COPD and Obstructive Sleep Apnea Overlap Syndrome Review
Chronic Obstructive Pulmonary Disease and Obstructive Sleep Apnea Overlap Syndrome: Narrative Review
Chebly Dagher, MD 1, Antarpreet Kaur, MD 2,3
- University of Connecticut Health Center, Farmington, CT 06032
- Pulmonary, Critical care and Sleep medicine, Assistant Professor of Medicine, University of CT and Quinnipiac university
- Saint Francis Hospital and Medical Center, Hartford CT 06105.
OPEN ACCESS
PUBLISHED: 31 December 2025
CITATION: Dagher, C., and Kaur, A., 2025. Chronic Obstructive Pulmonary Disease and Obstructive Sleep Apnea Overlap Syndrome: Narrative Review. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.7176
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i12.7176
ISSN 2375-1924
ABSTRACT
Chronic obstructive pulmonary disease (COPD) and obstructive sleep apnea (OSA) are among the most common respiratory disorders, and their coexistence, termed COPD-OSA Overlap Syndrome (OVS), poses unique diagnostic and therapeutic challenges. OVS affects approximately 1% of adults, yet it’s true prevalence is likely underestimated due to variable diagnostic criteria and under recognition. The combination of chronic airflow limitation, recurrent nocturnal hypoxia, and sleep fragmentation creates a synergistic pathophysiological burden characterized by heightened systemic inflammation, oxidative stress, endothelial dysfunction, and autonomic imbalance. These mechanisms contribute to an increased risk of pulmonary hypertension, right-sided heart failure, arrhythmias, and overall cardiovascular morbidity and mortality compared with either disease alone. Clinically, OVS patients experience more profound nocturnal desaturation, greater daytime hypercapnia, and a higher frequency of exacerbations and hospitalizations. Diagnosis relies on screening tools such as the STOP-Bang or NoSAS questionnaires, with confirmation by polysomnography, though oximetry and capnometry remain valuable alternatives in resource-limited settings. Optimal management integrates aggressive treatment of COPD, lifestyle modification, and positive airway pressure therapy, particularly continuous positive airway pressure, which improves oxygenation, reduces exacerbations, and restores survival rates comparable to those of COPD-only patients. Despite its clinical importance, OVS remains understudied, underscoring the need for standardized diagnostic criteria, multicenter prospective trials, and a phenotype-based approach to guide individualized management and improve outcomes in this high-risk population.
Keywords
COPD, obstructive sleep apnea, overlap syndrome, diagnosis, management
Introduction
Chronic obstructive pulmonary disease (COPD) and Obstructive sleep apnea (OSA) are among the most prevalent respiratory conditions 1. When both disorders coexist in the same individual, the condition is referred to as COPD-OSA Overlap syndrome (OVS), a term originally described in 1985 2. COPD is marked by persistent, incompletely reversible airflow limitation and impaired ventilation, while OSA is caused by complex changes in airway causing recurrent upper airway obstruction during sleep, leading to intermittent apneas and hypopneas, with subsequent oxygen desaturation 3. Common symptoms include loud snoring, gasping or choking during sleep, and excessive daytime sleepiness 4. Reported prevalence rates of OVS vary significantly across studies, likely due to differences in diagnostic criteria and study populations 5.
When COPD and OSA coexist, they can contribute to more severe and prolonged nocturnal oxygen desaturation than either condition alone, potentially heightening the risk for a range of associated comorbidities 6,7. In OSA, intermittent episodes of oxygen deprivation trigger a cascade of biological responses, including oxidative stress, autonomic imbalance, systemic inflammation, and endothelial dysfunction, mechanisms that have been implicated in the development of cardiovascular complications 8,9. Similarly, COPD is linked to cardiovascular disease through mechanisms such as heightened sympathetic activity and chronic, low-grade inflammation 10. While both conditions independently elevate cardiovascular risk, their combined effect, whether additive or synergistic, remains to be fully clarified. However, existing evidence suggests that patients with OVS experience poorer health-related quality of life, a greater burden of comorbidities, more frequent exacerbations, and higher mortality compared to those with either disease alone 11-13. These findings highlight the importance of early identification and support routine screening for OSA in patients with COPD to enable timely diagnosis and targeted management of OVS. This review aims to explore the underlying pathophysiological mechanisms, clinical consequences, and diagnostic and therapeutic challenges associated with this complex condition.
Background
PREVALENCE
Chronic obstructive pulmonary disease (COPD) affects about 5-10% of global population and about 5% of the US population (estimated 13.7 million people). Its increasing prevalence is explained by high rates of smoking, rising environmental pollution and an ageing population 1. The prevalence of OSA varies, but most studies report rates of approximately 17–24% in middle-aged males (50–75 years old) and 9% in middle-aged females 14-16. OVS, characterized by the coexistence of COPD and OSA, affects approximately 1% of adults 17. However, the true prevalence of one condition in the presence of the other remains a subject of ongoing debate, as it largely depends on the specific criteria used to define COPD and OSA 6,17. Studies report varying estimates, with the prevalence of COPD among individuals diagnosed with OSA ranging from 8% to 16% 18,19. Conversely, the prevalence of OSA among individuals with COPD demonstrates a much broader range, between 5% and 65% 1,20,21. These discrepancies may be attributed to differences in study populations, diagnostic methods, and thresholds used to identify each condition 1,22. Approximately 1 in 10 patients with one entity will have other by chance and causal association is not implied. Most of the studies for OVS that depict a lower prevalence looked at individuals with milder COPD 3,23. A study looking at moderate to severe COPD patient demonstrated the prevalence of 66% in these individuals 1. This variability highlights the complexity of accurately characterizing OVS and underscores the need for standardized definitions and diagnostic approaches to better understand its true burden in the general population 24.
UNDERSTANDING COPD AND OSA
Chronic obstructive pulmonary disease (COPD) is defined by long-standing limitations in airflow that show limited reversibility and are linked to atypical inflammatory activity within the pulmonary system 24,25. It refers to a spectrum of related respiratory disorders, such as chronic bronchitis and emphysema, that predominantly impact the airways, though the lung tissue and blood vessels may also be affected based on the underlying disease mechanism 25. These pathophysiologic changes are often more pronounced during physiological stressors, such as exercise or sleep, where the respiratory system is further challenged 24. The pathological effects of COPD are multifaceted and include mucous hypersecretion, which contributes to airway obstruction, and ciliary dysfunction, impairing the clearance of mucus and debris from the respiratory tract 26. Airflow obstruction and hyperinflation further compromise respiratory efficiency, while gas exchange abnormalities lead to impaired oxygenation and carbon dioxide elimination 25,27. Additionally, COPD can result in pulmonary hypertension due to increased vascular resistance and strain on the pulmonary circulation 27. Systemic effects, such as cachexia and polycythemia, also manifest in advanced stages, reflecting the widespread impact of the disease beyond the lungs 26. The mechanisms underlying respiratory dysfunction in COPD can be broadly categorized into two primary groups: mechanical impairment and ventilatory dysfunction. These categories represent distinct pathways that contribute to respiratory compromise, providing valuable insight into the diverse factors driving disease progression 24-27. Sleep in COPD patient presents physiological changes that further challenges the pulmonary mechanics. Loss of drive to breath during wakefulness, hypoventilation, worse elastic loading and inability to shorten the expiratory phase led to marked disturbances in breathing during sleep and hence, a poorer sleep quality 3.
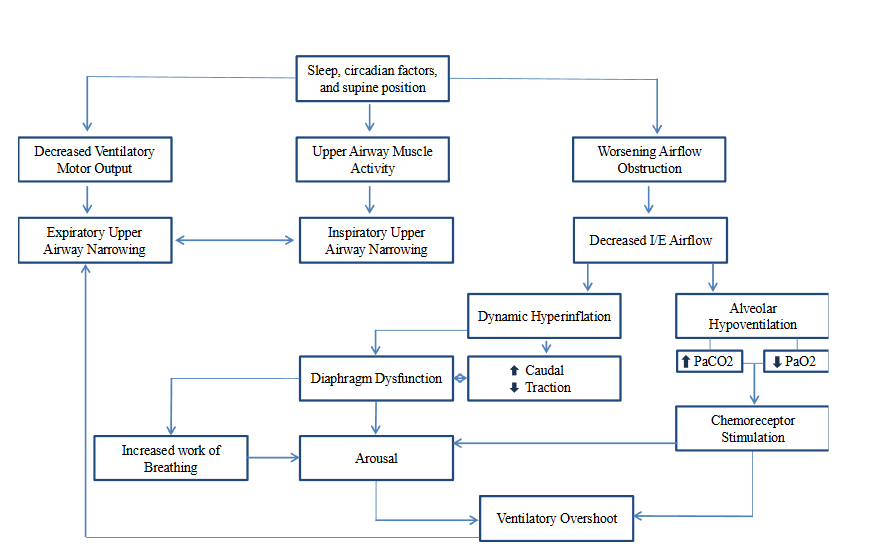
Obstructive sleep apnea (OSA) is a condition marked by repeated narrowing or closure of the pharyngeal airway during sleep, leading to periodic reductions in oxygen saturation and disrupted sleep patterns 28. During sleep, there is a physiological decline in respiratory drive compared to wakefulness, leading to decreased neuromuscular output to respiratory muscles 28,29. This attenuation particularly affects pharyngeal dilator muscles, such as the tensor palatini and genioglossus, which are essential for keeping the upper airway open 29. In at-risk individuals, these changes result in pharyngeal airway narrowing, increasing vulnerability to central apnea and airway collapse 30. Airway narrowing leads to increased upper airway resistance and turbulent airflow, which causes oscillations of soft tissues, such as the soft palate, during sleep 30,31. This increased resistance imposes an internal mechanical load on the lungs, which is compensated for by elevated respiratory effort during wakefulness 31. Recurrent respiratory events associated with OSA are characterized by cycles of hypoventilation and hyperventilation, leading to fluctuations in end-tidal CO2 and variable oxygenation 32. These events exacerbate oxidative stress and create significant gradients in partial oxygen pressure between the proximal and distal airways 33. This process can further impair oxygen delivery and contribute to systemic complications 32. Individuals with optimal upper airway anatomy generally maintain normal oxygen saturation and sleep patterns despite mild hypercapnia 34. However, anatomical predispositions such as inspiratory flow limitation, snoring, or structural airway collapse increase the risk of OSA, with snoring serving as a clinical marker 35. In obesity, excess adipose tissue around the pharyngeal airway, particularly in those with larger neck circumferences, further heightens the risk of airway collapse 34. These factors underscore the intricate relationship between anatomy, respiratory mechanics, and risk factors in the pathogenesis of OSA 32,34.
CLINICAL FEATURES AND RISK FACTORS OF OVS
Despite the uncertainty regarding which conditions overlap, the term “OVS” emphasizes that the resulting pathophysiology is more intricate than would be anticipated if the conditions were entirely separate, thereby justifying its classification as a syndrome. The clinical features of OVS frequently appear alongside the typical manifestations of both COPD and OSA 2. In addition to the hallmark symptoms of COPD and OSA, patients with OVS often exhibit more severe and prolonged hypoxemia, increased daytime hypercapnia, and metabolic derangements, including acidemia 36,37. Clinicians must recognize and distinguish between nocturnal hypoxemia caused by COPD and hypoxemia resulting from obstructive airway hypoventilation in OSA 36. As a result, they may present with morning headaches due to hypercapnia, cyanosis from hypoxemia, and peripheral edema, highlighting additional clinical findings associated with OVS 38.
There is evidence indicating a reciprocal relationship between OSA and COPD, where OVS may arise in individuals with COPD who later develop OSA or in those with OSA who are subsequently diagnosed with COPD 39. Factors linked to its development include older age, male gender, cigarette smoke and alcohol exposure, reduced physical activity, and obesity 2,40. Smoking, a shared risk factor for both COPD and OSA, exacerbates these conditions through oxidative stress and airway inflammation 38.
PROMOTING AND PROTECTIVE MECHANISMS IN OVS
In individuals with COPD, several factors can either promote or protect against the development of OSA 41. Promoting factors include coexistent cor pulmonale with fluid retention. The rostral shift of peripheral edema, where fluid redistributes from the lower extremities to the neck and upper airway during sleep, compromising airway patency 41,42. Other contributors include upper airway and skeletal myopathy associated with advanced COPD which reduces upper airway stability during sleep. Moreover, the use of corticosteroids, frequently prescribed in COPD, can further weaken muscles and promote weight gain, thereby increasing the risk of airway obstruction 42,43. Additionally, chronic cigarette smoking, commonly associated with COPD, contributes to airway inflammation, remodeling, and dysfunction, increasing the susceptibility to OSA 44.
Conversely, certain factors appear to protect individuals with COPD from developing OSA. A low body mass index (BMI), often seen in severe COPD due to increased metabolic demands and cachexia, reduces fat deposition in the upper airway, lowering the likelihood of obstruction 45. Lung hyperinflation, a characteristic feature of COPD, may mechanically stabilize the upper airway due to tracheal traction and reduce its collapsibility during sleep 46. Additionally, diminished REM sleep, a stage when OSA events are most prominent, decreases the exposure to periods of airway collapse 47. Other protective factors include older age, which paradoxically reduces OSA severity due to age-related changes in airway structure 48.
The influence of these promoting and protective factors can vary depending on the underlying COPD phenotype. For instance, patients with emphysema typically have hyperinflated lungs and lower BMI due to increased work of breathing, which may reduce their risk of OSA 38,46. In contrast, patients with chronic bronchitis often present with higher BMI, increasing the likelihood of OSA due to adipose tissue accumulation around the neck 46. Moreover, environmental and occupational exposures that drive the progression of COPD, particularly in resource-limited settings, can accelerate COPD progression and further influence the risk of OSA 49.
EVALUATION
Currently, no definitive guidelines exist for screening patients with COPD for OSA 50. However, the STOP-Bang Questionnaire (SBQ), which evaluates factors such as snoring, tiredness, observed apnea, blood pressure, BMI, age, neck circumference, and gender, is recommended as an initial screening tool for all COPD patients 51. Diagnostic testing is advised for patients identified as having an intermediate-to-high risk of OSA based on SBQ scores 50. Additional screening tools include the NoSAS Questionnaire, which assesses neck circumference, obesity, snoring, age, and sex, and the Berlin Questionnaire, both of which aid in identifying patients at increased risk for sleep apnea 52,53. Polysomnography remains the gold standard for evaluating sleep disturbances in patients with OVS, offering the most comprehensive assessment of sleep and breathing patterns 50,54. However, sleep studies have limitations in this scenario as well. Given the variety of physiological airway changes and sustained and severe hypoxemia in COPD patients, the apnea-hypopnea index (AHI), might be underestimation in prolonged hypoxic events 55. Moreover, sleep disturbances associated with hyperinflation, poor sleep quality, and nocturnal cough can likely cause breathing disturbances being misinterpreted as respiratory events 56. Conversely, frequent arousals might not lead to adequate desaturation and might lead to underestimation of hypopneas 55. Additionally, the correlation on AHI with clinical outcomes, including cardiovascular outcomes, symptom severity, and mortality, remains unknown 57. In COPD patients, sleep indices that determine hypoxemic load (e.g. deoxygenation nadir, duration of hypoxia, area under oxygen desaturation curve) might predict clinical outcomes, and further research is warranted in this field 3.
In resource-limited settings, oxygen desaturation monitoring and overnight oximetry may suffice for clinical evaluation 58. The American Thoracic Society (ATS) and European Respiratory Society (ERS) recommend overnight polysomnography for patients with mild COPD and clinical evidence of pulmonary hypertension to evaluate for OSA 7,50. While overnight oximetry can be a valuable screening method, particularly in resource-constrained settings where a sawtooth pattern on oximetry often indicates OSA in COPD patients, confirmation with polysomnography remains essential 59,60.
Comparison of other methods of nocturnal gas-exchange beyond pulse oximetry is an area of interest in COPD patients. Capnometry, in addition to sleep study, might help detect potential hypoventilation and help guide treatment choices as well 3.
SEVERITY AND PHYSIOLOGICAL IMPAIRMENTS IN OVS
Patients with OVS often exhibit higher scores on the COPD Assessment Test and the Epworth Sleepiness Scale compared to patients with COPD alone, reflecting greater symptom burden and sleep-related impairments 61. These clinical findings are closely associated with physiological changes, such as lung hyperinflation, which significantly impacts pulmonary function in OVS 62. A reduced inspiratory capacity to total lung capacity (IC/TLC) ratio is indicative of more severe hyperinflation and is considered a more accurate marker of disease severity than forced expiratory volume (FEV1) 63. Hyperinflation can impair sleep quality by increasing the work of breathing in the recumbent position, though this mechanism requires further investigation 62. Additionally, the severity of airflow obstruction, measured by the forced expiratory volume/forced vital capacity (FEV1/FVC) ratio, correlates with a higher likelihood of persistent hypoxemia 64. However, data comparing pulmonary function testing in OVS versus COPD alone remain limited.
Patients with OVS are especially prone to oxygen desaturation during sleep 38. Notably, even in the absence of upper airway obstruction, individuals with COPD and an awake oxygen saturation (SaO₂) between 90% and 95% are prone to substantial nocturnal desaturation. This is particularly pronounced during rapid eye movement (REM) sleep and is attributed to alveolar hypoventilation, impaired ventilation-perfusion matching, and reduced end-expiratory lung volume 36. Continuous overnight monitoring of oxygen saturations, with an emphasis on the oxygen desaturation index (ODI), is critical, particularly in resource-limited settings where polysomnography may not be available. The ODI, which reflects intermittent hypoxemia, is considered a more reliable predictor of systemic inflammation and cardiovascular comorbidities than the apnea-hypopnea index (AHI) 58.
OUTCOMES OF OVS AND IMPLICATION OF COMORBIDITIES
Individuals with both COPD and OSA are thought to face an elevated risk of cardiovascular disease due to shared and potentially synergistic pathological mechanisms 38. These mechanisms include oxidative stress, systemic inflammation, vascular endothelial dysfunction, and accelerated atherosclerosis, which may interact in a way that exacerbates cardiovascular harm 65-67. Supporting this hypothesis, multiple studies have demonstrated that OVS is associated with a higher cardiovascular risk and greater mortality compared to COPD or OSA alone 5,68. However, whether the coexistence of these two conditions results in a truly synergistic effect on cardiovascular outcomes remains unclear 5. OVS patients may experience more frequent and severe episodes of oxygen desaturation, deeper hypoxemia, and a higher incidence of cardiac dysrhythmias 6,69. Mechanistic pathways indicate intermittent hypoxia leading to pulmonary vasoconstriction and right ventricular load leading to increased RV mass in patients with OVS as compared to COPD alone 70. Myocardial fibrosis has been proven to be present with cardiac MRI imaging in patients with worse nocturnal hypoxia 71. Synergistic mechanisms leading to increased inflammation and impaired metabolism in COPD and OSA lead to worse chronic inflammation in OVS 70,72.
Consequently, OVS patients are susceptible to developing pulmonary hypertension 73, right-sided heart failure, vascular endothelial dysfunction 65, and accelerated atherosclerosis 66. These combined effects significantly increase the risk of long-term complications and reduce overall survival rates 5.
Interestingly, emerging evidence suggests that the severity of OSA may influence the impact of COPD on mortality and respiratory function 74. For instance, severe OSA has been associated with a reduced contribution of impaired lung function to mortality, as well as decreased gas trapping and a lower burden of emphysema in smokers with concurrent OSA 74,75. These findings highlight the complex interplay between COPD and OSA, particularly in how they interact to influence gas exchange, cardiovascular health, and overall mortality. Understanding these mechanisms, alongside the balance of promoting and protective factors, is critical for identifying individuals at high risk for OVS and optimizing their clinical management.
MANAGEMENT
Lifestyle Modifications and Exercise
Exercise and pulmonary rehabilitation improve sleep quality, symptoms, and quality of life in OVS, while smoking cessation reduces morbidity and mortality 7. Weight loss benefits obese OSA patients, but cachexia in advanced COPD increases mortality 7,50.
Supplemental Oxygen
Supplemental nocturnal oxygen is necessary for persistent desaturations despite optimized medications and does not increase the risk of hypercapnia 50. However, studies indicate it may elevate the risk of arrhythmias in COPD patients, particularly those with coronary artery disease or left ventricular dysfunction, requiring cautious use. Therefore, oxygen therapy alone is insufficient for managing OVS 76.
Pharmacologic Therapies
Long-acting bronchodilator therapies, including inhaled beta-agonist and anticholinergics, have demonstrated significant benefits in improving mean nocturnal oxygen saturation in COPD patients 77. Both therapies reduce the time spent in hypoxemia during sleep, helping to mitigate nocturnal desaturation and related complications 38,77. The effectiveness of inhaled corticosteroids (ICS) in managing OVS remains controversial 78. Some studies suggest that ICS can improve the AHI, nocturnal oxygenation, daytime hypercapnia and lung function by reducing airway inflammation 78. Conversely, other research indicates that ICS may contribute to an increased risk of myopathy, potentially exacerbating upper airway collapsibility 3,79.
Theophylline improves nocturnal oxygenation and reduces the AHI in OSA patients, likely by stimulating central respiratory drive, which may be particularly effective in central sleep apnea. However, its use is limited by potential side effects 80.
Sedatives, opioids, and respiratory stimulants like acetazolamide are not recommended for OVS due to potential risks and lack of clinical benefit 81,82.
Positive Airway Pressure and Noninvasive Ventilation
Positive airway pressure (PAP) therapy is the most effective and widely recognized treatment for OVS. While PAP treatments have been proven to improve sleep quality and cardiovascular outcomes in OSA patients, the use of non-invasive ventilation (bilevel positive airway pressure ventilation) has been shown to improve mortality in advanced COPD patients with hypercapnia 39.
Positive airway pressure treatment has been shown to decrease pro-inflammatory markers associated with cardiovascular disease, such as C-reactive protein (CRP) and tumor necrosis factor-α (TNF-α) 83, increase exercise capacity, respiratory and skeletal muscle strength 84,85, improve mean arterial pressure 86 and FEV1 87. Adherence to PAP therapy has been associated with fewer COPD exacerbations, reduced COPD-related hospitalizations, lower incidence of cardiovascular events, and decreased mortality 5.
The study by Marin et al. highlights the significant impact of long-term continuous positive airway pressure (CPAP) therapy on survival and hospitalization rates in patients with OVS 5. Patients treated with CPAP had survival rates comparable to those with COPD alone, demonstrating that CPAP effectively mitigates the additional mortality risk associated with OVS. In contrast, patients not receiving CPAP had a 1.79-fold higher risk of mortality and were 1.70 times more likely to experience severe COPD exacerbations requiring hospitalization compared to COPD-only patients. These findings underscore the critical role of CPAP in improving long-term outcomes by reducing mortality, severe exacerbations, and hospitalizations in OVS patients 5,88,89.
Similarly, a study on CPAP and survival in moderate-to-severe OSA and hypoxemic COPD reinforces these conclusions, showing that long-term CPAP therapy significantly improves survival in OVS patients 90. Individuals with moderate-to-severe OSA and hypoxemic COPD who adhered to CPAP therapy experienced better outcomes, including reduced mortality, compared to those who did not receive or adhere to CPAP treatment 90. Albeit a small study with limited individuals, these results further emphasize CPAP’s role in alleviating nocturnal hypoxemia and mitigating the combined risks of COPD and OSA, highlighting its importance in improving prognosis and long-term outcomes in this high-risk population 90.
Prognosis
Several studies have examined the prognosis of patients with OVS. For instance, a case-control study reported that the presence of COPD in patients with OSA was associated with a nearly sevenfold increase in mortality compared to those without COPD 12. Similarly, Marin et al. found that among individuals with COPD, the coexistence of OSA significantly increased the risk of all-cause mortality, cardiovascular-related deaths, and hospitalizations due to COPD exacerbations 5. Notably, treatment with CPAP is associated with improved survival and reduced hospital admissions 5.
Furthermore, patients with OVS tend to experience more frequent COPD exacerbations and hospitalizations with increased healthcare utilization. These patients experience greater sleep disruption, more severe nocturnal oxygen desaturation, lower health related quality of life indices and a higher burden of comorbidities than those with either condition alone 5,91,92. These factors contribute to a significantly poorer health-related quality of life. Collectively, these findings highlight the importance of early recognition and management of OVS and support routine screening for OSA in patients with COPD.
Conclusion
Patients with OVS experience a higher burden of comorbidities, increased mortality, and reduced quality of life compared to those with COPD or OSA alone, contributing to both clinical and economic challenges. These patients often present with more severe nocturnal desaturation, systemic inflammation, oxidative stress, and endothelial dysfunction, all of which raise their risk for cardiovascular complications. CPAP remains the first-line therapy for OVS, offering improvements in oxygenation, sleep quality, and COPD exacerbation frequency. Despite evidence supporting CPAP’s benefits, research on OVS remains limited and inconsistent, largely due to small sample sizes, regional differences, and the lack of large, multicenter prospective studies. Additionally, varying diagnostic criteria and the frequent exclusion of OVS patients from clinical trials hinder broader understanding and management. There is a clear need for standardized evaluation tools, phenotype-specific research, and stronger clinical guidelines to improve the prevention, diagnosis, and treatment of this underrecognized population.
References
- Soler X, Gaio E, Powell FL, et al. High prevalence of obstructive sleep apnea in patients with moderate to severe chronic obstructive pulmonary disease. Annals of the American Thoracic Society. 2015;12(8):1219-1225.
- Adler D, Bailly S, Benmerad M, et al. Clinical presentation and comorbidities of obstructive sleep apnea-COPD overlap syndrome. PloS one. 2020;15(7):e0235331.
- Malhotra A, Schwartz AR, Schneider H, et al. Research priorities in pathophysiology for sleep-disordered breathing in patients with chronic obstructive pulmonary disease. An official American Thoracic Society research statement. American journal of respiratory and critical care medicine. 2018;197(3):289-299.
- Veasey SC, Rosen IM. Obstructive sleep apnea in adults. New England Journal of Medicine. 2019;380(15):1442-1449.
- Marin JM, Soriano JB, Carrizo SJ, Boldova A, Celli BR. Outcomes in patients with chronic obstructive pulmonary disease and obstructive sleep apnea: the overlap syndrome. American journal of respiratory and critical care medicine. 2010;182(3):325-331.
- Chaouat A, Weitzenblum E, Krieger J, Ifoundza T, Oswald M, Kessler R. Association of chronic obstructive pulmonary disease and sleep apnea syndrome. American journal of respiratory and critical care medicine. 1995;151(1):82-86.
- Owens RL, Malhotra A. Sleep-disordered breathing and COPD: the overlap syndrome. Respiratory care. 2010;55(10):1333-1346.
- Butt M, Dwivedi G, Khair O, Lip GY. Obstructive sleep apnea and cardiovascular disease. International journal of cardiology. 2010;139(1):7-16.
- Parish JM, Somers VK. Obstructive sleep apnea and cardiovascular disease. Elsevier; 2004:1036-1046.
- Sin DD, Man SP. Chronic obstructive pulmonary disease: a novel risk factor for cardiovascular disease. Canadian journal of physiology and pharmacology. 2005;83(1):8-13.
- Mermigkis C, Kopanakis A, Foldvary‐Schaefer N, et al. Health‐related quality of life in patients with obstructive sleep apnoea and chronic obstructive pulmonary disease (overlap syndrome). International journal of clinical practice. 2007;61(2):207-211.
- Lavie P, Herer P, Lavie L. Mortality risk factors in sleep apnoea: a matched case–control study. Journal of sleep research. 2007;16(1):128-134.
- Shaya FT, Lin P-J, Aljawadi MH, Scharf SM. Elevated economic burden in obstructive lung disease patients with concomitant sleep apnea syndrome. Sleep and Breathing. 2009;13:317-323.
- Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle-aged adults. New England journal of medicine. 1993;328(17):1230-1235.
- Sharma SK, Kumpawat S, Banga A, Goel A. Prevalence and risk factors of obstructive sleep apnea syndrome in a population of Delhi, India. Chest. 2006;130(1):149-156.
- Udwadia ZF, Doshi AV, Lonkar SG, Singh CI. Prevalence of sleep-disordered breathing and sleep apnea in middle-aged urban Indian men. American journal of respiratory and critical care medicine. 2004;169(2):168-173.
- Bednarek M, Plywaczewski R, Jonczak L, Zielinski J. There is no relationship between chronic obstructive pulmonary disease and obstructive sleep apnea syndrome: a population study. Respiration. 2005;72(2):142-149.
- Resta O, Barbaro MPF, Brindicci C, Nocerino MC, Caratozzolo G, Carbonara M. Hypercapnia in overlap syndrome: possible determinant factors. Sleep and Breathing. 2002;6:11-17.
- Greenberg-Dotan S, Reuveni H, Tal A, et al. Increased prevalence of obstructive lung disease in patients with obstructive sleep apnea. Sleep and Breathing. 2014;18:69-75.
- Larsson L, Lindberg A, Franklin K, Lundbäck B. Obstructive Lung Disease in Northern Sweden Studies. Obstructive sleep apnoea syndrome is common in subjects with chronic bronchitis: report from the Obstructive Lung Disease in Northern Sweden studies. Respiration. 2001;68(3):250-255.
- Sanders MH, Newman AB, Haggerty CL, et al. Sleep and sleep-disordered breathing in adults with predominantly mild obstructive airway disease. American journal of respiratory and critical care medicine. 2003;167(1):7-14.
- Turcani P, Skrickova J, Pavlik T, Janousova E, Orban M. The prevalence of obstructive sleep apnea in patients hospitalized for COPD exacerbation. Biomedical Papers. 2015;159(3):422-428.
- Sharma B, Feinsilver S, Owens RL, Malhotra A, McSharry D, Karbowitz S. Obstructive airway disease and obstructive sleep apnea: effect of pulmonary function. Lung. 2011;189:37-41.
- Janson C, Marks G, Buist S, et al. The impact of COPD on health status: findings from the BOLD study. European Respiratory Journal. 2013;42(6):1472-1483.
- Sharma S, Hashmi M, Chakraborty R. StatPearls [Internet] StatPearls Publishing. Treasure Island (FL): Sep. 2021;18
- Han MK, Agusti A, Calverley PM, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. American journal of respiratory and critical care medicine. 2010;182(5):598-604.
- Boers E, Barrett M, Su JG, et al. Global burden of chronic obstructive pulmonary disease through 2050. JAMA Network Open. 2023;6(12):e2346598-e2346598.
- Eckert DJ, Malhotra A. Pathophysiology of adult obstructive sleep apnea. Proceedings of the American thoracic society. 2008;5(2):144-153.
- Choudhary SS, Choudhary SR. Sleep effects on breathing and respiratory diseases. Lung India. 2009;26(4):117-122.
- Edwards BA, White DP. Control of the pharyngeal musculature during wakefulness and sleep: implications in normal controls and sleep apnea. Head & neck. 2011;33(S1):S37-S45.
- Strohl KP, Butler JP, Malhotra A. Mechanical properties of the upper airway. Comprehensive Physiology. 2012;2(3):1853.
- Locke BW, Lee JJ, Sundar KM. OSA and chronic respiratory disease: mechanisms and epidemiology. International Journal of Environmental Research and Public Health. 2022;19(9):5473.
- Edwards BA, Eckert DJ, Jordan AS. Obstructive sleep apnoea pathogenesis from mild to severe: Is it all the same? Respirology. 2017;22(1):33-42.
- Dewald D, Strohl KP. Mechanical Properties, Anatomy, and Control of the Upper Airway. Upper Airway Stimulation Therapy for Obstructive Sleep Apnea: Medical, Surgical, and Technical Aspects. 2021:27.
- Yoshikawa F, Nozaki-Taguchi N, Yamamoto A, et al. Preoperative sleep-disordered breathing and craniofacial abnormalities are risk factors for postoperative sleep-disordered breathing in patients undergoing skin-flap oropharyngeal reconstruction surgery for oral cavity cancer: a prospective case-control study. Sleep and Breathing. 2024;28(2):797-806.
- Lewis CA, Fergusson W, Eaton T, Zeng I, Kolbe J. Isolated nocturnal desaturation in COPD: prevalence and impact on quality of life and sleep. Thorax. 2009;64(2):133-138.
- Ramsey R, Mehra R, Strohl KP. Variations in physician interpretation of overnight pulse oximetry monitoring. Chest. 2007;132(3):852-859.
- McNicholas WT. COPD-OSA overlap syndrome: evolving evidence regarding epidemiology, clinical consequences, and management. Chest. 2017;152(6):1318-1326.
- Macrea M, Oczkowski S, Rochwerg B, et al. Long-term noninvasive ventilation in chronic stable hypercapnic chronic obstructive pulmonary disease. An official American Thoracic Society clinical practice guideline. American journal of respiratory and critical care medicine. 2020;202(4):e74-e87.
- Nair S, Paul T, Mehta AA, Haridas N, Kunoor A, Sudhakar N. Prevalence of overlap syndrome in patients with obstructive sleep apnea in a quaternary care center of Kerala. Indian Journal of Public Health. 2022;66(Suppl 1):S12-S16.
- Redolfi S, Yumino D, Ruttanaumpawan P, et al. Relationship between overnight rostral fluid shift and obstructive sleep apnea in nonobese men. American journal of respiratory and critical care medicine. 2009;179(3):241-246.
- Poulain M, Doucet M, Major GC, et al. The effect of obesity on chronic respiratory diseases: pathophysiology and therapeutic strategies. Cmaj. 2006;174(9):1293-1299.
- Chaouat A, Weitzenblum E, Kessler R, et al. Sleep-Related O2 Desaturation and Daytime Pulmonary Haemodynamics in COPD Patients With Mild Hypoxaemia. Chest. 1998;114(6):A-22 (1)-A-22 (1).
- Steveling EH, Clarenbach CF, Miedinger D, et al. Predictors of the overlap syndrome and its association with comorbidities in patients with chronic obstructive pulmonary disease. Respiration. 2014;88(6):451-457.
- Kim V, Han MK, Vance GB, et al. The chronic bronchitic phenotype of COPD: an analysis of the COPDGene Study. Chest. 2011;140(3):626-633.
- He B-T, Lu G, Xiao S-C, et al. Coexistence of OSA may compensate for sleep related reduction in neural respiratory drive in patients with COPD. Thorax. 2017;72(3):256-262.
- Medicine AOSATFotAAoS. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. Journal of clinical sleep medicine. 2009;5(3):263-276.
- Silva MdS, Poyares D, Silva LO, et al. Associations of the severity of obstructive sleep apnea with age-related comorbidities: a population-based study. Frontiers in Neurology. 2022;13:802554.
- Hurst JR, Siddharthan T. Global burden of COPD: prevalence, patterns, and trends. Handbook of global health. 2020:1-20.
- Singh S, Kaur H, Singh S, Khawaja I. The overlap syndrome. Cureus. 2018;10(10)
- Martins EF, Martinez D, Cortes AL, Nascimento N, Brendler J. Exploring the STOP-BANG questionnaire for obstructive sleep apnea screening in seniors. Journal of clinical sleep medicine. 2020;16(2):199-206.
- Georgakopoulou VE, Pantazis N, Tsiafaki X, Nena E, Amfilochiou A, Steiropoulos P. Validation of NoSAS score for the screening of obstructive sleep apnea. Medicine international. 2023;3(2):1-10.
- Navarrete-Martínez E, Muñoz-Gómez R, Serrano-Merino J, et al. Validity and reliability of the Berlin questionnaire for the detection of moderate or severe obstructive sleep apnea in patients aged 40 years or older detected from primary care. Frontiers in Medicine. 2023;10:1229972.
- Medicine AAoS. Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep. 1999;22:667-689.
- Lurie A, Roche N. Obstructive sleep apnea in patients with chronic obstructive pulmonary disease: facts and perspectives. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2021;18(6):700-712.
- Celli BR, MacNee W, Agusti A, et al. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. European Respiratory Journal. 2004;23(6):932-946.
- Marin JM, Soriano JB, Marin-Oto M, et al. Sleep-disordered Breathing in Patients with COPD: Prevalence and Outcomes. Annals of the American Thoracic Society. 2025;(ja)
- Ryan S, Taylor CT, McNicholas WT. Selective activation of inflammatory pathways by intermittent hypoxia in obstructive sleep apnea syndrome. Circulation. 2005;112(17):2660-2667.
- Singh S, Khan SZ, Singh D, Verma S, Talwar A. The uses of overnight pulse oximetry. Lung India. 2020;37(2):151-157.
- Hang L-W, Wang H-L, Chen J-H, et al. Validation of overnight oximetry to diagnose patients with moderate to severe obstructive sleep apnea. BMC pulmonary medicine. 2015;15:1-13.
- Zhang P, Chen B, Lou H, et al. Predictors and outcomes of obstructive sleep apnea in patients with chronic obstructive pulmonary disease in China. BMC pulmonary medicine. 2022;22(1):16.
- Tsai SC, Lee-Chiong T. Lung hyperinflation and sleep quality in the overlap syndrome. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2009;6(6):419-420.
- French A, Balfe D, Mirocha JM, Falk JA, Mosenifar Z. The inspiratory capacity/total lung capacity ratio as a predictor of survival in an emphysematous phenotype of chronic obstructive pulmonary disease. International journal of chronic obstructive pulmonary disease. 2015:1305-1312.
- Eschenbacher WL. Defining airflow obstruction. Chronic Obstructive Pulmonary Diseases. 2016;3(2):515.
- Kato M, Roberts-Thomson P, Phillips BG, et al. Impairment of endothelium-dependent vasodilation of resistance vessels in patients with obstructive sleep apnea. Circulation. 2000;102(21):2607-2610.
- Schroeder EB, Welch VL, Evans GW, Heiss G. Impaired lung function and subclinical atherosclerosis: the ARIC study. Atherosclerosis. 2005;180(2):367-373.
- Drager LF, Bortolotto LA, Lorenzi MC, Figueiredo AC, Krieger EM, Lorenzi-Filho G. Early signs of atherosclerosis in obstructive sleep apnea. American journal of respiratory and critical care medicine. 2005;172(5):613-618.
- Shiina K, Tomiyama H, Takata Y, et al. Overlap syndrome: additive effects of COPD on the cardiovascular damages in patients with OSA. Respiratory medicine. 2012;106(9):1335-1341.
- Shepard Jr JW, Garrison MW, Grither DA, Evans R, Schweitzer PK. Relationship of ventricular ectopy to nocturnal oxygen desaturation in patients with chronic obstructive pulmonary disease. The American journal of medicine. 1985;78(1):28-34.
- Sharma B, Neilan TG, Kwong RY, et al. Evaluation of right ventricular remodeling using cardiac magnetic resonance imaging in co-existent chronic obstructive pulmonary disease and obstructive sleep apnea. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2013;10(1):4-10.
- Hu BZ, Jiang C, Ding YJ, et al. The clinical and hemodynamic characteristics of pulmonary hypertension in patients with OSA-COPD overlap syndrome. The American Journal of the Medical Sciences. 2024;367(6):375-381.
- Neilan TG, Bakker JP, Sharma B, et al. T1 measurements for detection of expansion of the myocardial extracellular volume in chronic obstructive pulmonary disease. Canadian Journal of Cardiology. 2014;30(12):1668-1675.
- Levi-Valensi P, Weitzenblum E, Rida Z, et al. Sleep-related oxygen desaturation and daytime pulmonary haemodynamics in COPD patients. European Respiratory Journal. 1992;5(3):301-307.
- Biselli P, Grossman PR, Kirkness JP, et al. The effect of increased lung volume in chronic obstructive pulmonary disease on upper airway obstruction during sleep. Journal of applied physiology. 2015;119(3):266-271.
- Krachman SL, Tiwari R, Vega ME, et al. Effect of emphysema severity on the apnea–hypopnea index in smokers with obstructive sleep apnea. Annals of the American Thoracic Society. 2016;13(7):1129-1135.
- Alford NJ, Fletcher EC, Nickeson D. Acute oxygen in patients with sleep apnea and COPD. Chest. 1986;89(1):30-38.
- McNicholas W, Calverley P, Lee A, Edwards J, Investigators TSSiC. Long-acting inhaled anticholinergic therapy improves sleeping oxygen saturation in COPD. European Respiratory Journal. 2004;23(6):825-831.
- Rezaeetalab F, Rezaeetalab F, Dehestani V. Inhaled steroids reduce apnea-hypopnea index in overlap syndrome. Pneumologia (Bucharest, Romania). 2013;62(4):212-214.
- Singh D, Agusti A, Anzueto A, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease: the GOLD science committee report 2019. European Respiratory Journal. 2019;53(5)
- Mulloy E, McNicholas WT. Theophylline in obstructive sleep apnea: a double-blind evaluation. Chest. 1992;101(3):753-757.
- Baillargeon J, Singh G, Kuo Y-F, Raji MA, Westra J, Sharma G. Association of opioid and benzodiazepine use with adverse respiratory events in older adults with chronic obstructive pulmonary disease. Annals of the American Thoracic Society. 2019;16(10):1245-1251.
- Adamson R, Swenson ER. Acetazolamide use in severe chronic obstructive pulmonary disease. Pros and cons. Annals of the American Thoracic Society. 2017;14(7):1086-1093.
- Wang Y, Su M, Zhang X. Effects of continuous positive airway pressure treatment of inflammatory factors in patients with overlap syndrome. Zhonghua yi xue za zhi. 2014;94(6):416-419.
- de Miguel J, Cabello J, Sánchez-Alarcos JM, Álvarez-Sala R, Espinós D, Álvarez-Sala JL. Long-term effects of treatment with nasal continuous positive airway pressure on lung function in patients with overlap syndrome. Sleep and Breathing. 2002;6:3-10.
- Nowiński A, Bieleń P, Jonczak L, Śliwiński P. Influence of treatment with continuous positive airway pressure on respiratory muscle function and physical fitness in patients with obstructive sleep apnoea and overlap syndrome. Advances in Respiratory Medicine. 2007;75(1):46-56.
- Toraldo DM, De Nuccio F, Nicolardi G. Fixed-pressure nCPAP in patients with obstructive sleep apnea (OSA) syndrome and chronic obstructive pulmonary disease (COPD): a 24-month follow-up study. Sleep and Breathing. 2010;14:115-123.
- Mansfield D, Naughton MT. Effects of continuous positive airway pressure on lung function in patients with chronic obstructive pulmonary disease and sleep disordered breathing. Respirology. 1999;4(4):365-370.
- Jaoude P, Kufel T, El-Solh AA. Survival benefit of CPAP favors hypercapnic patients with the overlap syndrome. Lung. 2014;192:251-258.
- Jaoude P, El‐Solh AA. Predictive factors for COPD exacerbations and mortality in patients with overlap syndrome. The clinical respiratory journal. 2019;13(10):643-651.
- Machado ML, Vollmer W, Togeiro S, et al. CPAP and survival in moderate-to-severe obstructive sleep apnoea syndrome and hypoxaemic COPD. European respiratory journal. 2009;35(1):132-137.
- Konstantinopoulos A, Karkoulias K, Perimeni P, Spyropoulos K, Perimenis P. The impact of long term conventional treatment for overlap syndrome (obstructive sleep apnea and chronic obstructive pulmonary disease) on concurrent erectile dysfunction: up-015. The Journal of Sexual Medicine. 2006;3:442-443.
- Zamarrón C, Paz VG, Morete E, del Campo Matías F. Association of chronic obstructive pulmonary disease and obstructive sleep apnea consequences. International Journal of Chronic Obstructive Pulmonary Disease. 2008;3(4):671-682.




