Hepatocellular Carcinoma Outcomes in Northern Tanzania
Clinico-demographic profile and Outcome of Hepatocellular Carcinoma Patients in Northern Tanzania – A 7-year Retrospective Review
Kennedy Misso, MD 1,2, Comfort M. Takang, MD, MPH 3, Elizabeth Mosha, MD 1, Julius Pius Alloyce, MSc 1, Yotham Gwanika, MSc 1, Salum Kalonge, MSc 1, Augustine Musyoka 1,2, Yvonne Datta, MD3, Arkadiusz Z. Dudek, MD, PhD 4, Randy Hurley, MD 5, Furaha Serventi, MD 1,2
- Kilimanjaro Christian Medical Centre (KCMC), Moshi, Tanzania
- Kilimanjaro Christian Medical Centre University (KCMCU), Moshi, Tanzania
- University of Minnesota Department of Hematology, Oncology, and Transplant, Minneapolis, Minnesota
- Division of Medical Oncology, Department of Medicine, Mayo Clinic, Rochester, Minnesota, USA
- Regions Hospital, HealthPartners System, St. Paul, Minnesota, USA
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Misso, K., Takang, CM., et al., 2026. Clinico- demographic profile and Outcome of Hepatocellular Carcinoma Patients in Northern Tanzania – A 7-year Retrospective Review. Medical Research Archives, [online]14(1). https://doi.org/10.18103/mra.v14i1.7092
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v14i1.7092
ISSN 2375-1924
Abstract
Background: Hepatocellular carcinoma (HCC) is a significant cause of cancer-related deaths worldwide, with sub-Saharan Africa, including Tanzania, experiencing a substantial burden of the disease. In northern Tanzania, HCC poses a considerable challenge due to the high prevalence of hepatitis B and C, combined with limited healthcare resources. However, there is limited knowledge about its impact in the region due to the scarcity of data. This study investigates the clinicodemographic profile, clinical presentation, treatment offered, and outcomes of patients diagnosed with HCC at a zonal hospital in northern Tanzania, aiming to support healthcare strategies for improved management and prevention of liver cancer.
Patients and Methods: We reviewed the data of patients aged 18 years or older who presented to KCMC with a diagnosis of HCC from January 1, 2016, to December 31, 2023. Diagnosis was established using American College of Radiology guidelines, incorporating triphasic CT or MRI findings and the Liver Imaging Reporting and Data System (LI-RADS); patients with cirrhosis and LI-RADS 5 were classified as HCC, while indeterminate lesions underwent biopsy. Of 124 identified cases, 94 met the inclusion criteria after excluding patients with incomplete imaging or insufficient diagnostic confirmation. Sociodemographic, clinical, laboratory, staging, treatment, and survival data were extracted from paper and electronic records, de-identified, and stored in REDCap for analysis. Statistical evaluation was performed using Stata 18, with descriptive statistics applied to continuous and categorical variables. Associations were tested using chi-square tests, t-tests, and ANOVA, while survival outcomes were assessed using Kaplan–Meier curves and log-rank tests. A p-value <0.05 was considered statistically significant.
Results: Most patients were male (67%) and aged 31–59 years (51%). Key risk factors included hepatitis B (44.7%), alcohol use (39.4%), and hepatitis C (6.4%). Advanced disease was common: 63.8% had ECOG performance status 2–3, and 42.5% had no liver cirrhosis. Only 18% received systemic therapy (mostly sorafenib); none underwent curative procedures. Median survival was 4.73 months, with an interquartile range (IQR) of 0.92-12.42 and a 13% one-year survival rate. Poor outcomes were statistically associated with high AFP levels, hepatitis B infection, lack of health insurance, and poor performance status.
Conclusion: Hepatocellular carcinoma is a significant health burden in northern Tanzania, with a high incidence of late-stage presentations, limited treatment options, and poor outcomes. Early detection through routine screening for high-risk populations, improved access to diagnostic and therapeutic services are critical to improving survival and quality of life for HCC patients. Public health interventions focused on reducing hepatitis B and C prevalence as well as alcohol use are essential for reducing the future burden of liver cancer in Tanzania.
Keywords
Hepatocellular carcinoma, Tanzania, clinicodemographic profile, survival, hepatitis B, hepatitis C
Introduction
According to the most recent Global statistics, Hepatocellular Carcinoma (HCC) is the sixth most common cancer and the third most common cause of cancer-related deaths worldwide. The rising global burden of HCC is significantly higher in low- and middle-income countries, with an estimate of over 80% of HCC cases worldwide reported in Sub-Saharan Africa (SSA) and Southeast Asia. Most cases of HCC in SSA are recorded among young adults, median 45 years (IQR 35–57), with an abysmal prognosis, marked by a median survival of 2.5 months from time of presentation. HCC is therefore a rising public health concern in SSA. Major risk factors for HCC in SSA include hepatitis B virus (HBV), hepatitis C virus (HCV), aflatoxin B1 exposure, and alcohol consumption, with metabolic dysfunction-associated fatty liver disease. In Tanzania, a national study on cancer mortality in 39 hospitals reported liver cancer as the third most common cause of cancer death nationwide. Another study published in 2014 on 142 HCC cases from a Hospital in Tanzania, most HCC cases presented at an advanced stage, with no patient receiving curative therapy as a result. Despite the significantly negative impact of HCC in Tanzania, the actual incidence of HCC in the country is unknown due to an overall paucity of data on the subject. We conducted a retrospective review spanning 7 years on the clinicodemographic profile and outcomes of HCC patients at a tertiary hospital in Northern Tanzania.
Methods
This retrospective study examined cases from Kilimanjaro Christian Medical Centre (KCMC), a key tertiary referral hospital in northern Tanzania, spanning from January 1, 2016, to December 31, 2023. KCMC serves as a zonal referral centre, offering specialised care to more than 15 million people. Following approval from the national and institutional review boards (IRBs), data were collected from all patients aged 18 years or older who presented to KCMC with a diagnosis of HCC. The study duration was selected as it coincided with the transition to electronic medical records and the strengthening of the cancer registry system at KCMC.
Diagnosis of HCC was based on the validated diagnostic guidelines from the American College of Radiology, which included the presence of a liver mass with characteristic HCC imaging findings on triphasic computed tomography (CT) or magnetic resonance imaging (MRI), and the use of the Liver Imaging Reporting and Data System (LI-RADS). Patients with cirrhosis and LiRADS V were considered HCC. Indeterminate lesions were biopsied. Patients with insufficient data to confirm HCC diagnosis were excluded from the study. We identified 124 patients. 12 patients had images done elsewhere and couldn’t be retrieved for LiRADS diagnosis confirmation, eight had low LiRADS grades without histological confirmation. A total of 94 patient records corresponding to the study time frame described met the study criteria and were accessed for review.
Study personnel obtained the required study information from the medical records of eligible patients. Both paper and electronic medical records were reviewed, and the data collected for the study were de-identified and stored electronically in REDCap, hosted at our institution, for further analysis. Information collected from the records of patients eligible for the study included sociodemographic data, clinical presentation, risk factors related to HCC (Alcohol use, Hepatitis B and C status, cirrhosis), liver-related blood tests, tumour stage, date of diagnosis, treatment modalities, start of treatment date, survival status, date of death or last follow-up date if alive.
Statistical analysis was conducted using Stata 18 software. For continuous variables, median and ranges were calculated, while proportions and frequency tables summarised categorical variables. Outcomes will be analysed as mean ± standard deviation and displayed through tables and charts. The chi-square (χ2) test was used to assess the significance of associations between categorical independent (predictor) and dependent (outcome) variables. For continuous outcome measures, the grouped sample t-test or one-way Analysis of Variance (ANOVA) was employed at a 95% confidence level. The significance level was set at P < 0.05. Survival analysis involved log-rank tests and Kaplan-Meier curves. P values less than .05 were considered statistically significant.
Results
CLINICODEMOGRAPHIC CHARACTERISTICS
Ninety-four patients with HCC were included in the final analysis. Among them, 67.0% were male. Most (51.1%) of the patients were between the ages of 31 and 59 years. Meanwhile, 35.1% were 60 years or older, and 13.8% were under 30 years of age. The most common risk factors were Hepatitis B infection (44.7%), Alcohol use (39.4%) and Hepatitis C infection (6.4%). Liver cirrhosis was noted in 12.8% of patients; however, this status was unknown in 44.7% patients. A small proportion (3.2%) of patients were reported to have fatty liver disease; meanwhile, the fatty liver disease status was unknown in 45.7% of cases. The majority of patients were categorised as Child-Pugh class B (30.8%), 16.0% as Child-Pugh class A, and 11.7% as Child-Pugh class C. Meanwhile, 41.5% were unclassified due to insufficient data. The majority of patients had an ECOG performance status of either II or III (63.8%) at the time of diagnosis, 7.5% had an ECOG performance status of 4, and 22.3% had an ECOG performance status of 1. Meanwhile, 6.4% had unknown ECOG status. The majority of patients were untreated (80.8%), 18.1% received sorafenib, and one patient (1.1%) received capecitabine. None of the patients received surgical resection or loco-regional therapy.
SURVIVAL ANALYSIS
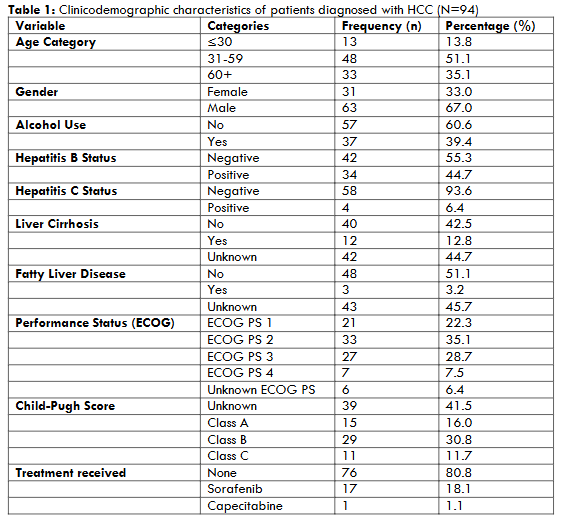
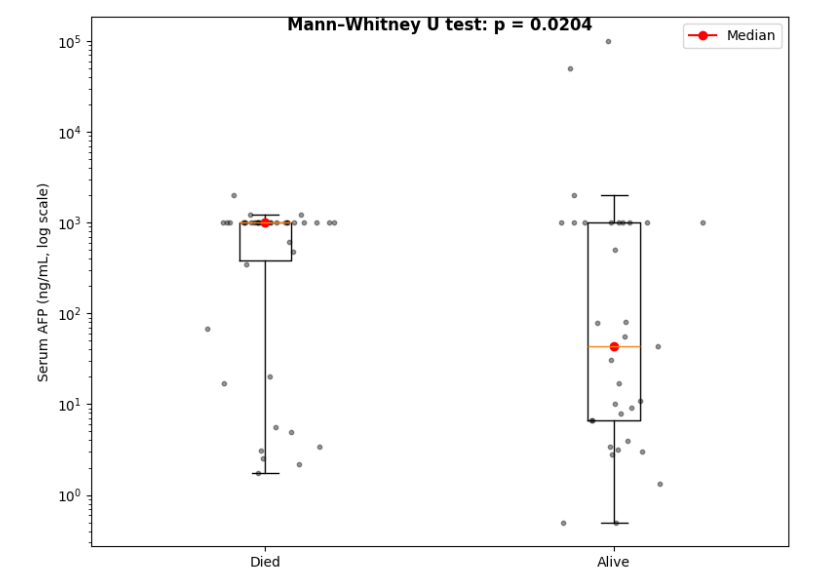
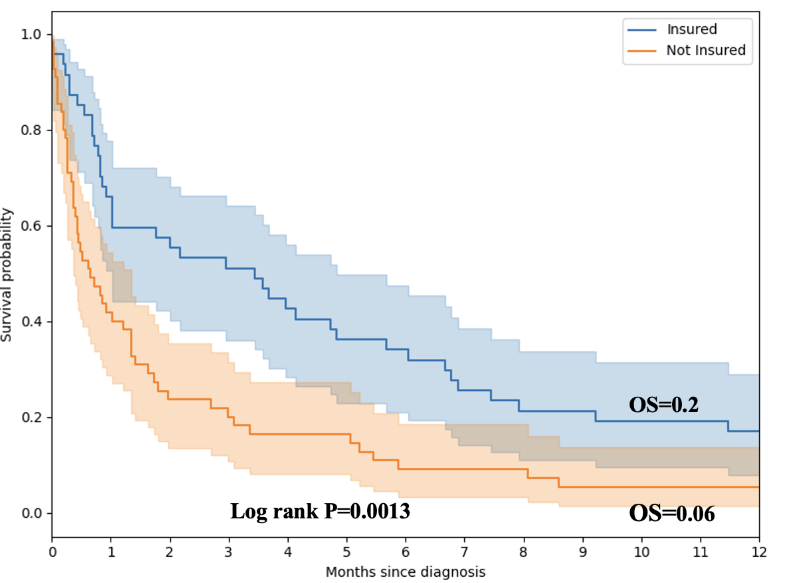
Median survival was 4.73 months (IQR 0.92-12.42), with a significant number of patients dying within the first month. Survival ranged from 1 day to 1378 days (~3.8 years). There was a statistically significant negative impact of high serum AFP levels. The median AFP was significantly higher among patients who died within one year compared to survivors (P=0.02). Figure 2 shows that the overall survival rate for patients diagnosed with HCC was 13% at one year. Those with health insurance had statistically better survival outcomes. Those with hepatitis B infection had a poor survival outcome.
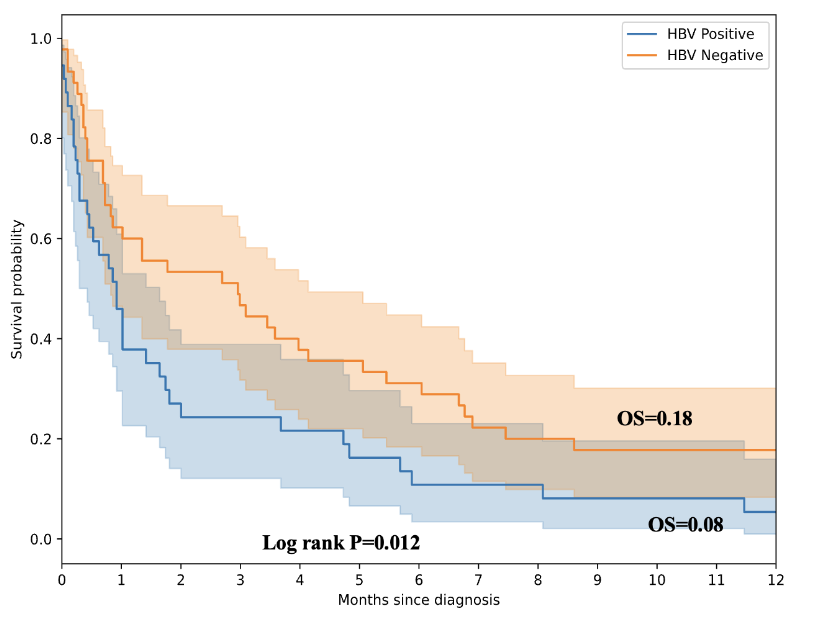
Patients aged 60 and older had a lower risk of early mortality (adjusted HR = 0.14, p = 0.12), although this was not statistically significant. A lack of health insurance was associated with a trend toward increased risk (adjusted HR = 4.22, p = 0.12). Hepatitis C positivity was initially associated with a higher risk (adjusted HR = 4.51, p = 0.61), but this finding was not statistically significant. Patients with a poor performance status (ECOG PS 4) had a significantly higher risk of early mortality (adjusted HR = 39.10, p = 0.05).
| Variable | Categories | Crude HR 95%(CI) | p-value | Adjusted HR (95% CI) | p-value |
|---|---|---|---|---|---|
| Age Category | ≤30 | 1 | 1 | ||
| 31-59 | 0.50 (0.17 – 1.45) | 0.20 | 0.28 (0.04 – 2.00) | 0.21 | |
| 60+ | 0.31 (0.10 – 0.92) | 0.04 | 0.14 (0.01 – 1.63) | 0.12 | |
| Gender | Female | 1 | 1 | ||
| Male | 1.71 (0.73 – 4.04) | 0.22 | 0.27 (0.04 – 1.82) | 0.18 | |
| Health Insurance | Yes | 1 | 1 | ||
| No | 2.72 (1.28 – 5.82) | 0.01 | 4.22 (0.70 – 25.56) | 0.12 | |
| Alcohol Use | No | 1 | 1 | ||
| Yes | 0.91(0.43 – 1.92) | 0.80 | 3.10 (0.55 – 17.43) | 0.20 | |
| Hepatitis B Status | Negative | 1 | 1 | ||
| Positive | 1.89 (0.85 – 4.22) | 0.12 | 1.82 (0.35 – 9.59) | 0.48 | |
| Hepatitis C Status | Negative | 1 | 1 | ||
| Positive | 5.64 (1.17 – 27.34) | 0.03 | 4.51 (0.01 – 1561.80) | 0.61 | |
| Fatty Liver Disease | No | 1 | 1 | ||
| Yes | 0.93 (0.12 – 7.10) | 0.95 | 4.28 (0.01 – 89.41) | 0.67 | |
| Unknown | 0.95 (0.44 – 2.03) | 0.89 | 0.35 (0.07 – 1.84) | 0.22 | |
| Performance Status (ECOG) | ECOG PS 1 | 1 | 1 | ||
| ECOG PS 2 | 1.13 (0.43 – 2.97) | 0.80 | 0.51 (0.08 – 3.22) | 0.48 | |
| ECOG PS 3 | 1.73 (0.60 – 5.01) | 0.31 | 1.94 (0.19 – 19.41) | 0.57 | |
| ECOG PS 4 | 10.14 (1.83 – 56.24) | 0.01 | 39.10 (0.99 – 1542.97) | 0.05 | |
| Unknown ECOG PS | 4.53 (1.11 – 18.48) | 0.03 | 20.44 (0.27 – 1546.62) | 0.17 | |
| Child Pugh Score | Class A | 0.59 (0.23 – 1.55) | 0.28 | 1 | |
| Class B | 0.86 (0.34 – 2.14) | 0.74 | 1.96 (0.21 – 18.61) | 0.56 | |
| Class C | 1.42 (0.39 – 5.22) | 0.60 | 24.28 (0.93 – 634.24) | 0.06 | |
| Sorafenib | No | 1 | 1 | ||
| Yes | 0.67 (0.31 – 1.49) | 0.33 | 2.17 (0.36 – 13.04) | 0.40 |
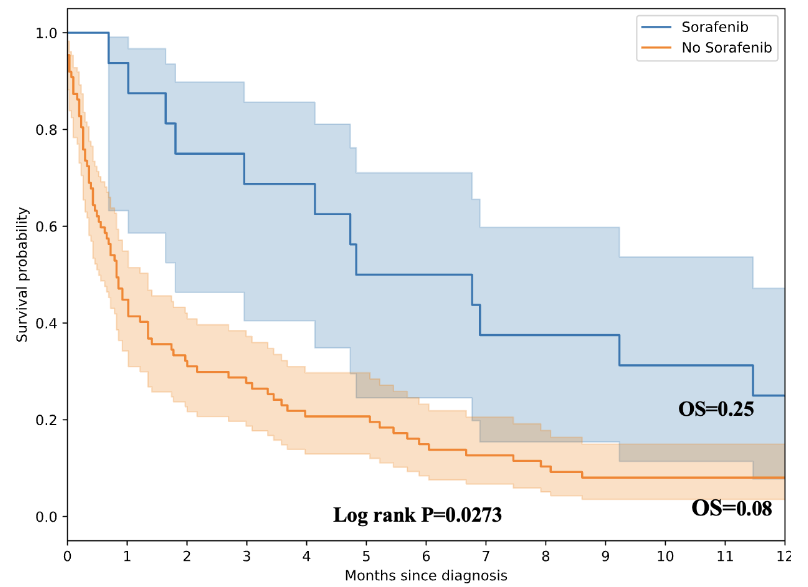
EFFECT OF SORAFENIB ON OVERALL SURVIVAL
Patients who received sorafenib had an overall survival rate of 25% at one year, compared to 8% who did not receive sorafenib.
Discussion
The clinicodemographic profile of patients with HCC in northern Tanzania aligns with global trends, with a higher incidence in males and middle-aged individuals. In our cohort, the median age at presentation was 51 years (interquartile range, IQR, 38–70), with more than half of the patients falling between 31 and 59 years of age. Males predominated (67%). Similar to other studies, a multicentre, multinational study of 2,566 patients in Africa reported a median age of 58 years in Egypt (IQR, 53–63) and 46 years (IQR, 36–58) in other African countries. Patients in our study were a decade older than most reports from sub-Saharan Africa (typically in the mid-40s), yet still younger than the median ages observed in developed countries. This difference likely reflects differences in population age structure, underlying etiologies, and surveillance practices: HBV-driven HCC in Africa tends to present earlier, whereas in high-income countries, HCC is increasingly associated with alcohol and metabolic dysfunction–associated steatosis liver disease, which manifests later in life. Our findings, therefore, occupy an intermediate position, underscoring both the demographic transition in our setting and the persistent contrasts with high-income regions. These interpretations should be viewed in light of our study’s retrospective, single-center design and modest sample size. Nonetheless, while male predominance appears consistent across settings, the age at presentation seems more sensitive to demographic and health system factors.
In this cohort, serum AFP levels showed a significant association, with patients who died within the first year having markedly higher AFP values compared to survivors, as confirmed by a Mann–Whitney U test (p= 0.02). This finding reinforces the role of AFP as a prognostic biomarker in hepatocellular carcinoma, consistent with prior reports linking elevated AFP to tumour aggressiveness, vascular invasion, and poorer outcomes. While AFP alone is not sufficient for diagnosis or prognostication, its correlation with survival in our setting highlights its potential utility in risk stratification, particularly in resource-limited environments where advanced imaging or molecular profiling are not readily available.
Treatment options in our study were minimal: 80% of patients received no therapy, 18% received sorafenib, and none underwent resection or loco-regional therapy. Sorafenib use was associated with markedly better short-term survival, although the multivariate analysis did not reach statistical significance. The significance of sorafenib was more apparent in the one-year survival analysis. Patients treated with sorafenib had statistically better survival outcomes (25% vs 8%), with a log-rank P value of 0.0273. This pattern reflects sorafenib’s cytostatic effect, which can delay early mortality even in advanced disease. The American society of Clinical oncologist (ASCO) and European Society of Medical Oncologist (ESMO) recommend immune checkpoint inhibitor–based combinations, atezolizumab + bevacizumab and durvalumab + tremelimumab (STRIDE regimen) in eligible patients as the first line treatment. The regimes are costly and not readily accessible in our clinical settings. Locoregional therapies, such as Transarterial Chemoembolization (TACE) and Radiofrequency Ablation (RFA), as well as interventional radiology, are unavailable. As a result, most patients receive palliative care. The African Liver Cancer Consortium reported similar findings: a low proportion of patients receiving treatment modalities and poor outcomes afterwards. In their ten-year retrospective review, treatment rates were significantly higher in Egypt (76%) in comparison to the rest of Africa (3%). In a retrospective analysis involving 142 HCC cases in northwest Tanzania, Jaka et al reported 100% of cases received palliative treatment with 46.5% in-hospital mortality, following diagnosis of HCC. They reported a median hospital stay of 14 days (IQR 10-16).
Our survival analysis reveals a notably poor outlook, with a median survival time of just 4.73 months (IQR 0.92-12.42). Indicates rapid disease progression for most patients. The fact that the majority did not live past one year underscores the substantial early mortality. In contrast, a few individuals survived for 3.8 years. High skewness and kurtosis values indicate a distribution heavily weighted toward short survival periods, with only a small number of long-term survivors. These results highlight both the aggressive course of the disease and the variability in patient outcomes, implying that while most are diagnosed late and succumb quickly, a small fraction may possess biological or therapeutic factors allowing for extended survival. This pattern has implications for clinical practice, statistical analysis, and research focused on uncovering predictors of longer-term survival. The African Liver Cancer Consortium found a median survival of 10.9 months (95% CI: 9.6–12.0) in Egypt, compared to 2.5 months (2.0–3.1) in other African countries (p < 0.0001). One-year survival was 44% in Egypt versus 10% in the rest of Africa, highlighting a significant difference. Similar reports from Tanzania National Hospital HCC study, Mwanga et al report only 5.6% had an observed tumour resection at the time of presentation. The difference is likely due to earlier presentation, availability of curative options, a well-established surveillance system, and better healthcare infrastructure in Egypt. On a systemic scale, these results underscore the need to enhance screening programs, referral systems, and treatment infrastructure to limit late-stage diagnoses.
Nearly half (44.7%) of our patients were hepatitis B positive. Hepatitis B infection was statistically associated with poor survival outcomes. Those with hepatitis B infection had an earlier presentation and a poor survival outcome. Patients with health insurance had significantly better one-year survival compared to those without coverage (20% vs 6%, log-rank p = 0.0013). Although the 30-day adjusted hazard ratio did not reach statistical significance. The trend toward improved outcomes suggests that insurance status may serve as a proxy for broader socioeconomic advantages, including earlier health-seeking behaviour, a greater likelihood of diagnostic work-up, and improved access to systemic therapy. Notably, sorafenib—the only systemic therapy available in our setting—is not covered by insurance, suggesting that indirect factors, such as the socioeconomic status (SES), transportation, or supportive care, may influence the observed benefit. Although SES was not examined in our cohort, individuals with health insurance are usually employed or have siblings with formal employment. Other insurance plans may provide coverage for individuals not in formal employment, which we postulate to be more common among those with higher socioeconomic status. These findings align with reports from high-income countries, where financial protection mechanisms have a significant impact on cancer outcomes. A retrospective analysis of 762 patients found that individuals with Medicaid or without insurance had a later diagnosis, less access to curative treatment, and lower survival rates in hepatocellular carcinoma compared to those with private insurance. An analysis of the SEER database from 2011 to 2015, which included 33670 patients with HCC, found that Patients with private insurance were more likely to be diagnosed at earlier stages, receive timely treatment, and achieve better survival outcomes, including quicker access to liver transplant waitlists. In comparison, those with Medicaid or no health insurance faced delayed diagnosis, less access to curative therapies, and significantly poorer outcomes, with financial and systemic barriers compounding these disparities. These parallels underscore that financial protection mechanisms are crucial not only in high-income countries but also in resource-constrained settings, such as Tanzania, where expanding insurance coverage and incorporating essential cancer therapies into benefit packages could substantially enhance outcomes.
Improving HCC outcomes in Tanzania requires a multipronged approach: scaling up hepatitis B vaccination and antiviral therapy, implementing affordable surveillance for high-risk groups, expanding diagnostic and treatment infrastructure, and strengthening health insurance coverage to include essential cancer care. Together, these measures would reduce late-stage presentation, improve survival, and address inequities in access to care.
Conclusion:
Hepatocellular carcinoma (HCC) constitutes a significant health burden in northern Tanzania, marked by frequent late-stage diagnosis, restricted availability of curative treatments, and low survival rates. This seven-year retrospective analysis at a zonal referral hospital found that most patients were middle-aged males. Hepatitis B infection and alcohol consumption were the predominant risk factors; the majority of patients presented with advanced-stage disease and reduced functional status. Only a small proportion received systemic therapy, mainly sorafenib. The median survival was 4.7 months, and only 13% survived longer than one year. Poor prognosis was significantly correlated with high serum alpha-fetoprotein (AFP) levels, hepatitis B infection, absence of health insurance, and reduced performance status. Patients treated with sorafenib demonstrated higher one-year survival rates compared to those who did not receive systemic therapy. Furthermore, health insurance coverage was associated with improved survival, emphasising the influence of socioeconomic status on access to treatment and patient outcomes. These results indicate an urgent requirement for improved early detection protocols, broader access to diagnostic and therapeutic resources, and reinforced public health initiatives addressing hepatitis B, hepatitis C, and alcohol consumption. Expanding health insurance coverage and incorporating essential cancer treatments into benefit plans may significantly increase survival rates and quality of life for patients with HCC in Tanzania.
Acknowledgements of research support for the study.
Foundation for Cancer Care in Tanzania (FCCT), Minneapolis, Minnesota, USA, for research funding support.
List of where and when the study has been presented in part elsewhere:
Not applicable.
Disclaimers:
None
References
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249. doi:10.3322/caac.21660
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71(1):7-33. doi:10.3322/CAAC.21654
- Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16(10):589-604. doi:10.1038/S41575-019-0186-Y;SUBJMETA
- Cancer Today. Accessed September 30, 2025. https://gco.iarc.fr/today/online-analysis-table?v=2018&mode=population&mode_population=regions&population=900&populations=900&key=asr&sex=0&cancer=11&type=0&statistic=5&prevalence=0&population_group=0&ages_group%5B%5D=4&ages_group%5B%5D=17&group_cancer=1&include_nmsc=1&include_nmsc_other=1
- Yang JD, Gyedu A, Afihene MY, et al. Hepatocellular carcinoma occurs at an earlier age in Africans, particularly in association with chronic Hepatitis B. American Journal of Gastroenterology. Nature Publishing Group. 2015;110(11):1629-1631. doi:10.1038/ajg.2015.289
- Spearman CW, Dusheiko G, Jonas E, et al. Hepatocellular carcinoma: measures to improve the outlook in sub-Saharan Africa. Lancet Gastroenterol Hepatol. 2022;7(11):1036-1048. doi:10.1016/S2468-1253(22)00041-3
- Villanueva A. Hepatocellular Carcinoma. Longo DL, ed. New England Journal of Medicine. 2019;380(15):1450-1462. doi:10.1056/NEJMra1713263
- Lyimo EP, Rumisha SF, Mremi IR, et al. Cancer Mortality Patterns in Tanzania: A Retrospective Hospital-Based Study, 2006-2015. JCO Glob Oncol. 2020;6:224-232. doi:10.1200/JGO.19.00270
- Jaka H, Mshana SE, Rambau PF, Masalu N, Chalya PL, Kalluvya SE. Hepatocellular carcinoma: Clinicopathological profile and challenges of management in a resource-limited setting. World J Surg Oncol. 2014;12(1):1-9. doi:10.1186/1477-7819-12-246/TABLES/5
- KCMC – Kilimanjaro Christian Medical Centre. Accessed October 5, 2025. https://www.kcmc.ac.tz/#
- Marks RM, Masch WR, Chernyak V. LI-RADS: Past, Present, and Future, From the AJR Special Series on Radiology Reporting and Data Systems. doi:10.2214/AJR.20.24272
- Liang Y, Xu F, Guo Y, et al. Diagnostic performance of LI-RADS for MRI and CT detection of HCC: A systematic review and diagnostic meta-analysis. Eur J Radiol. 2021;134:109404. doi:10.1016/j.ejrad.2020.109404
- ACR Liver Imaging Reporting & Data System (LI-RADS®). Accessed December 5, 2025. https://www.acr.org/Clinical-Resources/Clinical-Tools-and-Reference/Reporting-and-Data-Systems/LI-RADS
- Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: Building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/J.JBI.2019.103208
- Abou-Alfa GK, Lau G, Kudo M, et al. Tremelimumab plus Durvalumab in Unresectable Hepatocellular Carcinoma. NEJM Evidence. 2022;1(8). doi:10.1056/EVIDOA2100070
- Hwang SY, Danpanichkul P, Agopian V, et al. Hepatocellular carcinoma: updates on epidemiology, surveillance, diagnosis and treatment. Clin Mol Hepatol. 2024;31(Suppl):S228. doi:10.3350/CMH.2024.0824
- Gordan JD, Kennedy EB, Abou-Alfa GK, et al. Systemic Therapy for Advanced Hepatocellular Carcinoma: ASCO Guideline Update. J Clin Oncol. 2024;42(15):1830-1850. doi:10.1200/JCO.23.02745
- Vogel A, Chan SL, Dawson LA, et al. Hepatocellular carcinoma: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Annals of Oncology. 2025;36(5):491-506. doi:10.1016/j.annonc.2025.02.006
- Yang JD, Mohamed EA, Aziz AOA, et al. Characteristics, management, and outcomes of patients with hepatocellular carcinoma in Africa: a multicountry observational study from the Africa Liver Cancer Consortium. Lancet Gastroenterol Hepatol. 2017;2(2):103-111. doi:10.1016/S2468-1253(16)30161-3
- Park JW, Chen M, Colombo M, et al. Global patterns of hepatocellular carcinoma management from diagnosis to death: the BRIDGE Study. Liver Int. 2015;35(9):2155-2166. doi:10.1111/LIV.12818
- Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16(10):589. doi:10.1038/S41575-019-0186-Y
- Franco RA, Fan Y, Jarosek S, Bae S, Galbraith J. Racial and Geographic Disparities in Hepatocellular Carcinoma Outcomes. Am J Prev Med. 2018;55(5):S40-S48. doi:10.1016/j.amepre.2018.05.030
- Mak D, Babb de Villiers C, Chasela C, Urban MI, Kramvis A. Analysis of risk factors associated with hepatocellular carcinoma in black South Africans: 2000-2012. PLoS One. 2018;13(5):e0196057. doi:10.1371/journal.pone.0196057
- Li B, Liu A, Wen Y, et al. The prognostic values of serum markers in hepatocellular carcinoma after invasive therapies based on real-world data. J Clin Lab Anal. 2021;35(9). doi:10.1002/JCLA.23932
- Norman JS, Mehta N. The Role of AFP-L3 and DCP Biomarkers in the Diagnosis and Management of Hepatocellular Carcinoma. Current Hepatology Reports 2025 24:1. 2025;24(1):16-. doi:10.1007/S11901-025-00686-7
- Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378-390. doi:10.1056/NEJMoa0708857
- Mwanga A, Mwakipesile J, Kitua D, Ringo Y. A Comprehensive Overview of In-patients Treated for Hepatocellular Carcinoma at a Tertiary Care Facility in Tanzania. Journal of Renal and Hepatic Disorders. 2023;7(1):22-27. doi:10.15586/jrenhep.v7i1.153
- Yen YH, Kee KM, Hu TH, et al. Hepatitis B virus-related hepatocellular carcinoma has superior overall survival compared with other etiologies. PLoS One. 2024;19(3):e0290523. doi:10.1371/JOURNAL.PONE.0290523
- Sellers CM, Uhlig J, Ludwig JM, et al. The impact of socioeconomic status on outcomes in hepatocellular carcinoma: Inferences from primary insurance. Cancer Med. 2019;8(13):5948-5958. doi:10.1002/CAM4.2251
- Zheng Y, Zhang X, Lu J, Liu S, Qian Y. Association between socioeconomic status and survival in patients with hepatocellular carcinoma. Cancer Med. 2021;10(20):7347-7359. doi:10.1002/cam4.4223

