Sympathetic Response to Self-Observed Balance Loss
Sympathetic Nervous System response to observed balance loss while walking in young adults: Explorative study
Gil Meir 1, Amos Katz 2, Avi Ohana 1, Anat Reiner-Benaim 3, and Itshak Melzer 1
- Department of Physical Therapy, Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer-Sheva, Israel.
- Barzilai Medical Center and the Faculty of Health Sciences, and Ben-Gurion University of the Negev, Beer-Sheva, Israel.
- Department of Epidemiology, Biostatistics and Community Health Sciences, School of Public Health, Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer-Sheva, Israel.
OPEN ACCESS
PUBLISHED 31 January 2026
CITATION Meir, G., Katz, A., et al., 2026. Sympathetic Nervous System response to observed balance loss while walking in young adults: Explorative study. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7241
ISSN 2375-1924
ABSTRACT
Background: Video observation of one’s own loss of balance may evoke emotional and physiological arousal, yet little is known about how such stimuli affect Sympathetic Nervous System activity as measured by Electrodermal activity.
Research question: This study investigated whether observing video recordings of one’s own balance loss during treadmill walking induces a measurable electrodermal activity response? and whether such responses habituate over repeated exposures?
Methods: Thirty-four healthy young adults were presented with six video clips showing their own balance losses during treadmill walking. electrodermal activity was continuously monitored using finger electrodes. Skin conductance response (SCR) was quantified as the difference between pre- and post-observation skin conductance level (SCL) and normalized using a relative SCR/SCL (%) index.
Results: All participants exhibited significant phasic electrodermal activity responses (SCRs) following each observed perturbation (P≤0.05), with the strongest response during the first viewing (0.38±0.41 µS) and a progressive decline across subsequent trials (0.10±0.18 µS by trial six). Linear regression revealed a significant attenuation in absolute SCR values (R²=0.80, p=0.016), indicating rapid physiological habituation. Relative SCL deltas (SCR/SCL%) followed a similar trend (R²=0.58, p =0.079), further supported by a mixed-effects model showing a significant decline in log-transformed relative SCR values.
Significance: These findings demonstrate that video-based self-observation of balance loss reliably elicits sympathetic activation that rapidly habituates with repetition. The steep habituation trajectory suggests rapid cognitive appraisal and reclassification of the visual threat as non-threatening. This approach may serve as an objective, low-burden proxy for assessing fall-related concern and emotional reactivity, with potential applications in clinical screening, therapeutic desensitization, and fall-prevention programs.
Keywords: electrodermal activity; skin conductance; sympathetic nervous system; video observation; balance perturbation; habituation.
1. Introduction
Electrodermal activity, also known as skin conductance is a robust index of sympathetic nervous system activity and emotional arousal since the sweat glands are solely innervated by the sympathetic branch. Therefore, electrodermal activity is believed to represent a quantitative functional measure of sudomotor activity, and consequently, an objective assessment of arousal. It captures both tonic (skin conductance level, SCL) and phasic (skin conductance responses, SCRs) signals, offering objective, continuous tracking of involuntary arousal not accessible through self-report. Numerous studies have investigated electrodermal activity responses during video-based emotional stimulation. It was demonstrated increased electrodermal activity for clips of both high-arousal positive and negative valence compared to low-arousal clips, highlighting the modality’s sensitivity to emotional intensity. In another study, electrodermal activity showed increased intersubject correlation during fearful and amusing videos, suggesting consistent sympathetic responses, while relaxing videos showed decreased electrodermal activity synchrony, indicating more varied emotional experiences.
Electrodermal activity has also been linked to balance, postural challenges, threat, and fall-related anxiety. It was found earlier that tonic electrodermal activity (skin conductance level, SCL) correlate with sway in anxious individuals during quiet standing. Likewise, elevated tonic electrodermal activity was reported when participants stood at height or on narrow surfaces, conditions that simulate postural threat. For example, unperturbed standing in simulated and real heights. Unexpected perturbations have a dramatic effect on Sympathetic Nervous System activity. Previous studies found clear phasic spikes in response to balance-related tasks in perturbed standing, walking on uneven surfaces and climbing stairs in stroke survivors and people with amputation, suggesting electrodermal activity effectively indexes physiological arousal tied to perceived balance risk linked to arousal, attention. Recently, a laboratory induced balance loss during perturbed walking triggers phasic electrodermal activity elevation among young, and this response habituates after a series of 6 unannounced balance perturbations.
While prior research has established electrodermal activities’ sensitivity to emotional viewing and balance-related stress, little is known about how people physiologically respond to video observation of their own balance loss. Specifically, it is still scarce whether observing one’s own fall on video elicits physiological arousal, as indicated by a phasic increase in electrodermal activity, indicated by skin conductance level (SCL), and whether it habituates after a continuous exposure. In this study, we presented young adults with video clips of their own treadmill walking trials, including moments of balance losses and instability, while continuously monitoring electrodermal activity. This approach aims to evaluate whether: 1) Phasic electrodermal activity (skin conductance responses, SCRs) reliably spikes during emotionally or threat-relevant content; 2) Tonic electrodermal activity (skin conductance level, SCL) reflects residual arousal or worry across longer viewing periods. This electrodermal reactivity mirrors that seen in real-world balance challenges, suggesting a lab-based proxy for anxiety. By integrating media-driven emotional arousal with balance-specific electrodermal activity, our study seeks to uncover whether as-though movements after postural threats viewed via video can produce physiological signatures. We hypothesize that 1) there would be a phasic electrodermal activity (skin conductance responses, SCRs) response immediately post visual threatening condition; 2) The tonic electrodermal activity (skin conductance level, SCL) will gradually decrease during the observations, suggesting habituation.
2. Methods
2.1. PARTICIPANTS
A total of 33 healthy young adults (19 women and 14 men, average age 27.2 years) were recruited between February 2018 and May 2020 from the Faculty of Health Sciences at Ben-Gurion University of the Negev, Israel. Eligible individuals were between 20 and 40 years old. Volunteers were excluded if they had a history of cardiovascular conditions, implanted cardiac devices, or were taking medications known to influence autonomic function or heart rate, systolic blood pressure outside the 120–160mmHg range, resting heart rate above 100bpm, any diagnosed neurological, peripheral neuropathy, osteoporosis, unexplained dizziness or syncope. All participants provided written informed consent prior to participation. The study protocol was reviewed and approved by the Helsinki Committee of Soroka University Medical Center (Approval No. 0396-16-SOR).
2.2. EQUIPMENT AND EXPERIMENTAL PROCEDURE
EDA was recorded via standard finger electrodes during participants’ own video-viewing of treadmill-induced balance losses. Electrodermal activity was collected using the Biopac MP160 system under controlled lab conditions. Participants had previously experienced real-time perturbations while walking on a treadmill platform inducing unannounced balance challenges. For safety, participants wore a ceiling-mounted harness that allowed full mobility while preventing falls. Video clips shown included moments of their own slips or instability. The study included two conditions: (1) participants walked on a perturbation treadmill while experiencing six unexpected balance disturbances, and (2) later viewed video clips of their own balance loss. During the 6-minute walking task, participants responded naturally to platform translations (18 cm displacement, 1.2 m/s velocity, 3 m/s² acceleration) triggered semi-randomly every 45–60 seconds at heel contact, using a real-time MATLAB algorithm based on center of pressure (MathWorks, Cambridge, MA). Perturbation timing was synchronized with EDA recordings via the Biopac system (Biopac Systems Inc., Goleta, CA, USA).
2.3. DATA COLLECTION AND ANALYSIS
Electrodermal activity signals were acquired continuously during the video observation using a Biopac MP160 system and recorded at a sampling rate of 1,000 Hz.
Raw electrodermal activity signals, displayed in micro-Siemens (µS), were examined using AcqKnowledge software version 5.1 (Biopac Systems Inc., Goleta, CA, USA). Electrodermal activity signal captured in real-time, built-in smoothing high-pass filters: Attenuates frequencies below the cutoff, within the software were applied to improve signal clarity and reduce high-frequency noise. To assess electrodermal activity responses to the video observation perturbation events, three time-locked metrics were derived for each perturbation episode:
- Baseline skin conductance level (pre-perturbation): The average SCL during the 5 seconds immediately preceding the perturbation, used as a measure of tonic Sympathetic Nervous system activity during steady-state video observation of unperturbed walking.
- Peak SCL (post-perturbation): The maximum SCL value observed following the onset of the video observation of unannounced perturbations, reflecting the peak of the phasic response.
- Skin Conductance Response (SCR): Calculated as the difference between the peak post video observation of perturbation SCL and the baseline SCL. An SCR was considered present when the post video observation perturbation increases in SCL exceeded two standard deviations above the baseline mean within a 2–5 second window following the perturbation. This threshold-based criterion of 2 Standard Deviations (SD) above the mean: Threshold=μ+2σ: μ = mean of the signal/distribution; σ = standard deviation (SD) which is commonly used to identify event-specific autonomic responses.
To account for variability in individual baseline skin conductance, we computed a relative response ratio by dividing the SCR amplitude by the corresponding baseline SCL value during video observation of unperturbed, yielding an SCR/SCL (%) metric. This percentage was then log-transformed to approximate a normal distribution for subsequent statistical analysis. This ratio provides a standardized index of response intensity by expressing the phasic activation (SCR) as a proportion of the underlying tonic level (SCL). Mathematically, for each video observation of the perturbation event Pi, the SCR amplitude was defined as:
ΔSCLPi (%) = SCR ampPi (µS) / SCL PrePi(µS) × 100
Each Pi represents an individual video observation of unannounced perturbation event. The SCR amplitude refers to the absolute change in skin conductance level following the video observation of perturbation, while the pre-perturbation SCL denotes the mean value over the 5-second period prior to the video observed perturbation onset. Both the raw SCR amplitude and the normalized SCR/SCL (%) ratio were used to quantify the magnitude of phasic Sympathetic Nervous System activation in response to video observation of balance loss. In contrast, the average SCL values recorded before the video observation of perturbation served as indicators of the tonic (baseline) level of Sympathetic Nervous System activity during video observed unperturbed walking.
2.4. STATISTICAL ANALYSIS
Statistical analyses were performed using Predictive Analytics Software (PASW, IBM Corp., Armonk, NY, USA). The variables were normally distributed (Shapiro-Wilk test), thus were analysed using a linear mixed effect model. To test whether video observation of one’s own loss of balance evoke phasic sympathetic response, electrodermal activity was compared before (Baseline skin conductance level pre-perturbation) and after each of the six-video observation (Peak SCL post-perturbation) of one’s own balance perturbations during walking. In addition, we used a linear mixed effect regression model with the subjects as a random effect to test if there was a trend in the relative rise of SCL (i.e., SCRs in %) across repeated perturbations. The 0.05 level was used for significance.
3. Results
In the experiment, all participants were exposed to 6 unannounced balance loss and performed balance reactive stepping responses to avoid a fall and restore stability following each perturbation. It must be noted that balance was regained without falling within approximately four-five steps after the onset of perturbations (2-3 seconds post perturbation).
| # Perturbation: Observed | Pre-perturbation SCL (µS) | Post-perturbation SCL (µS) | Average SCR (µS)/ SCL Delta | Relative SCL delta (SCR / SCL in %) |
|---|---|---|---|---|
| 1st perturbation | 4.05 ± 3.27 | 4.44 ± 3.66 | 0.38 ± 0.41 | 8% ± 6 |
| 2nd perturbation | 4.01 ± 3.34 | 4.32 ± 3.70 | 0.31 ± 0.52 | 5% ± 7 |
| 3rd perturbation | 3.87 ± 3.37 | 4.01 ± 3.50 | 0.14 ± 0.28 * | 3% ± 5* |
| 4th perturbation | 3.63 ± 3.41 | 3.76 ± 3.54 | 0.13 ± 0.19 * | 3% ± 6 * |
| 5th perturbation | 3.53 ± 3.29 | 3.66 ± 3.42 | 0.13 ± 0.22 * | 4% ± 6 * |
| 6th perturbation | 3.69 ± 3.39 | 3.78 ± 3.42 | 0.10 ± 0.18 * | 3% ± 5* |
| Average value of all perturbations | 3.80 ± 3.35 | 3.99 ± 3.54 | 0.19 ± 0.18 | – |
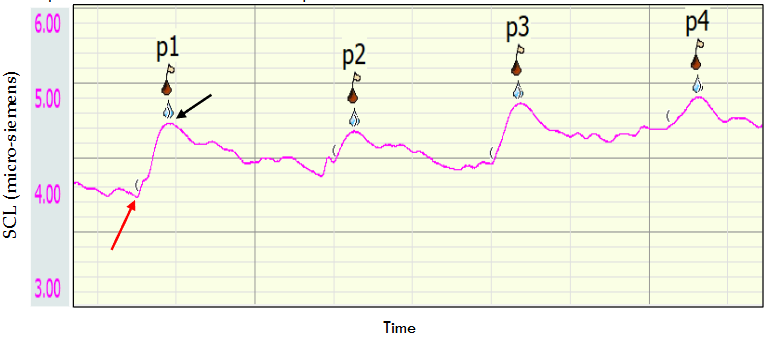
Figure 1: An Example of the Electrodermal Activity raw data received during a typical experiment. The pink line represents the skin conductance (in micro-siemens units). Each red flag represents the peak value of the response for each perturbation, and its serial number. The red and black arrows point at the skin conductance levels pre and post perturbation respectively. The delta between pre and post levels is the Skin Conductance Response.
THE SKIN CONDUCTANCE LEVEL–THE PHASIC COMPONENT.
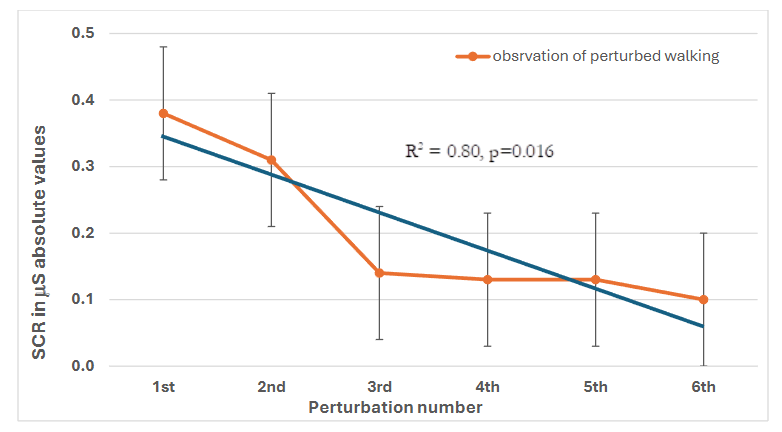
A significant increase in the average value of SCL was found before (pre) and immediately after (peak SCL post-perturbation) each of the six perturbation events with an average value of all perturbations 3.80 ± 3.35 µS vs. 3.99 ± 3.54 µS (p < 0.05, Table 1). A progressive decrease was observed in skin conductance response (mean SCR values ± SD) across the six perturbations, from 0.38 ± 0.41 µS, in the first perturbation to 0.10 ± 0.18 µS in perturbation 6 (see Average SCR (µS)/SCL Delta, Table 1). A linear regression analysis revealed a significant negative trend in SCR values across perturbations (R² = 0.80, p = 0.016, see Figure 2), further indicating a consistent attenuation in physiological arousal, i.e., mean SCR, or threat response over repeated exposures to balance perturbations.
Figure 2: Linear regression values of relative change in Skin Conductance Level. The absolute change in skin conductance level (SCL, µS), calculated as post- minus pre-perturbation values following observation of six self-experienced balance losses. The change in SCL progressively decreased across repeated perturbations, indicating habituation (R² = 0.80, p = 0.016).
RELATIVE SKIN CONDUCTANCE LEVEL
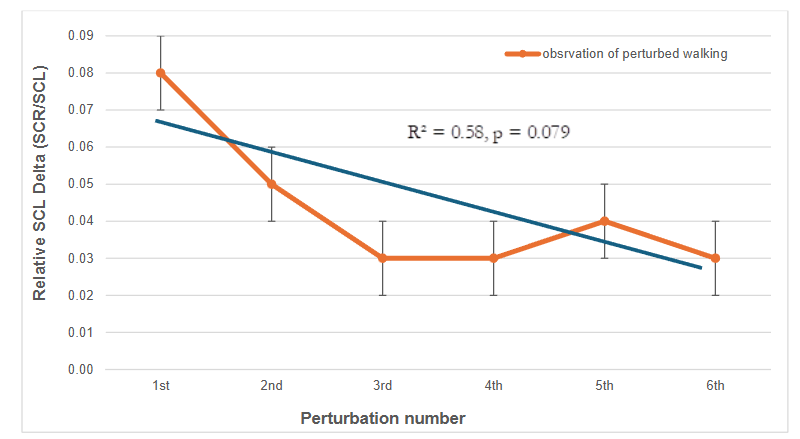
Relative changes in skin conductance level (SCL), expressed as SCR/SCL (%), showed a modest decline over the six perturbations. The mean values ± SD of the first perturbation 1 (8% ± 6) significantly differ from each of the subsequent perturbations (see Relative SCL delta (SCR / SCL in %, table 1). Linear regression indicated a decreasing trend in the relative SCL delta, though the relationship did not reach statistical significance (R² = 0.58, p = 0.079, Figure 3), suggesting a possible habituation effect.
Figure 3: Linear regression values of Relative Skin Conductance Level Delta. Relative SCL change (SCR/SCL, in %) across repeated six perturbations. Linear regression analysis revealed a non-significant trend of a decrease in relative autonomic arousal across perturbations (R² = 0.58, p = 0.079).
4. Discussion
Our findings show the robust impact of watching videos of one’s own balance-loss events on electrodermal activity, evident in a phasic raise in SCR. Across the six perturbations, SCR peaked at 0.38±0.41µS for the first video observation and declined steadily to 0.10±0.18µS by the sixth perturbation, reflecting rapid autonomic habituation. The electrodermal activity analyses revealed a significant decline in absolute SCR amplitude across perturbations (R²=0.80, p=0.016), and in relative SCL delta (SCR/SCL %) (R²=0.58, p =0.079). This aligns well with prior studies using emotionally evocative videos. Together, these findings support the notion that repeated exposure and cognitive appraisal contribute to reduced emotional and physiological responsiveness, aligning with habituation mechanisms observed in sympathetic nervous system responses. This suggests that the phasic electrodermal activity responses in our study were attributable to emotional response to the visual observation of balance loss, rather than actual physical instability. Together, these results suggest a rapid electrodermal activity response (SCR) as well as rapid habituation of the electrodermal activity response, a robust decline in absolute SCR amplitudes in the relative metrics (SCR/SCL%) over successive perturbations, indicating physiological and emotional adaptation to repeated viewing of video recordings depicting one’s own balance-loss events. This slope was significantly and negatively correlated with the visual observation of participants’ own balance loss, and was characterized by a steeper habituation pattern (R²=0.80, p=0.016) compared to previous findings, in which the participants exhibited a shallower habituation slope in response to a “real” treadmill-induced unexpected balance losses (R²=0.094, p = 0.00115). Our main finding reveals a link between the habituation of SCR to repeated observations of balance loss, suggesting that the events were cognitively and emotional appraised and quickly identified as non-threatening. This cognitive recognition likely led to a rapid reduction in perceived risk, emotionally stress response and a corresponding decline in physiological arousal. During the real balance loss condition however, all six perturbations were consistently perceived as threatening, with a less steep habituation in the sympathetic response.
Our results align with prior evidence of diminished SCR over repeated exposures to aversive stimuli (e.g., startle noises) in healthy individuals. The researcher demonstrated that repeated exposure to the same stressor resulted in a progressive reduction in electrodermal responses, consistent with classical habituation of sympathetic nervous system activity. Importantly, individuals who showed faster and more pronounced habituation also exhibited psychological traits associated with greater stress resilience, such as lower anxiety and better emotional regulation. In general the research on emotional stimuli presented via video has demonstrated that SCR amplitude significantly declines upon repeated exposure, reflecting an initial rise in autonomic arousal that diminishes as the stimuli become familiar. Studies involving facial expressions and emotion-related words reported an exponential decay in SCR amplitude across successive presentations, indicating strong initial engagement that habituates over time. Research into emotional video stimuli has demonstrated that SCR amplitude significantly decreases with repeated exposure, reflecting strong initial autonomic engagement that attenuates as novelty and arousal diminish.
Importantly, our findings extend this habituation phenomenon to a novel context self-observation of one’s own loss-of-balance events. Even though participants viewed personally relevant visual stimuli, their physiological responses habituated within six exposures, supporting the notion that memory retrieval alone are insufficient to sustain high sympathetic arousal, and that cognitive appraisal, may be the core of adaptations when the repetition of the observed loss of balance diminishes the perceived threat over time.
The present work contributes meaningfully to both electrodermal activity methodology and fall-related Sympathetic Nervous System research. The current study confirms that observed video-based stimuli of balance threatening conditions specifically self-balance loss are sufficient to elicit small yet significant SCR and SCL responses, positioning them as valid proxies for threat exposure research. While previous studies demonstrated increased electrodermal activity during postural perturbations, our findings show that, compared to real balance loss during walking, observing one’s own perturbations elicits a similar but significantly smaller arousal response (0.96±0.7 vs. 0.19±0.18). This reduction likely reflects the absence of actual physical performance demands during real-life balance loss, yet still indicates a small but significant emotional response, particularly during the first observation trial. These studies show that SCRs are highly sensitive to postural perturbations and that the responses are context-dependent threatening stimuli to the individual. It is therefore suggested that the sympathetic response is not triggered only by afferent peripheral sensory input, but rather by the emotional content and the perceived challenge. A review presents evidence of the associations between emotion-related areas in the brain (i.e., the limbic system, anterior cingulate) and electrodermal activity, further establishing the notion that emotions affect the SCL and very much influenced by context, emotion, and higher cortical areas, rather than “simple” afferent sensory input. The habituation trajectory also mirrors that observed with whole-body perturbations in real or simulated falls, suggesting that psychological threat attenuates rapidly with repetition.
5. Conclusions
While extensive research has examined electrodermal activity responses to balance-challenging situations during quiet standing, perturbed sitting, and perturbed standing and walking, consistently showing that externally induced perturbations elicit robust sympathetic responses. The present study demonstrates that observing one’s own balance-loss events also evokes sympathetic activation. Specifically, self-observation elicited clear phasic skin conductance responses and relative increases in skin conductance level, indicative of sympathetic arousal. Moreover, repeated exposures resulted in significant habituation of SCR amplitude and a trend-level decline in relative SCL change, consistent with established electrodermal activity habituation literature.
Statements and Declarations
CONFLICTS OF INTERESTS
No conflicts of interest, financial or otherwise, are declared by the authors.
ETHICAL STANDARD STATEMENT
This study was approved by the Helsinki ethics committee (Soroka University Medical Center, Beersheva, Israel. Approval number 0396-16-SOR). Prior to participation all subjects provided an informed consent.
SUBMISSION DECLARATION
This manuscript has not been published before and is not under consideration of publishment elsewhere.
AUTHOR CONTRIBUTION
G.M., A.K., and I.M. conceived and designed research; G.M., and A.O. performed experiments; G.M., A.O., A.R.-B., and I.M. analysed data; G.M., A.O, A.K., and I.M. interpreted results of experiments; G.M. and I.M. prepared figures; G.M., A.O, A.K., and I.M. drafted manuscript; G.M., A.K., A.R.-B., and I.M. approved final version of manuscript.
ROLE OF THE FUNDING SOURCE
N/A
CONSENT FOR PUBLICATION
All authors provide consent for publication.
DATA AVAILABILITY
Data will be made available upon reasonable request.
5. References
- Boucsein W, Fowles DC, Grimnes S, Ben-Shakhar G, Roth WT, Dawson ME, Filion DL; Society for Psychophysiological Research Ad Hoc Committee on Electrodermal Measures. Publication recommendations for electrodermal measurements. Psychophysiology. 2012;49:1017–34.
- Ellaway PH, Kuppuswamy A, Nicotra A, Mathias CJ. Sweat production and the sympathetic skin response: improving the clinical assessment of autonomic function. Auton Neurosci. 2010;155:109–14.
- Posada-Quintero HF, Florian JP, Orjuela-Cañón AD, Chon KH. Electrodermal activity is sensitive to cognitive stress under water. Front Physiol. 2018;8:1128.
- Aguado L, Fernández-Cahill M, Román FJ, Blanco I, de Echegaray J. Evaluative and psychophysiological responses to short film clips of different emotional content. J Psychophysiol. 2018;32(1):1–19.
- Kim I, Kim H, Kim J. Examining the consistency of continuous affect annotations and psychophysiological measures in response to emotional videos. Int J Psychophysiol. 2023;193:112242.
- Maki BE, McIlroy WE. Influence of arousal and attention on the control of postural sway. J Vestib Res. 1996;6:53–9.
- Sibley KM, Mochizuki G, Frank JS, McIlroy WE. The relationship between physiological arousal and cortical and autonomic responses to postural instability. Exp Brain Res. 2010;203:533–40.
- Sibley KM, Mochizuki G, Lakhani B, McIlroy WE. Autonomic contributions in postural control: a review of the evidence. Rev Neurosci. 2014;25:687–97.
- Cleworth TW, Horslen BC, Carpenter MG. Influence of real and virtual heights on standing balance. Gait Posture. 2012;36(2):172–6.
- Huffman JL, Horslen BC, Carpenter MG, Adkin AL. Does increased postural threat lead to more conscious control of posture? Gait Posture. 2009;30(4):528–32.
- Pollock CL, Carpenter MG, Hunt MA, Gallina A, Vieira TM, Ivanova TD, et al. Physiological arousal accompanying postural responses to external perturbations after stroke. Clin Neurophysiol. 2017;128(6):935–44.
- Clark DJ, Chatterjee SA, McGuirk TE, Porges EC, Fox EJ, Balasubramanian CK. Sympathetic nervous system activity measured by skin conductance quantifies the challenge of walking adaptability tasks after stroke. Gait Posture. 2018;60:148–53.
- Cho E, Wakeling JM, Pousett B, Pollock CL. Mapping of electrodermal activity (EDA) during outdoor community-level mobility tasks in individuals with lower-limb amputation. J Rehabil Assist Technol Eng. 2021;8:20556683211006837.
- Kawasaki T, Oda H, Sawaguchi Y, Kunimura H, Hiraoka K. Sympathetic response to postural perturbation in stance. Front Hum Neurosci. 2021;15:763582.
- Meir G, Katz A, Berdichevsky Y, Reiner-Benaim A, Melzer I. Sympathetic response following unannounced loss of balance during walking in young adults: laboratory study. J Neurophysiol. 2024;132(3):822–8.
- Boyd RL, Blackburn KG, Pennebaker JW. The narrative arc: revealing core narrative structures through text analysis. Sci Adv. 2020;6:eaba2196.
- Wise K, Alhabash S, Park H. Emotional responses during social information seeking on Facebook. Cyberpsychol Behav Soc Netw. 2010;13:555–62.
- Walker FR, Thomson A, Pfingst K, Vlemincx E, Aidman E, Nalivaiko E. Habituation of the electrodermal response – a biological correlate of resilience? PLoS One. 2019;14(1):e0210078.
- Okamoto S, Makieoka T, Tara I, Kosuge Y. Skin conductance response and habituation to emotional facial expressions and words. Acta Psychol (Amst). 2025;251:104573.
- Juuse L, Tamm D, Lõo K, Allik J, Kreegipuu K. Skin conductance response and habituation to emotional facial expressions and words. Acta Psychol (Amst). 2024;251:104573.
- Critchley HD. Electrodermal responses: What happens in the brain. Neuroscientist. 2002;8(2):132.
- Brown LA, Sleik RJ, Polych MA, Gage WH. Is the prioritization of postural control altered in conditions of postural threat in younger and older adults? J Gerontol A Biol Sci Med Sci. 2002;57(12):M785-M792.
- Sibley KM, Lakhani B, Mochizuki G, McIlroy WE. Perturbation-evoked electrodermal responses are sensitive to stimulus and context-dependent manipulations of task challenge. Neurosci Lett. 2010;485(3):217-221.
- Sibley KM, Mochizuki G, McIlroy WE. Perturbation-evoked electrodermal activity responds to instability, not just motor or sensory drives. Clin Neurophysiol. 2008;120(3):619-625.
- Kawasaki T, Oda H, Sawaguchi Y, Kunimura H, Hiraoka K. Sympathetic response to postural perturbation in stance. Front Hum Neurosci. 2021;15:763582.
- Sibley KM, Mochizuki G, Esposito JG, Camilleri JM, McIlroy WE. Phasic electrodermal responses associated with whole-body instability: presence and influence of expectation. Brain Res. 2008;1216:38-45.