Long-Term Outcomes of MSC Therapy in Psoriatic Arthritis
Case Report: Psoriatic Arthritis and Stem Cell Therapy – A Five-Year Follow-Up Report
Paul A. Dreschnack, MD, PC ¹; Ina Dreschnack, MS ²
- Paul A Dreschnack, MD, P.C.
- Harvard University
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Dreschnack, PA., and Dreschnack, I., 2025. Case Report: Psoriatic Arthritis and Stem Cell Therapy – A Five-Year Follow-Up Report. Medical Research Archives, [online] 13(4). https://doi.org/10.18103/mra.v13i4.6547
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6547
ISSN 2375-1924
ABSTRACT
Psoriatic arthritis (PsA) is a chronic inflammatory-immune mediated disease that can lead to debilitating musculoskeletal symptoms and, in severe cases, comorbidities such as osteoporosis, cardiovascular disease, and bowel inflammation. Traditional treatments, including NSAIDs, DMARDs, and biologics, offer symptom management but do not halt disease progression. Recent advances in regenerative medicine, particularly mesenchymal stem cell (MSC) therapy, have shown promise in addressing both inflammation and tissue regeneration. This case study evaluates the long-term outcomes of MSC therapy in a 68-year-old female patient with 25-year PsA history, and psoriatic skin lesions that covered 80% of her body. She received adipose-derived MSC treatment five years ago for debilitating arthritic joint pain and psoriasis. Initial responses were promising, with reduced pain, stiffness, and enhanced joint function. Five years post-treatment, the clinical observations on the patient report sustained improvements in mobility, minimal discomfort, and stable significant resolution of psoriatic skin lesions (from 80% to <5% body surface area). This case report highlights the potential of MSC therapy as a long-term treatment for PsA, offering promising outcomes in both joint function and skin manifestations, with a focus on the durability and stability of treatment effects over time.
Keywords: Psoriatic arthritis, mesenchymal stem cell, joint pain, inflammation, skin lesion.
Introduction
Psoriatic arthritis (PsA) is a heterogeneous chronic inflammatory-immune mediated disease that progresses in stages. It is characterized by musculoskeletal inflammation (arthritis, spondylitis, dactylitis, enthesitis), and generally occurs in patients with psoriasis. In severe cases, PsA is associated with osteoporosis, uveitis, cardiovascular disease, and bowel inflammatory disease. Current treatments of PsA such as non-steroidal anti-inflammatory drugs (NSAIDs), disease-modifying antirheumatic drugs (DMARDs), and biologics, help to slow progression and manage symptoms. However, advancements in our understanding of the immunopathogenesis of PsA have opened up new therapeutic alternatives centered around regenerative medicine. Currently, mesenchymal stem cell (MSC) therapy has gained attention as a potential treatment for psoriatic arthritis as it may help reduce inflammation and support tissue regeneration. In this case study we report the clinical observations on a five-year post-treatment with mesenchymal stem cell therapy on a 68-year-old patient diagnosed with PsA. Initially, she presented with debilitating arthritis, severely limiting daily activities and overall quality of life. Conventional treatments offered only temporary relief, prompting the exploration of regenerative options. To address the underlying joint degeneration and inflammation, the patient underwent stem cell therapy as part of a personalized treatment plan. Mesenchymal stem cells (MSC) harvested from autologous adipocytes were used during treatment therapy. The protocol included injections directly into her left knee and a systemic intravenous (IV) drip on three separate occasions. The initial response was encouraging, with notable reductions in pain, and stiffness and improvements in joint function. Over the months following the procedure, the patient reported increased mobility, decreased reliance on pain medications, and a return to previous activities. Current clinical observations show a persistent, sustained, and stable treatment outcome. Since her last stem cell treatments five years ago, her ability to ambulate has improved with minimal discomfort. She can ascend stairs at five years post-treatment which was her primary objective. There was a significant reduction in psoriatic skin lesions from 80% of body surface area to <5%. This outcome is still maintained and stable, even five years post-treatment. The patient has elected not to use any biological preparations available, and control outbreaks with topical clobetasol cream only, as she is most accustomed to this therapy and has felt it to be successful for her needs. In this follow-up assessment, we evaluate the long-term outcomes of mesenchymal stem cell therapy concerning this case. Beyond initial symptom relief, the focus is on sustained improvements, potential setbacks, and any adjustments needed in the patient’s ongoing management. Understanding how the treatment has held up over time provides valuable insight into the durability of stem cell interventions for arthritis and their role in long-term joint health.
Methods
Five years ago, we published a case report study on psoriatic arthritis and stem cell therapy. The patient is currently a 68-year-old female who has a 25-year history of psoriatic arthritis accompanied by left knee pain, which interfered with her ambulatory ability. Her dermatopathology initially appeared as eczema in her teen years, involving all extremities and her thorax anteriorly and posteriorly. Over time, the eczema progressed to psoriasis at about age 40 years, and the psoriasis progressed to psoriatic arthritis at approximately age 60. Eighty percent of her skin body surface area was affected by the skin disease. Methotrexate usage controlled her skin lesions initially, but as the disease progressed, it lost its efficaciousness after four years of usage and was discontinued by her physician. Attempts to control her skin lesions were abandoned due to her allergies to NSAIDs, and lack of continued responsivity to methotrexate. Clobetasol usually successfully reduced her lesions to erythema on her thorax, however, the extremity symptomology was somewhat more difficult to control. Her physician entertained the idea of starting her on Enbrel® (etanercept), however, she had a family history of lung cancer, so it was determined not to be a viable possibility. Ustekinumab (Stelara®), which blocks interleukins, also was not attempted. Neither were Apremilast (Otezla®) or Secukinumab (Cosentyx®) seemingly due to failed therapy with methotrexate in the past. Orthopedically, there was a history of pes planus and genu varum. There was also a history of hypertension, occasional benign preventricular contractions, and hypercholesterolemia. Medication at the time of examination included lisinopril, hydrochlorothiazide, and rosuvastatin. Allergies included non-steroidal anti-inflammatory drugs, and penicillin, both of which produced wheezing and urticaria. Lesions of the extremities had an erythematous base around a central plaque. The left knee showed mild pain on palpation of the joint medially and laterally, and on range of motion with full knee flexion and extension. There was a click on knee extension – a positive McMurray test, and a negative drawer sign. Her hands exhibited mild joint soreness, but overt joint deformities were absent. Clubbing was absent. Joint deformities were likewise absent on all extremities. Mesenchymal stem cells (MSC) isolated from autologous adipocytes were harvested with intravenous (IV) sedation utilizing a super-wet technique and the Tulip aspiration system. One hundred twenty ml of adipocyte lipoaspirate was sent to the laboratory for cell isolation. This was achieved via ultrasound separation at Advanced Therapeutics Labs in New York City. An intra-articular left knee injection was performed under 1% Lidocaine local anesthesia, and a drip of intravenous mesenchymal stem cells was administered. Cell counts determined by the lab were 336,000,000. Twenty-four hours later, the patient returned to work. Her discomfort rapidly improved, and reported that she could ascend steps with mild residual discomfort. One week postoperatively, she noticed that her skin lesions had markedly improved. By four weeks, all skin lesions had disappeared completely, although she still had mild left knee pain. Skin lesions were absent completely for seven months postoperatively. After this period, they began to return on the thorax and extremities. She did receive two additional interarticular injections into the left knee with “some improvement” in her discomfort but did not receive any additional systemic intravenous therapy (other than the single treatment at the time of cell harvest). When the skin lesions returned, they were not as severe as the usual outbreaks that occurred before stem cell treatment. After one year, they were limited to clusters on the lower extremities, elbows, and thorax.
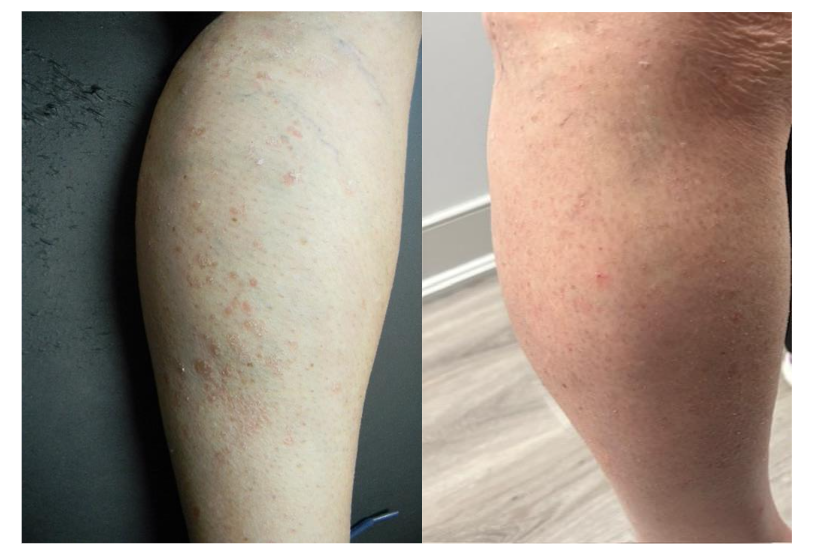
Over five years, no further treatment therapy utilizing stem cells was performed for her arthritic knee. Current clinical observations show a persistent, sustained, and stable treatment outcome. Since her last stem cell treatments five years ago, her ability to ambulate has improved with minimal discomfort. She can easily ascend stairs at five years post-treatment, which was her primary objective. There was a significant reduction in psoriatic skin lesions, reduced from 80% body surface coverage to <5% (Figure 1). This outcome is still maintained and stable, even at five years post-treatment. The patient has elected not to use any biological preparations available, and control outbreaks with topical clobetasol cream only, as she is most accustomed to this therapy and has felt it to be successful for her needs.
Discussion
The successful long-term outcome of this 68-year-old patient with psoriatic arthritis treated with stem cell therapy highlights the potential of regenerative medicine in managing chronic autoimmune conditions. Over five years, the patient maintained a significant reduction in psoriatic lesions, from 80% body coverage to just <5%, demonstrating both the durability and efficacy of the treatment. The sustained improvement suggests that stem cell therapy may modulate the immune response in a fashion that traditional treatments do not, offering a promising alternative for patients with severe, treatment-resistant psoriasis and arthritis. Additionally, the absence of major adverse effects in this case report supports the safety profile of stem cell interventions in autoimmune disease management. While this case presents compelling evidence for the long-term benefits of stem cell therapy, further research is necessary to validate these findings in larger, controlled clinical trials. The mechanisms behind the immune modulation observed in this patient remain an area for future investigation, particularly in understanding how stem cells influence chronic inflammatory pathways in psoriatic arthritis. Recent research indicates that MSC therapy has demonstrated encouraging long-term results across a wide range of therapeutic fields, such as tissue regeneration and immune system regulation. Research has highlighted that MSCs can self-renew, multi-differentiate, and secrete bioactive molecules that aid in tissue repair and modulate immune responses. For instance, they can differentiate into osteoblasts, chondrocytes, and adipocytes, epithelial cells, immune cells by promoting bone/cartilage repair, skin regeneration, and wound healing. Such features enable them to not only replicate themselves to sustain the stem cell population but also to generate specialized cells that aid in tissue repair and regeneration. Additionally, MSCs have powerful immunomodulatory properties that enable them to affect immune responses through the release of cytokines, chemokines, and growth factors that possess anti-inflammatory effects. They act as immunosuppressive agents by inhibiting the activation of T-cells, dendritic cells, and macrophages, which are involved in the inflammation seen in PsA. Key anti-inflammatory cytokines like interleukin-10 (IL-10), and transforming growth factor-beta (TGF-β) secreted by MSCs help reduce pro-inflammatory cytokines (TNF-α, IL-17, IL-23, IL-6, interferon-gamma) seen in PsA condition. Also, IL-10 inhibits the activation and proliferation of pro-inflammatory T-helper cells and promotes regulatory T-cell differentiation (anti-inflammatory) which is critical in PsA inflammation. Whereas TGF-β helps control the immune response by suppressing the activation/polarization of macrophages (shift from M1 pro-inflammatory to M2 anti-inflammatory). This shift creates an immunosuppressive microenvironment that helps resolve the inflammatory state seen in PsA pathology. Recent findings reveal that the positive effects of MSCs are also associated with the production of extracellular vehicles (EVs). These bioactive molecules play a key role in influencing cell behavior, and cell-cell communication in the surrounding tissue. Moreover, MSC-derived EVs can modulate inflammation by delivering anti-inflammatory signals to target cells which aids in decreasing systemic inflammation in PsA. Growth factors such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), insulin-like growth factor (IGF), and hepatocyte growth factor (HGF) reduce fibrosis, facilitate angiogenesis, cell proliferation, and migration at the lesion site. This process helps repair the synovial tissue and skin lesions which are characteristic of PsA pathology. Based on the clinical observations of this follow-up study, we suggest that MSC therapy on the patient may have played an essential role in promoting ongoing tissue repair (skin and synovial lesions) and reducing chronic inflammation in the long term. The patient showed a significant reduction in psoriatic lesions, with body coverage decreasing from 80% to just under 5% over time. We suggest that the migration characteristics of MSCs to sites of skin lesions may have played a role in this improvement. This migration may have suppressed the activity of immune cells like T cells and macrophages/dendritic cells in the skin, leading to a reduced production of their pro-inflammatory cytokines which play a key role in the development and progression of psoriasis. Furthermore, MSCs might have improved the healing of psoriatic lesions by stimulating the regeneration of epithelial cells and decreasing the excessive proliferation of keratinocytes. Since MSCs contribute to the remodeling of the extracellular matrix by releasing matrix metalloproteinases (MMPs) that break down damaged matrix components, we propose that this mechanism may have facilitated skin tissue healing and alleviated chronic inflammation in the affected joints. Expanding research efforts could refine patient selection criteria, optimize treatment protocols, and potentially lead to broader clinical applications of stem cell therapy in autoimmune diseases. This case underscores the transformative potential of regenerative medicine and supports the continued exploration of stem cell-based treatments for refractory psoriatic arthritis. The fact that the improvement was not complete but remained stable, indicates that the treatment provided a lasting but partial remission. The improvement suggests they may have altered the underlying immune dysregulation driving psoriatic arthritis, at least in the skin. This case is nonetheless a single-patient observation, so we cannot generalize the findings without more cases and controlled studies. Our patient was treated for an arthritic joint, and the skin outcome was an incidental finding, Further treatment, and follow-up research (e.g., biomarkers, imaging, and additional patients) would help determine mechanisms, optimal dosing, and long-term safety, which we strongly advocate for.
Conclusion
Initially, our patient sought medical attention not for dermatopathological resolution, but for joint pain relief from PsA. After being treated with adipose-derived mesenchymal stem cells significant improvements were noted. All of her psoriatic skin lesions disappeared within two months, and she remained asymptomatic for seven months without any further treatment. When her lesions did return, they were of much less severity than previously seen. Discomfort in the left knee was also improved and sustained. Over the five years of follow-up, the patient’s clinical reports show ongoing improvements in mobility, minimal discomfort, and a stable, substantial reduction in psoriatic skin lesions, with the affected body surface area decreasing from 80% to <5%. The outcome and patient satisfaction were better than expected. This case report is significant because the effect has been enduring and sustainable, and multiple treatments were not required after the initial encounter (three treatments). Our study not only highlights the benefits observed in a single patient but also contributes to the broader discussion of regenerative medicine in arthritis care. By analyzing the patient’s progress months or even years after treatment, we can better understand the factors influencing success and identify the next steps in optimizing stem cell applications for similar cases. However, additional research and clinical trials are needed to comprehensively determine the long-term benefits and limitations of MSC therapy, particularly in a clinical setting.
Conflict of Interest
The authors do not have any conflicts of interest in the preparation of this manuscript.
References
- Azuaga AB, Ramírez J, Cañete JD. Psoriatic Arthritis: Pathogenesis and Targeted Therapies. Int J Mol Sci. 2023;24(5):4901. Published 2023 Mar 3. doi:10.3390/ijms24054901
- Ocampo D V, Gladman D. Psoriatic arthritis. F1000Res. 2019;8:F1000 Faculty Rev-1665. Published 2019 Sep 20. doi:10.12688/f1000research.19144.1
- Kang EJ, Kavanaugh A. Psoriatic arthritis: latest treatments and their place in therapy. Ther Adv Chronic Dis. 2015;6(4):194-203. doi:10.1177/2040622315582354
- Dreschnack PA, M Scott. “Case Report: Psoriatic Arthritis and Stem Cell Therapy.” J Regen Med. 2020, 9:1
- Minev T, Balbuena S, Gill JM, Marincola FM, Kesari S, Lin F. Mesenchymal stem cells – the secret agents of cancer immunotherapy: Promises, challenges, and surprising twists. Oncotarget. 2024;15:793-805. Published 2024 Nov 22. doi:10.18632/oncotarget.28672
- Wu S, Sun S, Fu W, Yang Z, Yao H, Zhang Z. The Role and Prospects of Mesenchymal Stem Cells in Skin Repair and Regeneration. Biomedicines. 2024;12(4):743. Published 2024 Mar 27. doi:10.3390/biomedicines12040743
- Krampera M, Glennie S, Dyson J, et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood. 2003;101(9):3722-3729. doi:10.1182/blood-2002-07-2104
- Choi JJ, Yoo SA, Park SJ, et al. Mesenchymal stem cells overexpressing interleukin-10 attenuate collagen-induced arthritis in mice. Clin Exp Immunol. 2008;153(2):269-276. doi:10.1111/j.1365-2249.2008.03683.x
- Farzamfar S, Garcia LM, Rahmani M, Bolduc S. Navigating the Immunological Crossroads: Mesenchymal Stem/Stromal Cells as Architects of Inflammatory Harmony in Tissue-Engineered Constructs. Bioengineering (Basel). 2024;11(5):494. Published 2024 May 16. doi:10.3390/bioengineering11050494
- Dabrowska S, Andrzejewska A, Janowski M, Lukomska B. Immunomodulatory and Regenerative Effects of Mesenchymal Stem Cells and Extracellular Vesicles: Therapeutic Outlook for Inflammatory and Degenerative Diseases. Front Immunol. 2021;11:591065. Published 2021 Feb 5. doi:10.3389/fimmu.2020.591065
- Harting MT, Srivastava AK, Zhaorigetu S, et al. Inflammation-Stimulated Mesenchymal Stromal Cell-Derived Extracellular Vesicles Attenuate Inflammation. Stem Cells. 2018;36(1):79-90. doi:10.1002/stem.2730
- Zhang, B., Gao, S., Liu, S., Gong, X., Wu, J., Zhang, Y., Ma, L., & Sheng, L. (2024). Regenerative mechanisms of stem cells and their clinical applications for degenerative eye diseases. Journal of Research in Medical Sciences.29(1):42. DOI: 10.4103/jrms.jrms_358_23

