Faricimab’s Role in Diabetic Macular Edema Treatment
Revisiting Faricimab and Earlier Randomized Controlled Trials for Diabetic Macular Edema in Light of Newly Defined Pathogeneses
Avinoam Ophir, MD1
- Ophthalmology Clinic.
Correspondence: [email protected]
OPEN ACCESS
PUBLISHED: 28 February 2026
CITATION: Ophir, A., 2026. Revisiting Faricimab and Earlier Randomized Controlled Trials for Diabetic Macular Edema in Light of Newly Defined Pathogeneses. Medical Research Archives, [online] 14(2).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
The primary aim of therapy for center-involved diabetic macular edema (DME) is to achieve a long-lasting dry macula. High therapeutic efficacy correlates with targeting the underlying pathogenesis. Recent discoveries have identified two additional tractional pathogeneses that are undetectable by standard optical coherence tomography (OCT). These indicated that all “diffuse” DME in non-ischemic maculae is tractional in origin. Consequently, DME may be classified by pathogenesis as tractional DME and microaneurysm-related DME.
Faricimab (Vabysmo) inhibits vascular endothelial growth factor-A and angiopoietin-2, and is highly effective in abolishing leaking microaneurysms, altering their turnover. In the pivotal YOSEMITE and RHINE trials, two outcome assessments were reported. The first applied a numerical definition in which central subfield thickness <325µm was considered “no DME” (“dry” macula), irrespective of OCT anatomy. Using <325µm and visual criteria, injection intervals were extended to 12 and 16 weeks, and <325µm was found in ~83% of eyes over the 2-year trial. The second assessment was anatomical: at year-1 and -2, approximately half, ~40% and 47%, respectively, achieved ≥8 weeks of dry, fluid-free maculae. Hence, injection intervals were extended in 43% (83%-40%) and 36% of eyes, respectively, with edematous maculae, instead of prompt treatment.
Uniquely, analysis revealed a sharp clinical separation between two response patterns: (1) anatomical drying for ≥8 weeks, consistent with faricimab’s effect in microaneurysm-related DME, and (2) partial or absent response, consistent with tractional DME. Had DME been stratified before treatment and faricimab used only for microaneurysm-related disease, dryness could likely have been achieved for ≥8 weeks, while futile injections could have been avoided in tractional DME. Concerns regarding outcomes following missed stratification might also be relevant, in retrospect, to most earlier trials and real-world practice.
In conclusion, the well-defined clinical separation between the two DME pathogeneses supports identical earlier claims, and seems a landmark moment for DME therapy after decades. It follows the conduct: Never mix two different items for calculations. After excluding microaneurysm-related DME, the benchmarks for long-lasting resolution remain pars plana vitrectomy in treatment-naïve eyes or following failed grid laser photocoagulation; grid laser photocoagulation is efficacious in specific early conditions. Aligning future trials and clinical practice with pathogenetic enrollment, consistent with the Declaration of Helsinki, and anatomy-based endpoints is essential to move toward an effective, potentially curative therapy.
Keywords
Dry diabetic macular edema; Curative DME. Faricimab; Microaneurysm-related DME; Tractional DME; Diffuse DME; Focal DME; DME pathogenesis; DME classifications; Extrafoveal traction; 3D-OCT; Transitional-phase DME; Anti-VEGF.
Introduction
The aim of diabetic macular edema (DME) therapy is to achieve long-lasting dry maculae. The pivotal faricimab (6 mg; Vabysmo®, Genentech/Roche) randomized controlled trials (RCTs) YOSEMITE (n=940) and RHINE (n=951), conducted across 353 centers worldwide, were designed to treat DME efficaciously while reducing frequent intravitreal injections.
Faricimab inhibits vascular endothelial growth factor (VEGF)-A and angiopoietin-2 (Ang-2).
Ang-2 activity is enhanced in inflammatory milieus, promoting pericyte loss and endothelial junction destabilization ensuing microaneurysm (MA) formation, and amplifies VEGF responsiveness. Faricimab also abolishes leaking MAs and reduces new MA formation in MA-related (“focal”) DME (MAs-DME).
Achieving long-lasting dry maculae
Clinically meaningful RCT conclusions require alignment between study design and disease pathogenesis. When this is absent, management tends to default to a trial-and-error (T&Er) approach with reduced efficacy, repeated interventions [intravitreal medications and/or modified grid laser photocoagulation (GLP)], and cumulative long-term burden. In the two current trials, treatment was conducted by T&Er, as in earlier DME RCTs, merging MAs-DME with “diffuse” DME of previously unfamiliar pathogenesis into one group, “DME”.
Publications in recent years have proved that all “diffuse” DME in non-ischemic maculae are tractional in essence. These advances included the discovery of two tractional pathogeneses, both undetectable on standard OCT, in addition to vitreofoveal traction and epimacular membrane (ERM): (A) extrafoveal vitreoretinal traction (2010), emerging anywhere within the area centralis and best detected by three-dimensional spectral-domain OCT, and vitreopapillary traction, which is often under-judged; and (B) transitional-phase DME, the earliest build-up of a vitreo-tractional process, demonstrable only ultrastructurally following pars plana vitrectomy (PPV) and internal limiting membrane (ILM) peeling, whether traction is OCT-detectable or not. This process was described by Gandorfer et al. (2005), with further contributions by Hagenau et al. (2019). We presume that progressive tangential thickening of the membranous layer on top of the ILM during this phase might sometimes emerge as a tractional taut posterior hyaloid. However, the transitional-phase innovation did not attract broad clinical attention in the ophthalmic community until recently. After excluding MAs-DME and macular ischemia using fluorescein (or OCT) angiography, the researchers advocated PPV in all cases of DME in which traction is undetected. By contrast, we advised to initially look for extrafoveal traction and, if excluded, recommending GLP as first-line therapy.
Clinically, long-lasting macular dryness was achieved in 92–100% of treatment-naïve eyes (n=7–120; 6–24 months) with “diffuse” DME by early PPV with ILM peeling in eyes without OCT-visible traction, and often in >80% after failed GLP. Surgical efficacy, however, seems degraded after failed anti-VEGF therapy. The very high surgical benefit could also reflect an accompanying MAs-DME component, when present, potentially mediated by improved retinal oxygenation. In Kumagai’s study (n=157) of PPV in DME without detected traction (pre-OCT era), a visual acuity (VA) gain of ~10 letters was maintained throughout the 10-year study. In the only GLP study (n=18) after excluding extrafoveal traction, durable macular drying was achieved in 13 (72%) eyes over a mean of 15.9 (4–24) months, with mid-term emergence of tractional membranes in 4 of the 5 failing GLPs, and non-tractional adherent vitreoretinal membranes in two eyes. This concept of pathogenetic-related early surgical intervention was largely overlooked and uncited in the various RCTs, despite evidence that a single early procedure can often achieve long-term anatomical macular dryness.
Due to the common difficulties and confusion in differentiating “focal” and “diffuse” DME, the cumulative data on tractional DME and its exceptional outcomes following PPV claim for a novel, practical, pathogenesis-related classification of DME in non-ischemic maculae: MAs-DME and tractional DME.
Consequences of merging pathogeneses
Most prior DME trials and real-world studies, involving anti-VEGF agents and their biosimilars, steroids, and GLP, conducted by T&Er, typically merged MAs-DME with an enigmatic pathogenesis into a single cohort labeled “DME”, as discussed. This pathogenesis was later proved to be tractional DME. The merger resulted in identical treatment recommendations for two biologically distinct entities, fostering flawed extrapolation to further studies and clinical practice. No trial achieved long-lasting dry maculae. Rather, this unification obscured pathogenetic differences and the probability to reveal the causes of high efficacy in one subgroup versus failure in the other. Consequently, real-world practice thereafter faces substantial burden, cost, and adherence challenges, which may result in incomplete macular drying, chronic edema, progressive foveal injury, and deterioration of visual acuity (VA).
This merged, “one-basket” analytical tradition is not new. Early RCTs initially reported on “focal” and “diffuse” DME separately, undergoing different laser treatments, focal or focal/GLP, but then merged them into a single “DME” cohort for calculations and recommendations. The RESTORE trial, for example, diagnosed ~50% of eyes as “focal” (MAs-) DME. Over time, this unification became routine across trials and clinical practice. The identical conclusions and treatment recommendations issued for two biologically distinct forms of DME, MAs-DME and tractional DME, indicate that the trial strategy was fundamentally misaligned. Furthermore, in the current trials, threshold-based CST definitions can create an illusion of disease control by masking persistent anatomical edema within mean values, thereby encouraging inappropriate treatment extension and its edema-related consequences, including in real-world practice. Merging distinct pathogeneses also masks true treatment success in the MAs-DME subgroup. After achieving clinical separation, it appears that faricimab may be efficacious in MAs-DME for several months, though further direct and comparative studies are required.
Anatomical vs. numeric criteria of dry macula
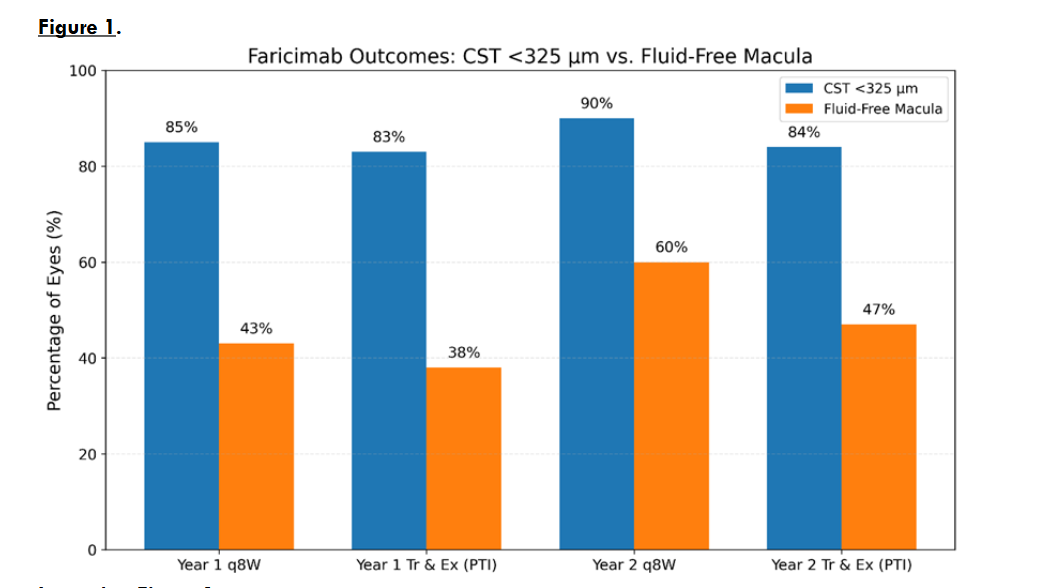
Moreover, the faricimab trials introduced, for the first time in DME, a numerical threshold as a surrogate for the anatomical OCT criterion of a dry macula. Central subfield thickness (CST) <325 µm was defined as “absence of DME”. Injection intervals were extended according to CST <325 µm and VA criteria, irrespective of macular anatomy. Using either fixed dosing every 8 weeks (q8W) or a personalized treat-and-extend approach [Tr&Ex; termed personalized treatment interval (PTI)], CST <325 µm was achieved in 83% (mean of the two trials) of eyes and increased to 90% at the ends of years 1 and 2, respectively. Under the Tr&Ex regimen, 73% (mean) and 78% of eyes received faricimab every 12 weeks (~3 months), and 52% and 62% received injections every 16 weeks (~4 months) at the ends of years 1 and 2, respectively. Nineteen eyes (2.0%) in YOSEMITE and 22 (2.3%) in RHINE never had their dosing interval extended beyond every 4 weeks.
A landmark in DME therapy
A unique singular aspect in DME trials was the concurrent reporting on faricimab outcomes according to OCT anatomy. The distinctive drying effect of faricimab on MAs-DME for ≥8 weeks resulted in strict clinical separation of the two DME types into pathogenesis-related outcomes. This landmark in DME therapy could be drawn for the first time during the data analysis: True anatomical dryness for ≥8 weeks was attained in ~40% of eyes (mean of the q8W and Tr&Ex subgroups: 43% and 38%, respectively) and ~54% at the ends of years 1 and 2, respectively. These likely representing the MAs-DME subgroup. The 83% prevalence meeting the CST <325 µm criterion at year-1 was more than twice the 40% anatomical dryness rate. This suggests that 43% (83% − 40%) of eyes classified as <325 µm, plus ~2% maintained on 4-week dosing throughout, still had anatomical macular edema. The remaining, edematous maculae likely represent tractional DME, which is refractory to pharmacologic drying.
Extending injection intervals under these edematous conditions would be expected to prolong edema duration. The discrepancy between the two analytic approaches highlights the limitations of a numeric definition of “no DME.”
Real-world studies
VA was stable during the two-year current trials. However, VA deterioration attributable to DME, often under real-world conditions, has been reported in ~25% of eyes by month-9 after treatment initiation, and often worsens, 50% during year 3, and ~70% by year-8. Nevertheless, most RCTs, including faricimab trials, prioritized VA as the primary endpoint. Yet, the faricimab investigators state that “individualized treatment decisions were primarily guided by CST findings.”
Real-world studies reinforce the outcome dichotomy. Rush et al. reported fluid-free maculae for ≥8 weeks in 39.2% (n=51) of eyes, closely mirroring the pivotal anatomical results. Conversely, El-Badawi et al. applied the <325 µm criterion (n=62) and extended or maintained dosing despite CST worsening by <10% or even 10–20% thickening, respectively. By 12 months, VA was not significantly changed from baseline, while 96.8% of eyes remained anatomically edematous, undermining the numerical criterion. Similarly, Galindo et al. (n=32) reported, in contrast to the RCTs, that BCVA remained stable at month 12 and that interval extension reached only ~8–9 weeks. Absolute standard deviation (SD) was reported at month-12 (403.6 ± 121.9 µm at baseline to 303.2 ± 105.2 µm), enabling estimation of edema prevalence at study end (via Z-score), unlike datasets reporting only change-from-baseline SD.
Here, when one is using a CST ≥280 µm threshold for edema, despite mean CST reduction below 325 µm, ~58.7% of maculae remained edematous at month-12.
Other trials
New extension datasets may further inflate the appearance of “success” when it is defined numerically. In the recent RHONE-X study, a nonrandomized open label 2-year extension of faricimab-treated completers. VA was maintained (~10 ETDRS letters gain) and CST <325 µm was documented in >90% of eyes by year 4; ~80% had ≥12-week dosing and ~67% had 16-week intervals. However, anatomical fluid-free rates or absolute final CST ± SD were not provided. Without anatomy-based stratification, such findings risk conflating numerical thresholds with true disease resolution. It would be important to evaluate the anatomical integrity and long-term adaptability of the foveal layers following four years of chronic edema during faricimab therapy.
The anatomical OCT outcomes demonstrate how a permissive threshold may misclassify edematous maculae as “no DME.” This concern is magnified when broader thresholds are adopted, such as <400 µm in the PHOTON trial for 8-mg aflibercept, where widening the “no DME” basket necessarily increases the proportion of anatomically edematous eyes eligible for interval extension. By week 96 in PHOTON, 93%, 72%, and 44% of eyes achieved 12-, 16-, and 20-week (3–5 months) dosing intervals, respectively, despite the expected frequent inclusion of tractional DME and the limited durability of pharmacologic effects in that subgroup.
These findings support a clear directional conclusion: DME should be stratified by pathogenesis before treatment, consistent with the Declaration of Helsinki. After excluding microaneurysm-related DME, long-lasting macular drying is most reliably achieved surgically with PPV and ILM peeling in treatment-naïve eyes or following GLP failure, while GLP may be durably efficacious in selected early (“transitional-phase”) cases. These approaches are cost-effective and globally applicable, including in low-resource settings. Aligning future trials and clinical practice with pathogenetic enrollment and anatomy-based endpoints is essential to move beyond trial-and-error care and toward a truly effective, potentially curative paradigm.
Conflict:
The author declares no conflict of interest
Acknowledgment:
I would like to thank the statisticians Mrs. Ira from Alpha Statistics Ltd and Prof. David Steinberg for their advice on the statistical issues in the manuscript.
References
- Sadda SR, Campbell J, Dugel PU, Holekamp NM, Kiss S, Loewenstein A, et al. Relationship between duration and extent of edema and visual acuity outcome with ranibizumab in diabetic macular edema: A post hoc analysis of Protocol I data. Eye (Lond) 2020;34(3):480-490.doi:10.1038/s41433-019-0522-z.
- Sakamoto A, Nishijima K, Kita M, Oh H, Tsujikawa A, Yoshimura N. Association between foveal photoreceptor status and visual acuity after resolution of diabetic macular edema by pars plana vitrectomy. Graefes Arch Clin Exp Ophthalmol. 2009;247(10):1325-1330. doi: 10.1007/s00417-009-1107-5.
- Cennamo G, Montorio D, Fossataro F, Fossataro C, Tranfa F. Evaluation of vessel density in disorganization of retinal inner layers after resolved diabetic macular edema using optical coherence tomography. PLoS One. 2021;16(1):e0244789. doi: 10.1371/journal.pone.0244789.
- Schmidt-Erfurth U, Garcia-Arumi J, Bandello F, Berg K, Chakravarthy U, Gerenda BS, et al. Guidelines for the Management of Diabetic Macular Edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica. 2017;237(4):185-222. doi: 10.1159/000458539.
- Wykoff CC, Abreu F, Adamis AP, Basu K, Eichenbaum DA, Haskova Z, et al. Efficacy, durability, and safety of intravitreal faricimab with extended dosing up to every 16 weeks in patients with diabetic macular oedema (YOSEMITE and RHINE): two randomized, double-masked, phase 3 trials. Lancet. 2022;399(10326);741-755. doi: 10.1016/S0140-6736(22)00018-6.
- Wong TY, Haskova Z, Asik K, Bağal CR, Csaky KG, Eter N, et al. Faricimab treat-and-extend for Diabetic Macular Edema: Two-year results from the randomized phase 3 YOSEMITE and RHINE trials. Ophthalmology. 2024;131(6):708-723. doi: 10.1016/j.ophtha.2023.12.026.
- Ferro Desideri L, Traverso CE, Nicolò M. The emerging role of the Angiopoietin-Tie pathway as therapeutic target for treating retinal diseases. Expert Opin Ther Targets. 2022;26(2):145-154. doi: 10.1080/14728222.2022.2036121.
- Heier JS, Singh RP, Wykoff CC, Csaky KG, Lai TYY, Loewenstein A, et al. The angiopoietin/tie pathway in retinal vascular diseases: A Review. Retina. 2021 Jan 1;41(1):1-19. doi: 10.1097/IAE.0000000000003003.
- Takamura Y, Yamada Y, Morioka M, Gozawa M, Matsumura T, Inatani M. Turnover of microaneurysms after intravitreal injections of faricimab for diabetic macular edema. Invest Ophthalmol Vis Sci. 2023 Oct 3;64(13):31. doi: 10.1167/iovs.64.13.31.
- Takamura, Y, Yamada, Y, Inatani, M. Role of microaneurysms in the pathogenesis and therapy of diabetic macular edema: A descriptive review. Medicina 2023,59, 435. https://doi.org/10.3390/medicina59030435.
- The DRCR.net, Elman MJ, Aiello LP, Beck RW, Bressler NM, Bressler SB, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117:1064-1077. doi:10.1016/j.ophtha.2010.02.031.
- Mitchell P, Bandello F, Schmidt-Erfurth U, Lang GE, Massin P, Schlingemann RO, et al. The RESTORE Study: Ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118(4): 615-625. doi: 10.1016/j.ophtha.2011.01.031.
- DRCR.net; Wells JA, Glassman AR, Ayala AR, Jampol LM, Aiello LP, Antoszyk AN, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. New Eng J Med. 2015;372(13):1193-1203. doi: 10.1056/NEJMoa1414264.
- Bressler SB, Barve A, Ganapathi PC, Beckmann K, Apte RS, Marcus DM, et al. Aflibercept biosimilar MYL-1701P vs. reference aflibercept in diabetic macular edema: The INSIGHT randomized clinical trial. Jama Ophthalmol. 2024:142(10):952-960. doi:10.1001/jamaophthalmol.2024.3458.
- Gillies MC, McAllister IL, Zhu M, Wong W, Louis D, Arnold JJ, Wong TY. Intravitreal triamcinolone prior to laser treatment of diabetic macular edema: 24-month results of a randomized controlled trial. Ophthalmology 2011;118:866-872. doi: 10.1016/j.ophtha.2010.09.029.
- Glassman AR, Wells III JA, Josic K, Maguire MG, Antoszyk AN, Baker C, et al. Five-year outcomes after initial Aflibercept, Bevacizumab, or Ranibizumab treatment for diabetic macular edema (Protocol T Extension Study). Ophthalmology. 2020;127(9):1201-1210. doi: 10.1016/j.ophtha.2020.03.021.
- Jhaveri CD, Glassman AR, Ferris FL 3rd, Liu D, Maguire MG, Allen JB, et al. Aflibercept monotherapy or bevacizumab first for diabetic macular edema. New Engl J Med. 2022; 387(8):692-703. doi: 10.1056/NEJMoa2204225.
- Sensoy E, Citirik M. Current biosimilar anti-VEGF drugs in retinal diseases. Eur Eye Res. 2024;4(3):237-244 | doi: 10.14744/eer.2024.29291.
- Blindbaek SL, Peto T, Grauslund J. Aflibercept and navigated versus conventional laser in diabetic macular oedema: a 12-month randomized clinical trial. Acta Ophthalmol. 2020 Jun;98(4):347-352. doi: 10.1111/aos.14266. Epub 2019 Oct 10. PMID: 31602811.
- Zhang J, Wang M, Chen L, Radke N. Diabetic blindness remains a big challenge despite all recent advancements in diagnostics and treatments. Asia Pac J Ophthalmol. 2024;13(5):100105. doi: 10.1016/j.apjo/2024/100105.
- Ophir A, Martinez MR, Mosqueda P, Trevino A. Vitreous traction and epiretinal membranes in diabetic macular edema using spectral-domain optical coherence tomography. Eye (London). 2010;24(10):1545-1553. doi: 10.1038/eye.2010.80.
- Ophir A, Trevino A, Fatum S. Extrafoveal vitreous traction associated with diabetic diffuse macular oedema. Eye (Lond). 2010;24(2):347-353. doi: 10.1038/eye.2009.106.
“`




