Pitfalls of Mycotoxin Testing in Clinical Practice
PITFALLS OF MYCOTOXIN TESTING: EVIDENCE-BASED FINDINGS CONFIRM MYCOTOXIN ASSAYS USING ANTIBODY TESTING AND URINARY ELISA ARE BASED ON FLAWED SCIENCE
Shoemaker R¹, McMahon S², Heyman A³, and Lark D⁴
- Progenedx, Pocomoke, MD
- Whole World Health Care, Roswell, NM
- School of Medicine and Health Sciences, George Washington University
- NSJ EnviroSciences Pty Ltd, Newcastle, Australia
OPEN ACCESS
PUBLISHED: 28 February 2026
CITATION: Shoemaker, R., et al., 2026. PITFALLS OF MYCOTOXIN TESTING: EVIDENCE-BASED FINDINGS CONFIRM MYCOTOXIN ASSAYS USING ANTIBODY TESTING AND URINARY ELISA ARE BASED ON FLAWED SCIENCE. Medical Research Archives, [online] 14(2).
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i2.7260
ISSN 2375-1924
ABSTRACT
With the recent focus on diagnosis and treatment in the United States of chronic illness stemming from exposure to the interior of water-damaged buildings, there has been greater use of unsupported laboratory testing. Prior extensive published studies have provided rigorous support for the causation of illness called Chronic Inflammatory Response Syndrome (CIRS). Still, we have not yet seen any studies of exposure to mycotoxins alone that have withstood peer review. The absence of control groups, prospective case-control studies, and placebo-controlled studies makes conclusions of causation impossible. In contrast, such basic studies are well represented in the peer-reviewed literature on CIRS.
Keywords
mycotoxins, CIRS, laboratory testing, chronic illness, water-damaged buildings
Introduction
Mycotoxins represent one of the most ubiquitous and underestimated threats to global food security and human health. These toxic secondary metabolites, produced primarily by fungi of the genera Aspergillus, Fusarium, and Penicillium, contaminate an estimated 25% of cereal crops worldwide, and exposure affects millions of individuals across both industrialized and developing nations. The World Health Organization acknowledges that mycotoxin contamination poses significant public health risks, with documented associations to liver cancer, immunosuppression, growth faltering in children, and neural tube defects.
Despite the well-documented health significance of mycotoxin exposure, several critical but often overlooked problems persist: the methodological inadequacy, analytical unreliability, and clinical misapplication of mycotoxin testing methods. This essay addresses the substantial gap between laboratory-based detection capabilities and clinically meaningful diagnostic interpretation, examining how current testing paradigms frequently generate misleading results that compromise both public health assessment and individual patient management.
Background and Scope of Mycotoxin Exposure
The fundamental challenge lies in the multiplicity and complexity of mycotoxin detection approaches, ranging from chromatographic instrumental methods to immunological assays and emerging biosensor technologies. Each methodology possesses inherent limitations regarding sensitivity, specificity, accuracy, and practical applicability. Moreover, the translation of analytical findings into meaningful health risk assessments remains profoundly hampered by insufficient harmonization of methods, inadequate reference standards, and a persistent lack of evidence linking circulating biomarkers to clinical health outcomes in human populations, with specific corrections to these parameters.
This comprehensive review critically examines the scientific and medical pitfalls endemic to mycotoxin testing, focusing on the gap between flawed methodology and misleading clinical interpretation that currently characterizes much of the field. Claims attributing chronic illness causation to fungal infection and advocating anti-fungal treatment are not supported by current evidence. CIRS is not an infectious disease. Studies have shown that inflammatory, metabolic, and immunologic parameters are present in all cases, and that specific therapies are required to correct the CIRS illness. We know that Actinobacteria are the source of 42% of cases confirmed by meeting the criteria of specific causation. Lipopolysaccharides, endotoxins derived from Gram-negative rods, result in an additional 28% of cases meeting specific causation criteria. Fungi are shown to result in 7% of cases.
As the peer-reviewed literature shows, neurosenescent effects of exposure to CIRS create cognitive impairment similar to Parkinson’s disease and Alzheimer’s. Now that we have the “Triple Positives of Parkinson’s,” a treatable transcriptomic basis for brain injury, it is even more incumbent on treating physicians to stop wasting time on costly but ineffective approaches to chronic illness acquired from exposure to water-damaged buildings. Concerns about the clinical interpretation of exposure biomarkers particularly when assays are promoted for individual diagnosis, treatment decisions, or medico-legal attribution without validated reference intervals or outcome-linked thresholds have also been discussed in the context of chronic inflammatory illness associated with water-damaged buildings and related diagnostic frameworks.
Analytical Methodology Pitfalls
CHROMATOGRAPHIC METHODS: LIMITATIONS DESPITE INSTRUMENTAL SOPHISTICATION
High-performance liquid chromatography combined with mass spectrometry (LC-MS/MS) has emerged as the gold standard for mycotoxin analysis, offering superior selectivity and sensitivity compared to earlier techniques. However, even these sophisticated instrumental approaches contain significant methodological limitations that compromise data reliability. The validation of LC-MS/MS-based methods for multi-mycotoxin determination reveals substantial variability in analytical performance across different food matrices.
A comprehensive analysis of mycotoxin detection methodologies revealed critical challenges with standardization. Among the most significant is the inconsistent recovery across analytes and matrices. In studies examining simultaneous determination of pesticides and mycotoxins, while overall accuracy and precision were satisfactory according to established guidelines, notable disparities emerged in recovery performance. More polar mycotoxins demonstrated matrix-dependent effects, with specific molecules showing 24% signal enhancement in one matrix but 46% signal suppression in another—differences that would lead to inaccurate quantification even with supposedly representative matrix-matched calibration.
The development of dilute-and-shoot approaches, intended to simplify sample preparation while simultaneously analyzing >500 mycotoxin metabolites, exemplifies both the promise and the peril of modern analytical chemistry. While apparent recoveries ranged from 70-120% for 53-83% of analytes, depending on the matrix, this wide variability means that, for nearly half of the analyzed compounds, quantification accuracy may be substantially compromised. The distinction between precision under repeatability conditions (85-97% of analytes meeting RSD criteria) versus intermediate precision (93-94%) highlights the substantial contribution of relative matrix effects to overall analytical uncertainty.
IMMUNOASSAY APPROACHES: PROBLEMATIC CROSS-REACTIVITY AND SPECIFICITY ISSUES
Enzyme-linked immunosorbent assay (ELISA) and lateral flow immunoassays have been promoted as practical alternatives to chromatographic methods, particularly for field-based or rapid screening applications. These methods offer apparent advantages of speed, portability, and reduced technical complexity. However, critical analytical limitations substantially undermine their reliability for diagnostic purposes.
Cross-reactivity with non-target mycotoxins and related compounds represents a persistent and poorly characterized problem in immunoassay-based detection. A recent validation study of lateral flow devices for total aflatoxin screening identified concerning limitations: a direct competitive ELISA for multiple mycotoxins showed monoclonal antibody specificity ranging from 2% to 11%. While manufacturers frame this as acceptable “specificity,” from a clinical diagnostic perspective, cross-reactivity rates of this magnitude are highly problematic. They can lead to false-positive results that mislead clinical decision-making.
The development of a validated direct competitive ELISA for the simultaneous detection of aflatoxin B1, deoxynivalenol, fumonisin, ochratoxin A, and zearalenone in human serum demonstrated lower limits of quantitation (LLOQ) of 0.15 ppb for zearalenone and up to 19.53 ppb for the other mycotoxins. However, recovery from human serum ranged from 73% to 106%, indicating that for a substantial proportion of samples, quantification uncertainty could exceed ±27%, a margin unacceptable for individual clinical diagnostics.
Importantly, commercial ELISA kits for mycotoxin detection exhibit significant inter-kit and between-manufacturer variability. Studies examining the robustness of lateral flow immunoassays under harsh environmental conditions reported that, while agreement between rapid strip tests and validated LC-MS/MS reference methods reached 86.4%, this still left nearly 14% of results discordant, with 1.8% yielding invalid results. Such discordance rates render these tests unsuitable as definitive diagnostic tools for individual clinical decision-making, though they may have limited utility in preliminary screening contexts.
BIOSENSOR AND EMERGING TECHNOLOGIES: PROMISES BUT UNMET VALIDATION REQUIREMENTS
Emerging biosensor technologies, including surface-enhanced Raman spectroscopy (SERS), electrochemical immunosensors, and aptamer-based systems, have been proposed as innovative alternatives offering improved sensitivity and potentially real-time detection. However, critical validation gaps substantially limit clinical applicability.
Surface-enhanced Raman spectroscopy (SERS), despite theoretical advantages, faces significant practical limitations. A recent comprehensive review documented that although SERS has demonstrated detection limits as low as 1.3 pg/mL for cyclopiazonic acid, limited studies establish detection limits using naturally contaminated samples. The disconnect between laboratory performance and field applicability remains substantial. Moreover, SERS has made little progress toward integration with European regulatory limits and remains largely inaccessible to end users in resource-constrained settings.
Electrochemical aptasensors and point-of-care testing devices show promise but remain in early development, with substantial validation gaps. A portable, integrated, duplex, label-free electrochemical immunosensor demonstrated excellent performance in controlled conditions, achieving detection limits of 7.1-4.0 pg/mL for zearalenone and deoxynivalenol, respectively. However, such studies typically involve small cohorts of clinical samples and often lack independent external validation in independent populations—a critical requirement for diagnostic test qualification.
Matrix Effects, Cross-Reactivity, Standardization Failures in Laboratory Analysis
MATRIX-DEPENDENT ANALYTICAL PERFORMANCE VARIABILITY
One of the most poorly appreciated yet critically important limitations of mycotoxin testing is the phenomenon of matrix effects—the alteration of analyte response by co-extracted components in complex biological or food samples. Matrix effects are not merely technical nuisances but rather fundamentally compromise the validity of quantification across different sample types.
Studies comparing analytical performance across multiple food matrices revealed that recoveries and matrix effects were constant over 23 orders of magnitude of analyte concentration in only some matrices (figs and maize). In contrast, limits of quantification varied by 2-90 for 90% of the investigated compounds. This variability means that limits of quantification established in one matrix do not reliably predict analytical performance in another, necessitating matrix-specific validation that is often not conducted in routine laboratory practice.
Ionization suppression or enhancement in mass spectrometry-based methods can alter measured analyte concentrations by 30% or more, depending on co-eluting substances. For example, in the analysis of mycotoxins in medicinal herbs, matrix effects resulted in signal enhancement of 24% in one herb species (Melissa officinalis) but 46% signal suppression in another (Malva sylvestris), indicating that matrix-matched calibration using a single herb matrix yields systematic quantification errors in the analysis of the other.
LACK OF HARMONIZED STANDARDS AND REFERENCE MATERIALS
The absence of internationally harmonized analytical standards and certified reference materials for mycotoxin analysis represents a systemic failure that undermines comparability across laboratories and over time. A survey of 19 control, commercial, and research laboratories from 12 countries participating in proficiency testing revealed substantial variability in analytical capabilities and methods.
Critically, definitions of the limits of detection (LOD) and limits of quantification (LOQ) were not harmonized across laboratories. The signal-to-noise ratio criterion, used by 40% of laboratories, represents only one of multiple possible definitions, leading to systematic non-comparability of results between laboratories. This fundamental definitional inconsistency means that an LOQ reported by one laboratory is not directly comparable to that reported by another, rendering comparative risk assessment across populations or time periods unreliable.
STANDARDIZATION FAILURES AND VALIDATION INADEQUACIES
Despite the existence of Commission Regulation 401/2006 in the European Union, which specifies analytical acceptance criteria for mycotoxin testing, substantial variability persists in actual laboratory practice. The use of mycotoxin methodology in practice demonstrates a need for systematic harmonization, particularly in sample preparation, cleanup procedures, and quality control.
A critical issue is the lack of standardized approaches for handling masked or modified mycotoxins—metabolites conjugated by plants or animals that conventional methods may fail to detect. The current systematic definition acknowledges four hierarchical levels of mycotoxin modification, yet most routine laboratories lack adequate procedures to detect these forms, leading to systematic underestimation of total human exposure.
Human Biomonitoring Challenges: From Sample Collection to Interpretation
BIOMAKER SELECTION AND TOXICOKINETIC UNCERTAINTIES
Human biomonitoring of mycotoxin exposure through analysis of blood, plasma, serum, or urine represents an appealing approach to assess individual-level exposure, theoretically circumventing limitations of food consumption data. However, this strategy encounters substantial methodological pitfalls that limit its clinical utility.
The selection of appropriate biomarkers involves complex toxicokinetic considerations that are incompletely characterized by most mycotoxins. While aflatoxin B1-lysine (AFB1-lys) and ochratoxin A (OTA) in plasma are among the most widely studied biomarkers, substantial uncertainties remain regarding their quantitative relationships to external dose, inter-individual variability in toxicokinetics, and the proportion of exposure reflected in circulating biomarkers. For many mycotoxins, toxicokinetic parameters including clearance rates, transfer rates to urine or milk, and tissue distribution remain poorly characterized in humans.
A large human biomonitoring study of pregnant women in The Netherlands illustrates both the prevalence and the interpretive limitations of urinary mycotoxin measurements. In that cohort, total deoxynivalenol, total zearalenone, and ochratoxin A were among the most frequently detected analytes, with high detection frequencies reported across samples.
However, frequent detection at the population level does not equate to a quantitative assessment of individual exposure or internal biologically effective dose. Variability in absorption, metabolism, excretion, and timing of sample collection relative to exposure introduces substantial uncertainty, such that measured urinary concentrations cannot be reliably translated into external intake, tissue burden, or health risk for individual subjects. These limitations constrain the clinical interpretability of such measurements, particularly when applied outside controlled epidemiological contexts.
CRITICAL GAPS IN REFERENCE DATA AND HEALTH-BASED GUIDANCE VALUES
A major limitation of human biomonitoring for mycotoxin exposure assessment involves the near-complete absence of established reference ranges for general populations or at-risk subgroups. Unlike well-characterized biomarkers for environmental contaminants such as lead or mercury, mycotoxin biomarker concentrations lack established clinical decision limits or guidance values that would distinguish between “normal” background exposure and levels associated with health concern.
The challenges in establishing such guidance values are substantial. First, the relationship between biomarker concentrations and actual health outcomes has been established for only a few mycotoxins in limited populations. While aflatoxin B1-albumin adducts have been linked to hepatocellular carcinoma risk through epidemiological studies, comparable evidence for most other mycotoxins and their biomarkers is lacking.
Second, inter-individual variability in toxicokinetics, influenced by genetic polymorphisms, nutritional status, age, and concurrent exposures, results in wide ranges of biomarker concentrations for identical external doses.
METHODOLOGICAL ISSUES IN SAMPLE COLLECTION AND PRE-ANALYTICAL HANDLING
Fundamental pre-analytical errors substantially compromise the validity of human biomonitoring results. Decisions regarding sample type (spot versus 24-hour urine, plasma versus serum), collection protocols, stabilization procedures, and storage conditions profoundly affect measurable mycotoxin concentrations, yet standardized approaches remain unavailable.
For urinary sampling, the choice between spot urine and 24-hour collection introduces substantial variability. Spot urine samples reflect only the mycotoxin concentrations at a specific time, potentially missing transient peaks in urinary excretion. Moreover, different studies employ variable creatinine-based adjustments to account for urine dilution, thereby introducing additional sources of analytical variability. The metabolic fate of major mycotoxins and their biomarkers—including reversible protein binding, enterohepatic circulation, and time-dependent biotransformation—means that the timing of sampling relative to exposure profoundly affects measured biomarker concentrations, a phenomenon that is incompletely characterized for most mycotoxins.
Clinical Applications and Diagnostic Accuracy: The Gap Between Laboratory And Clinical Medicine
LIMITED CLINICAL VALIDATION AND DIAGNOSTIC UTILITY
Despite extensive development of mycotoxin detection methods, substantial gaps persist between analytical capability and clinical diagnostic utility. Most mycotoxin testing methods have been analytically validated (i.e., demonstrated to measure mycotoxin concentrations in controlled conditions reliably) but lack clinical validation (i.e., demonstrations that test results meaningfully predict health status or guide clinical decision-making).
A systematic review of diagnostic biomarker translation revealed that of 107 molecular- or “omics”-based diagnostic tests described in 2006, only 28 (26.2%) appeared to have made progress toward clinical application by 2016. This abysmal translation rate, combined with the recognition that many biomarker discovery studies suffer from substantial methodological bias, raises serious concerns about premature clinical application of mycotoxin biomarkers.
CHALLENGES IN ESTABLISHING RISK ASSESSMENT ENDPOINTS
Risk assessment frameworks for mycotoxin exposure have historically relied on hazard quotients or margins of exposure, based primarily on animal toxicology data rather than human epidemiological evidence. While animal studies have established that mycotoxins cause hepatotoxicity, nephrotoxicity, immunosuppression, and genotoxic/carcinogenic effects, translating animal-derived no-observed-adverse-effect levels (NOAELs) into human reference doses involves multiple assumptions that may not be valid.
Assessment of multiple mycotoxin exposure and cumulative health risk assessment further complicates risk characterization. Most regulatory frameworks address individual mycotoxins in isolation, despite epidemiological evidence that co-occurrence of multiple mycotoxins is common. A comprehensive exposure assessment study in the Yangtze River Delta identified multiple mycotoxins co-occurring in 22.47% of urine samples, with deoxynivalenol, fumonisin B1, and zearalenone being the most frequent. However, toxicological data on the interactive effects of multiple mycotoxins remain limited, and risk assessment frameworks for cumulative exposure are largely unavailable.
CLINICAL INTERPRETATION AND THE PROBLEM OF ASYMPTOMATIC BIOMAKER POSITIVITY
A critical but under-recognized problem in mycotoxin testing is the interpretation of positive biomarkers results in asymptomatic individuals. Detection of mycotoxins or their metabolites in biological samples does not automatically indicate clinically significant exposure or present health risk. Yet commercial mycotoxin testing laboratories frequently report positive results with implications suggesting clinical concern, without adequate context regarding the relationship between biomarker presence and actual health impact.
For example, detection of ochratoxin A in human urine samples is nearly universal in some populations, with 95% of pregnant women in a rural Bangladesh cohort testing positive for OTA. Yet the presence of detectable OTA does not indicate overt kidney disease or measurable clinical pathology in most individuals. The distinction between background environmental exposure and toxicologically significant exposure is poorly characterized by most mycotoxins.
Regulatory Framework Inconsistencies And Health Implications
JURISDICTIONAL VARIABILITY IN REGULATORY LIMITS AND ENFORCEMENT
Substantial inconsistencies in regulatory limits for mycotoxins across different jurisdictions undermine both scientific credibility and public health protection. While the European Union has established maximum permitted levels for major mycotoxins in foods and animal feeds, regulatory limits in many developing nations remain either absent or inconsistently enforced.
This regulatory fragmentation creates economic and health disparities, particularly affecting populations in tropical and subtropical developing nations, where climate conditions favor mycotoxin production. Moreover, the scientific basis for regulatory limits frequently reflects expert judgment regarding acceptable risk rather than robust epidemiological data establishing health-protective thresholds.
GAPS BETWEEN REGULATORY STANDARDS AND ACTUAL LABORATORY PRACTICE
Even within regulatory jurisdictions with established testing requirements, substantial variability persists between official regulatory standards and actual laboratory implementation. Proficiency testing programs, intended to ensure analytical comparability, reveal that many laboratories fall substantially short of meeting acceptance criteria established in Commission Regulation 401/2006.
The regulatory acceptance of multiple methodological approaches (chromatographic, immunological, biosensor-based) without robust comparative validation means that identical food samples may yield substantially different quantified mycotoxin concentrations depending on the laboratory and methodology employed. This lack of methodological standardization renders regulatory compliance determinations potentially unreliable.
EVIDENCE GAPS IN CLINICAL APPLICATION OF MYCOTOXIN TESTING
Perhaps most critically, the regulatory and clinical frameworks governing mycotoxin testing in human clinical medicine remain poorly developed compared to established medical diagnostics. Most mycotoxin testing offered in clinical settings lacks FDA or CE-IVD approval, operates in a largely unregulated space, and often fails to meet the analytical and clinical validation standards expected for other medical diagnostic tests.
Commercial laboratories offering mycotoxin biomarker testing to individual patients frequently lack transparent documentation of analytical validation, quality assurance procedures, or clinical evidence supporting their use for individual health assessment. The proliferation of poorly validated commercial mycotoxin testing represents a substantial public health concern, generating potentially misleading clinical information that may drive unnecessary interventions or create unwarranted health anxiety.
Evidence-based medicine (EBM) provides the framework for evaluating the suitability of laboratory tests for clinical use. Within this framework, diagnostic assays must satisfy three sequential criteria: analytical validity, clinical validity, and clinical utility before they can be relied upon to inform diagnosis or guide treatment.
While many mycotoxin detection methods demonstrate analytical capability under controlled laboratory conditions, analytical performance alone is insufficient for clinical application. Clinical validity requires a reproducible and meaningful association between test results and defined disease states or health outcomes in human populations. For most mycotoxin assays currently offered in clinical settings, such associations have not been established.
Critically, validated population reference intervals—an essential prerequisite for clinical interpretation—are lacking for most mycotoxin biomarkers measured in blood or urine. In the absence of reference distributions derived from appropriately characterized populations, numerical test results cannot reliably distinguish background environmental exposure from abnormal or clinically relevant findings.
Clinical utility further requires evidence that test-directed interventions improve patient outcomes. At present, no prospective clinical studies demonstrate that interventions guided by antibody- or biomarker-based mycotoxin testing improve health outcomes. In this context, the routine clinical use of such tests exceeds the available evidence base.
Case Example (Illustrative, Not Diagnostic)
These considerations are not merely theoretical. Failure to adhere to EBM principles risks misinterpretation of laboratory findings, inappropriate clinical inference, and unnecessary or misguided interventions. The following case example illustrates how such departures from established diagnostic standards can occur in practice.
Case Example: Misinterpretation of IgG Antibodies to Small-Molecule Mycotoxins
A practical illustration of evidence-based medicine principles is provided by the interpretation of IgG antibodies to small-molecule mycotoxins. In some commercial contexts, detection of IgG antibodies has been presented as evidence of current or ongoing exposure. This interpretation is not supported by established immunological or laboratory principles.
Mycotoxins are low-molecular-weight secondary metabolites that function as haptens rather than complete antigens. Table 1 summarizes representative mycotoxins, their molecular weights, and the immunological implications for IgG antibody-based analysis.
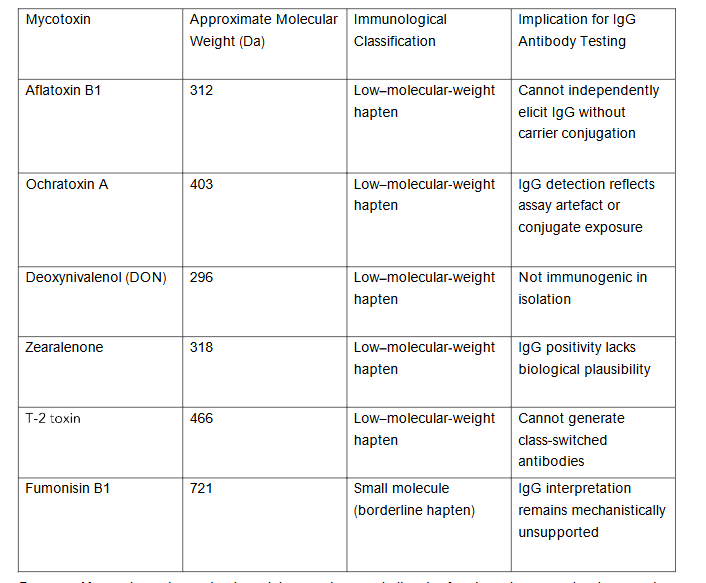
Footnote: Mycotoxins are low-molecular-weight secondary metabolites that function as haptens rather than complete antigens. Without covalent conjugation to a carrier protein, such compounds cannot elicit a class-switched IgG immune response. Accordingly, detection of IgG antibodies directed against isolated mycotoxins lacks an established immunological basis and does not meet criteria for analytical or clinical validity under evidence-based medicine frameworks. IgG antibodies are markers of acquired immunological memory and persistence, not of recent exposure. Interpreting IgG responses as indicators of current exposure departs from established immunological understanding and risks temporal misclassification.
From an analytical perspective, antibody assays targeting small molecules are inherently susceptible to cross-reactivity and non-specific binding, particularly in complex biological matrices such as serum or urine. These limitations further constrain interpretive reliability.
In the absence of validated population reference intervals or outcome-linked thresholds, results of such analyses cannot be reliably interpreted or used to guide clinical decision-making.
Conclusion
The comprehensive examination of mycotoxin testing methodologies reveals that substantial gaps persist between analytical capability and clinically meaningful application. While analytical techniques have become increasingly sophisticated, translating analytical findings into reliable health risk assessments remains profoundly limited by methodological pitfalls, inadequate standardization, incomplete toxicokinetic characterization, and the absence of robust clinical validation.
The field of mycotoxin testing exemplifies a broader phenomenon in modern medicine wherein technological sophistication in measurement has outpaced the development of evidence-based frameworks for clinical interpretation and application. Advanced chromatographic and immunological methods can reliably measure mycotoxin concentrations at extraordinarily low levels, yet the clinical significance of these measurements remains unclear for most mycotoxins and most individuals.
Substantial research and standardization efforts are urgently required to advance the field toward evidence-based practice. These efforts must prioritize:
- international harmonization of analytical standards and reference materials.
- comprehensive characterization of mycotoxin toxicokinetics and biomarker validity in human populations.
- establishment of health-based guidance values for mycotoxin biomarkers based on epidemiological rather than solely animal toxicology data.
- rigorous clinical validation studies demonstrating that mycotoxin biomarker testing meaningfully improves clinical decision-making; and
- establishment of regulatory frameworks governing the clinical application of mycotoxin testing comparable to those applied to other medical diagnostics.
Until such evidence-based advances are implemented, mycotoxin testing should be interpreted cautiously in clinical contexts, and practitioners should exercise substantial skepticism regarding commercial mycotoxin testing that claims to establish clinically significant exposure or to guide individual health interventions. Preventing mycotoxin contamination of food through comprehensive agricultural, storage, and processing interventions remains the most evidence-supported approach to reduce population-level mycotoxin exposure and its associated health consequences.
References
- Alshannaq AF, Yu JH. Occurrence, Toxicity, and Analysis of Major Mycotoxins in Food. International Journal of Environmental Research and Public Health. 2017;14(6):632.
- Turner PC, Flannery BM, Isitt C, Ali M, Pestka JJ. The role of biomarkers in evaluating human health concerns and fungal contaminants in food. Nutrition Reviews. 2012;70(6):1-13.
- Tittlemier S, Cramer B, DeRosa MC, et al. Developments in analytical techniques for mycotoxin determination: an update for 2023-24. World Mycotoxin Journal. 2025;18(1):71-97.
- Shoemaker RC. Urinary mycotoxins: A review of contaminated buildings and food in search of a biomarker separating sick patients from controls. Intern Med Rev. 2019;5(6):1-35.
- Shoemaker R, House DE. A time-series study of sick building syndrome: chronic, biotoxin-associated illness from exposure to water-damaged buildings. Neurotoxicol Teratol. 2005; 27:29-46.
- Shoemaker R, House DE. Sick building syndrome and exposure to water-damaged buildings: time series study, clinical trial and mechanisms. Neurotoxicol Teratol. 2006; 28:573-588.
- Shoemaker R, Lin K. Inside indoor air quality: Environmental Relative Moldiness Index (ERMI). Filtration News. 2007; 32:36.
- Shoemaker R, House D, Ryan J. Vasoactive intestinal polypeptide corrects chronic inflammatory response syndrome acquired following exposure to water-damaged buildings. Health. 2013;5(3):396-401.
- Shoemaker R, House D, Ryan J. Structural brain abnormalities in patients with inflammatory illness acquired following exposure to water-damaged buildings: a volumetric MRI study using NeuroQuant®. Neurotoxicol Teratol. 2014; 45:18-26.
- Berndtson K, McMahon S, Ackerley M, Rapaport S, Gupta S, Shoemaker RC. Consensus Statement, Part 1: Medically sound investigation and remediation of water-damaged buildings in cases of CIRS-WDB. Center for Research on Biotoxin-Associated Illness; Pocomoke, MD. 2016.
- Ryan C, Shoemaker RC. RNA-Seq on patients with chronic inflammatory response syndrome treated with vasoactive intestinal peptide shows a shift in metabolic state and innate immune functions that coincide with healing. Med Res Arch. 2016;4(7).
- Shoemaker R, Katz D, Ackerley M, Rapaport S, McMahon SW, Berndtson K, Ryan JC. Intranasal VIP safely restores volume to multiple gray matter nuclei in patients with chronic inflammatory response syndrome. Intern Med Rev. 2017:1-14.
- Shoemaker R, Heyman A, Lark D. Transcriptomics and brain volumetrics define the causes of cognitive impairment in patients with chronic inflammatory response syndrome and support the use of VIP in treatment. Med Res Arch. 2023;11(3).
- Singh J, Mehta A. Rapid and sensitive detection of mycotoxins by advanced and emerging analytical methods: A review. Food Science & Nutrition. 2020;8(3):1474-1499.
- Sulyok M, Stadler D, Steiner DJ, Krska R. Validation of an LC-MS/MS-based dilute-and-shoot approach for the quantification of >500 mycotoxins and other secondary metabolites in food crops. Analytical and Bioanalytical Chemistry. 2020;412:2489-2509.
- Fontana MEZ, da Silva RC, dos Santos ID, et al. Comprehensive assessment of clean-up strategies for optimizing an analytical multi-method to determine pesticides and mycotoxins in Brazilian medicinal herbs using QuEChERS-LC-TQ-MS/MS. Analytical Methods. 2024;16(31):2921-2940.
- Garg K, Villavicencio-Aguilar F, Solano-Rivera F, et al. Analytical Validation of a Direct Competitive ELISA for Multiple Mycotoxin Detection in Human Serum. Toxins. 2022;14(11):727.
- Cvak B, Warth B, Atehnkeng J, et al. Evaluating the Performance of Lateral Flow Devices for Total Aflatoxins with Special Emphasis on Their Robustness under Sub-Saharan Conditions. Toxins. 2021;13(11):742.
- Logan N, Cao C, Freitag S, et al. Advancing Mycotoxin Detection in Food and Feed: Novel Insights from Surface-Enhanced Raman Spectroscopy (SERS). Advanced Materials. 2024;36(3):2309625.
- Veenuttranon K, Lu X, Geng N, Zhang H, Chen J. A Portable Integrated Duplex Label-Free Electrochemical Immunosensor Constructed from a Nanozyme-Tuned Metal-Organic Framework for Dual Mycotoxin Detection. Analytical Chemistry. 2025;97(29):14209-14225.
- Cortese M, Gigliobianco MR, Magnoni F, et al. Compensate for or Minimize Matrix Effects? Strategies for Overcoming Matrix Effects in Liquid Chromatography-Mass Spectrometry Technique. Molecules. 2020;25(13):3047.
- Solfrizzo M, Alldrick AJ, van Egmond HP. The use of mycotoxin methodology in practice: a need for harmonization. World Mycotoxin Journal. 2009;2(3):1-10.
- M. Rychlik et al., “Proposal of a comprehensive definition of modified and other forms of mycotoxins including masked mycotoxins,” Springer Science Business Media, Jun. 2014, doi: https://doi.org/10.1007/s12550-014-0203-5.
- Arce-López B, Lizarraga E, Vettorazzi A, González-Peñas E. Human Biomonitoring of Mycotoxins in Blood, Plasma and Serum in Recent Years: A Review. Toxins. 2020;12(3):147.
- McKeon HP, Schepens MAA, van den Brand A, et al. Assessment of Mycotoxin Exposure and Associated Risk in Pregnant Dutch Women: The Human Biomonitoring Approach. Toxins. 2024;16(6):278.
- Cramer B, Visintin L, Maris E, Kuhn M, Degen GH, Turner PC, Humpf HU, De Saeger S. Human biomonitoring of mycotoxins: key challenges and future directions. Mycotoxin Res. 2025 Dec 23;42(1):13.
- Kensler TW, Roebuck BD, Wogan GN, Groopman JD. Aflatoxin: A 50-Year Odyssey of Mechanistic and Translational Toxicology. Toxicological Sciences. 2010;120(1): S28-S48.
- E. D. P. Nunes, B. A. D. Pestana, and B. B. B. Pereira, “Human biomonitoring and environmental health: a critical review of global exposure patterns, methodological challenges and research gaps,” Journal of Toxicology and Environmental Health. Part B, Critical Reviews, Jul. 2025, doi: 10.1080/10937404.2025.2529845.
- Parker LA, Chilet-Rosell E, Hernández-Aguado I, et al. Diagnostic Biomarkers: Are We Moving from Discovery to Clinical Application? Clinical Chemistry. 2018;64(11):2854.
- Huang Q, Jiang K, Tang Z, et al. Exposure Assessment of Multiple Mycotoxins and Cumulative Health Risk Assessment: A Biomonitoring-Based Study in the Yangtze River Delta, China. Toxins. 2021;13(2):103.
- N. Kyei, B. Cramer, H. Humpf, G. Degen, N. Ali, and S. Gabrysch, “Assessment of multiple mycotoxin exposure and its association with food consumption: a human biomonitoring study in a pregnant cohort in rural Bangladesh,” Archives of Toxicology, Apr. 2022, doi: 10.1007/s00204-022-03288-0.
- Benkerroum N. Retrospective and Prospective Look at Aflatoxin Research and Development from a Practical Standpoint. International Journal of Environmental Research and Public Health. 2019;16(19):3633.
- Sackett DL, Rosenberg WMC, Gray JAM, Haynes RB, Richardson WS. Evidence-based medicine: what it is and what it is not. BMJ. 1996;312(7023):71-72.
- Guyatt G, Rennie D, Meade MO, Cook DJ. Users’ Guides to the Medical Literature: A Manual for Evidence-Based Clinical Practice. 2nd ed. AMA Press; 2008.
- Bossuyt PM, Reitsma JB, Bruns DE, et al. Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. BMJ. 2003;327(7413):41-44.
- Clinical and Laboratory Standards Institute (CLSI). Defining, Establishing, and Verifying Reference Intervals in the Clinical Laboratory; Approved Guideline. CLSI document C28-A3. Wayne, PA; 2008.
- Pepe MS, Étions R, Feng Z, et al. Phases of biomarker development for early detection of disease. Biometrics. 2001;57(2):345-352.




