Modified Triple Test Score for Breast Lumps Diagnosis
Modified Triple Test Score for Palpable Breast Lumps: The Utility of Breast Ultrasound and Core Needle Biopsy in Resource-Constrained Settings
Gana SG¹, Bello N², Olarinoye-Akorede SA², Samaila MO³, Yusufu LMD¹
- Department of Surgery, Ahmadu Bello University Teaching Hospital, Zaria.
- Department of Radiology, Ahmadu Bello University Teaching Hospital, Zaria.
- Department of Pathology, Ahmadu Bello University Teaching Hospital, Zaria.
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Gana, SG., Bello, N., et al., 2025. Modified Triple Test Score for Palpable Breast Lumps: The Utility of Breast Ultrasound and Core Needle Biopsy in Resource-Constrained Settings. Medical Research Archives, [online] 13(4).
https://doi.org/10.18103/mra.v13i4.6482
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6482
ISSN 2375-1924
Abstract
Background: Triple assessment is the standard for diagnosing breast lumps, involving clinical examination, breast imaging, and tissue diagnosis. Developed by Arden Morris et al. in 1998, the Triple Test Score evaluates palpable breast masses through clinical examination, mammography, and cytology. We used a modified triple test score with clinical examination, breast ultrasound, and core needle biopsy, aiming for rapid and accurate diagnosis for effective patient management.
Aims: Compare the Modified Triple Test Score across two biopsy guidance techniques for managing palpable breast lumps at Ahmadu Bello University Teaching Hospital, Zaria.
Methods: This randomized controlled study at Ahmadu Bello University Teaching Hospital in Zaria, Nigeria, involved patients divided into Group A for palpation-guided core needle biopsy and Group B for ultrasound-guided core needle biopsy. All patients underwent surgical biopsies. True positives, defined by concordant malignant findings, were exempt from open surgical biopsy to initiate oncologic multimodal therapy, including mastectomy. Definitive mastectomy histology reports replaced surgical biopsy results, with data analyzed using SPSS version 21.
Results: Data from 80 patients were analyzed, with 40 patients in each core needle biopsy group. The ages ranged from 18 to 72, with the peak age group (20-29 years) comprising 29 patients (36.3%). The Modified Triple Test Score was calculated using clinical breast examination, ultrasound, and core needle biopsy. In the palpation-guided group, the clinical breast examination had a sensitivity of 90.0% and a specificity of 77.8%. The breast ultrasound scan had a sensitivity of 100% and a specificity of 90.9%, and the core needle biopsy showed a sensitivity of 94.7% and a specificity of 100%, achieving a combined Modified Triple Test Score of 100%. In the ultrasound-guided group, the sensitivity and specificity of the clinical breast examination were 90.5% and 100%, respectively. The breast ultrasound scan and core needle biopsy achieved 100% sensitivity and specificity, with a combined Modified Triple Test Score of 100%.
Conclusion: This study confirms the value of the modified triple test score in evaluating breast lumps, demonstrating high accuracy for both core needle biopsy techniques when combined scores are used.
Keywords: Palpable breast lump, Core needle biopsy, Palpation-guided CNB, Ultrasound-guided CNB, Modified Triple Test Score.
Introduction
Triple assessment is the gold standard in diagnosing palpable breast lumps. It consists of clinical breast examination (CBE), imaging (mammography or breast ultrasound), and pathology (cytology or histology). When performed together with concordant results, diagnostic accuracy approaches 100%. The goal is to provide a swift and accurate diagnosis for proper patient management.
Breast diseases significantly contribute to surgical outpatient referrals, primarily affecting women of reproductive age, with most cases being benign. The incidence of benign lesions peaks in the second to fifth decades, while malignant cases rise postmenopausally, albeit more slowly. In Nigeria, benign breast diseases peak at ages 20-29, with malignant rates increasing from 33.6-54.3 per 100,000 over two decades, currently estimated at 116 per 100,000. Breast cancer is the most common cancer among women globally. CBE, a low-cost test, can enhance breast cancer detection, especially in developing countries where it’s often the sole screening method. Preliminary data indicate improved cancer staging at diagnosis, although sensitivity may be low if performed by untrained personnel. Therefore, enhanced training and quality control are essential for CBE’s effectiveness.
BREAST ULTRASOUND
Ultrasonography is essential in breast imaging. Introduced in the 1950s using radar techniques, its role evolved from distinguishing cystic and solid masses to better characterization of solid masses. Simple cysts typically need no further work, but complex lesions and indeterminate solid masses require biopsies for differentiation. Advances in technology, including high-frequency transducers, compound imaging, and Doppler imaging, have enhanced the characterization of solid masses. It is the preferred imaging choice for young women, improving the assessment of shape, margins, and internal structure. For women under 30, breast ultrasound remains the primary imaging method for palpable masses.
Ultrasonography features suggesting a malignant lesion include spiculations, angular margins, acoustic shadowing, marked hypoechogenicity, microcalcifications, ductal extension, and microlobulations. Lesions are typically taller than they are wide, with a non-parallel orientation to the skin, and may be associated with thickened Cooper ligaments or skin. The benefits of ultrasound for guiding percutaneous breast biopsy include no ionizing radiation, accessibility to all breast areas, real-time visualization, shorter procedure duration, and multi-directional sampling. However, the lesion must be visible on ultrasound to undergo biopsy.
NEEDLE BIOPSY
Fine needle aspiration cytology (FNAC) is used in diagnosing oncologic lesions. It is safe, inexpensive, and minimally invasive but has high inadequacy and false negative rates. FNAC cannot distinguish in situ from invasive cancer, and immunohistochemistry, crucial for neo-adjuvant chemotherapy, is challenging with FNAC. It requires skilled pathologists for accurate diagnosis.
Core needle biopsy (CNB) involves removing small breast tissue samples with a hollow core needle. CNB is less invasive and leads to fewer complications, shorter recovery, and lower treatment costs compared to open surgical biopsy. While many women seek biopsy for diagnosis certainty, some find biopsy-related disfigurement difficult to accept. CNB offers better patient tolerance, cosmetic outcomes, and lower costs than open biopsy. It results in less breast tissue trauma and minimal scarring, aiding in future imaging assessments. Non-palpable breast lesions are typically biopsied under imaging guidance, while palpable masses can be biopsied by palpation or imaging. Several techniques exist for CNB, utilizing palpation or imaging to locate lesions. A vacuum-assisted core needle device can also be employed for tissue sample removal.
Palpation-guided CNB involves securing the breast lump with the surgeon’s non-dominant hand while introducing a CNB device percutaneously to collect biopsy cores. Its advantages include convenience, rapid tissue sampling for histology and immunohistochemistry, and a low complication rate. However, it requires operator experience to sample lesions correctly and may struggle with small lesions. Tumors with significant hemorrhage and necrosis yield poor tissue samples, and the false-negative rate ranges from 0–36%, with an insufficiency rate of 2–10%.
Ultrasound-guided CNB provides real-time visualization of the CNB needle, enhancing sampling accuracy. Limitations include the lack of USS machines and expertise in many primary and secondary healthcare facilities, long wait times, and bureaucratic issues in busy breast clinics.
Both methods must ensure high sensitivity and specificity, with false negatives being rare. Possible causes of false negatives include sampling errors, misinterpretation of slides, and the use of an inappropriate needle size. Underestimation often results from not sampling all critical lesion areas, such as carcinoma foci among atypical cells. Core-needle techniques can miss carcinoma cells, leading to underestimation and incorrect staging.
RADIOLOGY-PATHOLOGY CORRELATION
All CNB pathology results must correlate with the prebiopsy BI-RADS impression, which can be concordant or discordant. A benign result with a BI-RADS 4 (especially 4B or 4C) or 5 classification is considered discordant until proven otherwise. Close discussion with the pathologist is essential to explain mammographic findings, particularly for architectural distortion or a mass. A benign finding may also be discordant if calcifications are identified pathologically, due to potential sampling errors. A repeat CNB should target a different lesion area, or further imaging (e.g., USS, MRI, or molecular breast imaging) should be used to assess the most suspicious area for biopsy. The Royal College of Radiologists Breast Group (RCRBG) breast imaging classification employs a five-point scale to assess suspicion of malignancy in the UK. Categories 1 (normal) and 2 (benign findings) do not need sampling. Category 3 lesions have a low malignancy risk (0.5–2%) and warrant further investigation. The RCRBG system, while similar to BI-RADS, recommends biopsy for equivocal or probably benign findings (BI-RADS 3) for definitive diagnosis rather than follow-up, which may cause psychological distress. Core needle biopsy is routinely performed for category 4 (suspicious for malignancy) and category 5 (highly suspicious) lesions, associated with malignancy risks of 33–50% and 90%, respectively.
MODIFIED TRIPLE TEST SCORE
The triple test score (TTS), introduced by Arden Morris et al. in 1998, evaluates palpable breast masses. It relies on clinical breast examination, mammography, and cytology for diagnosis, following Johansen Christer’s framework from 1975. Each parameter receives a score of 1, 2, or 3 for benign, suspicious, and malignant results, respectively, yielding a TTS between 3 (benign) and 9 (malignant). The TTS reduces the need for open surgical biopsies, allowing long-term follow-up for benign lesions and definitive management for malignant ones. The lower sensitivity of mammography in women with dense breasts and a preference for ultrasonography in younger women led to ultrasound adoption in these cases. The limitations of fine needle aspiration, such as distinguishing between in situ and invasive cancers and the lack of tissue for immunohistochemistry, also support the use of core needle biopsies (CNB) for histopathologic assessment.
JUSTIFICATION
At Ahmadu Bello University Teaching Hospital (ABUTH), Zaria, a core needle breast biopsy can be guided by palpation or ultrasound. The diagnostic accuracy of these techniques has not been documented. This study aims to compare the accuracy of core needle biopsy (CNB) and assess the Modified Triple Test Score (MTTS) for managing breast lumps in female patients at ABUTH. To the researcher’s knowledge, this is the only study to evaluate the usefulness of the modified triple test score (MTTS) at our center. It aims to enhance the existing literature in clinical research practice.
AIM
To compare the Modified Triple Test Score using the two biopsy guidance techniques in managing palpable breast lumps in ABUTH Zaria.
Methodology
STUDY DESIGN
This was a randomized, controlled comparative study.
STUDY SETTING
This study was conducted at Ahmadu Bello University Teaching Hospital (ABUTH), situated in Zaria, Kaduna State, Northwestern Nigeria.
STUDY DURATION
This study was conducted over a 12-month period from July 2018 to June 2019.
STUDY POPULATION
The study included all adult female patients who presented to the General Surgery Unit of ABUTH Zaria with palpable breast lump(s).
INCLUSION CRITERIA
All consenting adult (18 years and older) female patients with palpable breast lump(s) were included in this study.
EXCLUSION CRITERIA
- Patients with fungating breast tumours and locally advanced skin involvement
- Patients with breast abscesses
- Patients with uncorrected bleeding disorders
- Patients with breast lumps less than 2cm in the widest diameter
SAMPLE SIZE
The minimum sample size for this study was calculated using the Yamane formula.
n = N / [1 + N (e)²]
Assuming the attrition is 10% Corrected sample size for each group = n / [1 – (% attrition/100)] = 33 / [1 – (10/100)] The corrected sample size for each group = 36.67 ≈ 37 This means there will be a minimum of 37 patients in each group. Since there will be two groups, the minimum number of patients to be recruited for this study will be 37 x 2 = 74.
STUDY PROTOCOL
All female patients with palpable breast lumps meeting the inclusion criteria were recruited for the study. Clinical breast examinations were conducted by the researcher on consenting patients after counseling, with a chaperone present, at the surgical outpatient department (SOPD). Each patient then underwent breast ultrasound scans of both breasts and axillae by the consultant radiologist or senior registrar. The same Mindray Ultrasound machine (DC-8 series, 2013, China) was used for ultrasound-guided biopsies. The researcher performed all core needle breast biopsies using a 14G Geotek® semi-automatic CNB device (reference number GSNA1420, Turkey). Patients were randomized into two groups using SPSS-generated numbers, with each group comprising half of the total patients. Group A received palpation-guided core needle biopsy (CNB), and Group B underwent ultrasound-guided CNB. Group A procedures were performed in the SOPD, while Group B procedures were conducted in the radiology department under real-time ultrasound guidance. The interventional team members were blinded to the patient groups and the pathology results. All patients had surgical biopsies after CNB results. Those with clinically-radiologically-pathologically concordant malignant findings and MTTS consistent with malignancy were considered true positives and exempt from surgical biopsy to start oncologic multimodal therapy, including mastectomy. Their definitive mastectomy histology reports replaced surgical biopsy results.
GROUP A PATIENTS:
The procedure followed the standard aseptic technique. The patient lay supine with the arm abducted at the shoulder and flexed at the elbow, hand tucked under the occiput. The ipsilateral breast was cleaned with 10% Povidone-iodine, exposing a 10cm area over the lump. The researcher, wearing sterile gloves, injected 5ml of 2% lidocaine into the skin at the incision site. After test-firing the core biopsy device, the researcher used a size 11 surgical blade to make a 5mm incision for the CNB needle. Holding the lesion with the left hand, the biopsy needle was inserted with the right hand until it abutted the margin of the lump. The patient was notified before triggering the CNB device, which was then withdrawn. The specimen was placed in a vial containing 10% Formalin. This was repeated until 5 adequate cores were collected. The researcher deemed grey-white cores that sank in the formalin as suitable. After compressing the biopsy site for 5 minutes, it was cleaned with povidone-iodine and dressed with sterile gauze. The labeled biopsy specimen was sent to the Histopathologist.
GROUP B PATIENTS:
The procedure was conducted under aseptic conditions. The patient lay comfortably supine with the ipsilateral arm abducted at the shoulder and flexed at the elbow, palm under her head. An ultrasound scan of the breast lesion was performed by the consultant radiologist using a Mindray machine with a high-frequency probe (7.5-13MHz) to document the lesion. The lump was localized, and orthogonal measurements were taken. The best approach to the lesion was then discussed. The researcher prepared the ipsilateral breast with 10% Povidone-iodine while wearing sterile gloves, then anaesthetized the needle entry point and pathway to the lesion with 5 ml of 2% lidocaine under ultrasound guidance. The researcher coupled and tested the biopsy device as the local anaesthetic took effect, as indicated by the numbness in the infiltrated skin. The patient was familiarized with the device’s ‘click’ sound during test firing to reduce anxiety. A 5mm incision was made on the numb skin with a size 11 surgical blade, and the biopsy device was inserted through the anaesthetized pathway under real-time ultrasound guidance. Needle tip positions relative to the lump were visualized to confirm adequate tissue sampling before firing the device, with pictures taken for documentation. The device was withdrawn, and the specimen was carefully retrieved and placed in 10% Formalin, repeating until 5 adequate cores were obtained. Firm pressure was applied to the wound between biopsies and for at least 5 minutes post-procedure with sterile gauze to ensure haemostasis. The wound was cleaned with povidone-iodine and dressed in sterile gauze. The specimen was labeled and sent to the Histopathologist.
SURGICAL BIOPSY
Open surgical biopsy was carried out for each patient 1 week following the core needle biopsy and after the histology report was out. The procedure was performed in the operating theatre following adequate patient counseling and preparation. Informed written consent was obtained, and routine asepsis was ensured. The biopsy specimen was properly oriented and completely submerged in 10% Formalin for onward transportation to the histopathology laboratory. Subsequently, proper procedural documentation and patient follow-up were undertaken as described for the core needle biopsy procedures.
ETHICAL CONSIDERATIONS
Approval for this study was obtained from the Health Research Ethics Committee (HREC) of Ahmadu Bello University Teaching Hospital (ABUTH) Zaria. The nature of this study was carefully explained to the recruited patients and their caregivers in a language they understood, and written informed consent was obtained. Patient selection was unbiased, and all patient examinations and biopsy procedures were carried out in the presence of a chaperone. Patient participation was fully voluntary, and non-consenting patients did not incur any punitive treatment. Patients reserved the right to withdraw consent at any point during this study.
DATA ANALYSIS
Data obtained was entered into a structured pro forma for each patient and analyzed using the Statistical Program for the Social Sciences version 21 (SPSS, Chicago, IL. USA). The chi-square test was used to analyze categorical variables. A confidence interval of 95% was employed, and the p-value, P<0.05, was considered significant.
Results
Eighty patients had their data analyzed. Forty of these belonged to the palpation-guided CNB group, while the other 40 patients belonged to the ultrasound-guided CNB group.
SOCIO-DEMOGRAPHIC CHARACTERISTICS
The age range of patients in this study was 18-72. The peak age group with palpable breast lumps was the 20-29-year-old group, with 29 patients (36.3%). The age distribution of the patients is shown in Table 1.
| Age Group | Palpation guided group (%) | Ultrasound guided group (%) | Total Number of Patients (%) |
|---|---|---|---|
| < 20 | 1 (2.5) | 7 (17.5) | 8 (10.0) |
| 20-29 | 17 (42.5) | 12 (30.0) | 29 (36.3) |
| 30-39 | 10 (25.0) | 8 (20.0) | 18 (22.5) |
| 40-49 | 9 (22.5) | 9 (22.5) | 18 (22.5) |
| 50-59 | 2 (5.0) | 2 (5.0) | 4 (3.8) |
| 60-69 | 0 (0.0) | 2 (5.0) | 2 (2.5) |
| 70 & Older | 1 (2.5) | 0 (0.0) | 1 (1.3) |
| Total | 40 (100%) | 40 (100%) | 80 (100%) |
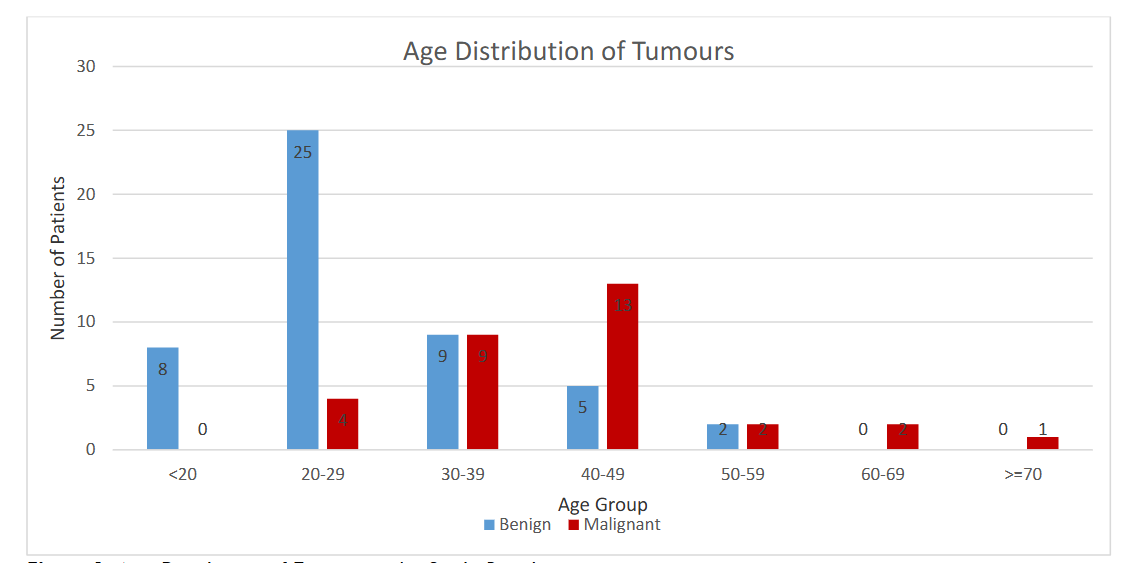
Twenty-five patients (51.0%) with benign lumps fall within the 20-29 years age group, while 13 patients (41.0%) with malignant lumps fall in the 40-49 years age group. Figure 1 shows the age distribution of benign and malignant tumours among the study participants.
MODIFIED TRIPLE TEST SCORE
The modified triple test score, computed using clinical breast examination (CBE), breast ultrasound scan, and core needle biopsy report, was computed for each patient in the 2 CNB arms in this study. Each parameter was scored independently in both groups.
CLINICAL BREAST EXAMINATION
In the Palpation-guided group, 20 patients scored 1 (Benign). Eighteen of these patients (90.0%) had benign histology reports on surgical biopsy, and 2 patients (10.0%) had malignant lesions. Eleven patients had a score of 2 (Suspicious). Five (45.5%) of them had benign lesions, while 6 (54.5%) had malignant lesions. Nine patients had a score of 3 (Malignant). Two (22.2%) of these had benign lesions, while the remaining 7 patients (77.8%) had malignant lesions. Table 7 summarizes the computed measures of accuracy for this group.
In the Ultrasound-guided group, 21 patients scored 1 (benign). Nineteen (90.5%) of these patients had benign lesions, while the other 2 (9.5%) had malignant lesions. Fifteen patients had a score of 2 (Suspicious). Five (33.3%) of these patients had benign lesions, while the remaining 10 (66.7%) had malignant lesions. Four patients had a score of 3 (Malignant). All of these 4 patients had malignant lesions following surgical biopsy.
BREAST ULTRASONOGRAPHY
In the Palpation guided group, 18 patients scored 1, and all (100%) had benign lesions. Eleven patients had a score of 2. Six (54.5%) of these had benign lesions, while 5 (45.5%) had malignant lesions. Eleven patients had a score of 3. One (9.1%) had a benign lesion, while 10 (90.9%) had malignant lesions.
In the Ultrasound-guided group, 17 patients scored 1, and all (100.0%) had benign lesions. Another 17 patients scored 2, 7 (41.2%) with benign lesions, while 10 (58.8%) had malignant lesions. Six patients scored 3, with all 6 (100.0%) having malignant lesions.
CORE NEEDLE BIOPSY
In the Palpation guided group, 19 patients scored 1 (Benign). Eighteen (94.7%) of these had a benign histology following surgical biopsy, while 1 (5.3%) had a malignant lesion. Eight patients had a score of 2 (Suspicious); 7 (87.5%) had benign lesions, while 1 (12.5%) had a malignant lesion. Thirteen patients scored 3 (Malignant), with all (100.0%) having malignant lesions.
In the Ultrasound-guided group, 24 patients scored 1, and all (100.0%) had benign lesions. Only 1 patient scored 2, and this patient (100.0%) had a malignant lesion. The final 15 patients scored 3, with all (100.0%) having malignant lesions.
Measures of Accuracy in the Palpation Guided CNB Group
| Measure of Accuracy | Clinical Breast Examination | Breast Ultrasonography | Core Needle Biopsy | MTTS |
|---|---|---|---|---|
| Sensitivity | 90.0% | 100.0% | 94.7% | 100.0% |
| Specificity | 77.8% | 90.9% | 100.0% | 100.0% |
Measures of Accuracy in the Ultrasound Guided CNB Group
| Measure Of Accuracy | Clinical Breast Examination | Breast Ultrasonography | Core Needle Biopsy | MTTS |
|---|---|---|---|---|
| Sensitivity | 90.5% | 100.0% | 100.0% | 100.0% |
| Specificity | 100.0% | 100.0% | 100.0% | 100.0% |
Figures 2 and 3 show the MTTS distribution for the Palpation-guided and Ultrasound-guided CNB groups, respectively.
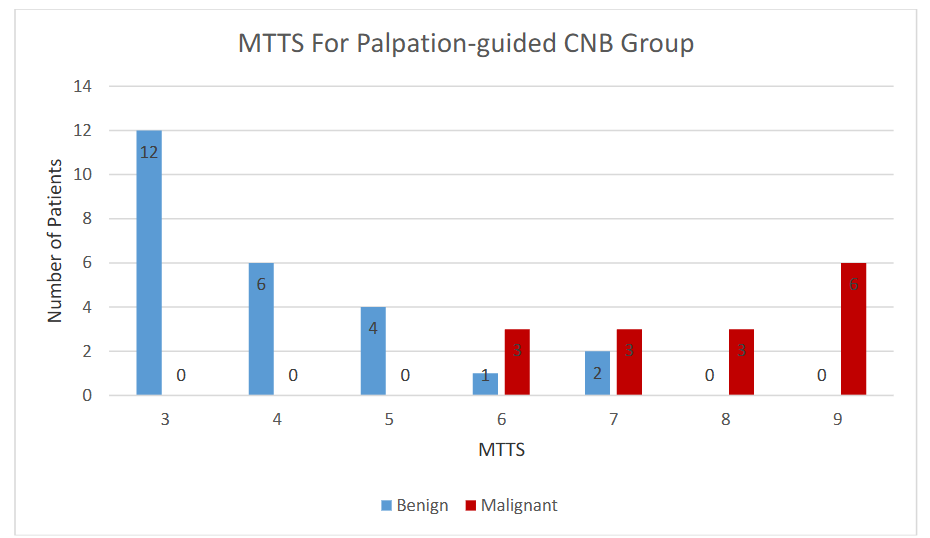
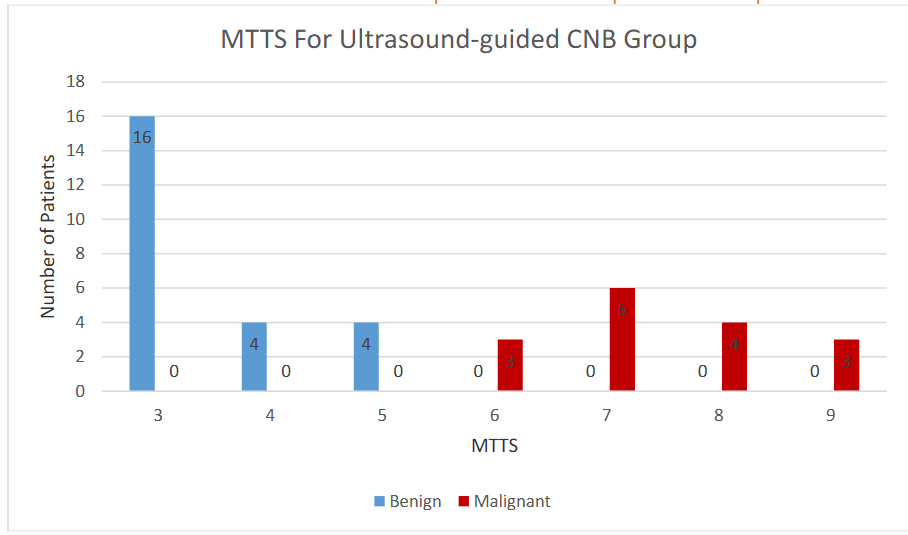
Distribution of MTTS and Histology Outcomes in the Study
| Score | Benign (%) | Malignant (%) | Total (%) |
|---|---|---|---|
| 3 | 28 (57.1) | 0 (0.0) | 28 (35.0) |
| 4 | 10 (20.4) | 0 (0.0) | 10 (12.5) |
| 5 | 8 (16.3) | 0 (0.0) | 8 (10.0) |
| 6 | 1 (2.0) | 6 (19.4) | 7 (8.8) |
| 7 | 2 (4.1) | 9 (29.0) | 11 (13.8) |
| 8 | 0 (0.0) | 7 (22.6) | 7 (8.8) |
| 9 | 0 (0.0) | 9 (29.0) | 9 (11.3) |
| Total | 49 (100.0) | 31 (100.0) | 80 (100.0) |
Figure 4 shows the overall MTTS distribution of the 80 patients in this study. Concordant benign (total score of 3) and malignant (total score of 9) showed a sensitivity and specificity of 100.0%, respectively. Total scores of 4, 5, and 8 also showed no overlap between benign and malignant lumps. Score 6 had 1 patient (2.0%) with benign and 6 patients (19.4%) with malignant lesions. Score 7 had 2 patients (4.1%) and 9 (29.0%) with malignant lesions.
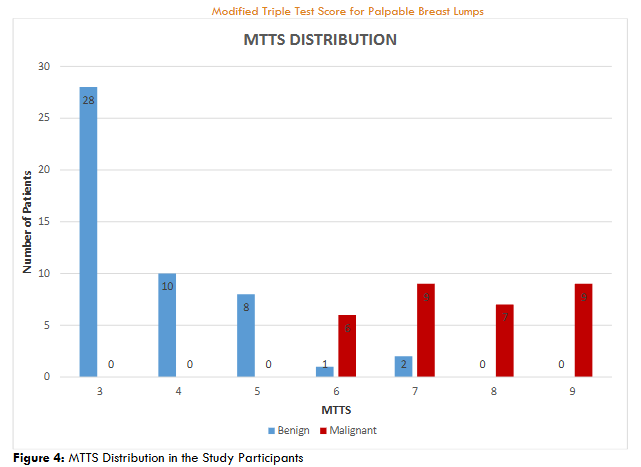
Figure 5 shows a breast mass (Fig. 5A) with real-time USS-guided CNB biopsy (Fig. 5B). The position of the needle tip is confirmed within the mass as representative tissue cores are obtained.
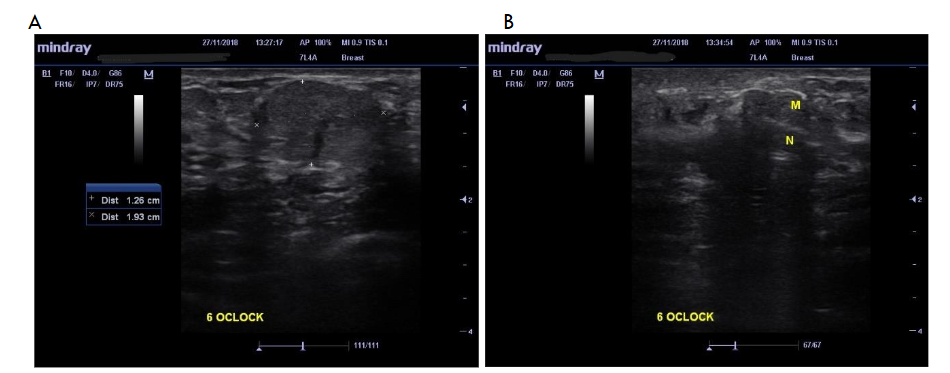
Figure 6 shows bilateral breast lumps in a patient. Fig. 6A illustrates a macrolobulated hypoechoic mass in the posterior aspect of the right breast at the 12 O’clock position. The mass is wider than tall, with posterior acoustic enhancement, and measures 2.96 x 1.92 cm in dimensions. Fig. 6B illustrates a well-defined oval-shaped hypoechoic mass in the left breast at the 3 O’clock position. It is wider than tall and measures 1.19 x 0.86 cm in dimensions.
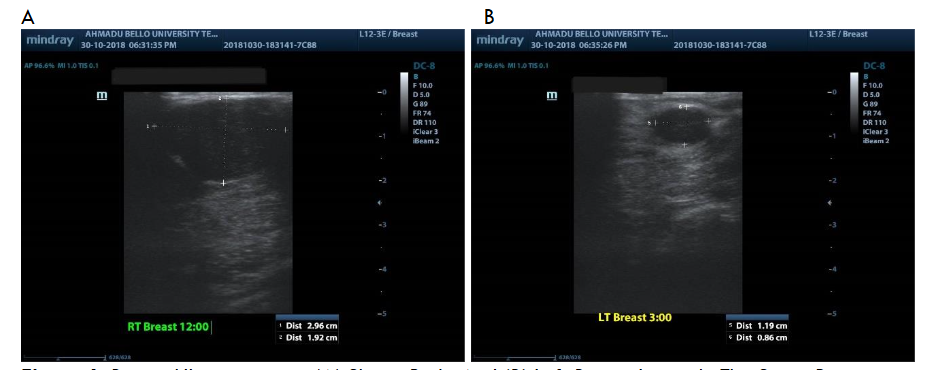
Figure 7(A) shows a CNB specimen displaying the distortion of the lobular architecture of the breast by a cellular tumour in diffuse sheets at x10 magnification. Figure 7(B) shows sheets of pleomorphic tumour cells with darkly stained nuclei and scant cytoplasm at x40 magnification.
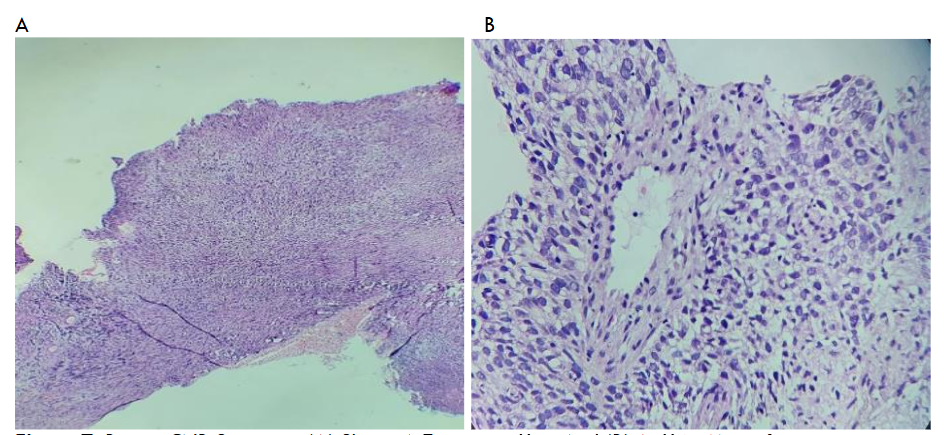
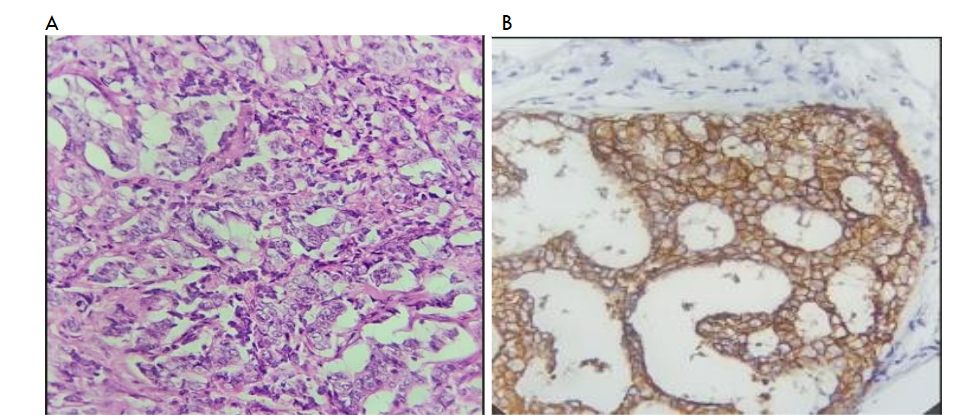
Figure 8(A) shows a surgical biopsy specimen with tumour cells at x40 magnification, while Figure 8(B) depicts breast immunohistochemistry showing complete membranous staining with HER2 (human epidermal growth factor receptor 2) positive breast tumour with a basketweave pattern at x40 magnification.
Discussion
The study revealed that the predominant age group with palpable breast lumps was the 20-29 year old group, which represented a significant 36.3% of the total patient population examined. Among these cases, benign breast lumps were particularly prevalent, constituting 51.0% of the instances, with fibroadenoma identified as the most commonly diagnosed benign histology. These findings echo those of multiple previous studies that have similarly reported heightened occurrences of breast lumps among women in their third decade of life, suggesting the need for awareness during this critical age period.
In contrast, malignant breast lumps were most frequently observed in the 40-49 age group, accounting for 41.9% of the malignant cases documented in this study. This aligns with the observations by Afolayan et al. in Ilorin, who noted comparable trends of breast cancer among Nigerian women. Additionally, international studies, such as those conducted by Larsen et al. in Oslo, Norway, indicated a higher peak incidence of breast cancer occurring within the 50-69 age range. In the United States, estimates from 2017 forecasted that the new cases of female breast cancer peaked primarily in the 60-69 years age group. Such data underscore the varying age-related patterns of breast cancer incidence across different populations and geographical regions.
Triple assessment is a very useful diagnostic tool to evaluate patients with breast lumps and to detect patients with breast cancer with an overall accuracy of 99.3%. This high level of diagnostic precision is contingent upon the correlation between clinical findings, imaging features, and pathological findings of the tissue sampled. The modified triple test score (MTTS) was developed to aid the interpretation of discordant triple assessment results. Wai et al. developed and reported a modified triple test score (MTTS), including CBE, Mammography, and/or Ultrasonography breast imaging, as well as FNAC and/or CNB for histology.
In our study, breast ultrasonography was used in place of mammography, while core needle biopsy (CNB) was used in place of fine needle aspiration cytology (FNAC). Each arm of the tripod retained the same scoring as the traditional triple test framework. Core needle biopsy (CNB) allowed for effective evaluation of receptor status in this study. It is associated with fewer complications and is more cost-effective than surgical biopsy. This advancement has the potential to improve the pathway from diagnosis to treatment in low- and middle-income countries (LMICs). While fine needle aspiration cytology (FNAC) offers a convenient option for pathological diagnosis in LMICs, there are challenges related to interpreting results in specific histological contexts. Additionally, the use of immunocytochemistry in pre-invasive carcinoma, along with FNAC’s false negative rate (FNR), may limit its widespread adoption compared to CNB. Ultrasound scans are widely available at comprehensive care facilities and do not use ionizing radiation, making them safe for repeated use, even in pregnant women. Women of all ages with palpable breast lesions can easily access breast ultrasonography. In contrast, mammography is less frequently available and more expensive, and stereotactic biopsies performed with this imaging technique can be more challenging than those performed with ultrasound-guided biopsies.
In the Palpation-guided CNB group of this study, clinical breast examination demonstrated a sensitivity of 90.0% and specificity of 77.8%. Breast ultrasonography showed a sensitivity of 100.0% and specificity of 90.9%. Furthermore, CNB had a sensitivity of 94.7% alongside a specificity of 100.0%. Rajan et al. reported a sensitivity and specificity for physical examination of 96.7% and 84.0%, respectively; 93.1% sensitivity and 95.9% specificity for breast ultrasonography; a 96.7% sensitivity and 100.0% specificity was reported for FNAC, which was used instead of CNB in their study carried out in India. Supporting these findings, Ngotho et al. reported a sensitivity of 100% and a specificity of 92.3% for CBE in Nairobi, Kenya, with corresponding metrics of 100% sensitivity and 94.2% specificity for breast ultrasonography, as well as 100% sensitivity and 98.1% specificity for FNAC. Moreover, Karim et al. reported a sensitivity of 95.5% and a positive predictive value (PPV) of 100% in a retrospective study conducted in Manchester, UK. In their study, they found CNB more reliable than FNAC.
In the ultrasound-guided CNB segment of our study, all parameters achieved 100.0% sensitivity and specificity, except for clinical breast examination, which had a sensitivity of 90.5%. The combined score (MTTS) for the concordant diagnostic arms (CBE, USS & CNB) across both groups yielded a sensitivity and specificity of 100.0%, respectively. This finding is consistent with that of Ngotho et al. who reported a similar MTTS outcome. Kharkwal et al. also endorsed the efficacy of the triple test score among the labour-class population in Delhi, India, in a prospective study. Their patients were 35 years of age and older.
In our analysis, clear distinctions emerged between MTTS scores 3-5, denoting benign lesions, and scores of 8-9, which indicated malignant conditions. Each respective group demonstrated congruence across clinical, radiological, and pathological results. However, instances of overlap reported in scores 6-7 predominantly were observed in the palpation group; for example, one benign and three malignant cases were found in the score 6 cohort, while the score 7 group included two benign and three malignant cases. There were no overlaps between benign and malignant lesions in the ultrasound-guided biopsy group. However, this difference was not statistically significant. This suggests that heightened attention to imaging characteristics was utilized during the ultrasound-guided procedures, significantly influencing the final imaging scores. This observation underscores the crucial role of interdisciplinary collaboration in comprehensive patient management, highlighting the need for further research to clarify the relationship between these diagnostic modalities with greater precision.
Wai et al. concluded that patients with an MTTS of 3-4 (indicative of benign lesions) required no further assessment, while those achieving scores of 8-9 (suggestive of malignancy) should proceed promptly to definitive management strategies. The high sensitivities and specificities demonstrated for both benign and malignant lumps in our study support this line of management, as patients with concordant malignant CNB results were appropriately directed towards definitive oncologic treatment, corroborated by surgical histology confirming their diagnoses. Accuracy measures reported by Gana et al. in 2023 showed comparable diagnostic success rates regardless of whether palpation-guided or ultrasound-guided methods were employed. Notably, a statistically significant distinction emerged regarding the occurrence of inconclusive CNB results, favouring the ultrasound-guided CNB technique. Integrating the MTTS with conventional diagnostic procedures yielded remarkable accuracy in identifying palpable breast lumps, offering an invaluable resource for clinical practice in Low and Middle-Income Countries (LMICs).
Limitations
- The study design did not accommodate repeat core needle biopsies. Considering the high accuracy of palpation-guided and ultrasound-guided CNB, the yield of repeat needle biopsies may be worth evaluating.
- The cost-effectiveness of either technique was not evaluated. This may be an important determinant of patients’ acceptance of either technique in our environment.
Conclusion
This study has established the modified triple test score as valuable in evaluating breast lumps in the breast clinic. The two CNB techniques have comparatively high accuracies in the combined scores.
Recommendations
The modified triple test score is an important tool in managing breast lumps. Its use can obviate the need for routine excisional biopsy for concordant benign lesions and expedite definitive therapy in concordant malignant breast lesions in our setting.
Conflicts of Interest Statement
The authors have no conflicts of interest to declare.
Acknowledgments
We recognize Professor Peter Pase Abur’s contributions and appreciate your encouragement and support during the conceptual phase of this work. We wish you a full recovery! Thank you, Dr. Sani Kamarudeen Owolabi of the Department of Pathology, for your diligence in preparing the photomicrographs.
References
- Nigam M, Nigam B. Triple Assessment of Breast – Gold Standard in Mass Screening for Breast Cancer Diagnosis. IOSR J Dent Med Sci. 2013;7(3):1-7.
- Batta A, Attri P.C, Jain P, Rashid Z,, Srivastava S CN. A Study of Modified Triple Test Score For Assessment of Palpable Breast Masses In Females In Western U.P. Ind J App Res. 2016;6(4):4-9.
- Guray M, Sahin AA, Objectives L. Breast Cancer Benign Breast Diseases: Classification, Diagnosis, and Management. Published online 2006:435-449.
- N WP, Amabra D. Benign Breast Lumps in A Tertiary Health Care Facility in. 2016;15(12):28-31. doi:10.9790/0853-1512032831
- Forae GD, Nwachokor FN, Igbe AP, Odokuma EI, Ijomone EA. Benign Breast Diseases in Warri, Southern Nigeria: A Spectrum of Histopathological Analysis. 2014;8(1):28-31. doi:10.4103/0331-3131.141026
- Akarolo-anthony SN, Ogundiran TO, Adebamowo CA. Emerging breast cancer epidemic: evidence from Africa. 2010;12(Suppl 4).
- Ibrahim NA, Popoola AO, Oludara MA, Omodele FO, Fadeyibi IO. Breast Cancer among Urban Nigerian Women: Appraising Presentation and the Quality of Care. 2011;4(4):388-392.
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: Sources, methods, and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;386(136):E359-E386. doi:10.1002/ijc.29210
- Provencher L, Hogue JC, Desbiens C, et al. Is clinical breast examination important for breast cancer detection? Curr Onc. 2016;23(4):332-339.
- Chiarelli AM, Majpruz V, Brown P, Thériault M, Shumak R, Mai V. The Contribution of Clinical Breast Examination to the Accuracy of Breast Screening. 2018;101(18). doi:10.1093/jnci/djp241
- Hooley RJ, Scoutt LM, Philpotts LE. Breast Ultrasonography: 2013;268(3).
- Lehman CD, Lee CI, Loving VA, et al. Ultrasound for Primary Imaging Women 30 – 39 Years of Age. 2012;(November):1169-1177. doi:10.2214/AJR.12.8842
- Liberman L. Percutaneous image-guided core breast biopsy. Radiol Clin North Am. 2002;40(3):483-500. doi:10.1016/S0033-8389(01)00011-2
- Johnson NB, Collins LC. Update on percutaneous needle biopsy of nonmalignant breast lesions. Adv Anat Pathol. 2009;16(4):183-195. doi:10.1097/PAP.0b013e3181a9d33e
- Hari S, Kumari S, Srivastava A, Thulkar S, Mathur S, Veedu PT. Image guided versus Palpation guided Core Needle Biopsy of Palpable Breast Masses: A prospective study. Indian J Med Res. 2016;143(5):597-604. doi:10.4103/0971-5916.187108
- Bruening W, Fontanarosa J, Tipton K, Treadwell JR, Launders J, Schoelles K. Systematic Review: Comparative Effectiveness of Core-Needle and Open Surgical Biopsy to Diagnose Breast Lesions. Ann Intern Med. 2010;152(4):238-246.
- Vega A, Garijo F, Ortega E. Core Needle Aspiration Biopsy of Palpable Breast Masses. Published online 2009. doi:10.3109/02841869509093635
- Rikabi A, Hussain S. Diagnostic usefulness of tru-cut biopsy in the diagnosis of breast lesions. Oman Med J. 2013;28(2):125-127. doi:10.5001/omj.2013.32
- Neal L, Sandhu NP, Hieken TJ, et al. Diagnosis and management of benign, atypical, and indeterminate breast lesions detected on core needle biopsy. Mayo Clin Proc. 2014;89(4). doi:10.1016/j.mayocp.2014.02.004
- Rageth CJ, O’Flynn EA, Comstock C, et al. First International Consensus Conference on Lesions of Uncertain Malignant Potential in the Breast (B3 Lesions). Breast Cancer Res Treat. 2016;159(2). doi:10.1007/s10549-016-3935-4
- Beagle G, Alexander PW, Toth-fejel S, Schmidt J, Vetto JT. Usefulness of the Triple Test Score for Palpable Breast Masses. 2001;136.
- Khoda L, Kapa B, Singh KG, Gojendra T, Singh LR. Evaluation of the Modified Triple Test (Clinical Breast Examination, Ultrasonography, and Fine-Needle Aspiration Cytology) in the Diagnosis of Palpable Breast Lumps. 2015;29(1):26-30. doi:10.4103/0972-4958.158927
- Kachewar S, Dongre S. Role of Triple Test Score in the Evaluation of Palpable Breast Lump. Indian J Med Paediatr Oncol. 2015;36(2):123-127. doi:10.4103/0971-5851.158846
- Yamane T. Statistics: An Introductory Analysis. 2nd ed. New York: Harper and Row; 1967.
- Ajay S, Micah B. Sampling Techniques & Determination of Sample Size in Applied Statistics Research: An Overview. 2014;II(11):1-22.
- Afolayan A, Olaoye O, Adesina M SR. Breast Cancer Trends In A Nigerian Population: An Analysis Of Cancer Registry Data. Int J Life Sci Pharm Res. 2012;2(3):29-34.
- Larsen I, Moller B, Johannesen T, et al. Cancer in Norway 2016- Cancer Incidence, Mortality, Survival and Prevalence in Norway. Oslo Can Reg Norw. 2017;1:1-114.
- Alteri R, Gansler T, Gaudet M, Gierach G, DeSantis C. Breast Cancer Facts and Figures. Amer Ca Soc. 2018;1:1-44.
- McDonald S, Saslow D, Alciati MH. Performance and Reporting of Clinical Breast Examination: A Review of the Literature. CA Cancer J Clin. 2004;54(6):345-361.
- O’Flynn EAM, Wilson ARM, Michell MJ. Image-guided breast biopsy: state-of-the-art. Clin Radiol. 2010;65(4):259-270. doi:10.1016/j.crad.2010.01.008
- Wai CJ, Al-Mubarak G, Homer MJ, et al. A Modified Triple Test for Palpable Breast Masses: The Value of Ultrasound and Core Needle Biopsy. Ann Surg Oncol. 2013;20(3):850-855. doi:10.1245/s10434-012-2710-y
- Gana S., Yusufu L, Abur P., Akorede-Olarinoye S., Bello N. Comparative Accuracy and Complications of Palpation-Guided Versus Ultrasound-Guided Core Needle Biopsy of Palpable Breast Lumps in Ahmadu Bello University Teaching Hospital, Zaria. J West Afr Coll Surg. 2023;13(2):28-36. doi:10.4103/jwas.jwas
- Pinto D, Schmitt FC. Immunohistochemistry Applied to Breast Cytological Material. Pathobiology. 2022;89(5):343-358. doi:10.1159/000522542
- Vaithianathan R, Sundaresan V, Santhanam R. Value of Modified Triple Test in the Diagnosis of Palpable Breast Lumps. Int J Cur Res Rev. 2013;05(5):125-134.
- Ngotho J, Githaiga J, Kaisha W. Palpable discrete breast masses in young women: Two of the components of the modified triple test may be adequate. SAJS. 2013;51(2):58-60. doi:10.7196/SAJS.1176
- Karim MO, Khan KA, Khan AJ, Javed A, Fazid S, Aslam MI. Triple Assessment of Breast Lump: Should We Perform Core Biopsy for Every Patient? Cureus. 2020;12(3):10-15. doi:10.7759/cureus.7479
- Kharkwal S, Sameer A, Mukherjee A. Triple test in carcinoma breast. J Clin Diagnostic Res. 2014;8(10):NC09-NC11. doi:10.7860/JCDR/2014/9237.4971

