Non-Invasive Acoustic Therapies for STEMI Reperfusion
A Review of Non-Invasive Acoustic Reperfusion Technologies for STEMI
Andrew K. Hoffmann, RDCS(a)1
- Ahof Biophysical Systems Inc. (Vancouver, B.C., Canada)
OPEN ACCESS
PUBLISHED: 31 December 2025
CITATION: Hoffmann, A.K., 2025. A Review of Non-Invasive Acoustic Reperfusion Technologies for STEMI. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v14i1.7190
COPYRIGHT: This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v14i1.7190
ISSN 2375-1924
ABSTRACT
St-Elevation Myocardial Infarction (STEMI), commonly an acute, occlusive blood clot in a major epicardial coronary artery, is the most serious of heart attacks, carrying a significant mortality and morbidity, and opening the occluded artery quickly with good distal reflow is the goal for best clinical outcomes. Slow arrival of Emergency Health Services (EHS), a lack of Cathlab availability in underdeveloped and rural areas, poor performance and bleeding risks with thrombolytic drug therapy, and a high rate of poor microvascular re-flow regardless of therapy, have prompted searches for alternative or adjunctive treatments. Mechanical forces or pressure waves imparted to the human body (herein described as Acoustic Therapies) have been well studied both in vitro and in catheter-based systems in their abilities to increase blood flow and disrupt and clear blood clots. However, the search for a non-invasive acoustic therapy suitable for first-line response in the treatment of acute thrombotic arterial occlusions, including STEMI, remains elusive. Indeed, due to the vast differences of acoustic penetration windows and variabilities of at-risk adjacent tissues, there has been a historic dilemma of threading the needle in finding a workable waveform and mode of delivery that is intense enough to provide adequate penetration with a clot-disruptive effect, while also avoiding harm to the patient. STEMI victims who reach professional care in view of Primary Percutaneous Coronary Intervention (PPCI) or pre-hospital IV thrombolysis, reportedly have a ~ 4-10% resultant in-hospital mortality, with the number approaching 50% if complicated by cardiogenic shock, hence there remains room for improvement. Indeed, acoustic reperfusion for STEMI could foreseeably add particular value as a stand-alone therapy or adjunctive to IV thrombolysis in rural or relatively poor areas where a Cardiac Cathlab is not readily available, as an early warning and treatment system for STEMI patients awaiting EHS, and, importantly, for treatment of the infamous “No-Reflow phenomenon” following PPCI, where many solutions have been tried and failed. This paper provides a history and commentary on the various forwarded non-invasive acoustic STEMI reperfusion strategies to date, including High Frequency diagnostic UltraSound (HFUS) with IV Microbubbles (MBs) – a technique commonly referred to as “Sonothrombolysis”, transcutaneous Low Frequency Ultrasound (LFUS), and little-known diastolic timed Low Frequency Vibration (dtLFV), which involves palpable, infrasonic to sonic frequency percussions to the chest wall as a stand-alone measure, or as an adjunct to IV thrombolysis. A discussion is provided on each of these therapies’ challenges, how they could fit in the modern paradigm of care, and where they are along their respective research and development pathways.
Keywords:
STEMI, Reperfusion, Sonothrombolysis, Ultrasound, Vibracoustic Therapy, Diastolic Timed Vibration
THE EUROPEAN SOCIETY OF MEDICINE
Medical Research Archives, Volume 13 Issue 12
REVIEW ARTICLE
Introduction
Acute Coronary Syndromes (ACS), typically involving thrombo-occlusive cardiovascular disease, remain the leading cause of death in the industrial world, and among these, ST-Elevation Myocardial Infarction (STEMI) is the most serious and carries a significant morbidity and mortality. With this, it is well established that rapid and complete restoration of blood flow to the occluded artery and its distal microvasculature are the main determinants of positive clinical outcome.
STEMI victims are preferentially sent to a Cardiac Cath lab, where emergency Primary Percutaneous Coronary Intervention (PPCI) is performed to open and stent the acutely thrombosed vessel. However, significant infrastructure is required for PPCI, hence, there are sometimes lengthy delays in delivering a patient to the Cath Lab, which leads to worse outcomes. Intravenous (IV) thrombolytic therapy, therefore, remains a common alternative therapy worldwide, particularly in rural areas, such as when a Cath lab is not available within 120 (and ideally 90) minutes. However, IV thrombolysis has a relatively low complete reperfusion rate and has worrisome potential hemorrhagic complications, including stroke. Importantly, both IV thrombolysis and PPCI, in between 30 to 60% of cases, suffer from incomplete microvascular reflow (in the Slow No-Reflow phenomenon), with small thrombotic fragments clogging the distal coronary vasculature, which leads to increased infarct size and co-morbidities.
In view of these challenges, there have been a variety of research initiatives involving the emergency application of transcutaneously applied mechanical force or pressure waves (herein described as Acoustic Therapies), to disrupt and clear acute coronary thrombosis to accelerate and ensure quality reperfusion in STEMI. These applications have been forwarded as stand-alone measures before PPCI, or adjunctive to IV thrombolysis, or used to treat the Slow to No Reflow phenomenon following PPCI. However, as will be shown as a repeating theme throughout this report, there is a common dilemma in the search for such non-invasive acoustic reperfusion systems, from lessons learned in the treatment of Acute Ischemic Stroke (AIS), and which we are slowly re-learning in STEMI as well.
The problem is that due to the extreme variabilities of quality of acoustic penetration windows, and variabilities to the at risk adjacent tissues and the arteries themselves, it is a particularly difficult challenge to thread the needle in finding a mode of delivery and standardized intensity (or dosing) of therapy, regardless of the frequency or waveform utilized, that is both reliably penetrative to reach the target thrombosed vasculature to provide a clot-disruptive effect, while also not harming the patients themselves.
Also, for the majority of people living in the developed world, the present-day Gold Standards of emergency PPCI and prehospital IV thrombolysis (Golden Hour) for survivors who make it to EHS, already fare modestly well with in hospital mortality rates of only ~ 4- 10%, with the number only approaching ~50% if the STEMI is associated with cardiogenic shock.
So, acoustic therapies used as an adjunct to these may have a difficult time, in requiring a high number of patients in any clinical trial, to prove statistically significant clinical benefits. However, non-invasive acoustic reperfusion therapies may, more so, find a clinically relevant niche in very poor or ultra rural areas where neither PPCI or IV thrombolysis is available, to promote survival and buy time for EHS transport which may take several hours after initiation of a 911 call. Also, beyond this, importantly, what is sorely needed, including in the urban scenario, is a treatment for the Poor to No-Reflow phenomenon following STEMI PPCI with many solutions tried and failed, and to this, acoustic therapies may prove to be vital, and could make a large, easily provable benefit.
Adding to this, even in the urban center, we tend to forget that up to about ~ 50% of heart attack victims don’t survive to make it to EHS, and many, reportedly, die because of unawareness of having a heart attack. So, society desperately needs a very early STEMI surveillance/detection method, and possibly with that, an ultra-early non-invasive acoustic therapy which could even be administered by the patients themselves to promote early action, early reflow, and hopefully improve initial survival rates to STEMI victims as a bridge to reaching EHS.
With all these factors in mind, the research (and proposed research) for a safe yet effective acoustic therapy system to enhance STEMI reperfusion continues and has been conducted (or suggested) at various frequencies, power amplitudes, and by varying delivery methods.
An Overview
It is well known since the invention of the Band-Aid that mechanical forces and friction should be avoided upon a new clot to prevent it from disaggregating and disadhering from its tissue interface. Indeed, that mechanical force can disrupt thrombosis in STEMI is easily exemplified in present-day PPCI, where it has become common practice to mechanically poke at an acute coronary clot to traverse it with a guide wire, which often, even before angioplasty, leads to a statistically significant improvement in the ST segment.
Low Frequency Vibration (LFV), consisting of mechanical oscillations in the infrasonic (1-16 Hz) to low sonic audible frequency range (< 250 Hz), was described as a clot-disruptive modality in 1978, with Wobser et al. disclosing a mechanical wave system oscillating as low as 50 Hz to disrupt big blood coagula during epigastric examinations. Years later, Will Dubrul in 1993 invented a Vibrating Catheter, which paved the way to today’s Trellis-8 peripheral infusion catheter, which produces between a 10 – 50 Hz oscillating motion to potentiate localized thrombolytic drug delivery and action in the dissolution of Deep Vein Thrombosis (DVT).
Low Frequency Ultrasound (LFUS, ~19 kHz – 500 kHz), consisting of tiny oscillations above the human hearing range, was studied in 1976 by G. Trubestein et al., who delivered LFUS by a rigid wire as a means to disrupt clots. With this, later studies showed that catheter-delivered LFUS has a synergistic effect with thrombolytic drugs, and Needelman et al., in 2005, showed in vitro that 20 kHz LFUS induces clot dissolution independently, even without a thrombolytic drug. In a breakthrough discovery, Tachibana et al., in 1995, demonstrated that LFUS (170 kHz) when accompanied by perfluorocarbon MicroBubbles (MBs), which oscillate and burst in response to ultrasound, even further potentiated thrombolysis.
Completing the frequency range, High Frequency Ultrasound (HFUS~ 0.5 MHz – 10 MHz) has been studied since the 1940s as a selective, high-intensity, focused means to destroy tissue targets such as cancerous tumors without causing damage to healthy, adjacent tissues. Regarding clot disruption, Rosenschein et al. in 2000 demonstrated that high intensity, focused HFUS at 0.5 MHz could disrupt thrombi in vitro without causing adjacent arterial damage.
HFUS is unique in that the waveforms can transmit as narrow, substantially non-divergent, side-by-side collimated beams, which can be reflected back to produce diagnostic images, as well as Doppler flow measurements. Power outputs allowed in diagnostic HFUS imaging are too low to cause tissue damage or clot lysis, however, in a major shift in care, Alexandrov et al. famously demonstrated in 2000 that continuous wave Doppler (2MHz) applied to the Middle Cerebral Artery (MCA) with IV tissue Plasminogen Activator (t-PA) in combination with contrast agent MBs enhanced reperfusion in AIS. Along the same lines, Xie and Porter et al. in 2009 demonstrated that transcutaneous 2D HFUS (1.5 MHz) imaging with IV-MBs safely disrupted blood clots in a canine model of femoral arterio-venous graft thrombotic occlusion.
Indeed, these repurposing of diagnostic ultrasonic scanning techniques with MBs for therapy in the treatment of acute thrombotic blockages have prompted a new medical term, theranostics, and, as will be discussed in detail later, have been dominating the research headlines of today.
Importantly, virtually the entire frequency range of acoustic therapies, from percussive infrasonic massage to ultrasound, is known to increase blood flow, purportedly through mechanical stimulation of the endothelium and red blood cells, which leads to an endogenous release of nitric oxide, a powerful vasodilator.
Hence, it would seem that there must be a good, preferably quick to administer, non-invasive acoustic therapy out there that can accelerate and ensure quality STEMI reperfusion. However, there is a big difference in the ability to apply acoustic energy invasively, or locally upon a clot, versus applying the same type of therapy, at a much higher amplitude transcutaneously outside the body. Indeed, with ultrasound (e.g. 19 Kz – 10 MHz), the worries come from overlying tissue heating along with arterial hemorrhage or spasm, and with lower frequency, palpable vibration massage (e.g. 1-250 Hz), while little is actually known, there comes worries of bruising to the chest wall or cracked ribs, along with knocking things loose to cause embolic concerns (such as calcified lesions within the heart), or perhaps, damage to the coronary endothelium itself, particularly if applied upon highly calcified lesions.
Nevertheless, the desire to save cardiac muscle and reduce mortality and morbidity has prompted the search for a safe and effective non-invasive acoustic therapy to improve reflow in STEMI, which began in the 1990s when IV thrombolysis was still struggling, as the Gold Standard of care.
Low Frequency Ultrasound (LFUS)
It was research Cardiologist Dr. Robert J Siegel from Cedars-Sinai, Beverly Hills California, who in the 1990s, famously led the initiative in exploring transcutaneously delivered LFUS as an adjunct to IV thrombolytic drug therapy for STEMI reperfusion, and in a first step his group confirmed in vitro that LFUS in the lower frequency range of 25 kHz – 50 KHz appeared the most effective at disrupting clots.
LFUS seemed a first reasonable choice, not only due to its success in catheter-assisted thrombolysis, but also due to its long, safe history in dentistry and cosmetics for plaque and cellulite removal, physiotherapeutic diathermy, and disruption of kidney stones by way of transcorporeal lithotripsy. Indeed, when contrasting ultrasonic frequencies, it has been found, generally, that both tissue penetration and the amount of cavitation and microstreaming produced within the body (chief mechanisms to assist thrombolysis) generally vary inversely with ultrasound frequency.
Siegel’s team quickly climbed the translational research pathway, first showing in 1996 that LFUS (26 kHz) applied to the groin with IV StreptoKinase (SK) accelerated thrombolysis in a rabbit model of iliofemoral artery thrombotic occlusion, and then in 2000 demonstrating that transthoracic, chest wall delivered LFUS (27 kHz) with IV t-PA improved recanalization rates, TIMI flow, and faster resolution of the ST segment, in a canine model of mid-LAD infarction. Moreover, histological examination of the animals following these trials was reportedly unremarkable, showing no adverse effects to the overlying chest wall, heart, lung or vessels treated. A LFUS prototype delivery system was created by TIMI3 Systems Inc., with LFUS beams (27kHz) expected to penetrate across the anterior chest wall and diverge to cover a substantial portion of the heart, hence would not require any special skill or targeting, making for an easy-to-use system applicable by a nurse or paramedic with minimal extra training.
The TIMI3 LFUS device sailed through a phase 1, clinical safety study of 25 patients performed in Brazil, with “No unanticipated major adverse events observed”, but then, unfortunately, in the Perfusion by ThromboLytic and Ultrasound (PLUS) study (a double-blind, randomized, controlled international clinical trial comparing 356 patients with STEMI receiving thrombolysis with LFUS or thrombolysis alone), the trial had to be discontinued for failure to show efficacy. With this, the simple act of increasing the intensity of the LFUS waveform was deemed unlikely an option, as potential burning of the skin was already a conceded worry of the investigators. Another consideration that would likely block the use of higher intensity transthoracic ultrasound would be a strong worry of adverse effects to the lung which is adjacent and partially overlies the heart, which, at least in animal studies has been shown to be extremely sensitive to Pulmonary Capillary Hemorrhage (PCH) secondary to even much weaker diagnostic intensity ultrasound.
However, in hindsight, a chief reason for the failure of the TIMI3 device may have been attributed to the fact that the LFUS beams were not visually targeted to ensure penetration to the heart, nor specifically to where a culprit epicardial clot would often reside via a verified acoustic penetration window. Indeed, a great percentage, particularly of the more proximal to mid aspects of the coronary anatomy, reside at the far medial and lateral basal aspect of the heart, which is, especially relative to an anteriorly placed ultrasonic emission site, crowded or acoustically shielded by overlying lung, which containing air, does not transmit ultrasound.
Perhaps the TIMI3 device would have fared better if applied to the apical position of the heart, where the LFUS beams, coming from alongside and underneath the lung, may have stood a better chance of hitting a greater percentage of the coronary tree. Indeed, modern-day theranostic HFUS sonothrombolysis trials such as MRUSMI and now REDUCE (discussed later) are being conducted from the apical views, likely for this wisdom. It might have also been interesting to have retested the TIMI3 device with the addition of IV-administered MBs, which would have possibly potentiated LFUS’s thrombolytic effect. However, Siegel’s canine model showed superior reperfusion with IV-tPA alone (without MBs), and the use of MBs to treat vascular blockages was still in its infancy, so perhaps for these reasons, they were not included in the protocol.
Shortly after, in 2004, Siegel’s team performed an open chest animal study showing that LFUS (27 kHz) significantly improved perfusion to an infarcting myocardial segment following acute occlusion of a diagonal branch, which suggested that LFUS may have at least vasodilated collateral vessels in view of a cardiac protective effect. However, this was perhaps too little too late, and it would seem that the world had moved on to other projects to explore.
Theranostic HFUS imaging with IV administered MBs, Sonothrombolysis
Years later, with the Cathlab now a major player in emergency PPCI, ultrasound made a stunning comeback whereby it was shown that diagnostic HFUS imaging techniques, with a co-administration of IV administered contrast MBs, a therapy now dubbed as HFUS theranostic sonothrombolysis, could be effective in accelerating and ensuring reflow in STEMI, and this even, without the need for an IV thrombolytic drug.
In HFUS sonothrombolysis, MB cavitation is induced by intermittently applied high-power Mechanical Index (MI) impulses that create shear forces that can dissolve coronary epicardial and microvascular thrombi and also stimulate endothelial and red blood cell nitric oxide release, which aims to augment microvascular flow. Importantly, since echo contrast techniques with MBs had already been deemed as safe, and having achieved regulatory approval in North America and Europe for purely diagnostic purposes, researching the technology as a method to treat STEMI would tend to fly through otherwise difficult regulatory hurdles.
Indeed, fast forward to the 2000s to early 2020s, several pre-clinical and small to medium-scale clinical trials in HFUS sonothrombolysis have shown preliminary success in enhancement of reperfusion in STEMI, many without adverse effects.
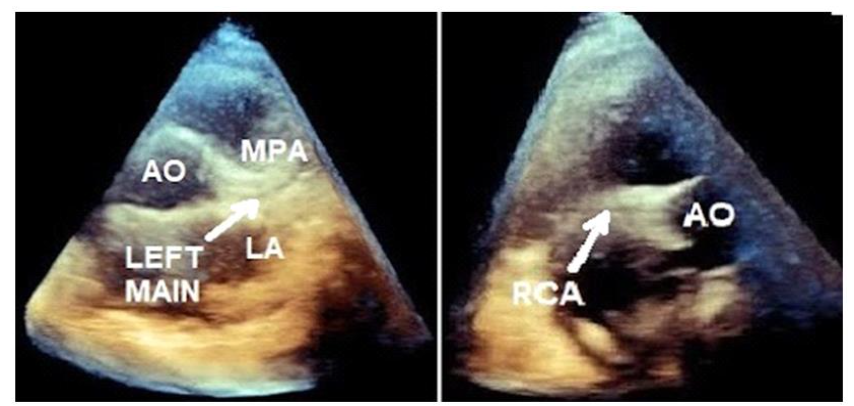
The first human clinical trial was the in-ambulance, single arm, pre-PPCI, STEMI Sonolysis trial (n =10), which used parasternally delivered Three Dimensional (3D) HFUS targeted to the aortic root and proximal coronary vasculature, which demonstrated the feasibility and safety of the technique and superior initial angiographic recanalization rates.
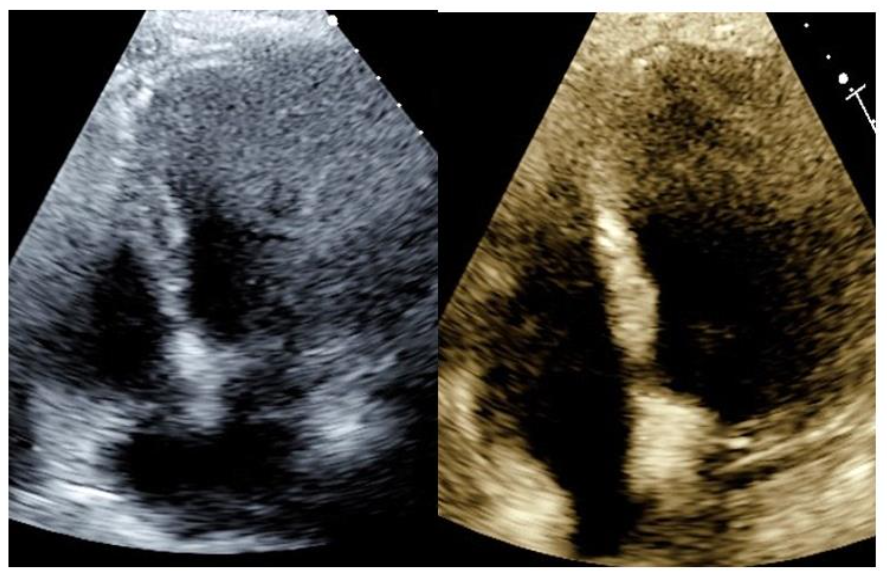
Following this, the Microvascular Recovery with UltraSound in Acute Myocardial Infarction (MRUSMI) trial (n = 100) was performed, with 50 patients receiving pre- and post-STEMI HFUS sonothrombolysis vs. 50 patients receiving PPCI only. This trial, which utilized Two-Dimensional (2D) HFUS applied to the apical views with intermittent high MI flashes (>= 5 microseconds pulse duration), showed an improved rate of initial angiographic patency, as well as enhanced post-procedure microvascular flow and improved outcome Left Ventricular (LV) function.
![Image (left) shows the placement of an echo transducer to the apical window. Image (right) shows echocardiographic images pre- and post-HFUS sonothrombolysis in a pre-PPCI STEMI victim showing successful reperfusion. Image taken from JACC, Vol 67, Issue 21, 31 May 2016, Page 2506-2515 [Open Access].](/pdf-to-wp-converter/uploads/images/non-invasive-acoustic-therapies-stemi-reperfusion-figure-3.png)
Moreover, the STEMI pre-PPCI SONOSTEMI trial, a single-arm Canadian trial (n=15) which utilized the MRUSMI protocol, confirmed the feasibility of the technique in a very fast PPCI response center, with the sonothrombolysis procedure taking only 12.5 +/- 9.3 minutes to perform. This trial also confirmed an impressive initial angiographic patency with 2 / 3 TIMI flow in 7 / 15 cases, with successes most commonly regarding the LAD, as well as a 100% rate of post-procedure TIMI 3 flow, and with 12 of 15 patients having an LV ejection fraction of >= 50% at 90 days follow-up.
However, in the ROMIUS clinical trial, with HFUS 2D ultrasound applied from the parasternal window using a higher pulse duration during high MI treatment flashes (20 microseconds vs the standard 5 microseconds), while impressively showing an initial angiographic patency of 50%, showed a higher incidence of severe vasoconstriction in the infarct vessel unresponsive to intracoronary nitroglycerin, which forced the trial to be discontinued.
Making matters worse, El Kadi et al., in a post-PPCI sonothrombolysis treatment of No-Reflow study recently published in 2024 (n = 67), with HFUS imaging also conducted from the apical views via a similar protocol as used in MRUSMI, showed no efficacy in improving the No-Reflow condition. This raised a particular alarm, because the focus of the therapeutic beams to a known and easily targeted ischemic ROI should be HFUS sonothrombolysis’s bread and butter, hence casting doubt on the entire technique.
Indeed, there may be good reasons to be suspicious that HFUS sonothrombolysis with IV-delivered MBs may not work consistently, or in larger-scale trials. Firstly, it is an inherent weakness of HFUS sonothrombolysis that it requires a skilled sonographer to ensure adequate therapeutic levels of ultrasound reach the epimyocardium of the heart, and with this, many heart attack victims are overweight and/or have a history of smoking (with hyperinflated lungs), whereby it can be very challenging to ensure penetrative echocardiographic views.
Moreover, it must be emphasized that not all sonographers are created equal. In this Author’s experience (including as an Echo Supervisor for five years at the Royal Columbian Hospital), only a better third of the most experienced members of an echo staff would have the required strength, skill, and tenacity to be trusted to ensure decent target HFUS delivery to the hearts of potentially technically difficult patients in challenging, rushed, technical environments. Indeed, in the failure of the Kadi et.al. trial for treatment of post-PPCI No-Reflow, how many sonographers were utilized, what was their level of experience, and how good were their images?
Furthermore, a dirty secret in contrast echocardiography, is that after finding a decent view via standard high MI HFUS imaging, the sonographer must continue to maintain imaging for the most part at low power MI settings (to preserve the MBs), which generally makes an already challenging image even more difficult to hold and maintain. Indeed, injuries in the field of sonography are common, and the emotional pressure to get penetrative theranostic images under technically difficult conditions could create a perfect storm for sonographer injury.
Secondly, targeting the apical window in echocardiography leaves imaging (and hence therapeutic exposures) to the more proximal, basally situated coronary arteries (which produce the biggest infarcts) to the far-field, hence subject to attenuation and acoustic shadowing, particularly from the MBs themselves, which accumulate in the LV cavity.
Thirdly, an acutely thrombosed coronary vessel has, by definition, no or very slow flow; hence, the MBs, which are typically administered systemically, would tend to go everywhere except to where they are most needed, with the MBs only trickling down the blocked artery by passive diffusion. Hence, the effectiveness of HFUS sonothrombolysis could be severely compromised due to a paucity of MBs expected to actually reach the compromised vasculature, particularly earlier on in therapy during the TIMI zero flow phase.
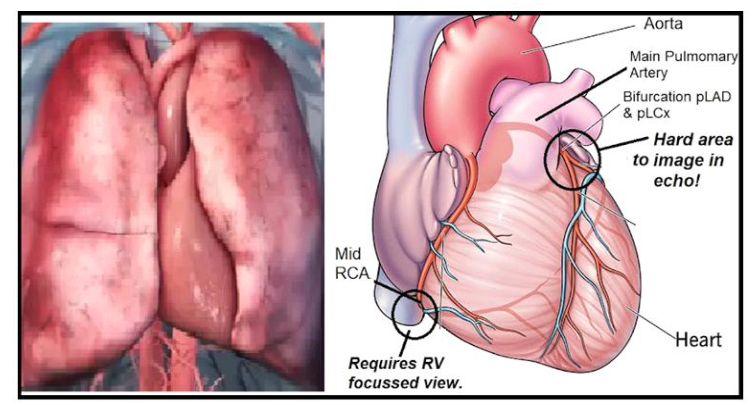
But finally, perhaps the main weakness of HFUS sonothrombolysis is that, at least at present, and assuming an IV administration of MBs, the exact location of the originating acute blood clot responsible for the initial occlusion cannot be directly seen by standard echocardiography. With this, it is only the distal at-risk ischemic myocardial ROI, which was explicitly targeted in MRUSMI and now REDUCE, and the instigating epicardial clot, which would likely be located within an imaging plane remote from its distal myocardial filling segment, is hit if at all by luck as the probe is rotated between the apical views. Moreover, for inferior STEMI, the protocol would likely benefit from the addition of an RV focused view, to ensure HFUS penetration to the proximal to mid-RCA, which is presently not the case.
However, on a bright note, there is now a note of optimism in the search for the treatment of the infamous post-PPCI Slow – to- No- Reflow phenomenon!
HFUS Sonothrombolysis with Intra-Coronary Administered MBs
Understanding the challenges of IV-delivered MBs for HFUS sonothrombolysis, Brush et al., just this year (2025) in a pioneering case study, successfully attempted the technique with an intracoronary injection of MBs down an acutely thrombosed LCx, which reportedly solved an otherwise refractory No-Reflow issue! Importantly, as this procedure addresses the chief theoretical drawbacks of HFUS sonothrombolysis with systemically delivered MBs (i.e., the paucity of MBs expected to fill the culprit thrombosed vasculature, and the inability for the sonographer to directly visualize the artery, including at its blockage point along with its distal tributaries), its success in promoting reflow would seem unlikely as a fluke!
As mentioned earlier, a solution to the No or Slow Reflow phenomenon has plagued emergency PPCI with many ideas tried and failed, so this new solution with MBs delivered intra-coronary could be potentially a very big deal (a game changer), and further testing in larger-scale trials to reproduce success in this technique certainly appears warranted.
Of course, it’s hard to get too excited with an apparent success of a single case study, and perhaps the biggest worry with this approach may be, again, from lessons learned that as an acoustic reperfusion therapy begins to work, an adverse effect, such as hemorrhage or spasm to the coronary artery may not be unexpected to come as a clinical price.
What about 3D HFUS Sonothrombolysis?
It would seem reasonable that if the goal in theranostic HFUS sonothrombolysis with IV MBs is to target an echocardiographically hidden epicardial clot to accelerate vessel patency (and prevent re-occlusion), that it might make more sense to consider the use of 3D HFUS, which would provide a greater overall catchment volume in improving the odds of providing therapeutic ultrasonic exposures to key areas.
Of course, particularly from the parasternal window it would often be impossible for a sonographer to capture the entire base of the heart in one 3D view, so, in a more practical solution the ultrasound probe could be selectively directed depending on the location of the ST elevation and prior identified ischemic ROI, with anterior and/or lateral infarcts tilting the probe anatomically leftward from the aortic root coronary origin, and for inferior infarcts especially with RV involvement (elevation in V4R), tilting the probe anatomically rightward.
To ensure a STEMI blockage involves a relatively proximal segment of a culprit artery, the method published by Slikkerveer et. al. in the Sonolysis trial could be adopted, which cleverly utilized a 12-lead ECG inclusion criteria, whereby patients must begin with a sum of ST elevation of 6 mm or more in combined leads, or 1 mm or more ST elevation in lead V4R in case of inferior infarction. It should be mentioned that 3D HFUS sonothrombolysis has also been studied from the apical window, with Qui et. al, in 2022 demonstrating in a porcine STEMI model of acute mid-LAD infarction, success in both enhanced recanalization of the epicardial infarct artery and distal reflow.
Hence, 3D HFUS sonothrombolysis, with, where appropriate, selective angulation of the probe to target an otherwise acoustically hidden epicardial clot, should not be ruled out. Indeed, foreshadowing the future, if intra-coronary MBs become the standard treatment for post-PPCI No-reflow, the future of urban HFUS sonothrombolysis with IV administered MBs may only rest in the pre-PPCI treatment to accelerate initial vessel patency, so use of the 3D Sonolysis trial protocol, which is particularly designed for that purpose, may make the most sense moving forward.
But has our obsession with ultrasound been blinding us to perhaps a much easier, more practical acoustic solution?
Diastolic Timed Low Frequency Vibration (dtLFV)
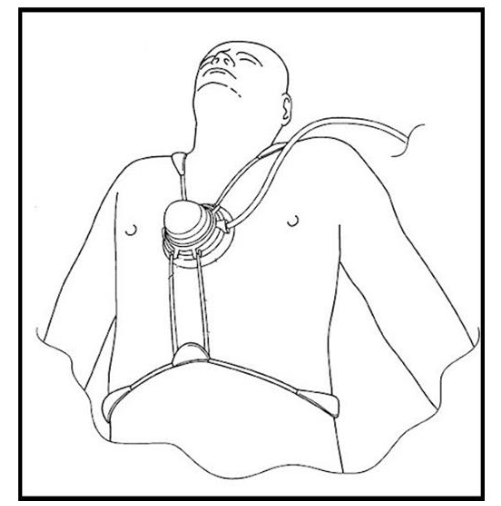
Recognizing the need for a lower tech, more practical STEMI reperfusion acoustic therapy which would not need a skilled sonographer to administer the therapy, the Author and his associates at the Engineering Department, of Simon Fraser University, SFU, Burnaby, BC, Canada have been researching the method of administering, diastolic timed Low-Frequency Vibration (dtLFV) in the infrasonic to sonic range, by use of an oscillation massager (e.g., ~ 50 impacts / second, ~ 4 mm stroke amplitude) applied upon a STEMI victim’s chest wall.
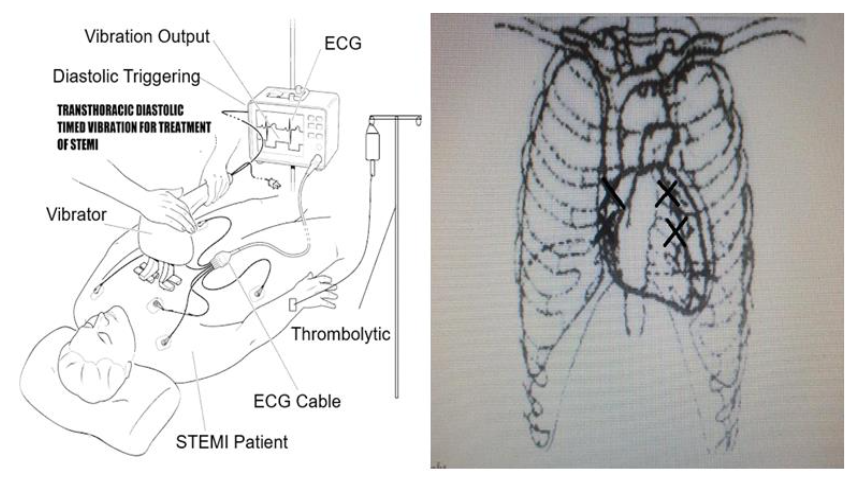
One normally associates LFV (20 – 250 Hz) with cheap massagers purchasable at the drug store, but more recently, under a new term of “Vibro-Acoustic Therapy”, its use has broadened as therapy for an entire host of disorders, including pain relief, improved balance, neuromodulated diseases, and accelerated muscle and bone recovery. Regarding the heart, an LFV platform was safely demonstrated for use in cardiac rehabilitation conditioning exercise in men following Myocardial Infarction (MI), and localized LFV (35 Hz, 6 mm stroke) applied to the upper back has been proposed, and showed feasibility (and preliminary safety with efficacy) in a case study as a possible outpatient means to stimulate coronary angiogenesis in treatment of chronic Coronary Artery Disease (CAD).
For most people, LFV massage is deemed safe, however, there are listed warnings in its habitual use based mostly on perceived risks. The literature available cites caution in use with: pregnancy, presence of a pacemaker or other electronic medical implants, history acute deep vein thrombosis (DVT) or atrial fibrillation not yet anticoagulated (as LFV may dislodge the clot), active cancer or tumors, recent surgery or unhealed wounds (e.g., surgical sites, skin rashes, open wounds), severe osteoporosis with high fracture risk, diabetes with complications (especially neuropathy, where sensation may be impaired), kidney or gallstones (which vibration may dislodge), and recent bone fractures or stress fractures.
With regards to proposed coronary reperfusion, LFV has been shown in-vitro and in catheter based systems to disrupt, fragment, and erode clots with or without an adjunctive thrombolytic drug agent, has vasodilation properties, and, while highly attenuating, has confirmed deep penetration capabilities across the chest wall to reach the heart. Notably, transthoracic, chest wall delivered dtLFV, with oscillations capturing the LV isovolumetric relaxation period and terminating before the isovolumetric contraction period, was extensively studied in the 1990s by a Japanese group led by research Cardiologist Dr. Y Koiwa for the treatment of ischemic heart failure, and it is from Koiwa’s work that much of dtLFV science for STEMI has arisen.
Koiwa showed not only that dtLFV applied at 50 Hz with a relatively gentle 2 mm stroke amplitude delivered by a vibrator with a single contact node applied to the left sternal margin of the third intercostal space, reliably penetrates from the chest wall to the heart in human volunteers, but the therapy also improved LV diastolic relaxation, enhanced coronary flow, and enhanced cardiac output/stroke volume purportedly by the Frank Starling mechanism.
![Figure 8: The application of dtLFV (50 Hz, 2 mm amplitude) penetrates from a human left precordial rib-space to the heart, measured from an intra-esophageal accelerometer and LV pressure transducer. Image taken with permission from Koiwa et. Al. [86], Modification of Human Left Ventricular Relaxation by Small Amplitude, Phase Controlled Vibration to the Chest Wall, Circulation 1997 (95): 156 - 162.](/pdf-to-wp-converter/uploads/images/non-invasive-acoustic-therapies-stemi-reperfusion-figure-8.png)
![Figure 9: Superimposed tracings of LV and LA Pressure (LVP, LAP) without vibration (dashed lines) and at the first application of dtLFV (solid lines) in a canine model. Diastolic Timed Low Frequency Vibration accelerated LV relaxation (point a), and caused a decrease in minimum LV diastolic pressure (point c). A marked increase in systolic pressure (signifying a sudden enhancement in stroke volume, following the first application of dtLFV) is easily seen (point d). Image taken with permission from Koiwa et. al. [87], (1992) Circulation Vol 86 No. 6 pp 1955 - 1964.](/pdf-to-wp-converter/uploads/images/non-invasive-acoustic-therapies-stemi-reperfusion-figure-9.png)
![Figure 10: Image (top) Shows the responses of LV Pressure (LVP), Coronary Perfusion Pressure (CPP), and Coronary Blood Flow (CBF - thick solid line) following application of vibration to the left ventricle with regional ischemia. Diastolic CBF increases due to vibration input, and this increase lasted for several beats after cessation of vibration. Image (bottom) shows a close-up superimposition of the effects of vibration (solid lines), versus no vibration (dashed lines), including enhanced CBF, increased LVP fall (-dp/dt), and a fairly constant CPP regardless of vibration. Images taken with permission from Koiwa et al. [91], Effects of Diastolic Vibration on Coronary Circulation, Japanese Journal of Medicine, Vol 32, (1994), Issue 3, pg. 180 - 187.](/pdf-to-wp-converter/uploads/images/non-invasive-acoustic-therapies-stemi-reperfusion-figure-10.png)
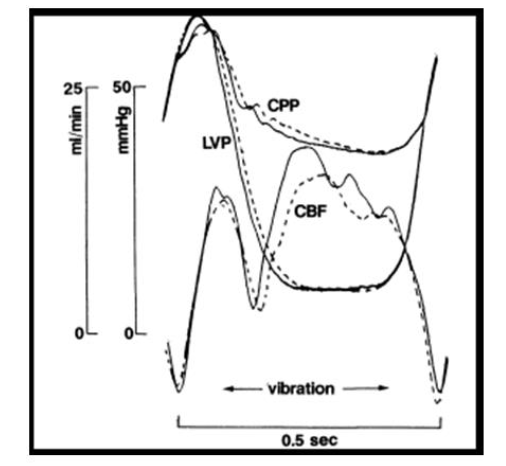 Figure 10: Image (top) Shows the responses of LV Pressure (LVP), Coronary Perfusion Pressure (CPP), and Coronary Blood Flow (CBF – thick solid line) following application of vibration to the left ventricle with regional ischemia. Diastolic CBF increases due to vibration input, and this increase lasted for several beats after cessation of vibration. Image (bottom) shows a close-up superimposition of the effects of vibration (solid lines), versus no vibration (dashed lines), including enhanced CBF, increased LVP fall (-dp/dt), and a fairly constant CPP regardless of vibration. Images taken with permission from Koiwa et al. [91], Effects of Diastolic Vibration on Coronary Circulation, Japanese Journal of Medicine, Vol 32, (1994), Issue 3, pg. 180 – 187.
Figure 10: Image (top) Shows the responses of LV Pressure (LVP), Coronary Perfusion Pressure (CPP), and Coronary Blood Flow (CBF – thick solid line) following application of vibration to the left ventricle with regional ischemia. Diastolic CBF increases due to vibration input, and this increase lasted for several beats after cessation of vibration. Image (bottom) shows a close-up superimposition of the effects of vibration (solid lines), versus no vibration (dashed lines), including enhanced CBF, increased LVP fall (-dp/dt), and a fairly constant CPP regardless of vibration. Images taken with permission from Koiwa et al. [91], Effects of Diastolic Vibration on Coronary Circulation, Japanese Journal of Medicine, Vol 32, (1994), Issue 3, pg. 180 – 187.
![Figure 11: Tracings of LV Pressure (LVP), ECG and dtLFV signal. Note characteristic instant decrease in LV diastolic pressures and increase in LVP systolic pressures during application of dtLFV, which indicates an increase in stroke volume, secondary to improved diastolic relaxation with increased LV filling, by the Frank-Starling mechanism. Image taken with permission from Koiwa et. a [92], (1992), Diastolic Vibration Improves Systolic Function in Cases of Incomplete Relaxation Circulation, Vol 86 No. 6 pp 1955 - 1964.](/pdf-to-wp-converter/uploads/images/non-invasive-acoustic-therapies-stemi-reperfusion-figure-11.png)
Importantly, LVF in the low sonic ranges (and expressly unlike ultrasound), has documented unique, internal transmission characteristics through body tissue, including along arteries and the epi-myocardium. This would theoretically allow a chest wall delivered dtLFV stimulus, even if only reaching the LV or a coronary vessel off mark of a culprit thrombosis site, to travel along the surface of the heart or along the coronary arteries themselves, to provide some level of therapeutic vibrations, or shaking to the entire coronary tree, even those aspects otherwise acoustically hidden under the lung. Hence, in theory, and unlike the LFUS TIMI3 device, this should allow for a skill-free placement of a dtLFV device over the anterior chest wall in the treatment of STEMI.
![Figure 12: Shows how an LFV input applied to the LV with induced regional ischemia readily transmits along the epi-myocardium, thereby enabling a therapeutic "shaking" response to a greater percentage of the coronary tree. Image taken from Elsevier Open Access. Smith D et. al. [97], Mechanical vibration transmission characteristics of the left ventricle implication with regard to auscultation and phonocardiography. J Am Coll Cardiol 1984, 4(3):517 521.](/pdf-to-wp-converter/uploads/images/non-invasive-acoustic-therapies-stemi-reperfusion-figure-12.png)
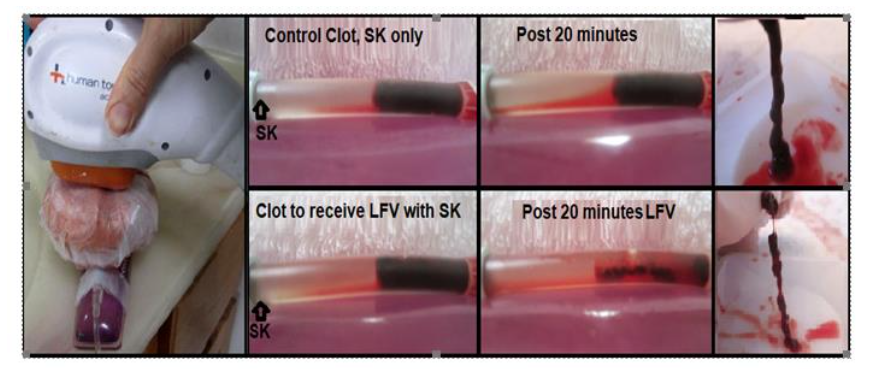
Adding to all this, LFV is known, both in catheter-based systems and by our own in vitro trial, to erode clots and potentiate the action of thrombolytic drug agents, presumably in part by increasing the mixing of systemically (or remotely) delivered thrombolytic agents down an otherwise zero-flow coronary artery (or flow system). Finally, as dtLFV has been shown to improve diastolic relaxation in the ischemic ventricle (including lowering diastolic pressures), this would increase the diastolic pressure gradient from epicardium to endocardium, and lower trans-myocardial vascular resistance, which should theoretically, in both cases, help microvascular flow in prevention or treatment of the No or Slow Reflow phenomenon.
So, while by many metrics dtLVF appears as a potentially ideal, first-line STEMI reperfusion system, best used as an adjunct to IV thrombolytics (or perhaps also pre-PPCI, drip and ship), there are strong reasons to suspect this therapy may not work or work safely. It must be stressed that while Dr. Koiwa and his team proved in human volunteers that dtLFV is penetrative to reach the heart, it is notable that an LFV waveform is expected to severely attenuate across the chest wall, with a resultant 95% loss of transmission of its applied force. Hence, dtLFV transthoracic therapy anticipates a very weak transmitted in-situ effect, which questions the aptitude of the therapy in achieving a helpful, thrombolytic effect.
In view of expected very weak penetration, it has been the position of the Author that for treatment of STEMI the intensity or “dosage” of dtLFV should be maximized, by varying how hard a paramedic presses down on the device upon the patient’s chest wall, to the limit of the patient’s tolerance (with the amount of force applied shown on a forcemeter readout upon the vibrator). This means that the goal would be to provide the most powerful thumping action upon the chest consented to by the patient, knowing that a very small amplitude would reach the target.
With this, there is a way to standardize or titrate the dose of a dtLFV to ensure adequate transthoracic transmission and a measured in-situ effect, by borrowing the technique of Koiwa et. al. whereby the patient could swallow a miniaturized transesophageal accelerometer disposed upon a wire, of measured length to rest behind the posterior LV of the heart. It would be the hope to record at least a 2 Gravity (G) measured acceleration (which, when measured with a 2 gram accelerometer, would be roughly equal to a force of ~0.04 Newtons, or ~ 4 grams of force per stroke), which would equate to gentle but intentional figure taps, actually felt by the heart.
The risks of dtLFV to an acutely ischemic heart are, of course, completely unknown; however, the chief worry, particularly in older patients, with chronic, severe, brittle calcified lesions (such as in severe Mitral Annular Calcification or Aortic Stenosis), would perhaps be the risk of dislodgement to cause embolic concerns, including stroke. Moreover, it is conceivable that a low-frequency percussive signal could damage the endothelium of the coronary arteries, particularly involving calcified plaques themselves. However, as the chest wall would be expected to take the significant brunt of therapy, the most common expected worry would likely be the potential of bruising on the chest or cracked ribs, particularly in older patients and especially if used as an adjunct to IV thrombolytic drug therapy.
All these concerns would need to be addressed should dtLFV ever reach clinical trials. With this, if research were to unfold, there would no doubt need to be progressive development of an inclusion/exclusion criteria, where STEMI patients would be first vetted before opting to receive dtLFV, such as having recent diagnostic testing showing no history of severe calcific valvular disease, transmural clot, or new AF not treated with anticoagulants (worry of LA clot).
Discussion
Mechanical impacts throughout the frequency spectrum all have proven abilities when employed in vitro and via catheter-based techniques to increase blood flow and break apart and clear blood clots. So, the question has become, can this effect be translated into a safe and effective non-invasive therapy to improve timely and quality reperfusion in STEMI, and under what niches could such improvements be best served?
A central common concern has been raised throughout this report, and is true for different reasons regardless of the frequency and amplitude of the proposed therapy, that it might be difficult, if not impossible to thread the needle in finding an intensity and mode of delivery of therapy which is sufficiently penetrative to cause reflow, while at the same time not harming the patient.
With regards to adjunctive acoustic therapy for STEMI, most recently, researchers have been placing their bets on HFUS imaging intensity sonothrombolysis with MBs, pre- and post-PPCI, which is considered a safe diagnostic procedure used for therapy. But the quality of acoustic penetration windows across the chest wall differs greatly and cannot always be anticipated from patient to patient, so it could very well turn out that the technique would fall into the awkward dilemma of not working in patients with poor acoustic windows, while causing adverse coronary effects with good windows. Moreover, the skill of the sonographer is another uncontrollable variable, in that an excellent sonographer, creating great penetration, may cause damage to the coronaries, and for a weaker sonographer, struggling to get an image, the technique may not work at all.
That said, certainly there is encouragement that HFUS sonothrombolysis might be safe and effective pre- and post-PPCI as per the MRUSMI trial, and to this we wait for the results of the slightly larger REDUCE trial. But the problem of poor reliability and safety of the technique has already started to rear its head, with an adverse effect on the coronaries (as identified as severe vasoconstriction during PPCI) in the ROMIUS trial, and with no efficacy in the post-PPCI No-reflow trial of El Kadi et al.
Putting things into perspective, acoustic therapy for STEMI can be divided into differing categories depending on at what stage the heart attack occurs, and the location where a patient hails from in terms of Cardiac Cath lab availability. In an urban center, the Gold Standard of course has become emergency PPCI, which offers, for the most part, good clinical outcomes for those who survive to meet EHS, so the possibilities for marginal improvements come from establishing earlier reflow and, of course, treating or preventing the infamous post-PPCI No-Reflow phenomenon.
Importantly, the calculus in the use of acoustic therapy for STEMI may have suddenly changed, with the landmark case study of Brush et. al., showing safety and effectiveness in treating No-Reflow by way of HFUS sonothrombolysis with an intracoronary injection of MBs. This technique, for the first time, enables exacting, targeted echocardiographic theranostics with a very high concentration of MBs deployed to the culprit vasculature, which deals perfectly with the major perceived theoretical weaknesses regarding the potential efficacy of HFUS sonothrombolysis with IV administered MBs. While one cannot get too excited with a single case study, nevertheless, given how the No Reflow Phenomenon has plagued PPCI, intra-coronary MB HFUS sonothrombolysis warrants an all-hands-on-deck research initiative, which should, in this Author’s opinion, start immediately in Cath labs throughout the nation.
However, again a note of caution. From lessons learned in both ultrasonic treatments of STEMI and AIS, the better the therapy works at establishing reflow, the higher the risks of adverse effects to the arteries and surrounding tissues. So, regarding this pioneering new treatment of No-Reflow, we will just have to wait and see.
Speaking optimistically, if HFUS sonothrombolysis with intracoronary MBs becomes the new standard of care for STEMI post-PPCI No-reflow, then the method of using IV administered MBs for STEMI, at least in the post-PPCI scenario, would become substantially mute. In this case, at least in the urban scenario the potential value of administering IV administered MBs would then rest only in its hopes of improving initial angiographic patency rates and early flow before arriving at the Cath lab, and whether this will influence ultimate STEMI clinical outcomes (with a good treatment for No-Reflow now, hopefully around the corner) still needs to be proven.
However, regarding care in rural areas and poorer countries where a Cardiac Cathlab may not be readily available within 90 minutes, perhaps HFUS sonothrombolysis with IV administered MBs, and continuing for a longer period of time to prevent re-stenosis and enhance microvascular reflow, as shown in the MRUSMI trial, may still be relevant. Indeed, in the absence of a Cardiac Cathlab, perhaps STEMI HFUS sonothrombolysis could also be tried as an adjunct to IV thrombolytic drug therapy, which hasn’t been tried in STEMI, but as referenced earlier has shown some preliminary effectiveness in the treatment of AIS.
The question of using 2D vs 3D imaging ultrasound, and by a more clever imaging protocol which attempts to target and focus therapeutic energy to where a culprit initiating epicardial clot is most likely to reside, are both factors which could greatly influence the potential effectiveness of IV-MB delivered HFUS sonothrombolysis, and these factors should be carefully considered if the REDUCE trial doesn’t fare well, especially with regards to initial angiographic patency.
There has also been some work with the use of 2b/3a platelet glycoprotein targeted MBs, which selectively bind activated platelets, and indeed in a porcine model of catheter-induced LAD infarction, Xie et al. showed superior initial recanalization and better distal microvascular reflow in using targeted MBs vs non-targeted. Ultimately, if HFUS sonothrombolysis is to ever become a standard of care, likely use of targeted MBs will certainly be considered, and if delivered intra-coronary, then they could advantageously be loaded with antiplatelet or thrombolytic drug agents, to further increase their thrombolytic effectiveness in the treatment of the No-reflow phenomenon, without increasing systemic bleeding risks.
But what about acoustic therapy for STEMI in rural areas or in poorer countries, where even HFUS sonothrombolysis may also be impractical or unaffordable? In these cases, one’s mind cannot help but think of dtLFV.
In terms of cost of procedure, chest wall dtLFV massage would not need MBs (which saves about $300 USD per procedure), plus if used in pre-hospital care, it would save the added expense of a fleet of echo machines and a ride-along sonographer’s salary. Indeed, perhaps, if nothing else is available, dtLFV could be used as a standalone measure, adjunctive only to aspirin (an oral antiplatelet). However, for greatest effectiveness, dtLFV seems tailor-made for use as an adjunct to IV thrombolytic drug therapy, including with the least expensive of lytics, such as StreptoKinase. Indeed, beyond offering independent agitative disruption and erosion of acute thrombosis, dtLFV would be expected, by production of convection currents, to improve IV thrombolytic drug mixing and diffusion down an otherwise TIMI 0 occluded coronary vessel, so it can be thought of, essentially, as a kinetically active drug delivery system.
Indeed, the adjunctive pairing of dtLFV and IV thrombolysis, if successful, could tip the balance of the now old battle between pre-hospital thrombolysis versus PPCI, or with IV thrombolytics as an adjunct to facilitated PCI (fPCI – drip and ship), which was otherwise settled as a failure due to higher rates of post-PPCI No-reflow in ASSENT 4. However, with Brush et. al.’s new treatment strategy for No-Reflow, perhaps ASSENT 4 could be revisited, with or without dtLFV, as there could now very well be a good way to clear up early clot disruption’s mess regarding distal thrombotic embolization.
Of course, the safety of dtLFV, especially if applied to older STEMI victims (with a higher likelihood of extensive calcification of the valves or coronaries themselves), would be a real question regarding potential; cracking of the ribs, bruising, embolic concerns, and also conceivably possible damage to the coronary endothelium itself. All these safety issues would need to be vetted and would most likely lead to comprehensive inclusion/exclusion criteria for STEMI patients being eligible for the therapy. Regarding future research, dtLFV could benefit from a basic, simple proof-of-principle animal study, which could be done in large, human-sized pigs, reindeer, or the great ape.
All three of dtLFV, LFUS, and HFUS sonothrombolysis offer potential for assisting distal capillary microvascular flow, whereby dtLFV would provide a clot disruptive and vasodilatory effect, plus relax the otherwise stiff myocardium (thereby lowering transmyocardial vascular resistance), LFUS would cause a clot disruptive and vasodilatory effect, and in HFUS sonothrombolysis, along with vasodilation, the MBs would travel through the arterioles and capillaries, clearing them upon each flash of High MI HFUS as they go. But again, it needs to be re-emphasized that in the urban scenario, very possibly the problem of no-reflow may be solved by intracoronary MB sonothrombolysis, and with this early clot disruption in view to recanalization of the vessel should not necessarily be feared (per lessons learned in ASSENT 4) and could quite possibly only carry an upside, by providing an earlier restoration of blood flow.
Taking a step back, it is notable that almost all research efforts so far in the delivery of acoustic STEMI reperfusion therapies are based on helping initial survivors, who do not succumb to sudden death before arrival of EHS. Indeed, we tend to forget that there are about 600,000 heart attacks per year in the United States, of which there are about 350,000 cardiac arrests of which 90% don’t make it to EHS to receive revascularization care, with many apparently only calling for help 2 hrs after first symptom, for not being aware they were even having a heart attack.
So, regarding STEMI care, it seems odd that a wearable STEMI alert system (like the Apple Watch for Atrial Fibrillation) is not at this time commercially available, and one would think, as heart attack is still ultimately the number one killer in the developed world, that by now it should be. Interestingly, a new, maverick first-line emergency method of utilizing patient-administered chest wall fist-strikes immediately following a STEMI alert by such a wearable has just been published. This concept, termed Thrombo-Agitative Percussion (TAP), aims to break apart the occlusive, epicardial clot at the earliest stages (while still a loosely bound platelet aggregate), to provide earliest possible (or plausibly instant) coronary recanalization and reflow as a bridge to survival in reaching EHS. Of course, for many reasons, this concept could be dangerous, and should not be widely disseminated to the public unless it were to be properly vetted in clinical trials.
Conclusions
There are many possible acoustic therapies at large, which, each, in its own way, could enhance STEMI reperfusion, but the question always becomes, at what price to safety, and would any alleged improvements be good enough, and under what niche, to translate to a net clinical benefit?
The use of HFUS sonothrombolysis with IV-administered MBs pre- and post-STEMI PPCI has shown preliminary success in the MRUSMI trial, and is presently being studied in the slightly larger REDUCE trial, where results should be available early in 2026. However, there are many technical reasons to suspect HFUS sonothrombolysis may not work reliably, due to a paucity of MBs expected to reach a zero flow acutely blocked coronary artery, the inability to echocardiographically image the location of a clot’s origin, and an inherently difficult patient STEMI population, many of whom are obese or smokers, with terrible acoustic windows.
The main headline of 2025 is the case study success of using transthoracic HFUS sonothrombolysis with an intracoronary administration of MBs reported by Brush et. al., as a means to treat No-Reflow following STEMI PPCI (with the patient still on the Cathlab table), which deals with many of the perceived issues of IV delivered MBs in sonothrombolysis, and we will have to wait to see if this technique pans out in larger studies.
Chest wall-directed dtLFV as a means to enhance STEMI reperfusion remains an interesting, easy-to-employ concept that is severely understudied, and it could potentially find a niche to accelerate and ensure the effectiveness of IV thrombolytic therapy, for No-Reflow, and/or in very poor countries where nothing else is available. However, transthoracic palpable percussion has perceived safety issues, particularly in embolic concerns by “knocking things loose” such as in calcified valves, or perhaps to the coronary endothelium itself, particularly if applied upon calcified lesions. Along with this, there are also issues of likely bruising to the chest wall and potential for cracked ribs if applied to older patients.
Regardless of the frequency and mode of delivery offered, this report predicts as much pessimism as optimism in finding a clinically safe and effective non-invasive acoustic therapy to assist STEMI reperfusion, as experience is continually teaching us in the difficulty of “threading the needle” in finding a solution that disrupts thrombosis to promote reflow while not unacceptably harming the adjacent tissues, the arteries, or more broadly the patient.
However, in the end, when it comes to heart attack, time is muscle, and knowing that acoustic therapies by many frequencies and modes of delivery can accelerate clot disruption and enhance blood flow, effort will continue to find a workable non-invasively delivered adjunctive or stand-alone solution, of which HFUS sonothromboysis is presently “up to bat”.
Affiliation Details:
The Author of this manuscript is the President and CEO of Ahof Biophysical Systems Inc., a private cardiovascular research and development corporation, located in Vancouver, B.C., Canada. ABS Inc. provided the funding and resources for this review.
Conflict of Interest:
The author, AH, has a patent pending regarding use of an ST-segment surveillance wearable, which incorporates use of “Thrombo-Agitative Percussion” (TAP) – inclusive of fist strikes to the chest, deep coughing, and upper back slams – as a prospective first-line, home or office-based emergency reperfusion method for STEMI as a bridge to promote survival to EHS.
References:
- Elendu C, Amaechi DC, Elendu TC, Omeludike EK, Alakwe-Ojimba CE, et al. Comprehensive review of ST-segment elevation myocardial infarction: Understanding pathophysiology, diagnostic strategies, and current treatment approaches. Medicine Baltimore. 2023; 102: 35687.
- Ibanez B, James S, Agewall S, Antunes MJ, Halvorsen S, et al. 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the Task Force for the Management of Acute Myocardial Infarction in Patients Presenting With ST-Segment Elevation of the European Society of Cardiology. Eur Heart J. 2018; 39: 119-177.
- Sabe MA, Kaeberlein FJ, Sabe SA, Kelly A, Summerfield T, et al. Emergency Chest Pain Center: A Novel Approach to Reduce Door to Balloon Time. JACC Adv. 2025; 4: 101774.
- Van de Werf F, Ardissino D, Betriu A, Cokkinos DV, Falk E, et al. Management of acute myocardial infarction in patients presenting with ST-segment elevation. The task force on the management of acute myocardial infarction of the European Society of Cardiology. Eur Heart J. 2003; 24: 28-66.
- Bhandari M, Vishwakarma P, Sethi R, Pradhan A. Stroke Complicating Acute ST Elevation Myocardial Infarction Current Concepts. Int J Angiol. 2019; 28: 226-230.
- Hillani A, Potter B: Intracoronary Thrombus and No-Reflow: one Size to Fit All? Canadian Journal of Cardiology. Feb 2021, Editorial, Vol 37, Issue 2, Pg. 202-205. DOI:https//doi.org/10.1016/j.cjca.2020.08.015
- Faridi KF, Wang Y, Minges KE, Smilowitz NR, McNamara RL, Kontos MC, Wang TY, Connors AC, Clary JM, Osborne AD, Pereira L, Curtis JP, Blankinship K, Mayfield J, Abbott JD. Predicting Mortality in Patients Hospitalized With Acute Myocardial Infarction: From the National Cardiovascular Data Registry. Circ Cardiovasc Qual Outcomes. 2025 Mar;18(3):e011259. doi: 10.1161/CIRCOUTCOMES.124.011259. Epub 2025 Jan 13. PMID: 39801472; PMCID: PMC11919567.
- Annibali G, Scrocca I, Aranzulla TC, Meliga E, Maiellaro F, Musumeci G. “No-Reflow” Phenomenon: A Contemporary Review. J Clin Med. 2022 Apr 16;11(8):2233. doi: 10.3390/jcm11082233. PMID: 35456326; PMCID: PMC9028464.
- American Heart Association, CPR Facts and Stats, American Heart Association CPR. Cares. Accessed November 2nd, 2025. https://mycares.net/sitepages/aboutcares.jsp
- Fang J. Public awareness of heart attack symptoms. What should we look for and how will it help? Future Cardiology, 2010, Volume 6, Issue 5, pp 563 – 565.
- Dickson, E. US patent No. 1612267, entitled, Surgical Dressing, 1925.
- Russhard P, Al Janabi F, Parker M, Clesham GJ. Patterns of ST-segment resolution after guidewire passage and thrombus aspiration in primary percutaneous coronary intervention (PPCI) for acute myocardial infarction. Open Heart. 2016;3:e000430. https://doi.org/10.1136/openhrt-2016-000430
- Wobser E, Stumpff U: Intragastral disintegration of blood coagula by mechanical vibration. Endoscopy 1978, 10:15-19. doi:10.1055/s-0028-1098254.
- Dubrul, et al: Vibrating Catheter, US Patent No. 5,713,848, 1998.
- Krajcer Z, Atmakuri, S. One step treatment with of DVT with the Trellis-8 Device. Apr. 2006, ENDOVASCULAR TODAY, pp. 69-74.
- Trubestein G., Engel C., Etzel F., Sobbe A., Cremer H., Stumpff U. Thrombolysis by ultrasound. Clin. Sci. Mol. Med. 1976;3:697s-698s. doi: 10.1042/cs051697s.
- Steffen W, Fishbein MC, Luo H, Lee DY, Nita H, Cumberland DC, Tabak SW, Carbone M, Maurer G, Siegel RJ. High intensity, low frequency catheter-delivered ultrasound dissolution of occlusive coronary artery thrombi: an in vitro and in vivo study. J Am Coll Cardiol. 1994;24:1571-1579.
- Nedelmann, Max & Brandt, Christian & Schneider, Felicitas & Eicke, Martin & Kempski, Oliver & Krummenauer, Frank & Dieterich, Marianne. (2005). Ultrasound-Induced Blood Clot Dissolution without a Thrombolytic Drug Is More Effective with Lower Frequencies. Cerebrovascular diseases (Basel, Switzerland). 20. 18-22. 10.1159/000086122.
- Tachibana K, Tachibana S. Albumin microbubble echo-contrast material as an enhancer for ultrasound accelerated thrombolysis. Circulation. 1995;92:1148-1150. doi: 10.1161/01.cir.92.5.1148.
- Yao R, Hu J, Zhao W, Cheng Y, Feng C. A review of high-intensity focused ultrasound as a novel and non-invasive interventional radiology technique. J Interv Med. 2022 Jun 22;5(3):127-132. doi: 10.1016/j.jimed.2022.06.004. PMID: 36317144; PMCID: PMC9617156.
- Rosenschein U, Furman V, Kerner E, Fabian I, Bernheim J, Eshel Y. Ultrasound imaging-guided noninvasive ultrasound thrombolysis: preclinical results. Circulation. 2000 Jul 11;102(2):238-45. doi: 10.1161/01.cir.102.2.238. PMID: 10889137.
- Alexandrov AV, Molina CA, Grotta JC, Garami Z, Ford SR, Alvarez-Sabin J, Montaner J, Saqqur M, Demchuk AM, Moyé LA, Hill MD, Wojner AW; CLOTBUST Investigators. Ultrasound-enhanced systemic thrombolysis for acute ischemic stroke. N Engl J Med. 2004 Nov 18;351(21):2170-8. doi: 10.1056/NEJMoa041175. PMID: 15548777.
- Xie F, Lof J, Everbach C, He A, Bennett RM, Matsunaga T, Johanning J, Porter TR. Treatment of acute intravascular thrombi with diagnostic ultrasound and intravenous microbubbles. JACC Cardiovasc Imaging. 2009 Apr;2(4):511-8. doi: 10.1016/j.jcmg.2009.02.002. PMID: 19580735.
- Needs D, Blotter J, Cowan M, Fellingham G, Johnson AW, et al. Effect of Localized Vibration Massage on Popliteal Blood Flow. J Clin Med. 2023; 12: 2047.
- Fateh HR, Nakhostin Ansari N, Nakhostin-Ansari A, et al. The effects of local calf vibration on balance, blood flow, and nerve conductivity in patients with diabetic peripheral neuropathy: a pilot study. Physiother Theory Pract Jul 2024;40(7):1397-1403. doi: 10.1080/09593985.2023.2173992
- Liao F, Zhang K, Zhou L, Chen Y, Elliott J, Jan YK. Effect of Different Local Vibration Frequencies on the Multiscale Regularity of Plantar Skin Blood Flow. Entropy (Basel). Nov 13 2020;22(11)doi: 10.3390/e22111288
- Espeit L, Lapole T. Effects of graduated compression stockings, local vibration and their combination on popliteal venous blood velocity. Phlebology. Aug 2020;35(7):505-512. doi: 10.1177/0268355520902000
- Ren W, Pu F, Luan H, et al. Effects of Local Vibration With Different Intermittent Durations on Skin Blood Flow Responses in Diabetic People. Front Bioeng Biotechnol 2019;7:310. doi: 10.3389/fbioe.2019.00310
- Lindblad LE, Lorenz RR, Shepherd JT, Vanhoutte PM: Effect of vibration on canine cutaneous artery. Heart Circ Physiol 1986, 19:H519-H523.
- Ljung B, Silvertsson R: Vibration-induced inhibition of vascular smooth muscle contraction. Blood Vessels 1975, 12:38-52.
- Hudlicka O, Wright A: The effect of vibration on blood flow in skeletal muscle in rabbit. Clin Sci Mol Med 1978, 55:471-476.
- Ichioka S, Yokogawa H, Nakagami G, Sekiya N, Sanada H: In vivo analysis of skin microcirculation and the role of nitric oxide during vibration. Ostomy Wound Manage. 2011 Sep; 57 (9): 40-7.
- Maloney-Hinds C, Petrofsky JS, Zimmerman G, Hessinger DA: The role of nitric oxide in skin blood flow increases due to vibration in healthy adults and adults