Cost Comparison of Alzheimer Drugs: Donanemab vs Evolocumab
The Cost of Alzheimer Drug Treatment
Hely M Guirgis1, MD, PhD
- University of California, Irvine
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Guirgis, HM., 2026. The Cost of Alzheimer Drug Treatment. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Introduction: The anti-amyloid drug donanemab is approved In the United States and Europe for treatment of early symptomatic Alzheimer’s disease (mild dementia) in adults with confirmed amyloid pathology. Evolocumab was also reported to lower the risks of heart attacks and Alzheimer. In view of longevity and continued early Alzheimer recognition, we planned to compare costs of donanemab and evolocumab with other drug classes.
Methods: Drug costs were obtained from Google search and yearly cost calculated.
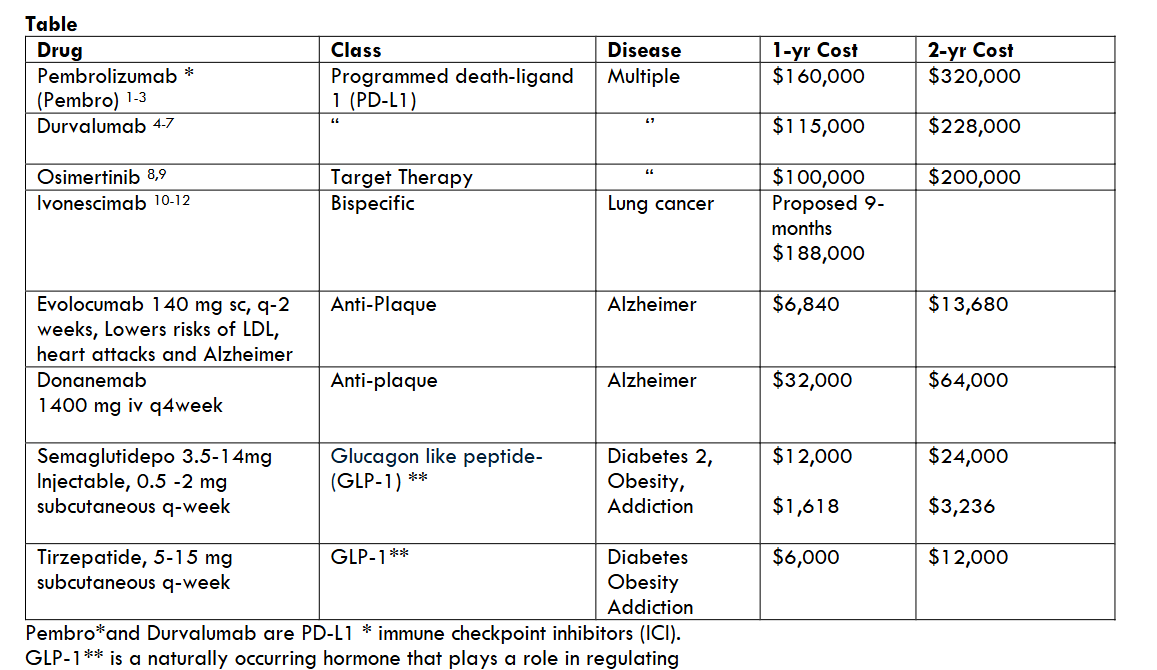
Results: Yearly costs of evolocumab were $6,840. Donanemab was more expensive at $32,000. Pembrolizumab was $160,00, durvalumab $115,000, Osimertinib $100,000, semaglutide $12,000, proposed 9-month ivonescimab $188,000.
Conclusions: Donanemab $32,000 yearly cost was higher than evolocumab at $6,840. Both drugs were reasonably priced as compared with other classes. Considering longevity, the overall worldwide Alzheimer future management would be too costly to be affordable.
Keywords
- Alzheimer’s Disease
- Donanemab
- Evolocumab
- Drug Costs
- Healthcare Economics
Introduction
According to the World Health Organization, dementia is one of the biggest diseases of our time. Ten million are diagnosed every year. More than 55 million people (about twice the population of Texas) worldwide live with a form of dementia. It is the seventh leading cause of death among all diseases. As population ages, the number of cases of dementia rises. While the deterioration of cognitive functioning is not caused by age itself, it does primarily affect the older generation. As a result, many elderly people suffer from disability and loss of independence. Psychological, social and economic implications result for them and their families, carers and society more broadly. We planned to report on costs of approved dementia drugs in comparison with classes of other diseases.
Methods
Drug costs and some other information were obtained from Google search. Drug yearly costs were calculated.
Results
Costs of various drug classes in the United States (US) were compared with the 2 anti-plaque drugs for Alzheimer treatment (Table). The 2-anti-plaque drug yearly costs were evolocumab $6,840 and donanemab $32,000.
One-year cost of the programmed death-ligand 1 (PD-L1) Pembrolizumab (Pembro) (1-3)) was $160,000 and Durvalumab $115,000 (4-7). Osimertinib (8,9), widely used in non-small-cell lung cancer, targets the epidermal growth factor receptors (EGFR) at a yearly $100,000 cost.
Ivonescimab (10-12), not yet approved, represents the bispecific class (3-5) at a proposed 9-month $188,000 cost.
Discussion
It is important to emphasize that drug costs vary widely, in contrast to the stability of value 13,14. Costs depend on the purchasing agent and the amount purchased. The most important factor in cost determination is the number of potential consumers.
According to US 2023 data, life expectancy is 81.1 years, higher for females compared to males at 75.8. It is estimated that ten million people with dementia are diagnosed worldwide every year. With continued longevity and early recognition of Alzheimer, disease, drug treatment costs are bound to multiply.
Alzheimer treatment validation approaches including radiation, radiology and drugs is a rational valid necessity, regardless of costs. Considering the present and future number of Alzheimer patients, treatment would be universally too expensive to afford.
The number of (glucagon like peptide-(GLP-1) drugs for diabetes mellitus and obesity is increasing and was therefore included for comparison.
The yearly cost of evolocumab was $6,840 and $32,000. There is no direct value or efficacy comparison between the 2 drugs and unlikely to see one in the future. Hence, it would be indeed wise to choose the cheaper one, namely evolocumab.
In summary, the number of Alzheimer patients is rapidly multiplying. The overall treatment costs of drugs, radiology and radiation modalities have also increased, sending a warning of unsustainable worldwide heavy economic burden. More efforts, finances, new drugs and plaque-focused radiation need to be devoted to speed up the pace of progress against Alzheimer.
References
- Garon EB, Rizvi NA, Hui R, et al. Pembrolizumab for the treatment of non-small cell lung cancer. N Engl J Med. 372:2018–28, 2015.
- Reck M, Rodriguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 375:823–1833, 2016.
- Herbst RS, Baas P, Kim D-W, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-postive, advanced non-small-cell lung cancer (KEYNOTE -010): A randomised controlled trial. Lancet 387: 1540-1550, 2016.
- Heymach JV, Harpole D, Mitsudomi T, et al. Perioperative durvalumab for resectable non–small-cell lung cancer. N Engl J Med. 2023;389(18):1672-1684 (Including Supplement and Protocol).
- IMFINZI® (durvalumab) [Prescribing Information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
- Heymach JV, Harpole D, Mitsudomi T, et al. Perioperative durvalumab for resectable NSCLC: updated outcomes from the phase 3 AEGEAN trial. Presented at: IASLC 2024 World Conference on Lung Cancer; September 7-10, 2024; San Diego, CA. Abstract OA1
- Cheng Y. et al. Durvalumab after Chemoradiotherapy in Limited-Stage Small-Cell Lung Cancer. N Engl J Med 2024; 391:1313-1327.
- Herbst RS, Wu Yi-long, John T (2023) Adjuvant Osimertinib for Resected EGFR-Mutated Stage IB-IIIA Non–Small-Cell Lung Cancer: Updated Results from the Phase III Randomized ADAURA Trial. J Clin Oncol 41(10): 1830-1840.
- Lv C, Fang W, Wu N, Jiao W, Xu S, et al. Osimertinib as Neoadjuvant Therapy for EGFR-Mutant Resectable Stage II-IIIB Lung Adenocarcinoma. Lung Cancer (2023), 178: 151-156.
- Zhou C, Chen J Wu L, et al: Phase 3 study of ivonescimab (AK112) vs pembrolizumab as first-line treatment for PD-L1-positive advanced NSCLC: Primary analysis of HARMONi-2. 2024 World Conference on Lung Cancer. Abstract PL02.04. Presented September 8, 2024.
- Wang L, Luo Y, Ren S, et al: A phase 1b study of ivonescimab, a programmed cell death protein-1 and vascular endothelial growth factor bispecific antibody, as first- or second-line therapy for advanced or metastatic immune-therapy-naive NSCLC. J Thorac Oncol 19:465-475, 2024.
- Guirgis, HM. Cost Comparison of The Bispecific Ivonescimab, Immune Check Point Inhibitors and Target Therapy in advanced/metastatic Lung Cancer. J Cancer Sci Treatment, 7(1): 228-231.
- Schnipper LE, Davidson NE, Wollins DS, et al. American Society of Clinical Oncology statement: A framework to assess the value of cancer treatment options. J Clin Oncol. June 22, 2015.
- Cherny NI, Sullivan R, Dafni U, et al. A standardized, generic, validated approach to stratify the magnitude of clinical benefit that can be anticipated from anti-cancer therapies. The European Society for Medical Oncology: magnitude of clinical benefit scale (ESM-MCBS): Oxford University Press; 2015.
- Guirgis. HM. Costs of Target Therapy and Proportionality to Number of Purchases: Propose Using Maintenance Dose and Limited Duration. ESMED 2024. Doi https://doi.org/10.18103/mra.v 12i1.4959. ISSN:2375-1924.




