward optimizing long-term outcomes while reducing treatment burden for children with VUR.
Keywords
- Vesicoureteral reflux
- pediatric urology
- diagnostic innovations
- risk stratification
- treatment trends
Introduction
Vesicoureteral reflux (VUR) is a common pediatric condition that affects approximately 1% of children and has long been a subject of ongoing debate regarding the most appropriate diagnostic and therapeutic approach. Historically, the presence of VUR has been associated with an increased risk of recurrent febrile urinary tract infections (UTIs), renal scarring, and the potential development of reflux nephropathy. As a result, prevention of renal injury was the central goal of VUR management. However, over the last two decades, our understanding of the disease process has evolved, shifting clinical practice toward a more individualized, risk-based approach.
Although VUR may resolve spontaneously, particularly in cases where the voiding cystourethrogram (VCUG) shows late filing or voiding-only, low-grade reflux, persistence is more likely in children with early filling, high-grade or bilateral reflux and in those with concurrent bladder and bowel dysfunction (BBD). The etiology of renal scarring is now understood to be multifactorial, involving not only infection but also congenital dysplasia and voiding dysfunction. These insights have reframed the role of VUR from a direct cause of renal injury to one component in a broader clinical picture.
Advances in imaging techniques have mirrored this evolving perspective. While voiding cystourethrography remains the gold standard for diagnosing and grading VUR, its invasive nature and radiation exposure have prompted growing interest in contrast-enhanced voiding urosonography (ceVUS), which has demonstrated high diagnostic accuracy without ionizing radiation and is now widely used in European centers. In addition, quantitative tools such as the ureteral diameter ratio (UDR) and the vesicoureteral reflux index (VURx) have emerged as valuable adjuncts to grade alone in predicting the likelihood of spontaneous resolution and breakthrough infections.
Therapeutic options have also shifted. Continuous antibiotic prophylaxis (CAP), once widely used in all grades of reflux, is now reserved for carefully selected patients due to growing concerns about antibiotic resistance, low adherence, and potential effects on the microbiome. While there will always be a role for traditional open surgery, the endoscopic correction using the Double HIT method is outpatient, minimally invasive, well-tolerated, and provides cure rates approaching those of open surgery. Robot-assisted ureteral reimplantation continues to expand in use, though it is associated with higher costs and a steeper learning curve, and its comparative effectiveness remains under evaluation.
This review aims to summarize current concepts and evolving approaches to the diagnosis and management of pediatric VUR in 2025, incorporating the latest evidence, updated clinical tools, and future directions toward more personalized and evidence-based care.
Epidemiology and Natural History
VUR is diagnosed in approximately 1% of the general pediatric population and in up to 30-40% of children evaluated for febrile UTIs. The condition demonstrates a clear familial and genetic predisposition, with siblings of affected children exhibiting VUR prevalence rates as high as 25-35% and offspring of affected parents reaching rates over 50%. These observations suggest a strong heritable component, although the specific genetic architecture remains incompletely defined. Several candidate genes involved in ureteric bud development (e.g., ROBO2, RET, and AGTR2) have been proposed, though no single causative mutation has been universally accepted. This has led to ongoing debate about the clinical value of proactive sibling screening, especially for asymptomatic children.
The natural course of VUR varies widely and depends on multiple clinical and anatomical factors. While not the only factor, spontaneous resolution may be more common in low-grade reflux (grades I-III), particularly in younger children, with annual resolution rates estimated between 10-20%. In contrast, high-grade VUR (grades IV-V), bilateral reflux, and abnormalities in bladder function, such as BBD, are associated with lower rates of resolution and a higher risk of recurrent infection and renal damage. The timing of reflux on the VCUG, particularly early filling reflux, has been proven to be a very critical risk factor in both reflux resolution and the risk of breakthrough UTI. In fact, the finding of early filling reflux as the sole factor evaluated has been shown to decrease the chance of resolution by 50%. Other factors, such as female gender, anomalies (e.g. duplex systems or periureteral diverticula), and high grade reflux render the chance for resolution even lower.
While early studies attributed much of the renal injury in VUR to ascending infections and scarring from pyelonephritis, more recent evidence suggests that congenital renal dysplasia plays a substantial role, especially in children with high-grade or prenatal reflux. Distinguishing between congenital dysplasia and acquired scarring can be challenging, but advances in imaging, particularly with DMSA scintigraphy, have improved the ability to characterize renal parenchymal abnormalities. However, the presence of renal scarring alone does not always correlate with clinical severity, and many children with radiographic findings remain asymptomatic with preserved renal function.
The clinical significance of VUR has therefore shifted from being viewed as a universal risk factor for renal disease to a marker for a subset of children at elevated risk. This evolving understanding has contributed to a more selective and individualized approach to both surveillance and intervention, with increased emphasis on patient age, reflux grade, recurrent febrile UTIs, presence of BBD, and renal imaging findings.
Diagnosis and Imaging
The diagnosis of VUR typically follows the evaluation of a child with a febrile UTI. Ultrasonography is often the initial imaging modality due to its noninvasive nature, but its sensitivity for detecting VUR is limited. VCUG remains the gold standard for diagnosing and grading VUR, particularly in children with recurrent febrile UTIs, abnormal renal ultrasound findings, or family history of reflux.
The VCUG remains the gold standard for VUR diagnosis and grading, offering direct visualization of reflux severity and timing during bladder filling and voiding. It also provides critical anatomic details, such as urethral morphology and bladder contour. Although widely used, VCUG presents several limitations: it is invasive, exposes children to ionizing radiation, and can be distressing for patients and families. Despite its utility, VCUG interpretation is subject to inter-rater variability. While some studies have reported favorable intraclass correlation coefficients in the range of 0.8 to 0.9, interobserver agreement rates as low as 50-60% have also been documented. To enhance reliability, it is critical to follow a standardized VCUG protocol. Key elements include consistent use of contrast agents, standardized infusion techniques, acquisition of high-quality spot images, documentation of bladder volume at the onset of reflux, and use of appropriate timing during both filling and voiding phases.
Performing more than one fill void cycle may be necessary to avoid missed diagnoses of intermittent reflux. Inconsistent adherence to these protocols remains a limitation in routine clinical practice and represents a clear opportunity for quality improvement. The use of VCUG has become more selective over the last decade, guided by risk stratification algorithms and clinical practice guidelines that aim to reduce unnecessary testing in low-risk children.
An important development in the field has been the increasing use of ceVUS, particularly in Europe. CeVUS offers a radiation-free alternative to VCUG with high diagnostic concordance and excellent sensitivity and specificity. CeVUS uses ultrasound contrast agents to visualize reflux in real time without ionizing radiation. While it has not been adopted as widely in North America, accumulating data support its safety and efficacy, and its use is expected to expand with broader access to trained personnel and contrast agents. However, while CeVUS reduces radiation exposure, it does not alleviate the pain and anxiety associated with catheterization.
Additionally, tools such as UDR and the vesicoureteral reflux index (VURx) have emerged to complement the international grading system in assessing reflux severity and predicting spontaneous resolution. These measures incorporate quantitative measures, allowing for more refined risk stratification, particularly in infants and young children. When applied consistently, these tools may reduce interobserver variability in VUR grading and help guide management decisions.
UDR, defined as the diameter of the distal ureter normalized to the distance between the L1 and L3 vertebral bodies on VCUG, has emerged as a reliable predictor of both spontaneous VUR resolution and the risk of breakthrough febrile urinary tract infection. Notably, each 0.1 unit increase in UDR is associated with a significantly higher likelihood of persistent VUR. In addition to its predictive value, UDR assessment has demonstrated superior inter-rater reliability compared to traditional VUR grading, with intraclass correlation coefficients of 0.95 versus 0.87, respectively.
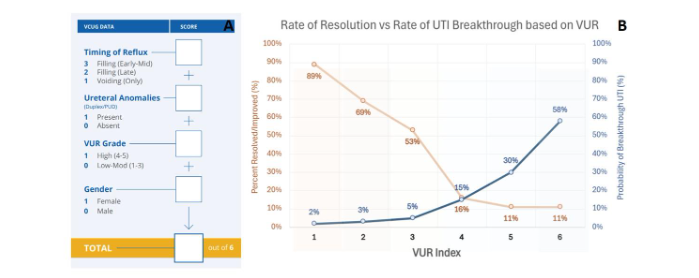
Growing understanding of the factors influencing spontaneous resolution of VUR led to the development of computational tools to enhance clinical prediction. One such tool, the VUR Index (VURx), was proposed to estimate the probability of spontaneous resolution based on individual patient characteristics, including sex, VUR grade, and timing of reflux during the voiding cycle. The VURx was initially derived from a cohort of 229 children under 2 years of age and subsequently validated in two independent cohorts: one with 369 children under 2 years and another with 271 children over 2 years. In addition to predicting resolution, the VURx has also demonstrated correlation with the risk of urinary tract infections (UTIs), providing added clinical value.
Radionuclide renal cortical scintigraphy with DMSA remains the most sensitive method for detecting renal parenchymal injury and is often used to assess for congenital dysplasia or scarring following febrile UTI. However, the routine use of DMSA scanning in follow-up has declined due to concerns over radionuclide availability, cost, limited influence on clinical management in many cases, and the cumulative radiation exposure from repeated studies.
The current diagnostic landscape for VUR increasingly emphasizes targeted imaging, individualized evaluation, and the reduction of unnecessary radiation exposure. As novel techniques, such as artificial intelligence-enhanced ultrasound interpretation and automated bladder dynamics analysis continue to evolve, the potential exists for further refining diagnostic accuracy in a child-centered, minimally invasive manner.
Treatment Approaches
The management of VUR has evolved considerably, reflecting a more individualized and risk-based approach to intervention. While spontaneous resolution remains common, especially in low-grade and low VURx reflux, the decision to initiate treatment depends on factors such as age, grade of reflux, sex, history of febrile UTIs, presence of renal abnormalities, and coexisting BBD.
Observation
Observation remains the cornerstone of management for many children with low-risk VUR. Children with a low VURx and the majority with low grade (I-II) VUR, and no history of febrile UTIs or renal abnormalities may be safely managed with observation alone in most cases. Studies have shown that many of these children experience spontaneous resolution over time, particularly if BBD is well controlled. Observation protocols typically include periodic renal ultrasounds and urinalysis, with prompt evaluation for any febrile episodes. In patients with intermediate risk, such as those with grade III reflux or minor renal abnormalities, a tailored follow-up plan may incorporate repeat imaging at intervals or surveillance VCUG in the setting of breakthrough UTI. In select cases of high-risk patients, repeat VCUG may be warranted to assess for resolution. The increasing use of predictive tools like UDR and VURx has improved the precision of these decisions.
Antibiotic Prophylaxis
CAP has been a longstanding component of VUR management. The rationale for CAP stemmed from early observations linking febrile UTIs to renal scarring, prompting widespread prophylactic use across all reflux grades. However, growing concerns over antimicrobial resistance, variable adherence, and emerging evidence about long-term renal outcomes have prompted a more selective application of this strategy.
The landmark Randomized Intervention for Children with Vesicoureteral Reflux (RIVUR) trial was instrumental in redefining the role of CAP. This multicenter, double-blind, placebo-controlled study demonstrated that trimethoprim-sulfamethoxazole prophylaxis reduced the incidence of recurrent UTIs by approximately 50% in children with grades I-IV VUR, particularly in those with prior febrile infections or BBD. However, CAP did not significantly reduce the risk of new or progressive renal scarring at two years, though it should be noted the trial was not powered to detect these differences. Studies have shown that CAP can alter the composition of the infant gut microbiome. A recent study demonstrated that long-term antibiotic exposure reduced microbial diversity and promoted the selection of resistance genes, raising concerns about the broader ecological and developmental impacts of CAP. These findings underscore the need for judicious prescribing and consideration of potential long-term effects, especially in otherwise healthy children with low-grade reflux. Current guidelines recommend CAP primarily for children with recurrent UTIs, high-grade reflux, or abnormal renal imaging, with an emphasis on careful patient selection and parental education. Shorter durations of CAP and targeted prophylaxis strategies are actively being studied. The American Academy of Pediatrics UTI guidelines are currently being reevaluated to better assess the risk of renal scarring from recurrent UTIs, with the goal of improving our ability to selectively identify children who should be evaluated for VUR.
Endoscopic Injection
Endoscopic correction using the Double HIT method is minimally invasive, well-tolerated, and provides cure rates approaching those of open surgery. Based on proven benefits of EI over the past 2 decades, open surgery has been challenged. In fact, the rate of open surgery has consistently decreased over the same time period. Despite these trends, open surgery may still be a preferred approach, particularly in young children with complex ureteral anatomy and refluxing megaureter. Clinical data from numerous studies have confirmed long-term safety and efficacy of endoscopic injection, though there is significant variation depending on surgeons and technique on the ultimate success rates. In a systematic review evaluating dextranomer/hyaluronic acid (Dx/HA) for pediatric VUR, the estimated aggregate success rate for endoscopic therapy, prior to widespread use of the double HIT method, was 72% with 89% success for grade I, 83% for grade II, 71% for grade III, 59% for IV and 62% for grade V reflux. More recently, a meta-analysis, including nine studies and more than 1400 renal units, the authors found no differences in UTIs for patients undergoing endoscopic management than vesicoureteral reimplantation for high grade VUR (grades III-V). In our own institutional experience, endoscopic injection for primary VUR demonstrates excellent intermediate and long-term clinical outcomes for grades I-IV VUR, with reported success rates exceeding 90% and low rates of secondary intervention even beyond five years post-procedure. Our institutional experience has also discouraged routine VCUGs as part of the post-operative protocol as clinical success was no different in patients who underwent post-operative VCUG compared to those who did not. While endoscopic therapy is highly effective for the treatment of most primary VUR using the double HIT method, its efficacy in cases of structural/functional bladder anomalies is not as well established, such as paraureteral diverticulum, posterior urethral valves and renal transplant.
Ureteral Reimplantation
Surgical correction remains the most definitive treatment for VUR and is indicated in cases of persistent high-grade reflux, breakthrough febrile UTIs, failure of endoscopic treatment, or parental preference. Open ureteral reimplantation, including the Cohen cross-trigonal and Politano-Leadbetter techniques, has long been the gold standard, with success rates exceeding 95%. In the most recent and largest study of over 1000 children undergoing open ureteral reimplantation, radiographic and clinical success rates were both 94%. In their study, risk factors for postop febrile UTI were female gender, the number of preop UTIs, high grade VUR, and renal scarring. Minimally invasive techniques, including laparoscopic and robot-assisted approaches, have become increasingly common. These techniques offer reduced postoperative pain, shorter hospital stays, and improved cosmesis, though they require specialized expertise and carry higher initial costs. Recent studies have demonstrated the safety and efficacy of robot-assisted ureteral reimplantation in pediatric patients, with comparable success to open techniques.
Certain populations, such as children with prior abdominal surgery, complex ureteral anatomy, or failed endoscopic treatments, may benefit from open approaches due to the technical demands of these cases. Conversely, older children and adolescents may be ideal candidates for robot-assisted ureteral reimplantation due to their body habitus and lower risk of postoperative urinary retention. Regardless of surgical technique, successful ureteral reimplantation requires optimization of bladder function prior to surgery. Untreated bladder and bowel dysfunction (BBD) has been associated with increased risk of postoperative UTIs and persistent symptoms, even in the absence of persistent reflux. Addressing BBD preoperatively remains a critical determinant of long-term success. Postoperative follow-up after reimplantation has also evolved. Routine post-op VCUG to confirm radiographic resolution is increasingly avoided in favor of symptom-based monitoring, particularly in asymptomatic patients with low risk of recurrence. This shift reflects a broader emphasis on clinical success and quality of life, rather than strictly radiographic endpoints.
In summary, ureteral reimplantation remains a highly effective option for children with complex or refractory VUR. The choice between open and robotic approaches should be individualized based on patient anatomy, surgeon experience, and institutional capabilities. With appropriate patient selection and BBD management, surgical correction offers excellent long-term outcomes with minimal recurrence and morbidity.
Future Directions
The future of VUR management is centered on innovation in diagnostics, risk stratification, and individualized therapy. A few areas of particular promise include:
- Artificial intelligence (AI): AI-assisted image interpretation and predictive analytics are being explored to enhance diagnostic accuracy, improve inter-rater reliability in reflux grading, and develop clinical decision-support tools for treatment planning.
- Biomarkers and genetic profiling: The identification of urinary, serum, and genetic biomarkers could help distinguish which children are most at risk for renal injury, guide treatment intensity, and predict treatment outcomes. Urinary neutrophil gelatinase-associated lipocalin (NGAL), for example, has been studied as a potential noninvasive marker of renal scarring in children with VUR.
- Microbiome research and targeted prophylaxis: The impact of antibiotics on the developing gut and urinary microbiomes is becoming more recognized. Research into microbiome preservation and alternative approaches to UTI prevention, such as probiotics, immunoprophylaxis, and vaccine development, may change the landscape of infection prevention in VUR.
- Guideline evolution and data registries: Updated guidelines from the AUA and EAU emphasize risk-based, individualized management and integration of emerging evidence. Collaborative multicenter registries and long-term outcome studies are helping define best practices and patient-centered outcomes.
Conclusions
The contemporary management of vesicoureteral reflux is defined by a shift from broad treatment strategies to nuanced, evidence-based care tailored to each patient’s risk profile. Advances in diagnostics, particularly in imaging and risk stratification tools, have refined clinical decision-making. Treatment options have expanded beyond traditional surgical intervention to include effective, less invasive modalities such as endoscopic injection, while observation and selective use of antibiotics remain appropriate for many.
Future innovations in artificial intelligence, biomarker discovery, and microbiome science promise to further personalize care and improve outcomes. As our understanding of VUR continues to evolve, the goal remains constant: to provide safe, effective, and individualized care that preserves renal function and quality of life for children affected by this condition.
Conflict of Interest Statement:
None.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Garcia-Roig M, Kirsch AJ. The Role of Antibiotic Prophylaxis in the Management of Vesicoureteral Reflux. The Journal of Urology. 2018;199(1):287-93.
- Läckgren G. Management of Vesicoureteral Reflux: What Have We Learned Over the Last 20 Years? Frontiers in Pediatrics. 2021;9:650326.
- Mattoo TK. Vesicoureteral Reflux and Reflux Nephropathy. Advances in Chronic Kidney Disease. 2011;18(5):348-54.
- Elder JS, Diaz M. Vesicoureteral Reflux Traditional Concepts and New Insights. Nature Reviews Urology. 2013;10(11):640-8.
- Kirsch AJ, Arlen AM. The Ureteral Diameter Ratio as a Predictor of VUR Resolution. Journal of Pediatric Urology. 2014;10(6):1249-54.
- Arlen AM, Kirsch AJ. Contemporary Outcomes of VUR Management in the United States. The Journal of Urology. 2020;204(3):572-7.
- Chua ME. Diagnostic Accuracy of CeVUS Versus VCUG: A Meta-Analysis. World Journal of Urology. 2019;37(11):2245-55.
- Ntoulia A. Contrast-Enhanced Voiding Urosonography: A Radiation-Free Option. Pediatric Radiology. 2018;48(2):216-26.
- Investigators RT. Antimicrobial Prophylaxis for Children with Vesicoureteral Reflux. New England Journal of Medicine. 2014;370:2367-76.
- Langley JM. Antibiotic Prophylaxis in Children: Resistance and Microbiome Impacts. Journal of the Pediatric Infectious Diseases Society. 2023;12(1):15-24.
- Williams G. Antibiotics for Preventing Recurrent UTI in Children. Cochrane Database of Systematic Reviews. 2019(2):CD001532.
- Kirsch AJ, Arlen AM. Evolving surgical management of pediatric vesicoureteral reflux: is open ureteral reimplantation still the ‘Gold Standard’? International braz j urol : official journal of the Brazilian Society of Urology. 2020;46(3):314-21.
- Peters CA. Robotic Ureteral Reimplantation: Contemporary Outcomes. The Journal of Urology. 2023;209(2):401-8.
- Sorensen MD. Cost-Effectiveness and Complications of Bilateral Robotic Reimplantation. Urology. 2024;175:67-73.
- Sargent MA. What is the normal prevalence of vesicoureteric reflux? Pediatric Radiology. 2000;30(9):587-93.
- Noe HN. The long-term results of prospective sibling reflux screening. Journal of Urology. 1992;148:1739-42.
- van Eerde AM, Duran K, van Riel E, de Kovel CGF, Koeleman BPC, Knoers NVAM, et al. Genes in the Ureteric Budding Pathway: Association Study on Vesico-Ureteral Reflux Patients. PLOS ONE. 2012;7(4):e31327.
- Westland R, Renkema KY, Knoers NVAM. Clinical Integration of Genome Diagnostics for Congenital Anomalies of the Kidney and Urinary Tract. Clinical Journal of the American Society of Nephrology. 2021;16(1):128-37.
- Peters CA, Skoog SJ, Arant BS, et al. Summary of the AUA guideline on management of primary vesicoureteral reflux in children. Journal of Urology. 2010;184:1134-44.
- Kirsch AJ, Arlen AM, Leong T, Merriman LS, Herrel LA, Scherz HC, et al. Vesicoureteral reflux index (VURx): a novel tool to predict primary reflux improvement and resolution in children less than 2 years of age. J Pediatr Urol. 2014;10(6):1249-54.
- Cooper CS. Diagnosis and management of vesicoureteral reflux. Pediatrics in Review. 2017;38(11):505-17.
- Elder JS. Imaging for vesicoureteral reflux: bottom-up, top-down or none of the above. J Urol. 2006;176(3):1063-9.
- Peters CA, Skoog SJ, Arant BS, et al. Summary of the AUA guideline on management of primary vesicoureteral reflux in children. J Urol. 2010;184:1134-44.
- ştaş CB, Kuzdan Ö, Erdoğan for Voiding Cystourethrograms in the Management of Vesicoureteral Reflux: An Interrater Comparison. Advances in urology. 2016;2016:1684190.
- Metcalfe CB, Macneily AE, Afshar K. Reliability assessment of international grading system for vesicoureteral reflux. J Urol. 2012;188(4 Suppl):1490-2.
- Ozaydin S, Celebi S, Caymaz I, Besik C, Karaaslan B, Kuzdan O, et al. Increasing the Reliability of the Grading System for Voiding Cystourethrograms Using Ultrasonography: An Inter-Rater Comparison. Nephro-urology monthly. 2016;8(5):e38685.
- Frimberger D, Bauer SB, Cain MP, Greenfield SP, Kirsch AJ, Ramji F, et al. Establishing a standard protocol for the voiding cystourethrography. J Pediatr Urol. 2016;12(6):362-6.
- Janssen KM, Kirsch AJ, Crisostomo-Wynne TC, Leong T, Cuda SP, Arlen AM. Standardized protocol for voiding cystourethrogram: Are recommendations being followed? J Pediatr Urol. 2021;17(1):66.e1-.e6.
- Polito C, Moggio G, La Manna A, Cioce F, Cappabianca S, Di Toro R. Cyclic voiding cystourethrography in the diagnosis of occult vesicoureteric reflux. Pediatric nephrology (Berlin, Germany). 2000;14(1):39-41.
- Subcommittee on Urinary Tract Infection RKB. Urinary tract infection: clinical practice guideline for the diagnosis and management of the initial UTI in febrile infants and children 2 to 24 months. Pediatrics. 2011;128(3):595-610.
- Chua ME, Ming JM. Diagnostic accuracy of contrast-enhanced voiding urosonography compared with voiding cystourethrography in children: a meta-analysis. World J Urol. 2019;37(11):2245-55.
- Kirsch AJ, Arlen AM. Utility of the ureteral diameter ratio and bladder volume in predicting vesicoureteral reflux resolution. J Urol. 2014;192(1):194-9.
- Ntoulia A. Contrast-enhanced voiding urosonography: a radiation-free alternative for diagnosing vesicoureteral reflux. Pediatr Radiol. 2018;48(2):216-26.
- Arlen AM, Kirsch AJ, Leong T, Cooper CS. Validation of the ureteral diameter ratio for predicting early spontaneous resolution of primary vesicoureteral reflux. Journal of Pediatric Urology. 2017;13(4):383.e1-.e6.
- Arlen AM, Leong T, Guidos PJ, Alexander SE, Cooper CS. Distal ureteral diameter ratio is predictive of breakthrough febrile urinary tract infection. Journal of Urology. 2017;198(6):1418-23.
- Cooper CS, Alexander SE, Kieran K, Storm DW. Utility of the distal ureteral diameter on VCUG for grading VUR. Journal of Pediatric Urology. 2015;11(4):183.e1-.e6.
- Cooper CS, Birusingh KK, Austin JC, Knudson MJ, Brophy PD. Distal ureteral diameter measurement objectively predicts vesicoureteral reflux outcome. Journal of Pediatric Urology. 2013;9(1):99-103.
- Swanton AR, Arlen AM, Alexander SE, Kieran K, Storm DW, Cooper CS. Inter-rater reliability of distal ureteral diameter ratio compared to grade of VUR. J Pediatr Urol. 2017;13(2):207.e1-.e5.
- Arlen AM, Garcia-Roig M, Weiss AD, Leong T, Cooper CS, Kirsch AJ. Vesicoureteral Reflux Index: 2-Institution Analysis and Validation. J Urol. 2016;195(4 Pt 2):1294-9.
- Garcia-Roig M, Ridley DE, McCracken C, Arlen AM, Cooper CS, Kirsch AJ. Vesicoureteral Reflux Index: Predicting Primary Vesicoureteral Reflux Resolution in Children Diagnosed after Age 24 Months. J Urol. 2017;197(4):1150-7.
- Arlen AM, Leong T, Wu CQ, Traore EJ, Cooper CS, Kirsch AJ. Predicting Breakthrough Urinary Tract Infection: Comparative Analysis of Vesicoureteral Reflux Index, Reflux Grade and Ureteral Diameter Ratio. J Urol. 2020;204(3):572-7.
- Hoberman A, Greenfield SP, Mattoo TK. Imaging studies after a first febrile urinary tract infection in young children. N Engl J Med. 2003;348(3):195-202.
- Khondker A, Kwong JC, Malik S, Erdman L, Keefe DT, Fernandez N, et al. The state of artificial intelligence in pediatric urology. Frontiers in Urology. 2022;Volume 2 – 2022.
- American Urological A. VUR clinical practice guideline. 2023.
- Skoog SJ. Pediatric vesicoureteral reflux guidelines: an update. Journal of Urology. 2010;184(3):1134-44.
- G W, JC C. Antibiotics for preventing recurrent urinary tract infection in children. Cochrane Database Syst Rev. 2019(2):CD001532.
- JM L. Antibiotic prophylaxis in children: resistance and microbiome impacts. J Pediatric Infect Dis Soc. 2023;12(1):15-24.
- M G-R, AJ K. The role of antibiotic prophylaxis in the management of vesicoureteral reflux. J Urol. 2018;199(1):287-93.
- TK M. Vesicoureteral reflux and reflux nephropathy. Adv Chronic Kidney Dis. 2011;18(5):348-54.
- Fouhy F. The effect of antibiotics on the infant gut microbiome. Gut Microbes. 2021;13(1):1939834.
- Mattoo TK. Contemporary management of vesicoureteral reflux. Pediatric Nephrology. 2022;37:1501-10.
- Peters CA, Skoog SJ, Arant BS, Jr., Copp HL, Elder JS, Hudson RG, et al. Summary of the AUA Guideline on Management of Primary Vesicoureteral Reflux in Children. The Journal of urology. 2010;184(3):1134-44.
- Kirsch AJ, Cooper CS, Läckgren G. Non-Animal Stabilized Hyaluronic Acid/Dextranomer Gel (NASHA/Dx, Deflux) for Endoscopic Treatment of Vesicoureteral Reflux: What Have We Learned Over the Last 20 Years? Urology. 2021;157:15-28.
- Garcia-Roig M, Travers C, McCracken CE, Kirsch AJ. National Trends in the Management of Primary Vesicoureteral Reflux in Children. The Journal of urology. 2018;199(1):287-93.
- Routh JC, Inman BA, Reinberg Y. Dextranomer/hyaluronic acid for pediatric vesicoureteral reflux: systematic review. Pediatrics. 2010;125(5):1010-9.
- Yap T-L, Chen Y, Nah SA, Ong CCP, Jacobsen A, Low Y. STING versus HIT technique of endoscopic treatment for vesicoureteral reflux: A systematic review and meta-analysis. Journal of Pediatric Surgery. 2016;51(12):2015-20.
- Mina-Riascos SH, Fernández N, García-Perdomo HA. Effectiveness and risks of endoscopic management compared to vesicoureteral reimplantation in patients with high-grade vesicoureteral reflux: systematic review and network meta-analysis. European journal of pediatrics. 2021;180(5):1383-91.
- Kalisvaart JF, Scherz HC, Cuda S, Kaye JD, Kirsch AJ. Intermediate to long-term follow-up indicates low risk of recurrence after Double HIT endoscopic treatment for primary vesico-ureteral reflux. Journal of Pediatric Urology. 2012;8(4):359-65.
- Lightfoot M, Bilgutay AN, Tollin N, Eisenberg S, Weiser J, Bryan L, et al. Long-Term Clinical Outcomes and Parental Satisfaction After Dextranomer/Hyaluronic Acid (Dx/HA) Injection for Primary Vesicoureteral Reflux. Front Pediatr. 2019;7:392.
- Arlen AM, Scherz HC, Filimon E, Leong T, Kirsch AJ. Is routine voiding cystourethrogram necessary following double hit for primary vesicoureteral reflux? Journal of Pediatric Urology. 2015;11(1):40.e1-.e5.
- Kirsch AJ, Arlen AM. Evaluation of new Deflux administration techniques: intraureteric HIT and Double HIT for the endoscopic correction of vesicoureteral reflux. Expert review of medical devices. 2014;11(5):439-46.
- Aydogdu O, Burgu B, Soygur T. Predictors of surgical outcome in children with vesicoureteral reflux associated with paraureteral diverticula. Urology. 2010;76(1):209-14.
- PEREZ-BRAYFIELD M, KIRSCH AJ, HENSLE TW, KOYLE MA, FURNESS P, SCHERZ HC. ENDOSCOPIC TREATMENT WITH DEXTRANOMER/HYALURONIC ACID FOR COMPLEX CASES OF VESICOURETERAL REFLUX. Journal of Urology. 2004;172(4 Part 2):1614-6.
- Puri P, Kumar R. Endoscopic correction of vesicoureteral reflux secondary to posterior urethral valves. The Journal of urology. 1996;156(2 Pt 2):680-2.
- Williams MA, Giel DW, Colleen Hastings M. Endoscopic Deflux injection for pediatric transplant reflux: a feasible alternative to open ureteral reimplant. Journal of pediatric urology. 2008;4(5):341-4.
- Vemulakonda VM, Koyle MA, Lendvay TS, Risk MC, Kirsch AJ, Cheng EY, et al. Endoscopic treatment of symptomatic refluxing renal transplant ureteroneocystostomies in children. Pediatric transplantation. 2010;14(2):212-5.
- Barrieras DJ. Comparison of open and laparoscopic ureteral reimplantation in children. Journal of Urology. 2001;166(2):726-9.
- Nelson CP, Hubert KC, Kokorowski PJ, Huang L, Prasad MM, Rosoklija I, et al. Long-term incidence of urinary tract infection after ureteral reimplantation for primary vesicoureteral reflux. J Pediatr Urol. 2013;9(1):92-8.
- Peters CA. Robotic ureteral reimplantation: contemporary outcomes. Journal of Urology. 2023;209(2):401-8.
- JC A, CS C. Diurnal urinary incontinence in children: a review of classification and treatment. J Urol. 2007;178(4 Pt 1):1213-9.
- L C-P, MP L. Pediatric dysfunctional elimination syndrome. Can J Urol. 2005;12(Suppl 1):40-2.
- SA K. Bladder and bowel dysfunction in children with vesicoureteral reflux and urinary tract infection diagnosis, treatment, and long-term followup. Urology. 2008;71(1):75-9.
- CA P. Robotic ureteral reimplantation: contemporary outcomes. J Urol. 2023;209(2):401-8.
- JS E, M D, AA C, et al. Pediatric vesicoureteral reflux guidelines panel summary report: clinical practice guidelines for the management of primary vesicoureteral reflux in children. J Urol. 2010;184(3):1134-44.
- SJ C, EG P, Jr WJ, DA B. Imaging after ureteral reimplantation: are we doing too much? J Urol. 2001;166(3):1074-7.
- Beksac AT. AI in pediatric urology: clinical applications and future directions. Journal of Pediatric Urology. 2023;19(1):65.e1-.e6.
- Khondker A. The state of artificial intelligence in pediatric urology. Frontiers in Urology. 2022;2:1024662.
- Aslan N. Neutrophil gelatinase-associated lipocalin as a marker of renal scarring in children with vesicoureteral reflux. Pediatric Nephrology. 2015;30(8):1383-9.
- Kassiri B. Role of the microbiome in urinary tract infections and treatment strategies. Diagnostics. 2023;15(1):93.
- Schlager TA. Probiotics and urinary tract infections in children. Current Opinion in Pediatrics. 2007;19(2):139-43.
- European Association of U. Guidelines on pediatric urology. 2023.
- Cooper CS. Real-world data in vesicoureteral reflux: insights from the VUR Research Network. Journal of Pediatric Urology. 2021;17(3):310.e1-.e7.

