Trends in Out-of-Hospital Deliveries and Infant Mortality
Births out of Hospital in France: Trends from 2013 to 2022, Infant Mortality and Risk Factors
Combier Evelyne 1, Cottenet Jonathan 1, Quantin Catherine 1,2
- Biostatistics and Bioinformatics (DIM), Dijon, France University Hospital, Dijon, France; Bourgogne Europe University, Dijon, France
- Université Paris-Saclay, UVSQ, Univ. Paris-Sud, Inserm, High-Dimensional Biostatistics for Drug Safety and Genomics, CESP, Villejuif, France
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Combier, E., Cottenet, J., et al., 2026. Births out of Hospital in France: Trends from 2013 to 2022, Infant Mortality and Risk Factors. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7195
ISSN 2375-1924
Abstract
Objectives of this study, based on an analysis of births and infant deaths in mainland France between 2013 and 2022, were: -a) to assess the annual number of births recorded in municipalities without maternity wards (out-of-hospital deliveries) and to analyze the trend in relation to the closure of maternity wards; -b) to estimate the excess risk of neonatal and infant mortality associated with out-of-hospital deliveries and the proportion of excess mortality attributable to them; -c) using routinely available data characterizing the environment, to identify the areas most at risk of out-of-hospital deliveries that would require specific interventions in the field of perinatal care.
Results: During this period, 0.48% of births (35,732 births) were recorded in 26,657 municipalities without a maternity ward. This rate increased from 0.37% in 2013 to 0.65% in 2022, while the number of towns with a maternity ward fell from 416 to 373. The risk of out-of-hospital delivery increases with the distance to the nearest maternity ward. A positive gradient is recorded from 30 km (1.7 from 31 km to 45 km; 2.4 from 46 km to 60 km and 3.0 from 61 km). The risk is multiplied by 1.3 in cases of difficulty accessing primary care services and by 1.8 when the woman lives in a rural municipality. The risks of out-of-hospital deliveries are unevenly distributed across the mainland area. In 2022, only 15 out of 96 departments had an out-of-hospital deliveries rate below the national average. These departments had fewer rural municipalities and more large urban areas, less difficulty accessing primary care, and shorter distances to the nearest maternity ward. The number of pregnant women living more than 30 km from a maternity ward was 6.5 times lower than in the rest of mainland France. The risks of infant and neonatal mortality, which are higher among out-of-hospital deliveries, are responsible for an excess of 204 deaths before the age of 1 and 180 neonatal deaths between 2013 and 2022. This excess neonatal mortality accounts for 2.71% of the excess neonatal mortality recorded in France between 2015 and 2017.
Keywords
out-of-hospital deliveries, infant mortality, neonatal mortality, perinatal care, France
Introduction
In France, the infant mortality rate defined as the number of deaths occurring before 1 year of age was estimated at 4.0‰ of live births in 2023. After decades of steady decline, this rate has plateaued and has even shown an upward trend over the past decade contributing to France’s fall in international rankings, from 7th place worldwide in 1989 to 25th in 2017. The rate increased further to 4.1‰ in 2024. This excess mortality is largely attributable to neonatal deaths, i.e., deaths occurring within the first 28 days of life, for which France ranked 22nd among 34 European countries. A recent comparative study estimated that, between 2015 and 2017, France recorded 2,292 excess neonatal deaths relative to the three highest-performing European countries (Finland, Norway, and Sweden).
This resurgence in infant mortality has occurred despite a succession of national perinatal plans intended to improve maternal and neonatal care. Substantial geographic disparities persist, not only between the overseas departments and mainland France but also within mainland regions. The objectives of these plans summarized in the 2005–2007 program by the principles of humanity, proximity, safety, and quality could only have been achieved if adequate healthcare services and professionals had been accessible to the entire population across the national territory, which has not been the case.
Like many countries, France has implemented a policy of perinatal care regionalization in the context of broader hospital restructuring and a growing shortage of medical specialists. These changes have contributed to the closure of numerous maternity units, particularly in rural areas thereby increasing the distance between women’s homes and the nearest maternity facility, reducing geographical access to obstetric care and heightening the risk of out-of-hospital deliveries (OHDs). Extensive evidence shows that delays in obstetric emergency care increase maternal and neonatal morbidity and mortality, and that long travel distances to delivery facilities are consistently associated with adverse outcomes for both mothers and newborns.
Inequalities in access to outpatient primary care further exacerbate these risks. A recent study highlighted the persistence of unequal access to general practitioners, specialists, nurses, pharmacies, and emergency services throughout France. Based on 2019 data, this analysis identified areas where targeted interventions would be necessary to restore equitable access to primary care nationwide. Despite full insurance coverage for pregnancy-related care, travel expenses remain the responsibility of pregnant women and are considerably higher in areas located far from outpatient services or maternity wards. Poverty, which is unevenly distributed across France, often overlaps with these medical deserts, and such financial and geographical barriers may lead to forgone care, inadequate pregnancy monitoring, and OHD, all of which contribute to increased perinatal morbidity and mortality.
Numerous French and international studies have shown that OHD is associated with increased neonatal complications and mortality, thereby contributing to the excess infant mortality observed in France. However, most existing analyses rely on hospital data and therefore include only newborns admitted after being born before arrival (BBA). These births include both unplanned OHD and planned home births complicated by perpartum events requiring transfer, making it difficult to isolate the risks specifically associated with unanticipated OHD.
In France, all births must be registered in the municipality where they occur, regardless of the place of delivery or subsequent hospitalization. As a result, any birth registered in a municipality without a maternity ward corresponds to a delivery outside a maternity facility. Although civil records do not distinguish between planned home births and unplanned OHD, comparing mortality among infants born in municipalities without maternity wards to those born elsewhere may provide an estimate of the excess neonatal and infant mortality associated with OHD. Furthermore, identifying the geographical distribution of these municipalities makes it possible to determine high-risk areas and analyze the environmental characteristics associated with them.
Objective
The objective of this study was to quantify the annual number of births registered in municipalities without maternity wards and describe their temporal evolution from 2013 to 2022 in the context of maternity unit closures, to estimate the excess risk of neonatal and infant mortality associated with out-of-hospital births and the proportion of excess mortality attributable to them, and to identify, using routinely available environmental data, the areas at greatest risk of OHD that may require targeted perinatal interventions.
Material and Methods
POPULATIONS STUDIED
Births
The study covered 7,396,441 live births and stillbirths recorded between 2013 and 2022 in all civil registries in municipalities in mainland France, regardless of the mothers’ place of residence. These civil registry data, which provide an exhaustive record of all births, were provided to us by INSEE (Institut National de la statistique et des Études Économiques). The data are anonymous and aggregated. For our analyses, we used available variables: year of birth, municipality of birth, mother’s municipality of residence, the status of the child at birth (live birth, stillbirth) or whether they were the subject of a declaratory judgment of birth (JDN). A JDN is issued by the court when the child has not been registered within the legal time limit.
Deaths
For deaths, we used data from the nominal files of deceased persons, which can be downloaded directly from the INSEE website. These files list all deaths recorded during the year in civil registries, regardless of age and place of death. They contain individual data such as date of birth, date of death, municipality of birth and municipality of death. Using the dates of birth and death, we calculated the age in days at the time of death and included infant deaths, i.e., those occurring before the age of 1. For each year of birth, deaths occurring before the age of 1 were searched for over three consecutive years (N, N+1, N+2). We only included deaths of children born in a municipality in mainland France, regardless of where they died.
ENVIRONMENTAL DATA
Access to local healthcare: To assess access to local healthcare, we used the IRDES score, which allows for the geographical identification of medical deserts at the municipal level. We grouped the seven clusters of the dendrogram used to construct the score into four classes (clusters 1-2, clusters 3-4, cluster 5, and cluster 6-7). Cluster 1-2 identifies and locates medical deserts. In contrast, clusters 6-7 and cluster 5 consist of municipalities where there were no difficulties in accessing healthcare or emergency services, while residents of municipalities in cluster 3-4 had difficulties accessing certain services.
As the score uses the localized potential accessibility (LPA) indicator established by the DREES for 2019, we only used 2019 data to search for environment-adjusted OHD risk factors.
Distance to the nearest maternity hospital: For all mainland municipalities and for each year separately, we calculated the distance in kilometers between the centroid of each municipality and the centroid of the municipality where the nearest maternity hospital is located. Maternity wards located in the same municipality are credited with the same distance. For municipalities where maternity wards are located, the distance is 0 km. To avoid taking into account changes related to municipal mergers and closures, we used the IGN map of municipal boundaries for the year 2020 for all years. For each year, the municipality where the maternity wards are located is that of the establishments listed by the DREES on December 31 of the current year. The median value of these distances was calculated for each year. Distances were calculated using CHRONOMAP© for MAPINFO© software and IGN Route 500® digital road network.
Urban areas/rural areas: We used the “municipal density grid” provided by INSEE to characterize the municipalities. This grid classifies municipalities according to the number of inhabitants and their distribution across the territory. It comprises seven levels, which are a subdivision of the three-category grid: 1) large urban centers, 2) “intermediate urban centers,” “urban belts,” and “small towns,” 3) rural municipalities, which include “rural towns,” “rural areas with scattered settlements,” and “rural areas with very scattered settlements.” For our analyses, we used the three-category classification.
Poverty rate by municipality: The poverty rate recorded in the population is calculated annually by INSEE at the level of municipalities and their groupings in public inter-municipal cooperation establishments (EPCI). As this information is confidential, this rate is only known for municipalities with a sufficient number of registered households. In order to be able to use this information for all municipalities, we assigned those with missing data the poverty rate attributed to the EPCI to which the municipality belongs. Municipalities were divided into three classes according to their position in relation to the mainland average poverty rate: 1) wealthy municipalities with a poverty rate below the average rate minus two standard deviations, 2) poor municipalities with an average rate above the average plus two standard deviations, and 3) an average level with a rate between the two standard deviations.
METHODS: DATA PROCESSING
Analysis of births
With regard to births, we first identified OHDs by using the annual census of maternity wards conducted by DREES to identify the municipalities of birth that had a maternity ward, the others being those where OHDs could be registered with the civil registry. We created an OHD variable that was coded, for each year, 1 for births in municipalities without maternity wards and 0 for births in municipalities with maternity wards. The distances to the nearest maternity ward were then assigned year by year to the municipalities of residence and grouped into four categories: 0-30 km, 31-45 km, 46-60 km, and 61 km and above.
Descriptive analysis
An initial descriptive analysis of the status of children at birth (live births, stillbirths, and JDN) was performed year by year on the entire database and then on the OHD population. We used a Somers’d test to evaluate trends over the years. We also evaluated the evolution of the OHD rate for the period 2013-2022. The impact of the decrease in the number of maternity wards on the evolution of the number of OHDs was highlighted in two ways: 1) by plotting the number of cities with maternity wards and the number of OHDs on the same graph, year by year, 2) by quantifying, using linear regression, the change in the annual OHD rate relative to the median distance to the nearest maternity ward in all mainland municipalities. For the regressions, we only took into account the period prior to the COVID-19 epidemic, i.e., the years 2013-2019.
Environmental risk factors for out-of-hospital deliveries
To determine the environmental risk factors associated with the risk of OHD, we used data from 2019, which is the year that the IRDES indicator of accessibility to local health services was constructed. The classes of the different factors were coded as 0/1. To calculate the relative risks associated with the environment, multivariate analyses were done using regressions based on Generalized Estimating Equations (GEEs) with a log link function and negative binomial distribution to consider the data correlations (municipalities and years) and over-dispersion. All variables were tested one by one in the regressions, but the model converged better with all variables included, according to the QIC statistic.
Statistical analyses were performed using SAS® version 9.4 (SAS Institute Inc., Cary, NC, USA). Statistical significance was defined as a p-value <0.05.
Empirical validation of risk factors for out-of-hospital deliveries
To enable empirical validation of OHD risk factors, we used the most recent data, i.e., data from 2022, and worked at the departmental level. To calculate OHD rates by department of residence of mothers, we excluded stillbirths and JDNs as well as births domiciled outside mainland France. We divided the departments into three groups based on their statistical difference from the mainland rate (high, low, average). Statistical significance was defined as a p-value <0.05.
Map representation
The three groups were represented on a map using MAPINFO© software. We created a map showing 1) departments with OHD rates statistically higher than the mainland average and 2) those with rates statistically lower than the average.
Validation of significant risk factors
For each of the significant risk factors, we verified that there were differences in the distribution of risk levels between the three groups and that the differences recorded were consistent with the results of the statistical analyses performed using the 2019 data.
Analysis of deaths
- For each year, we counted the number of deaths at D0, from D0 to D27, and from D0 to D365, for each municipality of birth.
- We extracted the subgroup of live births from the civil registry birth file and retained three variables: municipality of birth, the OHD variable, and the number of live births.
- In order to calculate neonatal and infant mortality rates, the live birth file was matched to the death file, by year, using the “municipality of birth” variable.
- Annual neonatal and infant mortality rates were calculated for mainland France, by dividing the total number of deaths by the total number of live births.
- We repeated the operation for only those municipalities of birth that did not have a maternity ward (OHD=1).
- By difference between the two rates, we calculated the mortality rates for children born in towns with a maternity ward (OHD=0). We used a Somers’d test to evaluate trends in mortality over the years.
- The crude relative risks of infant (D0-D365) and neonatal (D0-D27) excess mortality among OHDs were calculated year by year by comparison with births in towns with maternity wards.
- Excess mortality is the difference between the number of deaths recorded among OHDs and the number of deaths that would have been expected if the mortality rate had been the same as that recorded for births in towns with maternity wards (OHD=0).
Results
BIRTHS 2013 2022
From 2013 to 2022, the civil registry services of mainland France recorded 7,396,441 births, all newborn statuses combined (Table I (A)). Of these, 7,311,669 (98.85%) were live births, 79,480 (1.08%) were stillbirths, and 5,292 (0.07%) were subject to a declaratory judgment of birth (JDN) that did not specify the status of the child at birth. During the study period, except for 2021, there was a steady decline in the annual number of registrations, from 789,582 in 2012 to 694,778 in 2022. This decline relates solely to the number of live births registered, which fell from 781,240 in 2013 to 685,894 in 2022, with a decrease in the percentage of the stratum from 98.94% in 2013 to 98.72% in 2022. Conversely, the stillbirth rate increased from 1.01% in 2013 to 1.18% in 2022, with acceleration from 2020 onwards. As for JDNs, the rate doubled in 10 years, rising from 0.05% in 2013 to 0.10% in 2022. The trends are significant (p<10-4)
EVOLUTION OF OUT-OF-HOSPITAL DELIVERIES 2013-2022
During this period (Table I(B)), 35,732 births (0.48%) were recorded in 26,657 municipalities without maternity wards (OHDs). This rate rose from 0.37% in 2013 to 0.65% in 2022. The trend is significant (p<10-4). Among these births, 98.00% were live births, 1.26% were stillbirths, and 0.74% were JDNs. This distribution did not vary over the 10 years of the study (p=NS). Only 15.10% of these births were registered in a municipality other than the one declared as the municipality of residence. This percentage has been steadily decreasing. It was 16.87% in 2013 and 14.42% in 2022. The trend is significant (p=0.0021).
The number of cities with at least one maternity hospital fell from 416 to 373, leading to an increase in the annual medians for the distance in kilometers and travel time to the nearest maternity ward. These medians rose from 22.53 km and 22.90 min in 2013 to 23.94 km and 24.00 min in 2022, respectively. The trend is significant for both distances (p=0.0014) and travel times (p=0.0033). These medians are those of the distances and travel times calculated for each municipality in mainland France and for each year. The number of OHDs is negatively correlated with the number of cities that have maternity wards (R=-0.83).
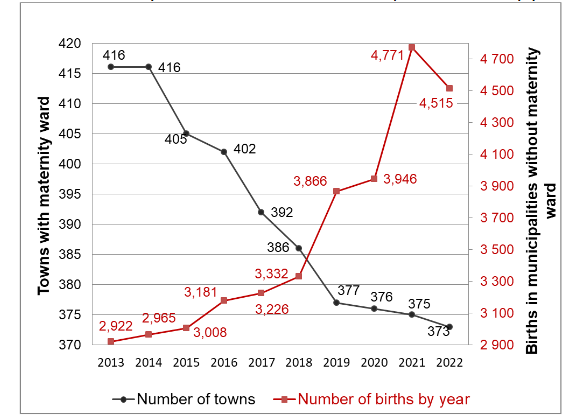
As for the evolution of the annual percentages of OHDs, for the years 2013-2019, it is a linear function of the median distance in kilometers to the nearest maternity ward, both for all births and for live births. The period of the COVID-19 epidemic is associated with a significant increase in the percentage of births in cities without maternity wards. This percentage remains high for 2022, although it is lower than that recorded in 2021.
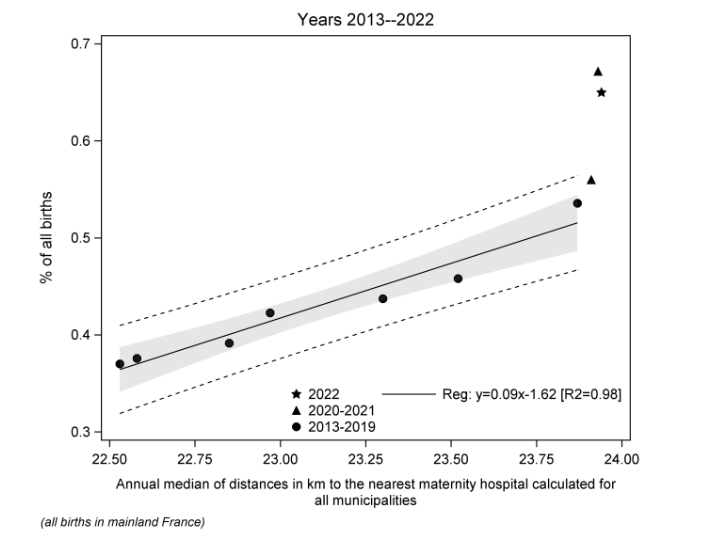
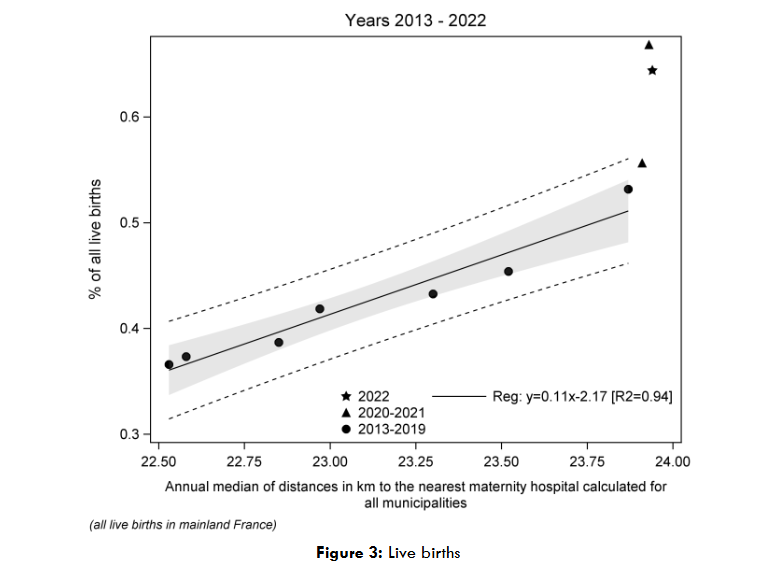
Figure 2: Change in out-of-hospital delivery rates according to increasing distances to maternity wards
CHANGES IN INFANT AND NEONATAL MORTALITY RATES BETWEEN 2013 AND 2022
Consultation of the INSEE 2013-2024 files of deceased persons enabled us to identify 25,409 deaths of children under 1 year of age who were born alive and whose births were registered in mainland France between 2013 and 2022. These deaths should be compared with the 7,311,669 live births recorded during the same period. The average infant mortality rate for 2013-2022 was 3.48‰ and the average neonatal mortality rate (D0-D27) was 2.52‰. We see the upward curves already reported for both infant mortality, which rose from 3.43‰ in 2013 to 3.64‰ in 2022 (p=0.0096), and neonatal mortality, which rose from 2.41‰ to 2.69‰ (p<10-4).
CHANGES IN MORTALITY RATES.
From 2013 to 2022, there were 326 infant deaths and 268 neonatal deaths for 35,015 OHDs, representing rates of 9.31‰ and 7.65‰, respectively.
- There was no increase in the annual infant mortality rate among the OHD population (p=0.3246), unlike that recorded among children born in cities with maternity wards (p=0.0142).
- As with infant mortality, no significant trend was recorded in the evolution of the neonatal mortality rate (p=0.0788) among OHDs, unlike the mortality rates recorded among children born in towns with maternity wards (p<10-4).
It should be noted that in 2022, the neonatal mortality rate was 5.43‰, which is lower than the average for the period (p<0.01) for OHDs.
INCREASED RISK OF MORTALITY AND POTENTIALLY PREVENTABLE MORTALITY AND DEATHS
Comparing the average mortality rates recorded from 2013 to 2022 among OHDs and children born in cities with maternity wards, we find that OHDs have an average excess risk of infant mortality of 5.85‰ and neonatal mortality of 5.16‰. This resulted in an excess of 204 deaths among children under 1 year of age and 180 deaths among infants in the neonatal period for the period 2013-2022.
For this period, the median relative risk is 2.9 (95% CI: 2.1-4.0) for infant mortality and 3.3 (95% CI: 2.3-4.6) for neonatal mortality. No relative risk was equal to or less than 1. The minimum risk was recorded in 2022: it was 2.1 (95% CI: 1.5-2.9) for infant mortality and 2.0 (95% CI: 1.4-3.0) for neonatal mortality.
PROPORTION OF INFANT AND NEONATAL MORTALITY DUE TO OUT-OF-HOSPITAL DELIVERIES
The 326 infant deaths and 268 neonatal deaths recorded between 2013 and 2022 among OHDs, relative to the total number of live births, account for 0.045‰ infant deaths and 0.037‰ deaths from D0 to D27 over the entire period. For infant mortality, there was an increase in the annual rate (p=0.0062) for OHDs, as well as for the rates calculated for the total population (p=0.0096) and for births in cities with maternity wards (p=0.0218).
Among OHDs, for death rates from D0 to D27 in 2013-2022, the trend test is not significant (p=0.1006) probably due to the low mortality rate recorded in 2022 (0.035‰), attributable to the low number of deaths reported on D0. In fact, in 2022, only 8 deaths were recorded on the day of birth (D0) for 4,416 births (1.81‰), whereas since 2013 the minimum number of deaths recorded was 10 in 2018 for 3,264 births (3.06‰). This decrease in the number of deaths at D0 in 2022 should be viewed in light of the increase in stillbirths in the same year compared to 2021 (68 vs 49), bringing the stillbirth rate for this population from 10.3‰ total births in 2021 to 15.1‰ in 2022.
Table 2: Distribution of infant and neonatal mortality rates by place of birth
| Years | All live births | OHD deaths | Other deaths | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Infant mortality | 2013 781,240 2,683 3.43 25 0.032 2,658 3.40 | 2014 780,818 2,570 3.29 31 0.040 2,539 3.25 | 2015 759,984 2,642 3.48 30 0.039 2,612 3.44 | 2016 744,310 2,504 3.36 28 0.038 2,476 3.33 | 2017 729,830 2,637 3.61 35 0.048 2,602 3.57 | 2018 719,180 2,595 3.61 30 0.042 2,565 3.57 | 2019 713,375 2,494 3.50 35 0.049 2,459 3.45 | 2020 695,942 2,295 3.30 37 0.053 2,258 3.24 | 2021 701,096 2,494 3.56 42 0.060 2,452 3.50 | 2022 685,894 2,495 3.64 33 0.048 2,462 3.59 |
| All 7,311,669 25,409 3.48 326 0.045 25,083 3.43 |
Neonatal mortality D0-D27
| Years | All live births | OHD deaths | Other deaths | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 2013 781,240 1,886 2.41 21 0.027 1,865 2.39 | 2014 780,818 1,782 2.28 27 0.035 1,755 2.25 | 2015 759,984 1,885 2.48 28 0.037 1,857 2.44 | 2016 744,310 1,823 2.45 25 0.034 1,798 2.42 | 2017 729,830 1,931 2.65 30 0.041 1,901 2.60 | 2018 719,180 1,850 2.57 21 0.029 1,829 2.54 | 2019 713,375 1,845 2.59 28 0.039 1,817 2.55 | 2020 695,942 1,735 2.49 31 0.045 1,704 2.45 | 2021 701,096 1,811 2.58 33 0.047 1,778 2.54 | 2022 685,894 1,842 2.69 24 0.035 1,818 2.65 |
| All 7,311,669 18,390 2.52 268 0.037 18,122 2.48 |
In 2015-2017, France recorded an excess neonatal mortality of 2,212 newborns. During the same period, excess mortality linked to OHDs was 60 neonatal deaths recorded in mainland France, representing 2.71% of the total national excess mortality.
ENVIRONMENTAL RISK FACTORS FOR GIVING BIRTH IN A TOWN WITHOUT A MATERNITY WARD
In 2019, the reference year for the use of the score developed by IRDES, of the 721,669 births registered with the civil registry in mainland France, 716,104 were also domiciled there. Of these, 3,866 (0.54%) were in municipalities that did not have a maternity ward.
Population density
Less than half of births (47.2%) were in large urban centers, 25.6% in other urban areas, and 27.2% in rural areas. While the OHD rate was 0.29% in large urban centers, it was 0.51% in other urban areas and 1.00% in rural areas. The unadjusted relative risks of OHD [RRc (95% CI)] was respectively [1.4(1.3-1.6)] and [2.5(2.3-2.8)].
Poverty rate of the municipality
Only 1.7% of births were registered in municipalities with a poverty rate more than 2 standard deviations below the mainland average and 19.3% in municipalities with a rate more than 2 standard deviations above it. The OHD rates were 0.61% for the richest municipalities, 0.33% for the poorest municipalities, and 0.59% for municipalities in the mainland average. The trend is significant (p<10-4). The unadjusted relative risk of OHD [RRc (95% CI)] compared to that of the middle-income reference group is significant for the poorest group [0.7(0.6-0.9)] and borderline significant for the richest group [0.8(0.6-1.0)].
Distance in km to the nearest maternity hospital
For 660,919 (92.3%) births, the declared place of residence was less than 31 km from a maternity hospital. This distance was greater than 45 km for 11,314 (1.6%) births and greater than 61 km for 2,077 (0.3%) of them. The OHD rates increased with the distance from the home to the nearest maternity ward. It was 0.46% from 0 to 30 km, 1.29% from 31 to 45 km, 2.02% from 46 to 60 km, and 2.57% at 61 km and above. The trend is significant (p<10-4). Compared to the 0-30 km distance class, the unadjusted relative risks [RRc (95%CI)] were [2.1 (1.9-2.4)], [3.3 (2.8-3.8)], and [4.0 (3.0-5.4)], respectively.
Access to primary and emergency care services
In 2019, 86.5% of births were in municipalities with no (clusters (6-7)) or few (cluster (5)) difficulties in accessing primary care. In contrast, for 9.9% of these (71,054 births), access to such care was difficult for certain specialties (clusters (3-4)), and 3.6% (25,914 births) were domiciled in medical deserts (clusters (1-2)). The OHD rate was higher when access to primary care and emergency services was more difficult. It was 1.27% in medical deserts compared to 0.41% for municipalities with no difficulty accessing care. Compared to these municipalities, there is a gradient of crude relative risks [RRc (95% CI)]: cluster (5) [1.2(1.1-1.3)], clusters (3-4) [2.0(1.8-2.1)] and clusters (1-2) [2.1(1.9-2.4)].
RISK FACTORS FOR OUT-OF-HOSPITAL DELIVERIES AFTER ADJUSTMENT
The degree of poverty in the municipality was no longer significant compared to the mainland average, whether the municipalities were poor [adjusted relative risk (RRa):1.0(0.9-1.2)] or rich [RRa:1.0(0.8-1.2)]. The other RRa for OHD were significantly higher than 1: for municipalities located in rural areas [RRa: 1.8 (1.6-2.1)] and for municipalities in urban areas other than large centers [RRa: 1.3 (1.2-1.5)], with large urban centers as the reference. For difficulties in accessing primary care limited to certain services [RRa:1.3(1.2-1.5)] or for medical deserts [RRa:1.3 (1.2-1.5)]. For distances greater than 30 km with a risk gradient: 31-45 km [RRa:1.7(1.5-1.9)], 46-60 km [RRa:2.4 (2.1-2.9)] and 61 km and above [RRa:3.0(2.3-4.1)].
Table 3: Increased risk of mortality among out-of-hospital deliveries
| Years | Births in towns with maternity | Births in towns without maternity ward (OHD) | Excess deaths | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Infant mortality rates | 2013 778,382 2,658 3.41 2,858 25 8.75 2.6 (1.7-3.8) 5.34 15 | 2014 777,903 2,539 3.26 2,915 31 10.63 3.3 (2.3-4.6) 7.37 21 | 2015 757,044 2,612 3.45 2,940 30 10.20 2.9 (2.1-4.2) 6.75 20 | 2016 741,195 2,476 3.34 3,115 28 8.99 2.7 (1.9-3.9) 5.65 18 | 2017 726,672 2,602 3.58 3,158 35 11.08 3.1 (2.2-4.3) 7.50 24 | 2018 715,916 2,565 3.58 3,264 30 9.19 2.6 (1.8-3.7) 5.61 18 | 2019 709,919 2,459 3.46 3,792 35 9.23 2.7 (2.1-4.1) 5.77 22 | 2020 692,070 2,258 3.26 3,872 37 9.56 2.9 (2.1-4.0) 6.30 24 | 2021 696,411 2,452 3.52 4,685 42 8.96 2.5 (1.9-3.4) 5.44 25 | 2022 681,478 2,462 3.61 4,416 33 7.47 2.1 (1.5-2.9) 3.86 17 |
| All 7,276,990 25,083 3.45 35,015 326 9.31 5.85 204 |
Neonatal mortality rates D0-D27
| Years | Births in towns with maternity | Births in towns without maternity ward (OHD) | Excess deaths | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 2013 778,382 1,865 2.40 2,858 21 7.35 3.1 (2.0-4.7) 4.95 14 | 2014 777,903 1,755 2.26 2,915 27 9.26 4.1 (2.8-6.0) 7.00 20 | 2015 757,044 1,857 2.45 2,940 28 9.52 3.9 (2.7-5.6) 7.07 21 | 2016 741,195 1,798 2.42 3,115 25 8.03 3.3 (2.2-4.9) 5.61 17 | 2017 726,672 1,901 2.62 3,158 30 9.50 3.6 (2.5-5.2) 6.88 22 | 2018 715,916 1,829 2.55 3,264 21 6.43 2.5 (1.6-3.9) 3.88 13 | 2019 709,919 1,817 2.56 3,792 28 7.38 2.9 (2.2-4.6) 4.82 18 | 2020 692,070 1,704 2.46 3,872 31 8.01 3.3 (2.3-4.6) 5.55 21 | 2021 696,411 1,778 2.55 4,685 33 7.04 2.8 (2.0-3.9) 4.49 21 | 2022 681,478 1,818 2.67 4,416 24 5.43 2.0 (1.4-3.0) 2.76 12 |
| All 7,276,990 18,122 2.49 35,015 268 7.65 5.16 180 |
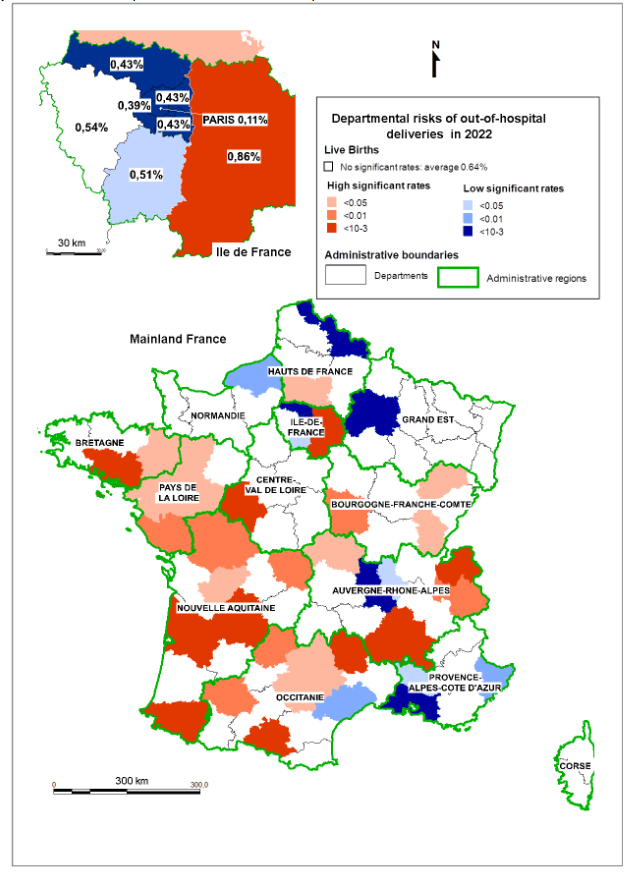
Empirical validation of risk factors: In 2022, 4,408 of the 4,416 records of live births made in the civil registries of municipalities without maternity wards (OHDs) were domiciled in all 96 departments of mainland France. The average rate of these births for the whole of mainland France (34,655 municipalities) was 0.64%. However, the rate varied according to the department of residence: 50 (20,221 municipalities) had an OHD rate that was not significant compared to the average (middle-class), 15 (3,652 municipalities) had a lower rate (low risk), and 31 (10,782 municipalities) had a higher rate (high risk).
Distribution of factors across risk levels
The differences in the distribution of the various risk factors across municipalities were statistically evaluated by successively comparing the High and Low-risk groups with the Middle-class group. Compared to the Middle-class group, access to primary care and emergency services was more difficult in the High-risk group and lower in the Low-risk group, at 61.7% and 38.6% vs. 58.4%, respectively.
Rural areas were underrepresented in the low-risk group (71.1% vs. 90.0%). There was no significant difference between the high-risk group and the middle class except for other urban areas, which were less represented in the high-risk group. The distances to the nearest maternity hospital, whether measured from each municipality or from the homes of women who gave birth, were shorter in the low-risk group compared to the middle-class group and greater in the high-risk group.
Map 1: Distribution of departmental risks of out-of-hospital deliveries in mainland France in 2022
Discussion
MAIN FINDINGS
In this study, out-of-hospital deliveries (OHDs) were defined as births registered in municipalities without maternity wards, rather than using the “place of delivery” variable available in INSEE birth data, which has a higher rate of missing information than the reported rate of OHDs and is subject to unknown self-reporting classification errors. This definition captures births that are unquestionably out of hospital and directly linked to the spatial organization of maternity services. It does not include births outside maternity wards recorded in municipalities that do have a maternity unit. Approximately 500 such births per year occur in midwife-led birth centers attached to maternity wards, which should be considered institutional births rather than OHDs.
Using this definition, we observed a marked increase in OHDs over a 10-year period, negatively correlated with the decrease in the number of municipalities hosting a maternity unit, consistent with international observations. Alongside the increase in the number of OHDs, there has been an increase in the number of towns without maternity wards where births have been registered, reflecting the widespread nature of the phenomenon across the country. This phenomenon is accompanied by an increase in the percentage of home births registered in the same municipality as the mother’s place of residence. This change may be due either to a greater number of women choosing to give birth at home, or to a change in the practices of emergency services professionals who prefer to deliver babies at home rather than during transport in an ambulance. As the data available does not allow us to distinguish between planned and unplanned home births, we have not been able to assess the change in the respective proportions of the two scenarios. The availability of the mother’s municipality of residence in civil registry data allowed us to calculate departmental OHD rates and to identify environmental risk factors associated with OHD.
Except for the municipal-level poverty indicator, all other variables studied influenced the risk of OHD. Distance to the nearest maternity ward was the strongest predictor, with a notable gradient starting at 31 km (RRa 1.7) and a threefold increase in risk from 61 km onward. Limited access to primary care increased the risk by 1.3. Risk was elevated throughout the territory outside major urban centers, rising by 1.3 in other urban areas and by 1.8 in rural municipalities.
The spatial distribution of departmental OHD rates aligns with the patterns described in the report from the National Academy of Medicine. Low-risk departments contained more large urban centers, enjoyed better primary care access, and had shorter distances to maternity wards, and were mainly located in regions known for better maternity access (Hauts-de-France, Île-de-France, Provence-Alpes-Côte d’Azur). High-risk departments were more rural, had poorer primary care access, and longer distances to maternity facilities. Two departments in Pays-de-la-Loire (Loire-Atlantique and Maine-et-Loire) were exceptions, as all municipalities are located within 45 minutes of a maternity unit. Their classification may be related to the spatial distribution of home-birth midwives and doulas, but no data covering the study period were available.
Regarding infant mortality, only the municipality of birth but not residence is recorded in death certificates. Although about 10% of women deliver outside their department of residence, rates of “leakage” can be substantial in rural areas after successive maternity closures, as illustrated for the Nièvre department. As births and resulting deaths are registered in neighbouring departments, mortality attributed to Nièvre appears underestimated. This underestimates the influence of environmental risk factors such as distance to maternity units and primary-care access. Because maternal residence was unavailable in the death registry, we could not analyse environmental determinants of mortality nor compute departmental mortality rates, but the registry did allow us to identify OHDs and calculate neonatal and infant mortality associated with them in mainland France.
Nevertheless, our analysis shows that OHDs accounted for 1.3% of total infant mortality over the study period (0.045‰ out of an average 3.48‰). However, they contributed 2.71% of excess neonatal mortality recorded between 2015 and 2017. With rising OHD numbers and no downward trend in OHD-specific mortality over 10 years despite national recommendations, continued closure of maternity units serving isolated areas is unlikely to reduce infant mortality. OHDs also increase risks of severe maternal morbidity and mortality, particularly from obstetric haemorrhage.
The stillbirth rate recorded in mainland France increased over the period as a whole, rising from 1.01% in 2013 to 1.18% in 2022. Although it was consistently higher among OHDs, no upward trend was recorded in this group.
These results are difficult to interpret as an indicator of the health status of a population, because civil registration of stillbirths is not mandatory and gestational age and reporting limits remain undefined. This is why the authorities have chosen to monitor the evolution of this indicator using hospital data. For the same reasons, we did not calculate stillbirth rates by department.
However, we did not exclude stillbirths when researching risk factors for OHDs because we were interested in the place of delivery and not the condition of the child at birth. These risk factors concern all pregnant women residing in mainland France, regardless of the outcome of their pregnancy.
INTERPRETATION
The progressive rise in OHDs seems to reflect the long-term consequences of maternity unit closures and the broader regionalization of perinatal care. Originally intended to improve outcomes for births <33 weeks of gestation, regionalization implemented through Decrees 98-899 and 98-900 following recommendations from the High Committee for Public Health encouraged closure of units performing fewer than 300 deliveries annually, with exemptions for isolated areas that have become increasingly rare. Current professional recommendations favour concentrating deliveries in high-volume units, and the National Academy of Medicine now recommends a minimum of 1,000 births per unit. According to our results, applying such thresholds without other compensating measures could intensify territorial inequalities in maternity access, particularly in rural areas already facing shortages of health services.
Arguments in favour of further regionalization often focus on the fact that 75.4% of excess neonatal mortality between 2015 and 2017 was attributable to extreme prematurity. Yet excess mortality is also observed at term, and our national healthcare PMSI data confirm that most OHDs occur at ≥37 weeks (89.7% from 2015–2017 and 90.3% from 2013–2022). Improving perinatal outcomes therefore should require attention to all pregnant women, not only those at risk of very preterm birth.
The geographic distribution of the population reinforces this observation: in 2019, over one-quarter (27.2%) of women giving birth lived in rural areas, 55,185 travelled more than 30 km (7.7%) to reach a maternity ward, and 11,314 (1.6%) travelled more than 45 km. Despite fewer births in 2022 than in 2019, the number of women living more than 30 km from a maternity unit increased to 56,867 (8.2%), with 12,300 (1.8%) living more than 45 minutes away. As distance is the strongest determinant of OHD, further closures would almost certainly worsen neonatal and maternal outcomes.
STRENGTHS AND LIMITATIONS
A major strength of this study is its use of civil registry data, in addition to hospital records, enabling exhaustive identification of all births occurring in municipalities without maternity wards over a decade. Unlike hospital-based datasets, which include only newborns admitted after being born before arrival (BBA), civil registry data allow capture of all OHDs, regardless of subsequent care. The data also provided access to fine-scale environmental context for mothers’ municipalities.
Several limitations should be noted. First, due to missing and potentially inaccurate information in the self-reported “place of birth” variable, OHDs had to be defined indirectly using municipal maternity availability. This ensures specificity but excludes OHDs occurring in municipalities with maternity units. Second, the absence of maternal residence in death certificates prevents assessment of environmental factors associated with mortality, especially in departments where interdepartmental leakage is high. Third, stillbirths were not analysed due to inconsistent civil registration. Finally, the absence of data on home-birth midwives and doulas prevented analysis of their contribution to local OHD patterns.
Despite these limitations, the study provides strong evidence that declining geographic accessibility to maternity services has contributed to a sustained increase in OHDs and that these births disproportionately influence excess neonatal mortality. Ensuring equitable territorial access to maternity care should therefore be considered a prerequisite for any effective national perinatal strategy.
WHAT SOLUTIONS ARE THERE TO ENSURE SAFE PREGNANCIES IN REMOTE AREAS?
To avoid OHDs, the first solution that was considered was to systematically induce labor at 39 weeks in low-risk women who live far from a maternity ward. However, this measure is not without risk. While studies show contradictory short and long-term outcomes for newborns, the same cannot be said for mothers, for whom several authors have reported an increased risk of hemorrhage, hysterectomy, and amniotic fluid embolism. This type of practice also raises ethical issues, particularly the use of a medical procedure that is neither necessary nor harmless to solve the travel time problems caused by the consolidation of maternity wards.
Following the model of maternity waiting homes established in certain remote areas of Scandinavian countries, the health insurance system offers pregnant women who live more than 45 minutes from a maternity ward the option of staying in accommodation close to the maternity ward for the five nights preceding their expected delivery date. However, this measure has its limitations, as only 70% of women gave birth at 39 weeks or more, and according to the PMSI, 10% of OHDs admitted to hospital between 2013 and 2022 were premature births.
Some experimentations have also been tried in some remote areas in France. For example, as part of the suspension of the Autun hospital center’s maternity license, in order to ensure the safety of unexpected deliveries at home or en route, a mobile emergency obstetric team attached to a structure called “SMUR obstétrical” has been set up on an experimental basis. This service was staffed with a sufficient number of midwives to provide 24/7 care, supplemented during interventions by an emergency physician trained in certain obstetric practices, an anesthetist nurse, and a paramedic. This team has a specially equipped vehicle to perform deliveries en route or at home. An incubator is available for births before 34 weeks. This system is attractive but also dangerous: it increases the time between the woman’s departure and arrival at the maternity ward, as the time taken by the SMUR to reach the woman’s home must be added to the journey time; unlike planned home births or births in birthing centers, women in labor are not necessarily low-risk, and possible perpartum accidents can occur, in particular amniotic fluid embolisms and catastrophic obstetric hemorrhages, which can lead to maternal death. In fact, five such cases were reported between 2016 and 2018 in the national survey on maternal deaths (one retroplacental hematoma, three uterine ruptures, and one amniotic fluid embolism). Finally, this service does not appear to be suited to the needs of geographically isolated women in labor, judging by the low number of interventions performed.
Unlike France, some Scandinavian countries such as Finland, Sweden, and Norway, which have the lowest recorded neonatal mortality rates, have chosen to maintain maternity wards that perform fewer than 500 deliveries to serve isolated areas. In Canada, there is a gradation of services with or without the possibility of cesarean sections on site, or services where cesarean sections are performed by trained general practitioners. In other countries with isolated populations, such as Australia, Denmark, New Zealand, and the United Kingdom, low-risk pregnancies can be managed in independent facilities run by midwives. A meta-analysis of maternal and perinatal outcomes by planned place of birth among women with low-risk pregnancies in high-income countries shows that the choice of place of birth has no impact on infant mortality and that there is a reduction in the risks of maternal morbidity and obstetric interventions when women give birth in a birth center or at home.
In France, there are birth centers run by midwives, but the law says they have to be next to a maternity that they have an agreement with. This kind of setup gives people more options, but it doesn’t meet the needs of women who live in rural areas more than 45 minutes away from a maternity ward. Rather than deploying obstetric SMURs, whose efficiency remains unevaluated, midwife-led birthing centers could be set up, the benefits of which have already been demonstrated in certain developed countries. In France, when a maternity ward is closed, the service is transformed into a “centre perinatal de proximité” (CPP). Midwives and at least one gynecologist-obstetrician provide prenatal and postnatal consultations, childbirth preparation classes, newborn care instruction, and family planning consultations. Only the practice of delivering babies is prohibited. There would be an opportunity to install midwife-led birthing centers in these CPPs. This is a hospital environment that pregnant women continue to frequent and where we regularly record deliveries by women who did not have time to reach the maternity ward. We counted them as OHDs in this study. Admittedly, there is no permanent obstetrician on staff, but there is also no obstetrician on the SMUR obstetric team, as it is the emergency physicians who have been trained in certain obstetric practices who intervene.
Conclusion
The risk factors for out-of-hospital delivery (OHD) identified in this study using routinely available data highlight substantial spatial inequalities in access to maternity services. These factors could serve as indicators of need, enabling public health authorities to identify areas where targeted interventions for low-risk pregnancies should be prioritized.
While improving the care of extremely preterm infants may require the continued regionalization of perinatal services, this process should not come at the expense of pregnant women living in underserved areas. No maternity unit should be closed without the prior implementation of an adequate and accessible alternative.
The development of midwife-led birthing centers successfully adopted in several countries may represent one such alternative. However, their implementation would require changes to current regulatory standards governing maternity services and to existing models of health facility financing.
Conflicts of Interest Statement
The authors have no conflicts of interest to declare.
Acknowledgements
We would like to thank the National Institute of Statistics and Economic Studies (Institut national de la statistique et des études économiques – INSEE), as well as the Public Statistics Data Archives (Archives de Données Issues de la Statistique Publique – ADISP) and for the data used in this study:
- Monthly municipal civil status statistics (births) – 2021, INSEE [producer], ADISP [distributor] (État civil communal mensuel (naissances) – 2021, INSEE [producteur], ADISP [diffuseur])
- Civil status – Births: Custom tabulation, INSEE [producer], ADISP [distributor] (État civil – Naissances : Tabulation sur mesure, INSEE [producteur], ADISP [diffuseur])
References
- Trinh N, de Visme S, Cohen JF, et al. Recent historic increase of infant mortality in France: A time-series analysis, 2001 to 2019. Lancet Reg Health – Eur. 2022;16:100339. doi:10.1016/j.lanepe.2022.100339
- Papon S. La mortalité infantile est stable depuis dix ans après des décennies de baisse – Insee Focus – 117. Accessed May 19, 2025. https://www.insee.fr/fr/statistiques/3560308
- Sartorius V, Philibert M, Klungsoyr K, et al. Neonatal Mortality Disparities by Gestational Age in European Countries. JAMA Netw Open. 2024;7(8):e2424226. doi:10.1001/jamanetworkopen.2024.24226
- INED. Natalité, mortalité, mortalité infantile – Europe et pays développés – Les chiffres. Published online July 2024. Accessed May 5, 2025. https://www.ined.fr/fr/tout-savoir-population/chiffres/europe-pays-developpes/natalite-mortalite-mortalite-infantile/
- Cortes A, Leurquin S. 4,1 Le scandale des accouchements en France. Buchel Chastel; 2025.
- Cour des Comptes. La politique de périnatalité. Des résultats sanitaires médiocres, une mobilisation à amplifier; 2024. Accessed May 17, 2025. https://www.ccomptes.fr/fr/documents/69727
- Vigneron E. Inégalités de santé, inégalités de soins dans les territoires français. Bull Académie Natl Médecine. 2012;196(4):939-952. doi:10.1016/S0001-4079(19)31781-9
- Puech F. Le temps, les temporalités et l’état de l’organisation des soins en périnatalité en France. Périnatalité. 2019;11(2):73-77. doi:10.3166/rmp-2019-0049
- Coldefy M, Com-Ruelle L, Lucas-Gabrielli V. Distances et temps d’accès aux soins en France métropolitaine. IRDES – Quest Déconomie Santé. 2011;(164). Accessed November 28, 2025. http://www.irdes.fr/Publications/2011/Qes164.pdf
- Haut Comité de Santé Publique. La sécurité et la qualité de la grossesse et de la naissance: pour un nouveau plan périnatalité. ENSP; 1994.
- NHS University of York. Hospital volume and health care outcomes,cost and patient access. Eff Health Care. 1996;2(8). Accessed October 29, 2025. https://www.york.ac.uk/media/crd/ehc28.pdf
- Heller G, Richardson DK, Schnell R, Misselwitz B, Künzel W, Schmidt S. Are we regionalized enough? Early-neonatal deaths in low-risk births by the size of delivery units in Hesse, Germany 1990–1999. Int J Epidemiol. 2002;31(5):1061-1068. doi:10.1093/ije/31.5.1061
- Ville Y, Rudigoz RC, Hascoët JM. Rapport 23-05. Planification d’une politique en matière de périnatalité en France : organiser la continuité des soins est une nécessité et une urgence. Bull Académie Natl Médecine. 2023;207(5):560-575. doi:10.1016/j.banm.2023.03.017
- CNGOF. Pérennité des équipes, quel avenir pour la continuité des soins en gynécologie obstétrique?; Commission démographie: rapport 2022.
- Charreire H, Combier E, Michaut F, et al. Une géographie de l’offre de soins en restructuration : les territoires des maternités en Bourgogne. Cah Géographie Qué. 2011;55(156):491-509. doi:10.7202/1008891ar
- Blondel B, Drewniak N, Pilkington H, Zeitlin J. Out-of-hospital births and the supply of maternity units in France. Health Place. 2011;17(5):1170-1173. doi:10.1016/j.healthplace.2011.06.002
- Combier E, Charreire H, Le Vaillant M, et al. Perinatal health inequalities and accessibility of maternity services in a rural French region: closing maternity units in Burgundy. Health Place. 2013;24:225-233. doi:10.1016/j.healthplace.2013.09.006
- Kildea S, McGhie AC, Gao Y, Rumbold A, Rolfe M. Babies born before arrival to hospital and maternity unit closures in Queensland and Australia. Women Birth. 2015;28(3):236-245. doi:10.1016/j.wombi.2015.03.003
- Pilkington H. Risques cachés de l’accouchement en France : les défis des naissances inopinées hors maternité. The Conversation. doi:10.64628/AAK.r4add9utm
- Engjom HM, Morken NH, Høydahl E, Norheim OF, Klungsøyr K. Increased risk of peripartum perinatal mortality in unplanned births outside an institution: a retrospective population-based study. Am J Obstet Gynecol. 2017;217(2):210.e1-210.e12. doi:10.1016/j.ajog.2017.03.033
- Huotari T, Rusanen J, Keistinen T, et al. Effect of centralization on geographic accessibility of maternity hospitals in Finland. BMC Health Serv Res. 2020;20(1):337. doi:10.1186/s12913-020-05222-5
- Bergonzoni A, Simon M. La part des femmes en âge de procréer résidant à plus de 45 minutes d’une maternité augmente entre 2000 et 2017 |. DREES Etudes Résultats. 2021;(1201). Accessed May 6, 2025. https://drees.solidarites-sante.gouv.fr/publications/etudes-et-resultats/la-part-des-femmes-en-age-de-procreer-residant-plus-de-45-minutes
- Örtqvist AK, Haas J, Ahlberg M, Norman M, Stephansson O. Association between travel time to delivery unit and unplanned out-of-hospital birth, infant morbidity and mortality: A population-based cohort study. Acta Obstet Gynecol Scand. 2021;100(8):1478-1489. doi:10.1111/aogs.14156
- Grzybowski S, Stoll K, Kornelsen J. Distance matters: a population based study examining access to maternity services for rural women. BMC Health Serv Res. 2011;11:147. doi:10.1186/1472-6963-11-147
- Pilkington H, Blondel B, Carayol M, Breart G, Zeitlin J. Impact of maternity unit closures on access to obstetrical care: The French experience between 1998 and 2003. Soc Sci Med. 2008;67(10):1521-1529. doi:10.1016/j.socscimed.2008.07.021
- Gunnarsson B, Smárason AK, Skogvoll E, Fasting S. Characteristics and outcome of unplanned out-of-institution births in Norway from 1999 to 2013: a cross-sectional study. Acta Obstet Gynecol Scand. 2014;93(10):1003-1010. doi:10.1111/aogs.12450
- Viisainen K, Gissler M, Hartikainen AL, Hemminki E. Accidental out-of-hospital births in Finland: incidence and geographical distribution 1963-1995. Acta Obstet Gynecol Scand. 1999;78(5):372-378.
- Ovaskainen K, Ojala R, Tihtonen K, Gissler M, Luukkaala T, Tammela O. Unplaned out-of-hospital deliveries in Finland: A national register study on incidence, characteristics maternal and infant outcomes. Acta Obstet Gynecol Scand. 2020;99(12):1691-1699. doi:10.1111/aogs.13947
- Hemminki E, Heino A, Gissler M. Should births be centralised in higher level hospitals? Experiences from regionalised health care in Finland. BJOG Int J Obstet Gynaecol. 2011;118(10):1186-1195. doi:10.1111/j.1471-0528.2011.02977.x
- Engjom H, Morken NH, Norheim O, Klungsøyr K. Availability and access in modern obstetric care: a retrospective population-based study. BJOG Int J Obstet Gynaecol. 2014;121(3):290-299. doi:10.1111/1471-0528.12510
- Combier E, Roussot A, Chabernaud JL, Cottenet J, Rozenberg P, Quantin C. Out-of-maternity deliveries in France: A nationwide population-based study. PloS One. 2020;15(2):e0228785. doi:10.1371/journal.pone.0228785
- Aoshima K, Kawaguchi H, Kawahara K. Neonatal mortality rate reduction by improving geographic accessibility to perinatal care centers in Japan. J Med Dent Sci. 2011;58(2):29-40.
- Ravelli A, Jager K, De Groot M, et al. Travel time from home to hospital and adverse perinatal outcomes in women at term in the Netherlands: Travel time and adverse perinatal outcomes in women at term. BJOG Int J Obstet Gynaecol. 2011;118(4):457-465. doi:10.1111/j.1471-0528.2010.02816.x
- Levaillant M, Garabédian C, Legendre G, et al. In France, the organization of perinatal care has a direct influence on the outcome of the mother and the newborn: Contribution from a French nationwide study. Int J Gynecol Obstet. 2024;164(1):210-218. doi:10.1002/ijgo.15004
- Beeram M, Solarin K, Young M, Abedin M. Morbidity and Mortality of Infants Born Before Arrival at the Hospital. Clin Pediatr (Phila). 1995;34(6):313-316. doi:10.1177/000992289503400604
- Bonal M, Padilla C, Chevillard G, Lucas-Gabrielli V. A French classification to describe medical deserts: a multi-professional approach based on the first contact with the healthcare system. Int J Health Geogr. 2024;23(1):5. doi:10.1186/s12942-024-00366-7
- Lapinte A, Legendre B. Renoncement aux soins : la faible densité médicale est un facteur aggravant pour les personnes pauvres. DREES Etudes et résultats N°1200. July 28, 2021. Accessed May 19, 2025. https://www.drees.solidarites-sante.gouv.fr/publications/etudes-et-resultats/renon

