Holistic Obesity Assessment in Pregnancy for Health Outcomes
Holistic Assessment of Obesity in Pregnancy: considering metabolic health
Cecilia M. Jevitt, PhD, CNM, APRN, RM, FACNM
- Midwifery/Department of Family Practice, Faculty of Medicine, University of British Columbia, Vancouver, Canada
OPEN ACCESS
PUBLISHED: 28 February 2026
CITATION: Jevitt, C.M., 2026. Holistic Assessment of Obesity in Pregnancy: considering metabolic health. Medical Research Archives, [online] 14(2).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Obesity is the most common complication to pregnancy with 16% to 30% of women worldwide living with high weights. Obesity has been defined solely by body mass index (BMI) without consideration of individual metabolic health. Neither most research nor research-based perinatal management guidelines have distinguished between metabolically healthy individuals and those with obesity related diseases. Several international medical organizations now recommend holistic health assessments that include screening for metabolic disorders in addition to BMI measurement, along with care recommendations tailored to metabolic health or disease. This article reviews research that studied outcomes for pregnant individuals living with obesity, who were stratified by metabolic health or disease, and their pregnancy outcomes. Additionally, methods for holistic health assessment during pregnancy are reviewed along with management for preclinical and clinical obesity based on international guidelines.
Keywords
- Obesity
- Pregnancy
- Metabolic health
- Holistic assessment
Introduction
Obesity is the most common complication to pregnancy with 16% to 30% of women worldwide living with high weights. Perinatal obesity research literature has focused on identifying maternal, fetal, and newborn complications associated with obesity (Table 1) and management to ameliorate these poor reproductive outcomes, making antenatal care weight and risk-centric, ignoring the large numbers of childbearing people living with obesity who have no obesity-associated disease and their pregnancy outcomes. Relying on the perinatal obesity research to date is problematic. Some studies mix individuals categorized as overweight with individuals in all obesity classes. Few studies stratify subjects by obesity class or divide individuals with obesity into those who are metabolically healthy and those with obesity-related disease. A smaller, growing body of literature has identified individuals living with obesity who are metabolically healthy and have uncomplicated pregnancy outcomes.
Outside of pregnancy, researchers have pivoted toward promoting a more comprehensive assessment of obesity, with patient centred health that is focused on the outcomes desired by the patient for healthy living, not weight loss. The American Medical Association, The European Association for the Study of Obesity, The Canadian Medical Association, and The American College of Endocrinology all support a health assessment that is more than only selecting a body mass index (BMI) range, urging one that reviews medical complications and functional status related to obesity. These holistic health assessments leading to individualized risk determination and care planning have not been described in the perinatal or midwifery literature. Targeted health recommendations that consider metabolic health can reduce overtreatment and potential iatrogenic harm. Pregnancy presents an unparalleled primary care opportunity for clinicians to support individuals living with obesity with health education and lifestyle strategies that can promote an uncomplicated pregnancy and prolong their metabolic health beyond the baby s birth. This manuscript reviews studies documenting metabolic health for pregnant individuals living with obesity. The methods used to determine metabolic health are reviewed and applied to holistic assessment of health in obesity, and antenatal health teaching to support that health.

METABOLICALLY HEALTHY OBESITY
Obesity defined only by body mass index (wt[kg]/height2[m2]) correlates poorly with individual adiposity. World Health Organization experts agree that there is no percent body fat cut-off that defines obesity even as research using WHO BMI ranges as indicators of health proliferates. Adipose tissue is the body s largest endocrine organ. It regulates energy homeostasis for the entire body and modulates immune function. Outside of pregnancy, metabolically healthy obesity (MHO) for women has been defined as a systolic blood pressure less than 130 mmHg, no blood pressure lowering medication, no diabetes, and a waist to hip ratio less than 0.95. Researchers using this definition with National Health and Nutrition Survey III (NHANES III) data from the United States (US) found that 40% of adults living with obesity were metabolically healthy and at low risk for cardiac disease and related mortality. Higher mortality was observed for people with BMIs > 40 regardless of metabolic health (MH). These researchers acknowledged that Asians were under-represented in their samples and that waist to hip ratios might not be a reliable screen for them.
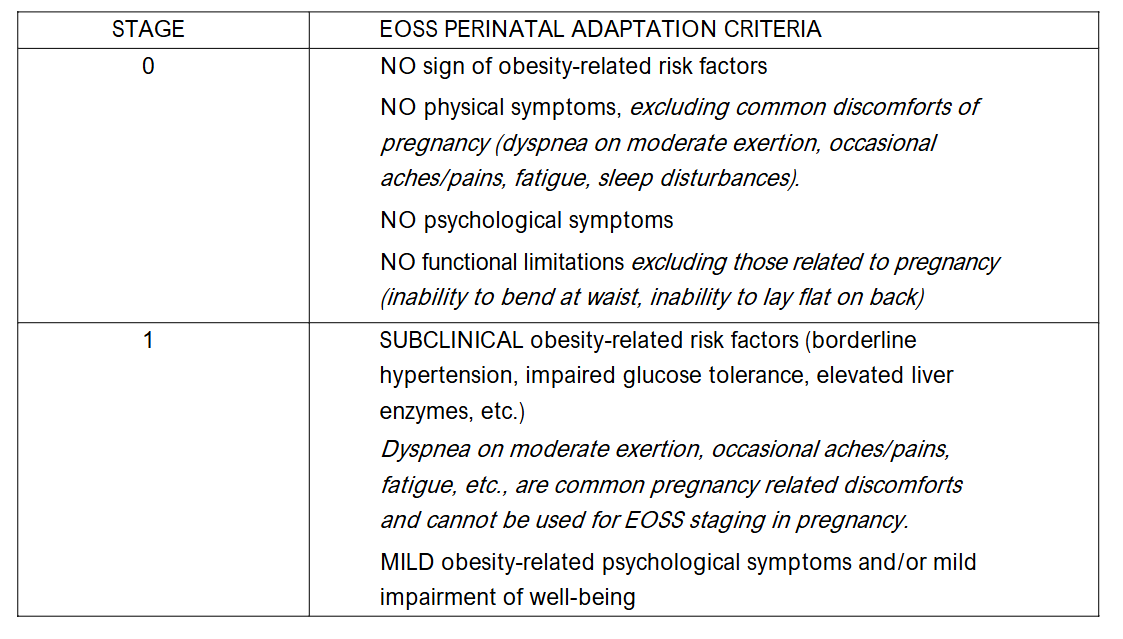
Metabolically healthy obesity is the basis for the Edmonton Obesity Staging System (EOSS). Developed originally to prioritize individuals living with obesity who would benefit from bariatric surgery, the EOSS divides individuals with obesity into 5 stages based on obesity-related symptoms and disease (Table 2). In the original EOSS application to United States NHANES III data, 27% of adults in obesity class I were in EOSS stages 0 and 1 (metabolically healthy), 22% in obesity class II, and 19% in obesity class III, demonstrating that metabolic health is imperfectly related to body mass index. The NHANES subjects were adults of all ages, male and female. Demsky, et al., first developed a perinatal adaptation of the EOSS which removes obesity-related symptoms, such as dyspnea, that occur during healthy, uncomplicated pregnancies to women of all sizes (Table 2). The perinatal adaptation of the EOSS can be used to provide a more holistic assessment of health, and guide risk discussions and prenatal health education with those in EOSS stages 0 and 1 needing supportive health education more than interventive management.
CLINICAL AND PRECLINICAL OBESITY
The Lancet Diabetes and Endocrinology Commission spent several years reviewing and redefining obesity. Their 2025 publication said that defining obesity as a disease led to overdiagnosis. Obesity (a BMI > 30) was divided into preclinical obesity and clinical obesity. Preclinical obesity is a BMI > 30 with no obesity-related disease. Clinical obesity, a BMI > 30 with obesity related metabolic disease, is a chronic disease specifically caused by excess adiposity. This conceptualization of preclinical and clinical obesity is consistent with the Edmonton Obesity Staging System s levels of obesity. Individuals with preclinical obesity have been treated with the same interventions as those with clinical obesity for decades. The Lancet Commission recommends monitoring with or without prophylactic interventions for those with preclinical obesity. Interventions related to metabolic disease and obesity are advised for individuals with clinical obesity. Metabolic disease-based approaches along with outcomes research have not been applied to antenatal care.
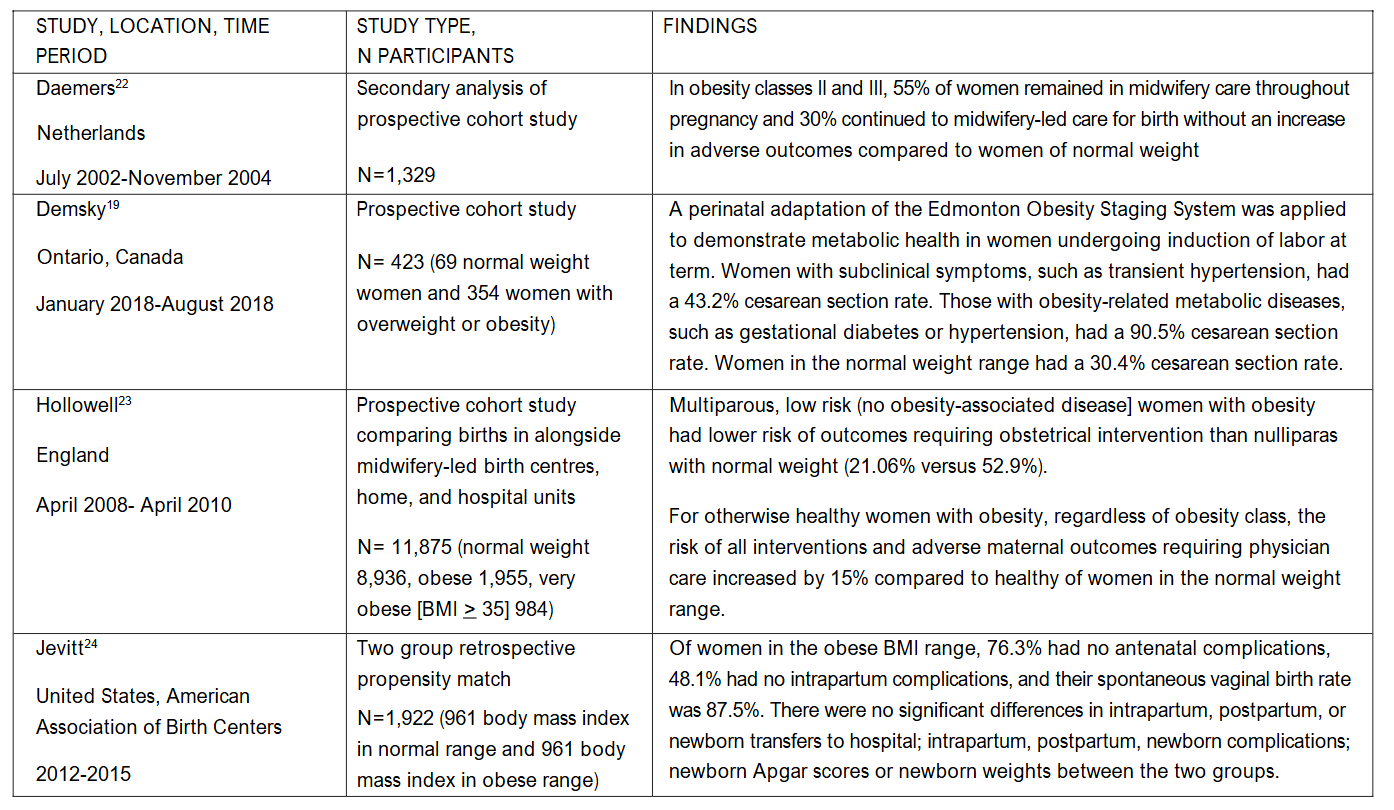
Aging is associated with increased adiposity and obesity. Premenopausal, reproductive age individuals with obesity may have higher rates of metabolic health because they have not yet been subject to the effects of aging which increase visceral fat deposition and increase insulin resistance, inflammation and impaired immune function, which in turn increase risk for type 2 diabetes, hypertension, hyperlipidemia and heart disease. Age may be the factor that explains the high numbers of pregnant people who are metabolically healthy in the studies reviewed in Table 3. In those studies, midwifery care or births in out of hospital settings are markers for the absence of diabetes, hypertension, preeclampsia or other obesity-related disease, because in all countries, childbearing people with obesity-related disease receive care management from physicians.
Holistic Prenatal Health Assessment
Antenatal care providers must balance the ethical responsibility to inform childbearing people of the perinatal risks associated with obesity with an unbiased, non-stigmatizing approach to health education. Obesity historically has been viewed as a personal responsibility with those living with high weights viewed as lazy or gluttonous. With several psychiatric medications and endocrine disrupting chemicals known to increase weight along with social stressors including racism, poor sleep, and a nonnutritious food supply, obesity must be viewed as an intergenerational physical, epigenetic adaptation to multiple socio-economic disparities, social and physical environments that alter the hypothalamic pituitary adrenal axis (HPA) and appetite regulating systems. Antenatal support for individuals living with obesity includes holistic assessment of health at the first prenatal visit, determination of an optimal weight gain target, assessment of usual eating and activity patterns and advice on optimal eating and activity for pregnancy. Life-style changes made during pregnancy can forestall the onset of obesity-related disease and improve future health.
The World Health Organization definition of health, a state of complete physical, mental and social well-being and not merely the absence of disease or infirmity should inspire clinicians to understand not only the pregnant person s physical being but also the daily environment in which that person lives. The Problem Oriented Medical Record (POMR) developed and promoted in the United States since the 1970s, reduces individuals to a list of their past and present physical and mental health illnesses. Trained in use of this system, clinicians see each problem separately as something to be cured, often lacking the holism to see the interrelatedness of many conditions and their socioeconomic roots. Holistic prenatal assessment begins with an understanding of the childbearing person s physical and metabolic health but must then extend to an understanding of how that person s life is centered in particular socio-economic conditions.
Using the Edmonton Obesity Staging System begins a holistic assessment that takes into consideration adiposity and metabolic health. A perinatal application of the EOSS stages childbearing people according to their metabolic health and enables the clinician to more accurately inform the patient of potential risks and ameliorating care. The perinatal EOSS adaptation does not use symptoms commonly occurring during pregnancy such as dyspnea on exertion, gastro-esophageal reflux, or mobility limitations. Another consideration is that individuals in EOSS stages 3 and 4 (end organ disease and failure) often have infertility, leaving EOSS stages 1-2 as the most used during pregnancy. A clinician does not have to do a complete EOSS staging during a prenatal assessment. Deciding on metabolic health is as simple as considering, Does the pregnant person have diabetes, hypertension, heart disease or other obesity-related morbidity? Individuals with obesity-related disease (EOSS stages 2-4 or clinical obesity) are at the highest risk for perinatal complications and need extra medical support; however, then can benefit from the same nutrition and activity advice as individuals who are metabolically healthy. Figure 1 presents an algorithm for presenting risk-ameliorating medical treatments.

The presence of obesity-related disease and high weight must both be considered during informed choice discussions with patients. Individuals with preclinical, metabolically healthy obesity can be offered risk ameliorating strategies and health education that aims to prevent obesity-related disease. Those with obesity-related disease (clinical obesity) should receive stronger recommendations to use risk-reducing treatments. Weight will change during pregnancy and metabolic health might change, necessitating revisiting the best self-care for patients and optimal support from antenatal care providers at each antenatal visit. The prenatal advice that is common to all pregnant individuals includes optimal weight gain, ideal nutrition, regular physical activity, and sufficient sleep. These are often the most difficult conversations for clinicians to have with patients.
Guideline-Based Prenatal Care for Individuals With Obesity
OPTIMAL WEIGHT GAIN TARGET
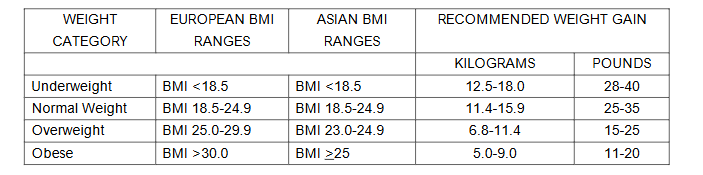
Clinicians often fear offending patients when discussing weight gain during pregnancy, particularly since weight gain recommendations became based on BMI. Weight gains that are consistent with 2009 United States Institute of Medicine Recommended Prenatal Weight Gain Guidelines have been shown to improve pregnancy outcomes. It is important to recognize that there are BMI categories for Asian populations based on increased risk for obesity-related diseases in Asian populations at lower BMI ranges than in other populations. Asian BMI categories lower the recommended antenatal weight gain for individuals from Asia (Table 4). Assisting patients with prenatal weight gain depends on two actions: supportive, unbiased health education and periodic weighing with non-judgmental analysis of weight loss or gain.
Until the 1960s in the English-speaking world, women were told to restrict weight gain during pregnancy to prevent preeclampsia and birth complications such as macrosomia. Maternal weighing, once a part of routine prenatal visits, was abandoned in the United Kingdom, Australia, New Zealand and Canada during the 1990s following research that demonstrated poor links between weighing and preventing low birth weight newborns or diagnosing preeclampsia. Guidelines for antenatal weight gain were introduced by the United States Institute of Medicine in 1990 that aimed to reduce low birth weights. These guidelines, still the current practice standard, required periodic weighing to determine the success of the nutrition and weight gain advice. Controversy about weighing persisted with routine weighing remaining part of prenatal visits in the United States. In other places, clinicians worried that weighing was embarrassing and stressful for patients and routine weighing was dropped. To recommend an antenatal weight gain target, clinicians will need to measure height and weight at the first prenatal visit. Patients not desiring these measurements can give prepregnancy measurements. An initial weight can be useful as a baseline if patients experience excessive nausea and vomiting with dehydration or insufficient weight gain is a concern. Patients can be offered regular weighing with antenatal visits. Weights should be taken in a private area, ideally with patients weighing themselves and reporting the weight to the clinician. Periodic weighing during pregnancy has been shown to assist patients in optimizing weight gain while avoiding excessive gain. How patients want their size used in conversation varies person to person from large body, high weight, and person of size. Surveys of adults found that the three top preferred terms were weight, unhealthy weight, and overweight, with responders universally disliking the terms fat and obesity. If a patient has a BMI > 30, the clinician can ask, How do you prefer to talk about your weight? Overweight? Large body? Large size?
Motivational Interviewing (MI) is a counseling style that has been used successfully during antenatal care to begin conversations about tobacco use, physical activity, and weight management. MI assumes a patient-centred approach with the plans and goals of patient being primary, not the rigid guidelines that could be imposed by clinicians. MI starts with the clinician asking permission to talk about a topic. For example, Would it be alright with you if we talked about what you know about weight gain in pregnancy? In that sentence, the clinician both asks permission and acknowledges that most patients have some ideas about their own health. If the patient does not want to talk about weight, the antenatal care provider still has an obligation to present evidence-based information about the risk for perinatal complications associated with high weights. The clinician could say, Based on your health today, I have to let you know that there is increased risk for diabetes during pregnancy and high blood pressure. The clinician would add community appropriate risk statistics, describe the screening for disease that will be done, and then ask if the patient has any questions. If the patient has no questions, the clinician can assure the patient that questions are welcome at any time.
ASSESSMENT OF EATING AND ACTIVITY PATTERNS
When doing nutritional counseling, the word diet is best avoided. Diet connotes food restriction and is associated with negative experiences for many. Diet can be replaced by eating or nourishment. The clinician could ask, What do you usually eat? or What are your favorite foods? Assumptions should not be made that individuals with high weights eat large amounts of high sugar or high fat foods. Activity can be assessed by asking what a usual day or workday is like for a patient. Is there a regular activity that they do such as walking a dog? Activity assessment includes usual sleep patterns and hours of sleep per day.
OPTIMAL EATING DURING PREGNANCY
Weight gains within the 2009 Institute of Medicine Guidelines depend on optimal eating and nutrition. No pregnancy-specific diets have been developed; however, the Mediterranean diet, the Nordic diet, the flexitarian diet, the DASH diet (Dietary Approaches to Stop Hypertension), the United States My Plate diet, and low glycemic index diets all include fiber rich fruits and vegetables, whole grains, peas, beans, and nuts, eggs, dairy products, plant-based oils and lean meats such as fish. All these eating patterns can provide the macro- and micro-nutrients needed for a healthy pregnancy and fetal growth. Use of the Mediterranean and DASH diets has been shown to reduce the risk for gestational diabetes by 15-38%. Approximately 100 kcal per day more per day are needed during the first 16-20 weeks of pregnancy, increasing to 300 kcal per day for the last half of pregnancy. Weight loss during pregnancy is not recommended, even for those with high prepregnancy weights.
OPTIMAL ANTENAL ACTIVITY
Exercise is another term that may be off-putting to some individuals because of its association with the grueling work done by professional athletes. Physical activity is a term that includes work activity, housework, walking, swimming and other low impact activities. More than 90 minutes of physical activity per week has been shown to reduce the odds for gestational diabetes by 21-46%. Regular physical activity including walking, approximately 150 minutes per week, has been shown to reduce the risk for cesarean section. Sleep is not often thought of as activity even though lack of sufficient sleep is associated with insulin resistance, high leptin levels, higher daily intake, weight gain and obesity. Six to seven hours of nighttime sleep recommended. A 2022 survey of sleep in American adults showed that 30% of those working 40 or fewer hours per week and 50% of those working more than 60 hours per week slept less than 7 hours during a 24 hour period.
MANAGEMENT STRATEGIES FOR INDIVIDUALS WITH CLASS III OBESITY OR OBESITY RELATED DISEASE
Risk ameliorating strategies vary country to country, however, folic acid 4-5mg per day oral supplementation in the first trimester to prevent neural tube defects and low dose aspirin to reduce the incidence of preeclampsia and as thrombus prophylaxis are recommended. Because adipose deposition may obscure fetal palpation, an ultrasound to document fetal position in labour is recommended. Some guidelines recommend screening for obstructive sleep apnea and a prenatal anesthesia consult. A moving and handling risk assessment and moving to prevent pressure sores if mobility is limited during labour can be considered when BMI exceeds 39 (Class III obesity). Multiparas who are metabolically healthy are candidates for low-risk, midwifery-led units. Venous access should be discussed with patients at the start of labour. Because the risk of stillbirth increases with a BMI greater than 39, consideration should be given to induction of labour at 39 to 40 weeks gestation. Postpartum chemoprophylaxis to prevent deep vein thrombosis needs to be considered particularly if mobility is limited.
Conclusions
Conceptualizations of obesity have evolved during the last two decades. Future perinatal research will need to continue the search for eating and activity patterns that support health without a stigmatizing focus on weight alone. This research will need to stratify participants by preclinical or clinical obesity and by WHO BMI groupings and obesity classes. Clinicians have newer concepts to apply such as metabolically health obesity or preclinical obesity, and assessment tools such as the Edmonton Obesity Staging System that assist in a nuanced evaluation of health. Use of a holistic health assessment paired with informed choice discussions that more accurately portray weight-related perinatal risks, will enable clinicians and patients to form individualized plans of care that support health and growth in pregnancy without focusing on weight alone.
Author Contributions:
Conceptualization, research, writing, diagrams and figures, writing including original draft preparation, review, and editing done by CMJ. The author read and agreed to the published version of the manuscript.
Funding:
This writing received no external funding.
Institutional Review Board Statement:
Not applicable
Informed Consent Statement:
Not applicable
Data Availability Statement:
Not applicable
Conflicts of Interest:
The author declares no conflicts of interest.
References:
- Denison FC, Aedla NR, Keag O, Hor K, Reynolds RM, Milne A, et al. Care of Women with Obesity in Pregnancy: Green-top Guideline No. 72. BJOG. 2019;126(3):e62 e106.
- Maxwell C, Gaudet L, Cassir G, Nowik C, McLeod NL, Jacob CE, et al. Guideline No. 391-Pregnancy and Maternal Obesity Part 1: Preconception and Prenatal Care. J Obstet Gynaecol Can. 2019;41(11):1623 40.
- Maxwell C, Gaudet L, Cassir G, Nowik C, McLeod NL, Jacob CE, et al. Guideline No. 392-Pregnancy and Maternal Obesity Part 2: Team Planning for Delivery and Postpartum Care. J Obstet Gynaecol Can. 2019;41(11):1660 75.
- Ferris L, de Vries B, Sweeting A. Management of obesity in pregnancy. Aust N Z J Obstet Gynaecol. 2022;62(5):623 5.
- Sagi-Dain L. Obesity in Pregnancy: ACOG Practice Bulletin, Number 230. Obstet Gynecol. 2021;138(3):489.
- Kent L, McGirr M, Eastwood KA. Global trends in prevalence of maternal overweight and obesity: A systematic review and meta-analysis of routinely collected data retrospective cohorts. Int J Popul Data Sci. 2024;9(2):2401.
- Wharton S, Lau DCW, Vallis M, Sharma AM, Biertho L, Campbell-Scherer D, et al. Obesity in adults: a clinical practice guideline. Canadian Medical Association Journal. 2020;192(31):E875 E91.
- Busetto L, Dicker D, Frühbeck G, Halford JCG, Sbraccia P, Yumuk V, et al. A new framework for the diagnosis, staging and management of obesity in adults. Nature Medicine. 2024;30(9):2395 9.
- Garvey WT. American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity. Endocrine practice.22(supplement 3):1 203.
- Schmidt M, Schaefer-Graf UM. The German guideline “Obesity in pregnancy”: comparison with the international approach. Arch Gynecol Obstet. 2024;309(5):1699 705.
- American Medical Association. AMA Proceedings: Support Removal of BMI as a Standard Measure in Medicine. 2023.
- Institute of Medicine, National Research Council Committee to Reexamine Weight Gain During Pregnancy. The National Academies Collection: Reports funded by National Institutes of Health. In: Rasmussen KM, Yaktine AL, editors. Weight Gain During Pregnancy: Reexamining the Guidelines. Washington (DC): National Academies Press (US) Copyright © 2009, National Academy of Sciences.; 2009.
- Kominiarek MA, Chauhan SP. Obesity Before, During, and After Pregnancy: A Review and Comparison of Five National Guidelines. Am J Perinatol. 2016;33(5):433 41.
- De Lorenzo A, Gratteri S, Gualtieri P, Cammarano A, Bertucci P, Di Renzo L. Why primary obesity is a disease? Journal of Translational Medicine. 2019;17(1).
- World Health Organization. Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. World Health Organ Tech Rep Ser. 1995;854:1 452.
- Olson SM, Muñoz EG, Solis EC, Bradford HM. Mitigating Weight Bias in the Clinical Setting: A New Approach to Care. Journal of Midwifery & Women’s Health. 2024;69(2):180 90.
- Zembic A, Eckel N, Stefan N, Baudry J, Schulze MB. An Empirically Derived Definition of Metabolically Healthy Obesity Based on Risk of Cardiovascular and Total Mortality. JAMA Network Open. 2021;4(5):e218505.
- Padwal RS, Pajewski NM, Allison DB, Sharma AM. Using the Edmonton obesity staging system to predict mortality in a population-representative cohort of people with overweight and obesity. Canadian Medical Association Journal. 2011;183(14):E1059 E66.
- Demsky AN, Stafford SM, Birch D, Sharma AM, Schulz JA, Steed H. The Edmonton Obesity Staging System Predicts Mode of Delivery After Labour Induction. Journal of Obstetrics and Gynaecology Canada. 2020;42(3):284 92.
- Hollowell J, Pillas D, Rowe R, Linsell L, Knight M, Brocklehurst P. The impact of maternal obesity on intrapartum outcomes in otherwise low risk women: secondary analysis of the Birthplace national prospective cohort study. BJOG: An International Journal of Obstetrics & Gynaecology. 2014;121(3):343 55.
- Jevitt CM, Stapleton S, Deng Y, Song X, Wang K, Jolles DR. Birth Outcomes of Women with Obesity Enrolled for Care at Freestanding Birth Centers in the United States. Journal of Midwifery & Women’s Health. 2021;66(1):14 23.
- Pétursdóttir Maack H, Larsson A, Axelsson O, Olovsson M, Wikström AK, Sundström Poromaa I. Pregnancy in metabolic healthy and unhealthy obese women. Acta Obstetricia et Gynecologica Scandinavica. 2020;99(12):1640 8.
- Relph S, Guo Y, Harvey ALJ, Vieira MC, Corsi DJ, Gaudet LM, et al. Characteristics associated with uncomplicated pregnancies in women with obesity: a population-based cohort study. BMC Pregnancy and Childbirth. 2021;21(1).
- Rowe R, Knight M, Kurinczuk JJ. Outcomes for women with BMI > 35kg/m2 admitted for labour care to alongside midwifery units in the UK: A national prospective cohort study using the UK Midwifery Study System (UKMidSS). PLOS ONE. 2018;13(12):e0208041.
- Vieira MC, White SL, Patel N, Seed PT, Briley AL, Sandall J, et al. Prediction of uncomplicated pregnancies in obese women: a prospective multicentre study. BMC Medicine. 2017;15(1).
- Relph S, Ong M, Vieira MC, Pasupathy D, Sandall J. Perceptions of risk and influences of choice in pregnant women with obesity. An evidence synthesis of qualitative research. PLOS ONE. 2020;15(1):e0227325.
- Jevitt CM. Obesity and Socioeconomic Disparities: Rethinking Causes and Perinatal Care. J Perinat Neonatal Nurs. 2019;33(2):126 35.
- World Health Organization. WHO Constitution. 2025.
- Gaudet-Blavignac C, Rudaz A, Lovis C. Building a Shared, Scalable, and Sustainable Source for the Problem-Oriented Medical Record: Developmental Study. JMIR Medical Informatics. 2021;9(10):e29174.
- Jevitt CM, Ketchum K. Pairing Evidence-Based Strategies With Motivational Interviewing to Support Optimal Nutrition and Weight Gain in Pregnancy. J Perinat Neonatal Nurs. 2024;38(1): 25 36.
- Christenson A, Johansson E, Reynisdottir S, Torgerson J, Hemmingsson E. Shame and avoidance as barriers in midwives’ communication about body weight with pregnant women: A qualitative interview study. Midwifery. 2018;63:1 7.
- Christenson A, Johansson E, Reynisdottir S, Torgerson J, Hemmingsson E. “or else I close my ears” How women with obesity want to be approached and treated regarding gestational weight management: A qualitative interview study. PLOS ONE. 2019;14(9):e0222543.
- Harinath M, Smith AM, Binstock AB, Pollak KI, Levine MD, Chang JC. Starting Conversations About Gestational Weight Gain: How Weight and Weight Gain Communication Is Initiated in the First Prenatal Visit [ID 2683652]. Obstetrics & Gynecology. 2024;143(5S):64S 5S.
- Kominiarek MA, Peaceman AM. Gestational weight gain. American Journal of Obstetrics and Gynecology. 2017;217(6):642 51.
- Zafar MI, Mills KE, Zheng J, Peng MM, Ye X, Chen LL. Low glycaemic index diets as an intervention for obesity: a systematic review and meta‐analysis. Obesity Reviews. 2019;20(2):290 315.
- Mijatovic-Vukas J, Capling L, Cheng S, Stamatakis E, Louie J, Cheung NW, et al. Associations of Diet and Physical Activity with Risk for Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. Nutrients. 2018;10(6):698.
- Teede HJ, Bailey C, Moran LJ, Bahri Khomami M, Enticott J, Ranasinha S, et al. Association of Antenatal Diet and Physical Activity Based Interventions With Gestational Weight Gain and Pregnancy Outcomes. JAMA Internal Medicine. 2022;182(2):106.
- Brady EM, Bodicoat DH, Hall AP, Khunti K, Yates T, Edwardson C, et al. Sleep duration, obesity and insulin resistance in a multi-ethnic UK population at high risk of diabetes. Diabetes Res Clin Pract. 2018;139:195 202.
- Capers PL, Fobian AD, Kaiser KA, Borah R, Allison DB. A systematic review and meta-analysis of randomized controlled trials of the impact of sleep duration on adiposity and components of energy balance. Obes Rev. 2015;16(9):771 82.
- Itani O, Jike M, Watanabe N, Kaneita Y. Short sleep duration and health outcomes: a systematic review, meta-analysis, and meta-regression. Sleep Med. 2017;32:246 56.
- U.S. Centers for Disease Control. QuickStats: Percentage of Employed Adults Aged 18 Years Who Slept 7 Hours per 24-Hour Period, by Sex and Number of Work Hours per Week-United States, 2022. MMWR Morbidity and Mortality Weekly Report. 2024;73(16):385.

