MRI vs. MDCT in Diagnosing Abdominal Tuberculosis
3 Tesla MRIs Diagnostic Edge over MDCT Ushering a New Era in Abdominal Tuberculosis Imaging: Value of cross-sectional Analysis
Dr.(Prof.)Sachin Khanduri, Dr. Vibhor Dhingra, Dr. Akshay Aggarwal, Dr. Archiya Habib, Dr. Nisha, Dr. Nishat Ara, Dr. Mugheer Ahmad, Dr. Saddam Hussain, Dr. Siddiqua Abdullah, Dr. Namra Mahmood
OPEN ACCESS
PUBLISHED: 31 January 2025
CITATION: Khanduri, S., et al., 2025. 3 Tesla MRIs Diagnostic Edge over MDCT: Ushering a New Era in Abdominal Tuberculosis Imaging: “Value of Cross-Sectional Analysis”. Medical Research Archives, [online] 13(12).
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Background: Tuberculosis (TB) remains a global health challenge, especially in developing regions. Abdominal tuberculosis, a form of extrapulmonary TB, poses diagnostic difficulties due to nonspecific symptoms overlapping with other conditions. Imaging techniques like MDCT and MRI have become crucial tools for diagnosing abdominal TB. This study compares the diagnostic effectiveness of these two methods.
Objectives: To compare Multidetector Computed Tomography (MDCT) and Magnetic Resonance Imaging (MRI) findings in Abdominal Tuberculosis.
Materials and methods: A cross sectional study was carried out over a period of twenty-four months in the Department of Radiodiagnosis, in collaboration with Department of Medicine and Surgery. Patients who met the clinical criteria were clinically examined, and their age, sex and clinical profiles were noted. All the patients were then subjected to imaging evaluation using USG, MDCT and MRI to diagnose abdominal tuberculosis. Diagnostic efficacy of MDCT and MRI were analysed against final diagnosis using FNAC/Biopsy, Ascitic fluid AFB positivity, Ascitic fluid culture positivity and on basis of positive therapeutic response to ATT treatment.
Results: MDCT was 82.9% sensitive and 76.5% specific, while MRI was 88.6% sensitive and 82.4% specific in diagnosis abdominal tuberculosis. Diagnostic accuracy using MCDCT alone was found out to be ~ 80.8% and that using MRI alone was found out to be ~ 86.5%.
Conclusion: The study highlights that both MDCT and MRI are highly sensitive in diagnosis of abdominal tuberculosis but MRI has a diagnostic edge over MDCT in the same.
Keywords: Abdominal tuberculosis, Multidetector Computed Tomography, Magnetic Resonance Imaging, Extrapulmonary tuberculosis
INTRODUCTION
Tuberculosis (TB) has been a global health priority for the World Health Organization (WHO) since its inception in 1948. Despite continued efforts, TB remains a major health challenge, with an estimated 6-7 million new cases annually. During the COVID-19 pandemic, global TB incidence briefly declined, but post-lockdown cases surged, reaching 7.5 million in 2022. TB remains a leading cause of death worldwide, second only to COVID-19 in 2023.
Over the years, TB prevalence and mortality have increased, reflecting a persistent global challenge. For instance, there were 8.6 million new cases and 1.3 million deaths in 2012, and by 2015, these numbers rose to 10.8 million cases and 1.8 million deaths.
This steady rise raises concerns about the effectiveness of current strategies to curb TB. Early diagnosis and adherence to treatment are crucial to reducing TB’s impact, as untreated TB patients can infect 10-15 others.
The rise of drug-resistant TB is particularly concerning, with approximately 450,000 new cases of drug-resistant TB reported in 2021. Countries like China, India, and Russia accounted for nearly half of these cases. While pulmonary TB remains the most common form, extrapulmonary TB (EPTB) affects around one in six TB patients, with abdominal TB being a common subtype. Abdominal TB can affect several gastrointestinal organs, including the intestines, peritoneum, and liver, accounting for 3-5% of all TB cases globally and 11-16% of EPTB cases.
This condition is more prevalent in developing regions, such as South Asia and Sub-Saharan Africa, where factors like poverty, malnutrition, and HIV co-infection contribute to the TB burden.
Abdominal TB can cause complications like intestinal obstruction, perforation, and abscess formation, often requiring surgical intervention. Diagnosing abdominal TB early is critical for managing such complications. However, the nonspecific symptoms of abdominal TB such as abdominal pain, weight loss, and fever can delay diagnosis since they overlap with other gastrointestinal disorders, such as Crohn’s disease. Diagnosis relies on clinical evaluation and the integration of laboratory tests, imaging techniques, molecular diagnostics, and histopathological analysis. While laboratory tests often reveal nonspecific signs, imaging plays a pivotal role in confirming the diagnosis.
Ultrasound is a first-line imaging tool for abdominal TB, detecting ascites, lymphadenopathy, and bowel thickening. It is also valuable for guiding biopsies. Computed Tomography (CT) offers detailed imaging for identifying lymphadenopathy, bowel involvement, and complications such as abscesses. Magnetic Resonance Imaging (MRI) is superior for soft tissue contrast and lacks ionizing radiation, making it suitable for use in children and pregnant women. MRI is particularly effective for assessing abdominal masses and disease extent in abdominal TB cases. Recent advancements in multidetector CT (MDCT) and MRI have significantly improved the accuracy of diagnosing abdominal TB. MDCT provides high-resolution imaging, crucial for evaluating disease extent and guiding further interventions. MRI excels in soft tissue characterization, making it invaluable for evaluating lymph nodes, bowel involvement, peritoneal disease, and solid organ lesions.
Given the increasing importance of MDCT and MRI in diagnosing abdominal tuberculosis, this study aims to compare the effectiveness of these imaging modalities in managing abdominal TB.
AIM
To evaluate and compare the diagnostic utility of 3 Tesla Magnetic Resonance Imaging (MRI) and Multidetector Computed Tomography (MDCT) in the detection and assessment of abdominal tuberculosis, aiming to identify the most effective imaging modality for accurate diagnosis and clinical management.
OBJECTIVES
To systematically compare the diagnostic performance of Multi-detector Computed Tomography (MDCT) and 3 Tesla Magnetic Resonance Imaging (MRI) in detecting abdominal tuberculosis, using cross-sectional imaging analysis and evaluating sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and overall accuracy.
MATERIAL & METHODS
A cross-sectional study was conducted at Department of Radiodiagnosis in collaboration with departments of Medicine and Surgery for a duration of twenty-four months from May, 2022 to May, 2024. Clearance for carrying out the study was obtained from the Institutional Ethical Committee of tertiary care centre (approval letter numbered R-Cell 2023/60) and an informed consent was obtained from all the patients.
The sampling frame was bound by the following inclusion criteria:
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| All patients referred from Medicine & Surgery department for suspected abdominal tuberculosis on the basis of clinical signs and symptoms, ascitic fluid examination, chemotherapy response to ATT, biopsy proven abdominal tuberculosis. | Allergic to iodinated dye |
| Deranged kidney function test | |
| Patient with known malignancy | |
| Critically ill patient | |
| Pregnancy |
The sample size was calculated at Department of Social & Preventive Medicine using following formula:
n = [DEFF*Np(1-p)]/ [(d2/Z21-α/2*(N-1)+p*(1-p)] where
- n = Sample size
- DEFF = Design effect (for cluster surveys = 1)
- N = Population size (for finite population correction factor or fpc)(N): 1,000,000
- P = Estimated proportion (Percentage of Abdominal TB cases in cases of extra-pulmonary tuberculosis (EPTB) (p): 16±10)
- Q = 1-p
- D = desired absolute precision or absolute level of precision
- z = level of confidence according to the standard normal distribution (for a level of confidence of 95, z = 1.96)
- Confidence limits as of 100 (d): 10
Considering 95 confidence interval, total calculated sample size is 52.
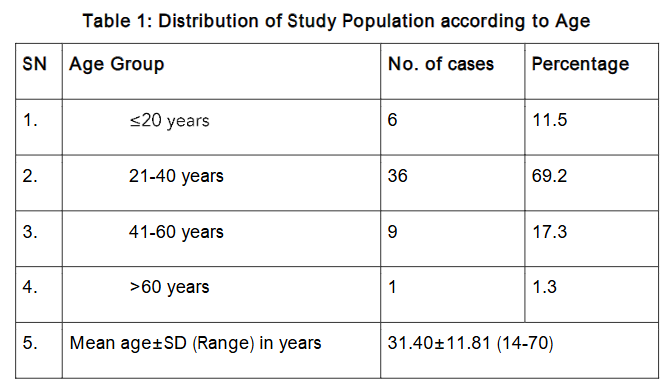
Methodology: Fifty-two patients falling in sampling frame and fulfilling the eligibility criteria were clinically examined and demographic details, nature of presenting complaints, medical history, history of ATT intake, personal habits and family history of tuberculosis were noted. Patients were classified according to their age and sex. As far as age and sex profile is concerned, in the present study, our patients were age range 14 to 70 years and had a dominance of male.
All the patients were then subjected to imaging evaluation using USG, MDCT and MRI to diagnose abdominal tuberculosis. All CT scans were performed on a 384 slice Dual Energy CT scanner (Somatom Force, Seimens Healthcare), MRI scans with a 3T MR scanner (Magnetom Vida, Seimens Healthcare), and all the images were post processed on a work station using Syngovia software that allows analysis of images using three material decomposition. Subjective assessment of images of both MDCT and MRI were assessed by two experienced radiologists to prepare the CT diagnosis. Mutual agreement of the two observers was considered as final. In case of a disagreement, it was referred to the third experienced radiologist and decision made by the third radiologist was considered as final.
Data Analysis: The statistical analysis was done using SPSS (Statistical Package for Social Sciences) Version 21.0 statistical Analysis Software. The values were represented in Number (%), Mean±SD and Median.
RESULT
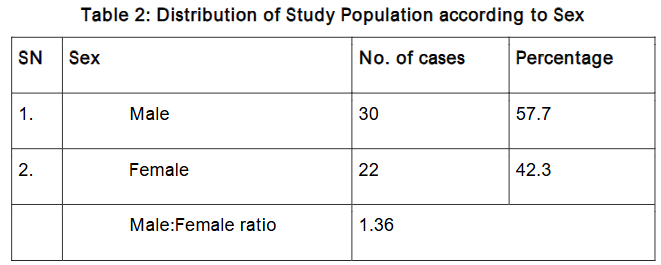
The study enrolled a total of 52 patients presenting with suspected abdominal tuberculosis. Among the most frequently reported symptoms were abdominal colicky pain (80.8%), significant weight loss (76.9%), fever (65.4%), night sweats (63.5%), and anemia (59.6%). Vomiting was documented in 53.8% of the cases, indicating the varied clinical presentation of abdominal TB. Furthermore, risk factors contributing to the disease included tobacco use (28.8%), alcohol consumption (23.1%), a previous history of pulmonary tuberculosis (21.2%), and a family history of tuberculosis (23.1%).
The final diagnosis was based on the basis of positive therapeutic response to ATT treatment and/or a positive AFB culture, positive biopsy and/or FNAC. The data so collected was fed into computer using Microsoft Excel 2013 software and was subjected to statistical analysis.
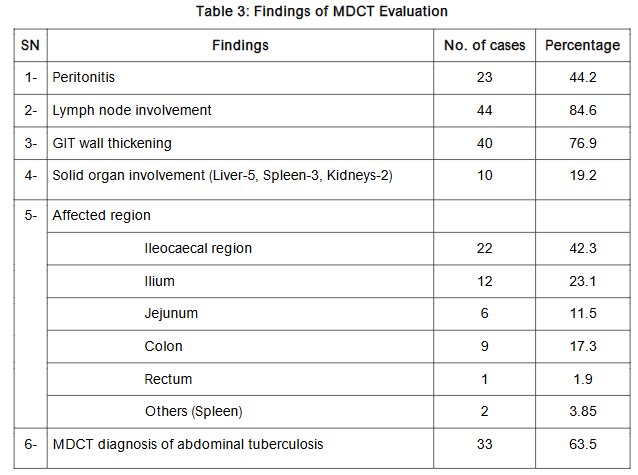
Multidetector Computed Tomography (MDCT) findings revealed lymph node involvement in a substantial 84.6% of patients, gastrointestinal (GIT) wall thickening in 76.9%, and peritonitis in 44.2%. Notably, solid organ involvement was observed in 19.2% of cases, affecting the liver, spleen, and kidneys. The ileocecal region emerged as the most frequently involved area (42.3%), followed by the ileum (23.1%) and colon (17.3%). MDCT diagnosed abdominal TB in 63.5% of patients, yielding a sensitivity of 82.9%, specificity of 76.5%, and an overall diagnostic accuracy of 80.8%.
| MDCT Diagnosis | Final Diagnosis | Total |
|---|---|---|
| Abdominal TB | 29 | 33 |
| No Abdominal TB | 6 | 19 |
| Total | 35 | 52 |
Sensitivity (%) Specificity (%) PPV (%) NPV (%)
82.9 76.5 87.9 68.4
Accuracy 80.8%
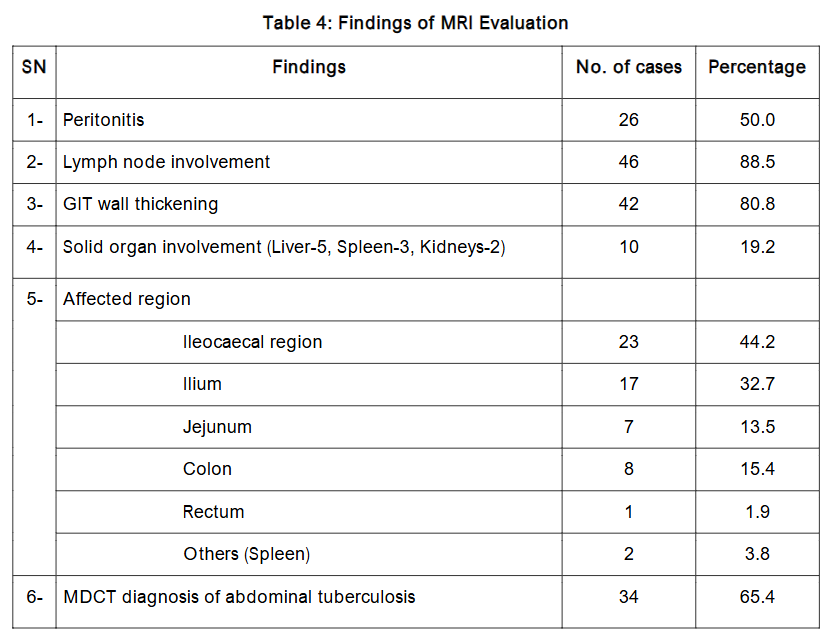
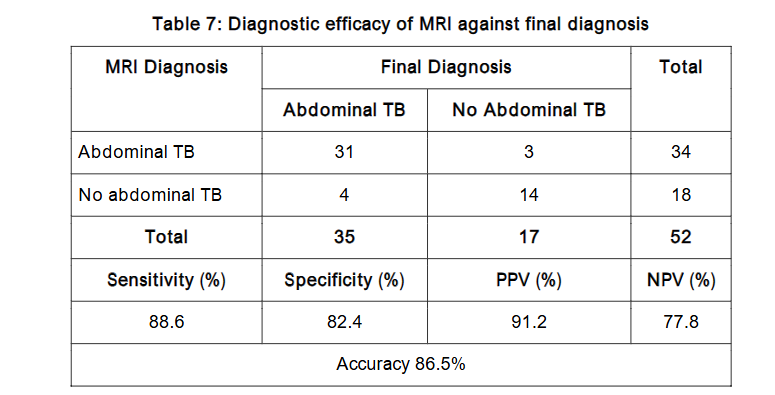
Conversely, Magnetic Resonance Imaging (MRI) demonstrated superior diagnostic performance compared to MDCT. MRI identified lymph node involvement in 88.5% of cases, GIT wall thickening in 80.8%, and peritonitis in 50%. It successfully diagnosed abdominal TB in 65.4% of patients, achieving a sensitivity of 88.6%, specificity of 82.4%, and overall accuracy of 86.5%.
| MRI Diagnosis | Final Diagnosis | Total |
|---|---|---|
| Abdominal TB | 31 | 34 |
| No Abdominal TB | 4 | 18 |
| Total | 35 | 52 |
Sensitivity (%) Specificity (%) PPV (%) NPV (%)
88.6 82.4 91.2 77.8
Accuracy 86.5%
This study emphasizes the crucial role of accurate imaging in diagnosing abdominal TB. While both imaging modalities were effective, MRI consistently outperformed MDCT in terms of sensitivity and diagnostic accuracy. As such, MRI is positioned as a more reliable and effective tool for the assessment and management of abdominal tuberculosis, enabling healthcare providers to make informed treatment decisions and ultimately improving patient outcomes. These findings advocate for the integration of MRI as a standard practice in the timely diagnosis and management of abdominal TB.
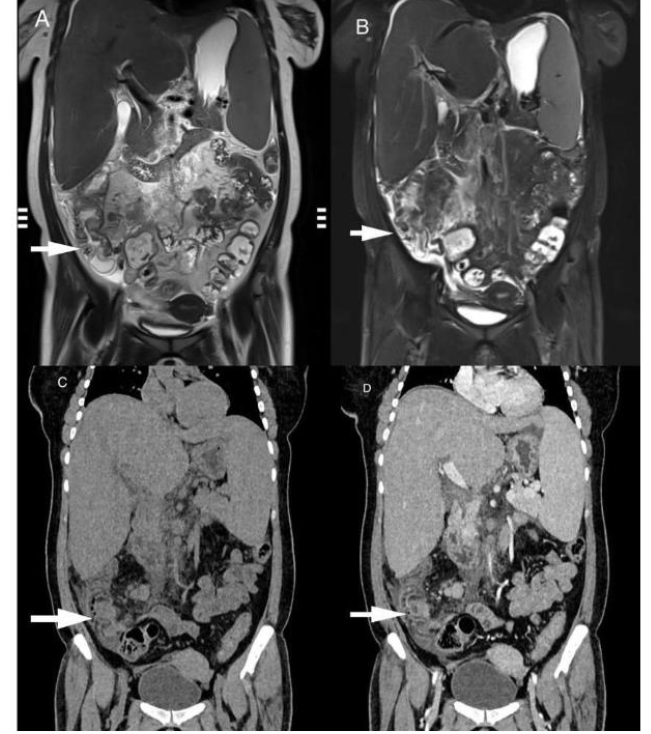
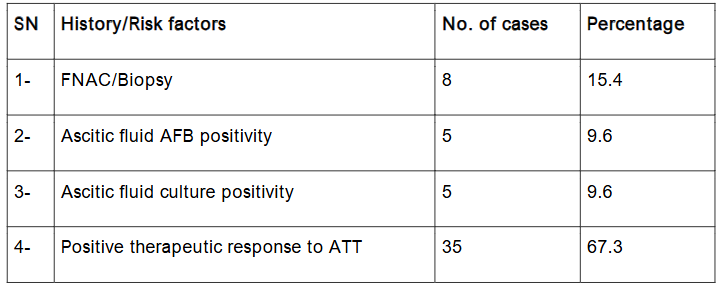
Figure 1: T2W coronal images without and with FS (A, B), and CT coronal images pre and post contrast (C, D), reveal circumferential mural thickening of caecum, ascending colon and ileocecal region (white arrow), showing homogeneous enhancement on CT coronal post contrast image.
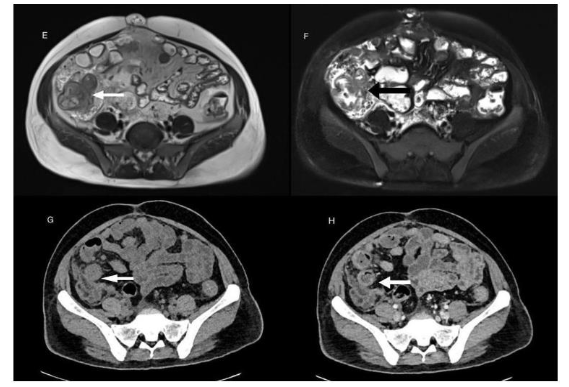
Figure 2: T2W axial images with and without FS (E, F), and CT axial images pre and post contrast (G, H), reveal circumferential mural thickening of terminal ileum, caecum, and ileocecal region (arrow), showing homogeneous enhancement on CT axial post contrast image.
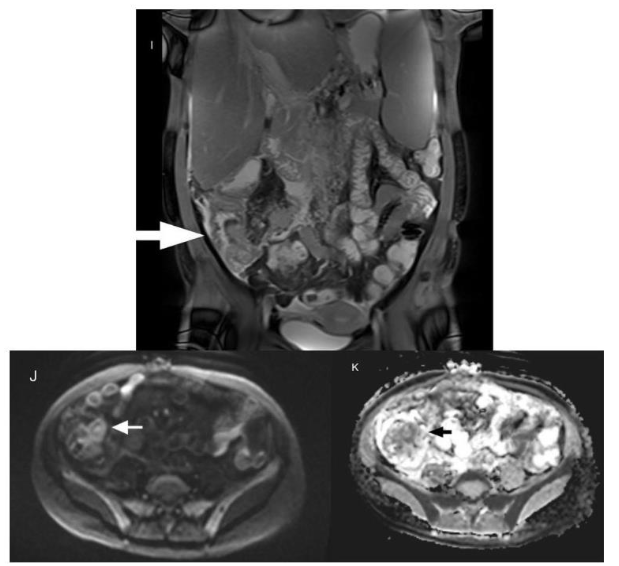
Figure 3: T2W coronal image (I) reveals circumferential mural thickening of ileocecal region showing homogeneous enhancement (white arrow). The ileocecal region (white arrow) appears bright on high b value Diffusion image (J) and appears dark (black arrow) on ADC maps (K) s/o diffusion restriction.
This study emphasizes the crucial role of accurate imaging in diagnosing abdominal TB. While both imaging modalities were effective, MRI consistently outperformed MDCT in terms of sensitivity and diagnostic accuracy. As such, MRI is positioned as a more reliable and effective tool for the assessment and management of abdominal tuberculosis, enabling healthcare providers to make informed treatment decisions and ultimately improving patient outcomes. These findings advocate for the integration of MRI as a standard practice in the timely diagnosis and management of abdominal TB.
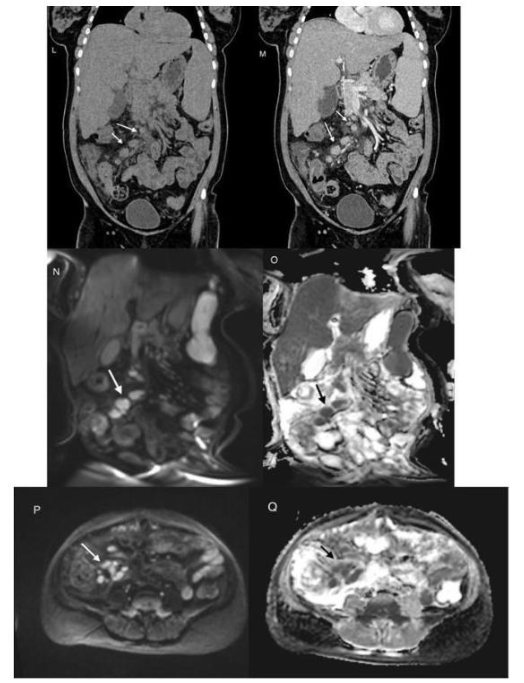
Figure 4: CT coronal pre-contrast (L) and post contrast images show multiple discrete and confluent lymph nodes (arrow) showing homogeneous enhancement on post contrast image.
Note that the nodes are well appreciated on diffusion-weighted coronal (N) and axial (P) images as bright round-ovoid lesions (white arrow) with corresponding drop in signal on ADC maps (O and Q), consistent with diffusion restriction.
DISCUSSION
Diagnosing abdominal tuberculosis is difficult due to nonspecific symptoms like abdominal pain, weight loss, and fever, which overlap with conditions such as Crohn’s disease and colorectal cancer. Histopathology is key but often invasive, and diagnostic yields from AFB staining and mycobacterial culture are low due to the disease’s paucibacillary nature. Molecular techniques, like PCR, offer better sensitivity but are costly and not universally available.
Imaging, including MDCT and MRI, plays a critical role, but features such as ascites, bowel thickening, and lymphadenopathy can mimic other diseases. MDCT allows detailed imaging and reconstruction, while MRI provides superior soft tissue contrast and avoids radiation exposure. Both modalities have strengths but are not 100% accurate. MRI is increasingly favored, particularly for its ability to avoid unnecessary radiation, especially in children and pregnant women, though more research is needed to confirm its superiority as it becomes faster and more affordable. The 3T field strength significantly increases the Signal-to-Noise Ratio (SNR), allowing for higher spatial resolution and the detection of subtle peritoneal implants and bowel wall thickening that may be volume-averaged and missed on MDCT.
This study compared Multidetector Computed Tomography (MDCT) and Magnetic Resonance Imaging (MRI) in diagnosing abdominal tuberculosis in 52 patients. Key symptoms included colicky pain (80.8%), weight loss (76.9%), and fever (65.4%). MDCT confirmed tuberculosis in 63.5% of cases, while MRI confirmed 65.4%. MRI was more sensitive (88.6%) and specific (82.4%) than MDCT. Both imaging techniques were effective, but MRI had better diagnostic performance, particularly in soft tissue contrast, and is safer for young patients needing multiple follow-ups and pregnant females due to the absence of ionising radiation exposure.
While the cost and scan duration of MRI are often cited as barriers in developing nations, the cost-effectiveness of accurate early diagnosis must be considered. Misdiagnosing TB as Crohn’s disease can lead to the inappropriate use of immunosuppressants, which can be catastrophic. The higher Positive Predictive Value (PPV) of MRI (91.2%) reduces the need for unnecessary invasive surgeries or equivocal therapeutic trials. Furthermore, modern 3T systems with parallel imaging techniques have significantly reduced acquisition times, making the modality more feasible for high-volume centers.
Limitations of this study include the relatively small sample size and the reliance on therapeutic response as a gold standard in a subset of patients, which is a common constraint in TB research due to the difficulty of obtaining biopsy samples from deep abdominal sites. Larger, multi-center studies are recommended to further validate the quantitative ADC cut-offs for differentiating tubercular from reactive etiologies.
REFERENCES
- Raviglione M, Pio A. Evolution of WHO policies for tuberculosis control, 1948 2001. The Lancet. 2002;359(9308):775-780. doi:10.1016/s0140-6736(02)07880-7
- Global Tuberculosis Report 2022. https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2022. Published October 27, 2022.
- Global Tuberculosis Report 2023. https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2023. Published November 7, 2023.
- World Health Organization: WHO. Treatment of tuberculosis: guidelines for national programmes. Treatment of Tuberculosis: Guidelines for National Programmes. https://www.who.int/news/item/07-05-2010-treatment-of-tuberculosis-guidelines-for-national-programme. Published May 7, 2010.
- Global Programme on Tuberculosis and Lung Health (GTB). Global tuberculosis report 2014. https://www.who.int/publications/i/item/9789241564809. Published October 23, 2014.
- Global Programme on Tuberculosis and Lung Health (GTB). Global tuberculosis report 2016. https://www.who.int/publications/i/item/9789241565394. Published December 10, 2016.
- Park K. Tuberculosis. In: Park’s Text Book of Preventive and Social Medicine. 20th ed. Jabalpur (M.P.), India: M/s Banarsidas Bhanot; 2013:159-175.
- Global Programme on Tuberculosis and Lung Health (GTB). Global tuberculosis report 2020. https://www.who.int/publications/i/item/9789240013131. Published October 15, 2020.
- Chu P, Chang Y, Zhang X, et al. Epidemiology of extrapulmonary tuberculosis among pediatric inpatients in mainland China: a descriptive, multicenter study. Emerging Microbes & Infections. 2022;11(1):1090-1102. doi:10.1080/22221751.2022.2054367
- Tobin EH, Khatri AM. Abdominal Tuberculosis. StatPearls – NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK556115. Published February 6, 2025.
- Engin G, Acunaş B, Acunaş G, Tunaci M. Imaging of extrapulmonary tuberculosis. Radiographics. 2000;20(2):471-488. doi:10.1148/radiographics.20.2.g00mc07471
- Debi U. Abdominal tuberculosis of the gastrointestinal tract: Revisited. World Journal of Gastroenterology. 2014;20(40):14831. doi:10.3748/wjg.v20.i40.14831
- Khan R, Abid S, Jafri W, Abbas Z, Hameed K, Ahmad Z. Diagnostic dilemma of abdominal tuberculosis in non-HIV patients: An ongoing challenge for physicians. World Journal of Gastroenterology. 2006;12(39):6371. doi:10.3748/wjg.v12.i39.6371
- Rana S, Farooqui MR, Rana S, Anees A, Ahmad Z, Jairajpuri ZS. The role of laboratory investigations in evaluating abdominal tuberculosis. Journal of Family and Community Medicine. 2015;22(3):152. doi:10.4103/2230-8229.163029
- Chow KM, Chow VCY, Hung LCT, Wong SM, Szeto CC. Tuberculous Peritonitis Associated Mortality Is High among Patients Waiting for the Results of Mycobacterial Cultures of Ascitic Fluid Samples. Clinical Infectious Diseases. 2002;35(4):409-413. doi:10.1086/341898
- Heller T, Goblirsch S, Wallrauch C, Lessells R, Brunetti E. Abdominal tuberculosis: sonographic diagnosis and treatment response in HIV-positive adults in rural South Africa. International Journal of Infectious Diseases. 2010;14:e108-e112. doi:10.1016/j.ijid.2009.11.030
- Ha HK, Jung JI, Lee MS, et al. CT differentiation of tuberculous peritonitis and peritoneal carcinomatosis. American Journal of Roentgenology. 1996;167(3):743-748. doi:10.2214/ajr.167.3.8751693
- De Backer AI, Mortelé KJ, De Keulenaer BL, Henckaerts L, Verhaert L. CT and MR imaging of gastrointestinal tuberculosis. PubMed. https://pubmed.ncbi.nlm.nih.gov/16999319/. Published August 1, 2006.
- Omprakash AR, D Souza RC, Sudarshan SH, Yadav V, Aithala PS, Pai JR, et al. Multi-Detector Computed Tomography in Abdominal Tuberculosis. Journal of Evolution of Medical and Dental Sciences. June 2022. doi:10.14260/jemds
- Joshi AR, Basantani AS, Patel TC. Role of CT and MRI in abdominal tuberculosis. Current Radiology Reports. 2014;2(10). doi:10.1007/s40134-014-0066-8
- Sharma R, Madhusudhan KS, Ahuja V. Intestinal tuberculosis versus Crohn’s disease: Clinical and radiological recommendations. Indian Journal of Radiology and Imaging – New Series/Indian Journal of Radiology and Imaging/Indian Journal of Radiology & Imaging. 2016;26(02):161-172. doi:10.4103/0971-3026.184417
- Dasgupta A, Singh N, Bhatia A. Abdominal Tuberculosis: A Histopathological Study with Special Reference to Intestinal Perforation and Mesenteric Vasculopathy. Journal of Laboratory Physicians. 2009;1(02):056-061. doi:10.4103/0974-2727.59700
- Jha DK, Pathiyil MM, Sharma V. Evidence-based approach to diagnosis and management of abdominal tuberculosis. Indian Journal of Gastroenterology. 2023;42(1):17-31. doi:10.1007/s12664-023-01343-x
- Sharma MP, Bhatia V. Abdominal tuberculosis. PubMed. https://pubmed.ncbi.nlm.nih.gov/15520484/. Published October 1, 2004.
- Sharma K, Sinha S, Sharma A, et al. Multiplex PCR for rapid diagnosis of gastrointestinal tuberculosis. Journal of Global Infectious Diseases. 2013;5(2):49. doi:10.4103/0974-777x.112272
- Maulahela H, Simadibrata M, Nelwan EJ, et al. Recent advances in the diagnosis of intestinal tuberculosis. BMC Gastroenterology. 2022;22(1):89. doi:10.1186/s12876-022-02171-7

