Individual Variations in Blood Loss During ECLS
Individual variations in hemostasis, thrombosis and blood loss during extracorporeal life support
Wayne L. Chandler MD1
Keywords
- extracorporeal life support
- hemostasis
- thrombosis
- blood loss
- platelets
Introduction
While extracorporeal life support (ECLS) can be lifesaving for patients needing short-term cardiopulmonary support, it is associated with an increased risk of blood loss, the need for recurrent transfusion support and thrombus formation in the ECLS circuit.¹⁻⁷ Current studies report that patients on ECLS show a continuing loss of red cells, platelets and coagulation factors. Pediatric patients on ECLS lose blood cells even faster than adults. This persistent blood loss leads to recurrent, at times daily, transfusion support including red cell, platelet and plasma transfusions. Increased blood loss and transfusions are associated with worse outcomes in ECLS. Thrombosis in the ECLS circuit is also a common problem. Anticoagulant medications are used to reduce the risk of circuit thrombosis, but anticoagulation leads to an increased risk of bleeding and transfusion support.
While we have a good understanding of the overall risks of blood loss, transfusion and circuit thrombosis during ECLS, there are two areas that require further study if we are to reduce these problems. First, we need to better understand what causes blood loss and circuit thrombosis. Not all blood loss during ECLS is due to bleeding. Some ECLS patients show only minor bleeding, but still require major transfusion support. Second, there is a substantial individual variation in the response of different patients to ECLS. Some ECLS patients require little transfusion support and show only minimal circuit thrombosis, while others require daily platelet or red cell transfusions and undergo several circuit changes due to thrombosis. We need a better understanding of the individual patient hematologic response to ECLS.
How a patient responds to ECLS depends of multiple factors including 1) the patient’s underlying problem and clinical condition before starting ECLS, 2) the response of the patient’s blood when exposed to the cannula, tubing, oxygenator and pumps of the circuit and 3) clinical progress while on the circuit including patient improvement or worsening and clot deposition in the circuit (Figure 1).⁸
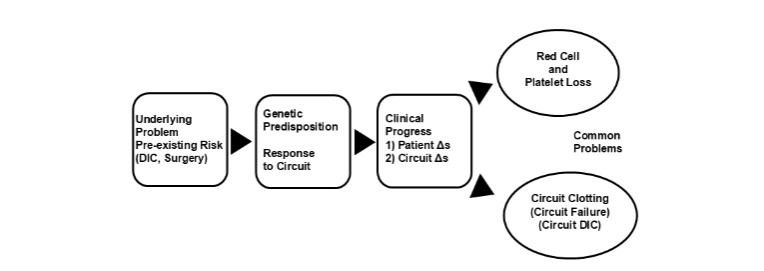
Figure 1. Patient response to extracorporeal life support. DIC = disseminated intravascular coagulation, Δs = changes.
To understand how a patient responds to ECLS we must separate what is happening in the patient versus what is happening in the ECLS circuit. This review will focus on hemostatic, hematologic and thrombotic aspects of the patient response to ECLS including effects on the coagulation system, platelets, red cells, endothelium and leukocytes, and what is known about different mechanisms of blood loss including bleeding, phlebotomy, hemolysis, cell activation and sublethal cell damage. The goal is to summarize how hematologic systems respond to ECLS, what factors affect this response and what future research is needed to minimize ECLS side effects and improve outcomes.
References
1. Thomas J, Kostousov V, Teruya J. Bleeding and Thrombotic Complications in the Use of Extracorporeal Membrane Oxygenation. Semin Thromb Hemost. 2018;44:20-29.
2. Dalton HJ, Reeder R, Garcia-Filion P, et al. Factors Associated with Bleeding and Thrombosis in Children Receiving Extracorporeal Membrane Oxygenation. Am J Respir Crit Care Med. 2017; 196:762-771.
3. Muszynski JA, Reeder RW, Hall MW, et al. RBC Transfusion Practice in Pediatric Extracorporeal Membrane Oxygenation Support. Crit Care Med. 2018;46:e552-e559.
4. Aubron C, Cheng AC, Pilcher D, et al. Factors associated with outcomes of patients on extracorporeal membrane oxygenation support: a 5-year cohort study. Crit Care. 2013;17:R73.
5. Dalton HJ, Garcia-Filion P, Holubkov R, et al. Association of bleeding and thrombosis with outcome in extracorporeal life support. Pediatr Crit Care Med. 2015;16:167-174.
6. Nellis ME, Saini A, Spinella PC, et al. Pediatric Plasma and Platelet Transfusions on Extracorporeal Membrane Oxygenation: A Subgroup Analysis of Two Large International Point-Prevalence Studies and the Role of Local Guidelines. Pediatr Crit Care Med. 2020;21:267-275.
7. Karam O, Nellis ME. Transfusion management for children supported by extracorporeal membrane oxygenation. Transfusion. 2021;61:660-664.
8. Chandler WL. Platelet, Red Cell, and Endothelial Activation and Injury During Extracorporeal Membrane Oxygenation. Asaio j. 2021;67:935-942.
9. Andrew M, Paes B, Milner R, et al. Development of the human coagulation system in the full-term infant. Blood. 1987;70:165-172.
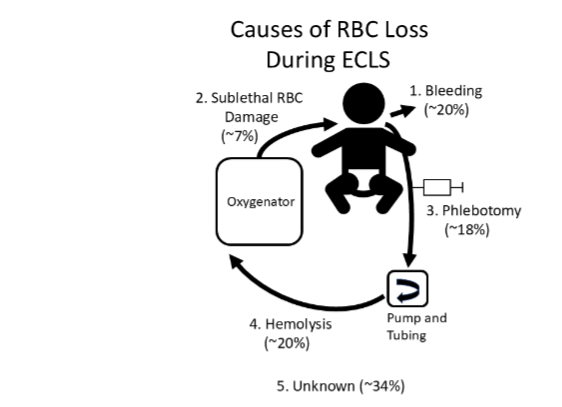
10. Bilodeau KS, Saifee NH, Chandler WL. Causes of red blood cell loss during extracorporeal membrane oxygenation. Transfusion. 2023.
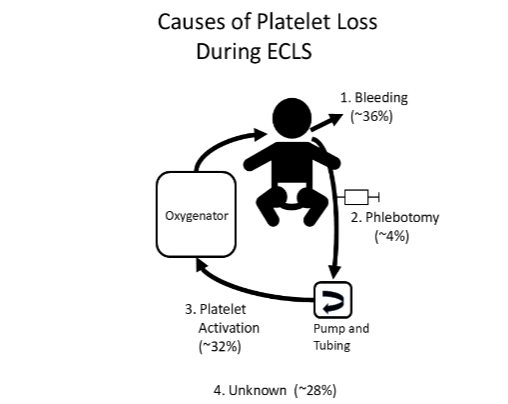
11. Atchison C, Widdershins A, Chandler WL. Causes of platelet loss during extracorporeal life support. Artif Organs. 2022.
12. Abrams D, Baldwin MR, Champion M, et al. Thrombocytopenia and extracorporeal membrane oxygenation in adults with acute respiratory failure: a cohort study. Intensive Care Med. 2016;42:844-852.
13. Kim HS, Cheon DY, Ha SO, et al. Early changes in coagulation profiles and lactate levels in patients with septic shock undergoing extracorporeal membrane oxygenation. J Thorac Dis. 2018;10: 1418-1430.
14. Figueroa Villalba CA, McMullan DM, Reed RC, Chandler WL. Thrombosis in Extracorporeal Membrane Oxygenation (ECMO) Circuits. Asaio j. 2022;68:1083-1092.
15. Smith SA, Travers RJ, Morrissey JH. How it all starts: Initiation of the clotting cascade. Crit Rev Biochem Mol Biol. 2015;50:326-336.
16. Negrier C, Shima M, Hoffman M. The central role of thrombin in bleeding disorders. Blood Rev. 2019;38:100582.
17. Al-Fares A, Pettenuzzo T, Del Sorbo L. Extracorporeal life support and systemic inflammation. Intensive Care Med Exp. 2019;7:46.
18. Plotz FB, van Oeveren W, Bartlett RH, Wildevuur CR. Blood activation during neonatal extracorporeal life support. J Thorac Cardiovasc Surg. 1993;105:823-832.
19. Sniecinski RM, Chandler WL. Activation of the hemostatic system during cardiopulmonary bypass. Anesth Analg. 2011;113:1319-1333.
20. Gorbet MB, Sefton MV. Biomaterial-associated thrombosis: roles of coagulation factors, complement, platelets and leukocytes. Biomaterials. 2004;25: 5681-5703.
21. Doyle AJ, Hunt BJ. Current Understanding of How Extracorporeal Membrane Oxygenators Activate Haemostasis and Other Blood Components. Front Med (Lausanne). 2018;5:352.
22. Chandler WL, Velan T. Secretion of tissue plasminogen activator and plasminogen activator inhibitor 1 during cardiopulmonary bypass. Thromb Res. 2003;112:185-192.
23. Chandler WL, Velan T. Plasmin generation and D-dimer formation during cardiopulmonary bypass. Blood Coagul Fibrinolysis. 2004;15:583-591.
24. Graulich J, Sonntag J, Marcinkowski M, et al. Complement activation by in vivo neonatal and in vitro extracorporeal membrane oxygenation. Mediators Inflamm. 2002;11:69-73.
25. Zhuo R, Siedlecki CA, Vogler EA. Autoactivation of blood factor XII at hydrophilic and hydrophobic surfaces. Biomaterials. 2006;27:4325-4332.
26. Chandler WL. Coagulation activation during extracorporeal membrane oxygenation (ECMO). Thromb Res. 2022;211:154-160.
27. Chandler WL, Velan T. Estimating the rate of thrombin and fibrin generation in vivo during cardiopulmonary bypass. Blood. 2003;101:4355-4362.
28. Hékimian G, Masi P, Lejeune M, et al. Extracorporeal Membrane Oxygenation Induces Early Alterations in Coagulation and Fibrinolysis Profiles in COVID-19 Patients with Acute Respiratory Distress Syndrome. Thromb Haemost. 2021;121: 1031-1042.
29. Meyer AD, Gelfond JA, Wiles AA, Freishtat RJ, Rais-Bahrami K. Platelet-derived microparticles generated by neonatal extracorporeal membrane oxygenation systems. Asaio j. 2015;61:37-42.
30. Meyer AD, Rishmawi AR, Kamucheka R, et al. Effect of blood flow on platelets, leukocytes, and extracellular vesicles in thrombosis of simulated neonatal extracorporeal circulation. J Thromb Haemost. 2020;18:399-410.
31. Helms J, Curtiaud A, Severac F, et al. Fibrinolysis as a Causative Mechanism for Bleeding Complications on Extracorporeal Membrane Oxygenation: A Pilot Observational Prospective Study. Anesthesiology. 2024;141:75-86.
32. Cheung PY, Sawicki G, Salas E, Etches PC, Schulz R, Radomski MW. The mechanisms of platelet dysfunction during extracorporeal membrane oxygenation in critically ill neonates. Crit Care Med. 2000;28:2584-2590.
33. Bleilevens C, Grottke O, Tillmann S, et al. Twelve Hours In Vitro Biocompatibility Testing of Membrane Oxygenators. Asaio j. 2015;61:548-555.
34. Robinson TM, Kickler TS, Walker LK, Ness P, Bell W. Effect of extracorporeal membrane oxygenation on platelets in newborns. Crit Care Med. 1993;21: 1029-1034.
35. Mutlak H, Reyher C, Meybohm P, et al. Multiple electrode aggregometry for the assessment of acquired platelet dysfunctions during extracorporeal circulation. Thorac Cardiovasc Surg. 2015;63:21-27.
36. Siegel PM, Bender I, Chalupsky J, et al. Extracellular Vesicles Are Associated With Outcome in Veno-Arterial Extracorporeal Membrane Oxygenation and Myocardial Infarction. Front Cardiovasc Med. 2021;8:747453.
37. Siegel PM, Hentschel D, Bojti I, et al. Annexin V positive microvesicles are elevated and correlate with flow rate in patients receiving veno-arterial extracorporeal membrane oxygenation. Interact Cardiovasc Thorac Surg. 2020;31:884-891.
38. Karam O, Goel R, Dalton H, Nellis ME. Epidemiology of Hemostatic Transfusions in Children Supported by Extracorporeal Membrane Oxygenation. Crit Care Med. 2020;48:e698-e705.
39. Lou S, MacLaren G, Best D, Delzoppo C, Butt W. Hemolysis in pediatric patients receiving centrifugal-pump extracorporeal membrane oxygenation: prevalence, risk factors, and outcomes. Crit Care Med. 2014;42:1213-1220.
40. Gbadegesin R, Zhao S, Charpie J, Brophy PD, Smoyer WE, Lin JJ. Significance of hemolysis on extracorporeal life support after cardiac surgery in children. Pediatr Nephrol. 2009;24:589-595.
41. Figueroa Villalba CA, Saifee NH, Chandler WL. Red Cell Damage During Extracorporeal Life Support. ASAIO J. 2024.
42. Lutz HU, Bogdanova A. Mechanisms tagging senescent red blood cells for clearance in healthy humans. Front Physiol. 2013;4:387.
43. Dalton HJ, Cashen K, Reeder RW, et al. Hemolysis During Pediatric Extracorporeal Membrane Oxygenation: Associations With Circuitry, Complications, and Mortality. Pediatr Crit Care Med. 2018;19:1067-1076.
44. Olia SE, Maul TM, Antaki JF, Kameneva MV. Mechanical blood trauma in assisted circulation: sublethal RBC damage preceding hemolysis. Int J Artif Organs. 2016;39:150-159.
45. Walsh-Sukys MC, Cornell DJ, Stork EK. The natural history of direct hyperbilirubinemia associated with extracorporeal membrane oxygenation. Am J Dis Child. 1992;146:1176-1180.
46. Kameneva MV, Antaki JF, Borovetz HS, et al. Mechanisms of red blood cell trauma in assisted circulation. Rheologic similarities of red blood cell transformations due to natural aging and mechanical stress. Asaio j. 1995;41:M457-460.
47. Sandza JG, Jr., Clark RE, Weldon CS, Sutera SP. Subhemolytic trauma of erythrocytes: recognition and sequestration by the spleen as a function of shear. Trans Am Soc Artif Intern Organs. 1974;20 b:457-462.
48. Nellis ME, Dalton H, Karam O. Quantifiable Bleeding in Children Supported by Extracorporeal Membrane Oxygenation and Outcome. Crit Care Med. 2019;47:e886-e892.
49. Figueroa Villalba CA, Brogan TV, McMullan DM, Yalon L, Jordan DI, Chandler WL. Conversion From Activated Clotting Time to Anti-Xa Heparin Activity Assay for Heparin Monitoring During Extracorporeal Membrane Oxygenation. Crit Care Med. 2020;48:e1179-e1184.
50. Zhang J. Biomarkers of endothelial activation and dysfunction in cardiovascular diseases. Rev Cardiovasc Med. 2022;23:73.
51. Kappelmayer J, Bernabei A, Edmunds LH, Jr., Edgington TS, Colman RW. Tissue factor is expressed on monocytes during simulated extracorporeal circulation. Circ Res. 1993;72:1075-1081.
52. Fischer M, Sperling C, Tengvall P, Werner C. The ability of surface characteristics of materials to trigger leukocyte tissue factor expression. Biomaterials. 2010;31:2498-2507.
53. Hundalani SG, Nguyen KT, Soundar E, et al. Age-based difference in activation markers of coagulation and fibrinolysis in extracorporeal membrane oxygenation. Pediatr Crit Care Med. 2014;15:e198-205.
54. Hastings SM, Ku DN, Wagoner S, Maher KO, Deshpande S. Sources of Circuit Thrombosis in Pediatric Extracorporeal Membrane Oxygenation. Asaio j. 2017;63:86-92.
55. Lehle K, Philipp A, Zeman F, et al. Technical-Induced Hemolysis in Patients with Respiratory Failure Supported with Veno-Venous ECMO – Prevalence and Risk Factors. PLoS One. 2015;10: e0143527.
56. Hastings SM, Deshpande SR, Wagoner S, Maher K, Ku DN. Thrombosis in centrifugal pumps: location and composition in clinical and in vitro circuits. Int J Artif Organs. 2016;39:200-204.
57. Lehle K, Philipp A, Gleich O, et al. Efficiency in extracorporeal membrane oxygenation-cellular deposits on polymethylpentene membranes increase resistance to blood flow and reduce gas exchange capacity. Asaio j. 2008;54:612-617.
58. Panigada M, L’Acqua C, Passamonti SM, et al. Comparison between clinical indicators of transmembrane oxygenator thrombosis and multidetector computed tomographic analysis. J Crit Care. 2015;30:441.e447-413.
59. Lubnow M, Philipp A, Foltan M, et al. Technical complications during veno-venous extracorporeal membrane oxygenation and their relevance predicting a system-exchange–retrospective analysis of 265 cases. PLoS One. 2014;9:e112316.
60. Wilson JM, Bower LK, Fackler JC, Beals DA, Bergus BO, Kevy SV. Aminocaproic acid decreases the incidence of intracranial hemorrhage and other hemorrhagic complications of ECMO. J Pediatr Surg. 1993;28:536-540; discussion 540-531.
61. Downard CD, Betit P, Chang RW, Garza JJ, Arnold JH, Wilson JM. Impact of AMICAR on hemorrhagic complications of ECMO: a ten-year review. J Pediatr Surg. 2003;38:1212-1216.
62. Stiller B, Lemmer J, Merkle F, et al. Consumption of blood products during mechanical circulatory support in children: comparison between ECMO and a pulsatile ventricular assist device. Intensive Care Med. 2004;30:1814-1820.
63. Henríquez-Henríquez M, Kattan J, Chang M, et al. Blood component usage during extracorporeal membrane oxygenation: experience in 98 patients at a Latin-American tertiary hospital. Int J Artif Organs. 2014;37:233-240.
64. Jackson HT, Oyetunji TA, Thomas A, et al. The
impact of leukoreduced red blood cell transfusion on mortality of neonates undergoing extracorporeal membrane oxygenation. J Surg Res. 2014;192:6-11.
65. Smith A, Hardison D, Bridges B, Pietsch J. Red blood cell transfusion volume and mortality among patients receiving extracorporeal membrane oxygenation. Perfusion. 2013;28:54-60.
66. Cashen K, Dalton H, Reeder RW, et al. Platelet Transfusion Practice and Related Outcomes in Pediatric Extracorporeal Membrane Oxygenation. Pediatr Crit Care Med. 2020;21:178-185.
67. Saifee NH, Brogan TV, McMullan DM, et al. Monitoring Hemostasis During Extracorporeal Life Support. Asaio j. 2020;66:230-237.
68. Yuan S, Ferrell C, Chandler WL. Comparing the prothrombin time INR versus the APTT to evaluate the coagulopathy of acute trauma. Thromb Res. 2007;120:29-37.
69. Shimono T, Makinouchi K, Yada I, Nosé Y. New method of evaluating sublethal damage to erythrocytes by blood pumps. Artif Organs. 1996; 20:568-571.
70. MacLaren G, Monagle P. Platelet Transfusion During Extracorporeal Membrane Oxygenation: Possible Harm, Ongoing Uncertainty. Pediatr Crit Care Med. 2020;21:208-209.
71. Wallisch M, Lorentz CU, Lakshmanan HHS, et al. Antibody inhibition of contact factor XII reduces platelet deposition in a model of extracorporeal membrane oxygenator perfusion in nonhuman primates. Res Pract Thromb Haemost. 2020;4:205-216.

