Remote Monitoring of Novel Cardiac Devices in Heart Failure
Utilizing Remote Patient Monitoring Tools to Evaluate Novel Implanted Cardiac Devices for Heart Failure Patients – A Case Series
Pratik Doshi¹, David Valdivia¹, Ubayd Haq¹, Julia Huang¹, Michael W. Fong²*
- Department of Internal Medicine, Keck School of Medicine of USC, University of Southern California
- Professor of Clinical Medicine, Division of Cardiovascular Medicine, Keck School of Medicine of USC, University of Southern California
Email: [email protected]
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Doshi, P., Valdivia, D., et al., 2025. Utilizing Remote Patient Monitoring Tools to Evaluate Novel Implanted Cardiac Devices for Heart Failure Patients – A Case Series. Medical Research Archives, [online] 13(4). https://doi.org/10.18103/mra.v13i4.6414
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6414
ISSN 2375-1924
ABSTRACT
Heart failure (HF) remains a leading cause of morbidity, mortality, and healthcare expenditures. A surge in novel technologies targeting different components of chronic HF pathophysiology has emerged, including the CardioMEMS pulmonary artery pressure monitor, the carotid barostimulator (Barostim Neo™) and the Bodyport cardiac scale. Despite the growing availability of remote solutions, the limited availability of robust studies has led to lower-level recommendations by national cardiology societies which may limit their adoption. One way to generate practical application data is by integrating established real-time hemodynamic monitoring devices with novel innovations, providing real-world evidence to support emerging solutions. In this case series, we review remote patient monitoring tools and their ability to evaluate novel implanted cardiovascular devices. This study aims to illustrate the benefit of clinical integration of these technologies and advocate for further use of this approach to enhance their real-world applicability in patient care.
Keywords
Heart failure, remote patient monitoring, CardioMEMS, Barostim Neo, Bodyport cardiac scale, cardiovascular devices
Introduction
Heart failure (HF) remains a leading cause of morbidity, mortality, and healthcare expenditures. Along with advances in guideline-directed therapies, the prevalence of HF is rising with projections estimating over 8 million affected adults in the United States by 2030. Effective outpatient management is essential to reduce hospitalizations. The complexity of HF management is compounded by patient adherence, financial constraints, and sociodemographic disparities requiring a multidisciplinary team-based approach. As prevalence continues to grow, interventions needed to continue improving mortality and quality of life may be limited as identification of decompensated HF remains rooted in physical examination findings which have limited positive predictive value.
To address this limitation, the use of remote patient monitoring (RPM) tools allows patients and providers to identify decompensation earlier in the course, and titrate treatment appropriately. The CardioMEMS pulmonary artery pressure monitor is an implantable wireless monitoring system that provides direct hemodynamic change detection and is well-established in HF management. The Bodyport Cardiac Scale is an emerging technology that employs a technology-enabled weight scale to track changes in multiple noninvasive biomarkers that generate a fluid vector to inform worsening volume status and alert impending heart failure exacerbations; potentially offering complementary information. Despite the growing availability of remote solutions, the limited availability of robust studies has led to a 2b recommendation of CardioMEMS by the American College of Cardiology and the American Heart Association. Nevertheless, these RPM devices continue garnering attention from experts to supplement the efforts of reducing HF hospitalization rates.
A surge in novel technology targeting treatment through different components of chronic HF pathophysiology has emerged. Recent innovations that have captured significant attention from the HF academic community and industry partners include: Barostim Neo™ and V-Wave interatrial shunt. The Barostim Neo™ device utilizes carotid baroreceptor stimulation through activation of the parasympathetic nervous system to improve cardiac function. The V-Wave interatrial shunt is another device that works by physically offloading the failing left heart, allowing for pressure and volume relief.
Although there has been a growth in the number of novel technological interventions, there is hesitation from patients, clinicians, and health systems to adopt them given the limited ability to assess clinical benefit in a practical setting. To this end, we aim to demonstrate through this case series how an integrative approach combining established RPM devices can be used to evaluate the efficacy of new technologies and augment evidence of their practical use.
Methods
We reviewed two cases demonstrating the utility of novel technologies, the Barostim Neo™ neurostimulator, and the V-Wave interatrial shunt, using two different RPM devices, the CardioMEMS monitoring system and the Bodyport Cardiac Scale. We also reviewed a case comparing the noninvasive and invasive RPM devices.
Cases
#1: RPM TOOL EVALUATING NOVEL NEUROHORMONAL STIMULATION DEVICE
Baroreceptor activation therapy (BAT) stimulates carotid baroreceptors to increase parasympathetic activity, leading to a reduction in afterload and cardiac remodeling. BAT is FDA approved for use in patients with LVEF≤ 35%, yet device use has been limited partly due to a lack of data demonstrating long-term clinical efficacy. CardioMEMS, a pulmonary artery pressure sensor with RPM capacity that is implanted in the pulmonary artery, has achieved widespread clinical use for remote monitoring in HF patients.
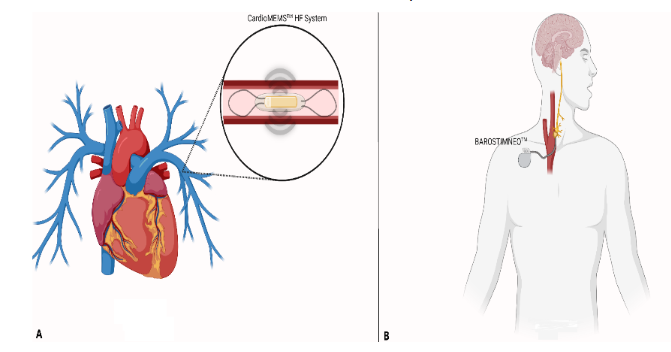
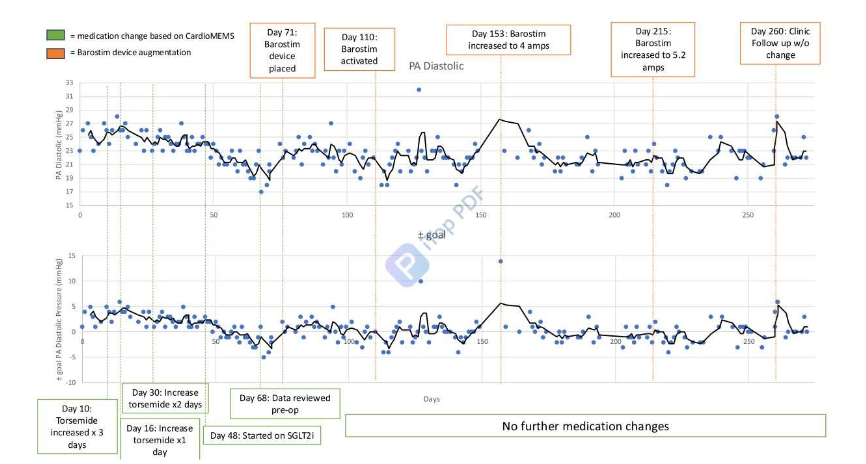
An 88-year-old (y/o) male with a history of ischemic cardiomyopathy (LVEF 34%), status post CardioMEMS, presented with severe tricuspid regurgitation, however, was deemed not to be a surgical candidate for repair. He was subsequently approved for and underwent BAT device placement, illustrated in Figure 1B below. Prior to BAT, he had chronically elevated PA diastolic pressure (PADP) requiring nearly weekly adjustment to diuretic therapy. Following implantation of BAT, the patient required no additional medication adjustments to maintain PADP goal of 22 mmHg. BAT demonstrated clinical efficacy in reducing vascular congestion without additional medication titration as demonstrated by the consistent PADP data presented via the CardioMEMS device, captured in Figure 2.
#2: INVASIVE VERSUS NONINVASIVE RPM TOOLS FOR HEART FAILURE
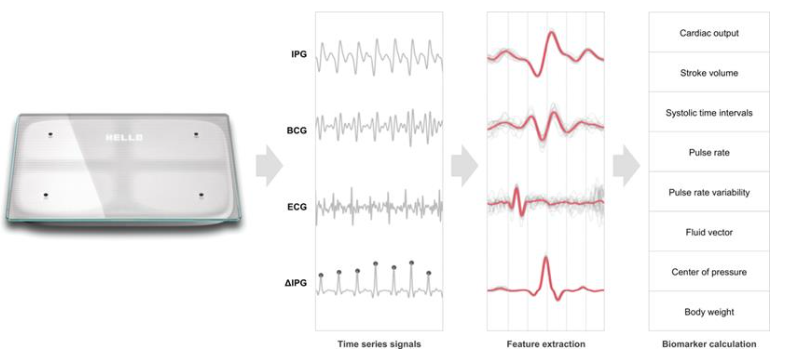
The Bodyport Cardiac Scale measures hemodynamic biomarkers via embedded sensors in a weight scale. In this case study, we qualitatively compared the performance of the Bodyport Cardiac Scale (weight, fluid vector, heart rate, illustrated in Figure 3) and CardioMEMS in the longitudinal management of a patient with HF.
A 71 y/o female with HFpEF (NYHA III, Stage C) and group 2 pulmonary hypertension was monitored for evidence of volume overload, with data captured on Figure 4 below. On six occasions the patient had an increased fluid vector and weight which improved with additional bumetanide for 1-7 days. The pulmonary artery diastolic pressure (PADP) as measured by the CardioMEMS device remained elevated with wide variability at each timepoint. On day 89, the patient reported shortness of breath with an increased fluid vector; however, her PADP of 25mmHg was below her average of 26mmHg. Increased diuretic for 2 days improved her fluid vector, PADP, and symptoms.
In a patient with HFpEF and pulmonary hypertension, chronically elevated (>25mmHg) and highly variable PADP made management of her volume status challenging despite there being an implanted device. The fluid vector from the cardiac scale trended with the patient’s clinical status and enhanced the team’s ability to remotely manage her HF.
#3: NONINVASIVE RPM TO EVALUATE INTRACARDIAC DEVICE EFFICACY
Managing heart failure in patients with complex structural heart disease often requires a multimodal approach that integrates remote monitoring strategies with advanced therapies to optimize care. The MitraClip, a transcatheter mitral valve repair device, has been shown to improve outcomes in patients with mitral regurgitation and heart failure by reducing symptoms and hospitalizations. Similarly, the V-Wave interatrial shunt aims to alleviate left atrial pressure and mitigate congestion in HF patients.
Complementing these interventional therapies, the Bodyport scale provides non-invasive, remote fluid status monitoring, enabling early detection of congestion and timely intervention.
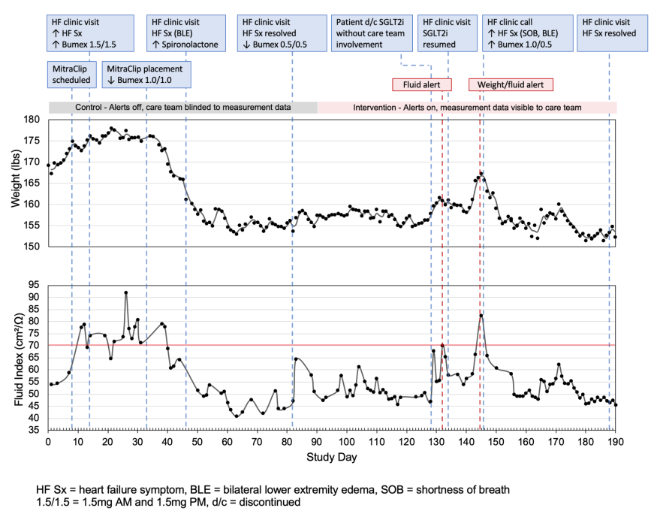
An 81 y/o male with a medical history including ischemic HFrEF (EF 26%), coronary artery bypass grafting with redo procedure, severe aortic stenosis now status post (s/p) transcatheter aortic valve replacement (TAVR) procedure, s/p V-wave interatrial shunt placement (2019), and s/p CRT-D placement, presented with shortness of breath secondary to volume overload. The patient was noted to have had three separate admissions within a six-month period for HF exacerbations. Subsequently, this patient was enrolled in the USC and Bodyport Remote Heart Failure Management Study (NCT04975633) for closer fluid status monitoring, with eventual MitraClip placement for moderate-to-severe mitral regurgitation contributory to HF.
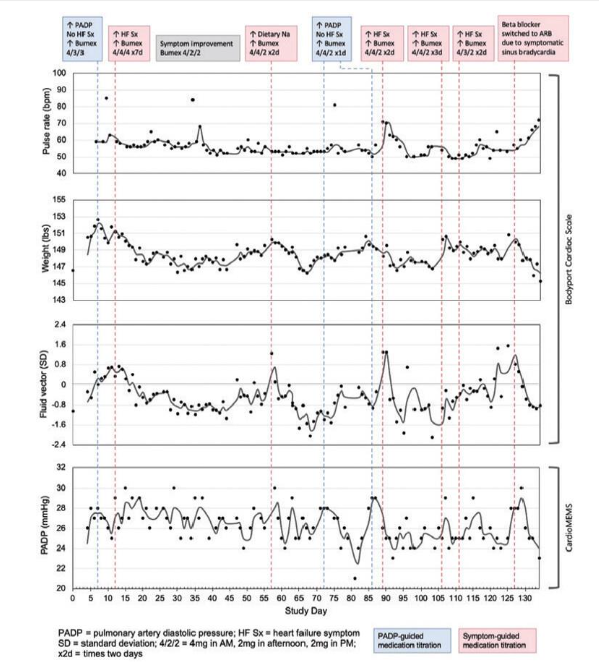
In this patient with recurrent presentations for HF and MitraClip placement for contributory valvular disease, BodyPort monitoring of fluid index and weight was crucial in titration of home diuretics and GDMT post-procedurally, as shown in Figure 5 below. The patient felt well after MitraClip placement and independently discontinued SGLT2i and decreased home diuretic dosing but managed to avoid hospitalization with the benefit of prudent intervention based on remote monitoring.
Discussion
This case series demonstrates practical applications of remote patient monitoring tools, both invasive and non-invasive, to evaluate the real world applicability of novel or investigational medical devices in the management of patients with HF. Each case provides specific insights into the integration of RPM with advanced therapies, offering actionable lessons for clinicians.
In Case 1, the integration of CardioMEMS, an invasive RPM tool, with BAT demonstrated the utility of RPM in objectively assessing the efficacy of a novel device outside of a standard clinical trial setting. This case involves an 88-year-old male with HFrEF and severe tricuspid regurgitation who was not a surgical candidate. He underwent implantation of a baroreflex activation device to improve symptomatic management of volume management. Prior to BAT, the patient required nearly weekly adjustment of his diuretic therapy. The stabilization of his PADP following BAT initiation without the need for diuretic adjustment provides clear evidence of BAT’s clinical benefit in reducing vascular congestion. This evidence is important as previously published clinical studies regarding BAT excluded patients with a similar profile (e.g. elderly patients who are poor surgical candidates). This underscores the value of continuous hemodynamic monitoring in optimizing device therapy for advanced HF patients, particularly in real-world settings where traditional clinical trial data may be lacking.
The head-to-head comparison of invasive (CardioMEMS) and noninvasive (Bodyport Cardiac Scale) further exemplifies how predicate RPM can be used to evaluate new medical technologies and devices. Case 2 involves a 71-year-old female with HFpEF and group 2 pulmonary hypertension with an existing CardioMEMS PA pressure monitoring device, who was given a noninvasive Bodyport scale that can measure fluid index through biometric sensors along with a patient’s weight. While CardioMEMS provided direct hemodynamic data, Bodyport offered actionable insights into fluid status through weight and fluid vector trends. Notably, Bodyport’s ability to detect early signs of fluid overload—even in the presence of variable PADP—demonstrates the potential of non-invasive tools to enhance remote HF management and guide therapeutic decisions.
Case 3 involved a complex patient with multiple cardiac interventions; a noninvasive RPM device (Bodyport Cardiac Scale) played a critical role in post-procedural monitoring following transcatheter mitral valve replacement (MitraClip). This case involves an 81-year-old male with ischemic HFrEF, with multiple inpatient admissions for HF exacerbation. He was monitored via the Bodyport scale and eventually underwent a MitraClip placement. The detection of fluid index and weight trends enabled timely adjustments to diuretics and guideline-directed medical therapy (GDMT), preventing hospitalization. This case exemplifies how non-invasive RPM can support the evaluation of structural heart interventions and improve outcomes in high-risk HF patients.
The cases presented here underscore several key principles for leveraging RPM to evaluate novel HF technologies in clinical practice:
- COMBINING NON-INVASIVE AND INVASIVE RPM TOOLS
As newer RPM tools enter the market, being able to compare the data generated by these tools with existing, validated RPM tools is critical for both patients and clinicians. We propose that, whenever possible, pairing non-invasive RPM (e.g., Bodyport Cardiac Scale) with invasive devices (e.g., CardioMEMS) provides a more comprehensive approach to HF management. While invasive tools offer precise hemodynamic data, non-invasive tools enhance accessibility, patient comfort, and longitudinal monitoring. This dual approach can improve the evaluation of novel devices by capturing both physiological and patient-centered outcomes. - INTEGRATING PATIENT SYMPTOM MONITORING
While not always presented in clinical studies as a primary endpoint, patient-reported outcomes (PROs) such as symptom management or quality of life are important markers to assess if a device has true real world applicability. Stated simply, if patients cannot experience a perceived improvement in their disease process, the uptake and distribution of these novel devices or technologies should be limited. For example, Bodyport’s ability to detect early signs of fluid overload, even in the absence of significant PADP changes, highlights the importance of integrating symptom monitoring into RPM strategies. - FACILITATING TIME-LIMITED TRIALS
RPM can support time-limited trials of novel devices by providing continuous, objective data on their efficacy. For instance, the use of CardioMEMS to monitor PADP post-BAT implantation provided clear evidence of clinical benefit within a defined follow-up period. Such trials can help address hesitancy among patients, clinicians, and health systems by generating real-world evidence of device performance.
This case series sought to demonstrate examples of the transformative potential of RPM in evaluating novel HF technologies. By pairing established RPM tools with investigational devices, clinicians can generate real-world evidence to support their adoption and integration into clinical practice. The cases highlight the importance of combining invasive and non-invasive diagnostics, incorporating patient symptom monitoring, and leveraging RPM for diuretic and outcome measurement. As the field of HF management continues to evolve, RPM will play an increasingly critical role in bridging the gap between clinical trials and real-world practice. Future research should focus on standardizing RPM protocols, developing predictive algorithms, and addressing barriers to implementation, such as cost and patient adherence.
Conclusion
In conclusion, RPM represents a powerful tool for evaluating and optimizing novel HF therapies, and may ultimately improve outcomes for patients with this complex and heterogeneous condition. Its integration into clinical practice can provide clinicians with the evidence needed to confidently adopt innovative technologies, ensuring that patients benefit from advancements in HF care.
Conflict of Interest:
Michael Fong MD is a speaker for CVRx.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Heidenreich PA, Albert NM, Allen LA, Bluemke DA, Butler J, Fonarow GC, Ikonomidis JS, Khavjou O, Konstam MA, Maddox TM, Nichol G, Pham M, Piña IL, Trogdon JG; American Heart Association Advocacy Coordinating Committee; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Radiology and Intervention; Council on Clinical Cardiology; Council on Epidemiology and Prevention; Stroke Council. Forecasting the impact of heart failure in the United States: a policy statement from the American Heart Association. Circ Heart Fail. 2013 May;6(3):606-19. doi: 10.1161/HHF.0b013e318291329a. Epub 2013 Apr 24. PMID: 23616602; PMCID: PMC3908895.
- Solomon SD, Dobson J, Pocock S, Skali H, McMurray JJ, Granger CB, Yusuf S, Swedberg K, Young JB, Michelson EL, Pfeffer MA; Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity (CHARM) Investigators. Influence of nonfatal hospitalization for heart failure on subsequent mortality in patients with chronic heart failure. Circulation. 2007 Sep 25;116(13):1482-7. doi: 10.1161/CIRCULATIONAHA.107.696906. Epub 2007 Aug 27. PMID: 17724259.
- Creaser JW, DePasquale EC, Vandenbogaart E, et al. Team-based care for outpatients with heart failure. Heart Fail Clin. 2015;11:379–405.
- Gilchrist SC, Barac A, Ades PA, Alfano CM, Franklin BA, Jones LW, La Gerche A, Ligibel JA, Lopez G, Madan K, Oeffinger KC, Salamone J, Scott JM, Squires RW, Thomas RJ, Treat-Jacobson DJ, Wright JS; American Heart Association Exercise, Cardiac Rehabilitation, and Secondary Prevention Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Peripheral Vascular Disease. Cardio-Oncology Rehabilitation to Manage Cardiovascular Outcomes in Cancer Patients and Survivors: A Scientific Statement From the American Heart Association. Circulation. 2019 May 21;139(21):e997-e1012. doi: 10.1161/CIR.0000000000000679. PMID: 30955352.
- Fudim M, Yazdi D, Egolum U, Haghighat A, Kottam A, Sauer AJ, Shah H, Kumar P, Rakita V, Centen C, Ozonat K, Smith S, DeVore AD. (2023). Use of a Cardiac Scale to Predict Heart Failure Events: Design of SCALE-HF 1. Circulation: Heart Failure, 16(5), e010012. doi:10.1161/CIRCHEARTFAILURE.122.010012.
- Abraham WT, Adamson PB, Bourge RC, et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: a randomised controlled trial. Lancet. 2011;377:658–666.
- Ciotola F, Pyxaras S, Rittger H, Buia V. (2024). MEMS Technology in Cardiology: Advancements and Applications in Heart Failure Management Focusing on the CardioMEMS Device. Sensors (Basel, Switzerland), 24(9), 2922. doi:10.3390/s24092922.
- Manavi T, Zafar H, Sharif F. (2024). An Era of Digital Healthcare—a Comprehensive Review of Sensor Technologies and Telehealth Advancements in Chronic Heart Failure Management. Sensors (Basel, Switzerland), 24(8), 2546. doi:10.3390/s24082546.
- Heart Failure Society of America (HFSA). (2024). HFSA Scientific Statement: Update on Device-Based Therapies in Heart Failure. Available at: https://hfsa.org/hf-stats-2024-heart-failure-epidemiology-and-outcomes-statistics (Accessed: 27 February 2025).
- Reed GW, Harmon EK, Harb S, et al. Design and Rationale of the v-Wave Shunt MitraClip Study. Am J Cardiol. 2024;227:29-36. doi:10.1016/j.amjcard.2024.06.023.
- Fudim M, Centen C, Mahoney D, et al. Surveillance and alert-based multiparameter monitoring to reduce worsening heart failure events: results from SCALE-HF 1.J Card Fail. 2024;30(2):145-153. doi:10.1016/j.cardfail.2023.11.005.
- Zile MR, Abraham WT, Weaver FA, et al. Baroreflex activation therapy in patients with heart failure and a reduced ejection fraction: long-term outcomes. Eur J Heart Fail. 2024;26(4):1051-1061. doi:10.1002/ejhf.3232.
- Abraham WT, Stevenson LW, Bourge RC, Lindenfeld JA, Bauman JG, Adamson PB. Sustained efficacy of pulmonary artery pressure to guide adjustment of chronic heart failure therapy: complete follow-up results from the CHAMPION randomized trial. Lancet. 2016;387(10017):453-461. doi:10.1016/S0140-6736(15)00723-0.
- Zile, M, Lindenfeld, J, Weaver, F. et al. Baroreflex Activation Therapy in Patients With Heart Failure With Reduced Ejection Fraction. J Am Coll Cardiol. 2020 Jul, 76 (1) 1–13. https://doi.org/10.1016/j.jacc.2020.05.015
- Radhoe, S. P., Clephas, P. R. D., Mokri, H., & Brugts, J. J. (2023). The CardioMEMS Heart Failure System for chronic heart failure – a European perspective. Expert Review of Medical Devices, 20(5), 349–356. https://doi.org/10.1080/17434440.2023.2196400
- Doshi, P, Valdivia, D, Pham, V. et al. ASSESSING THE EFFICACY OF BARORECEPTOR ACTIVATION THERAPY USING REMOTE PATIENT MONITORING: A CASE STUDY. J Am Coll Cardiol. 2024 Apr, 83 (13_Supplement) 3962. https://doi.org/10.1016/S0735-1097(24)05952-7
- Goodman, R, Aldea, D, Huang, C. et al. CASE OF CARDIAC SCALE AUGMENTING CARDIOMEMS TO ASSESS VOLUME STATUS IN OUTPATIENT HEART FAILURE MANAGEMENT. J Am Coll Cardiol. 2023 Mar, 81 (8_Supplement) 3467. https://doi.org/10.1016/S0735-1097(23)03911-6
- Stone GW, Lindenfeld J, Rodés-Cabau J, Anker SD, Zile MR, Kar S, Holcomb R, Pfeiffer MP, Bayes-Genis A, Bax JJ, Bank AJ, Costanzo MR, Verheye S, Roguin A, Filippatos G, Núñez J, Lee EC, Laufer-Perl M, Moravsky G, Litwin SE, Prihadi E, Gada H, Chung ES, Price MJ, Thohan V, Schewel D, Kumar S, Kische S, Shah KS, Donovan DJ, Zhang Y, Eigler NL, Abraham WT; RELIEVE-HF Investigators. Interatrial Shunt Treatment for Heart Failure: The Randomized RELIEVE-HF Trial. Circulation. 2024 Dec 10;150(24):1931-1943. doi: 10.1161/CIRCULATIONAHA.124.070870. Epub 2024 Sep 23. PMID: 39308371; PMCID: PMC11627316.

