Contraceptive Use in Women with Migraine: A Review
Contraceptive use in women with migraine
Keywords
- migraine
- contraceptive methods
- women’s health
Introduction
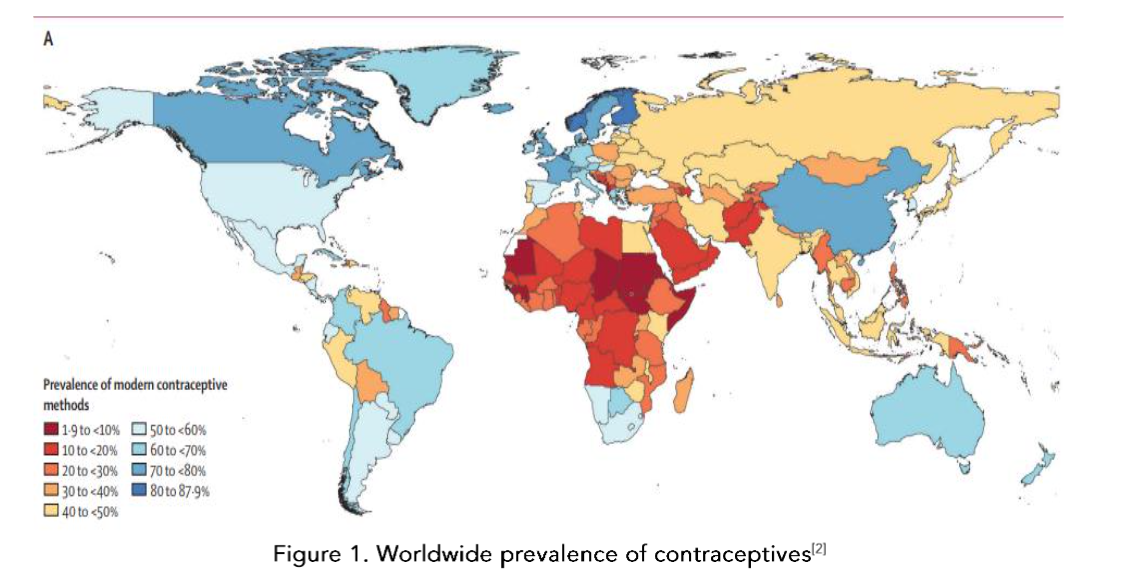
About one in four women of childbearing age has migraine[1] and many of these women will require a contraceptive. Contraceptives are used throughout the world, albeit in some regions more than others, so this is a global healthcare topic[2]. The use of contraception in women with migraine is often an issue of contentious debate and the guidelines vary and are constantly changing. In this review I will attempt to provide some context and clarification around these issues.
Figure 1. Worldwide prevalence of contraceptives[2]
Methods of contraception
Hormonal methods are either “combined”, which means a combination of oestrogen and progestogen or progestogen only. The most widely used combined contraceptive formulation is the oral contraceptive pill. There is also a vaginal ring or a patch but these are much less commonly used. With regards to progestogen only methods there is an oral version, there are also injectables; a depo injection, an implant and an intrauterine device, which releases levonorgestrel.
Important drug interactions
The most important contraceptive interaction in a women with migraine of childbearing age is topiramate as it is a teratogenic drug. Topiramate interacts with most of the contraceptive formulations except the depo progesterone or the intrauterine devices. Most of the data suggests that the interaction occurs at levels of 200 mg or higher[3]. Much lower doses are used in migraine as prophylaxis than in epilepsy but because the risk is one of significant teratogenicity, patients must be advised that if they are not on contraceptives or using oral contraceptives they need to take extra precautions, such as adding barrier methods or switching to an alternative method of contraception.
WHAT IS THE ROLE OF OESTROGEN/ PROGESTEROGEN IN MIGRAINE?
Fluctuations in oestrogen occur around menarche and the menopause and these correlate with peaks in migraine frequency[4]. Data suggests that oestrogen has a significant role, at least the drop in oestrogen has a significant role in triggering menstrual migraine (Figure 2)[5][6].
![Figure 2. Menstrual and perimenopausal susceptibility to migraine. (Onello et al [7])](/pdf-to-wp-converter/uploads/images/contraceptive-use-women-migraine-review-figure-2.png)
Figure 2. Menstrual and perimenopausal susceptibility to migraine. (Ornello et al [7])
There are estrogen receptors throughout the brain in areas that are thought to be important in migraine, such as the hypothalamus, pons, periaqueductal grey, locus coeruleus[8]. In animal studies oestrogen triggers cortical spreading depression which is thought to be the pathophysiological correlate of migraine aura. When a woman starts an oestrogen-containing contraceptive pill or initiates hormone replacement therapy or is pregnant, these are high oestrogen states and these can sometimes result in aura presenting for the first time[9], which correlates well with animal studies showing oestrogen triggering cortical spreading depression[10].
The role of progestogens in migraine is less well understood. It is known that allopregnanolone, which is a derivative of progesterone, enhances GABA activity in the central nervous system, whereas oestrogen has a glutaminergic effect, which is excitatory. Progestogens may inhibit neuronal excitability. One study looking at allopregnanolone levels in women with migraine suggested that they were lower in women with migraine[11]. Further research is needed in this area.
The effect of contraceptives on migraine
THE EFFECT OF COMBINED CONTRACEPTIVES
A common question asked by patients is “what will happen to my migraine if I take the contraceptive pill?” The problem with this question is that there are many different types of contraceptives. Even when one considers the combined oral contraceptive pill, this is not just a single entity and it consists of varying doses of oestrogen which have changed over the years very significantly. Going back to the last century, doses of 100 micrograms were used and this then decreased to about 50 micrograms and now doses as low as 15-20 micrograms are used. The fact that the dose of oestrogen has changed quite significantly throughout the years is a very important factor to bear in mind. Secondly, the progestogen component of the pill varies as well. There are first, second, third and fourth generations. So there is no simple answer to this question.
There is a paucity of good studies in this area because much of the data that is published looked
at “headache” as a side effect in women starting a contraceptive. These are not well designed studies looking specifically at whether this is migraine with or without aura or if it meets international headache society (IHS) criteria for migraine. Consequently the data available suggests migraine can get worse in 18 – 50%, improve in 8 to 35% and is not changed in 39 to 65% in women with migraine on combined contraceptives[12], so not very informative. In women with migraine with aura there may be an exacerbation of migraine with the combined contraceptive pill[12], which is perhaps logical if one relates back to the effect of oestrogen on cortical spreading depression.
When a woman is on the combined oral contraceptive pill, she will usually take it for 21 days and then she has a seven day break, where she has her menstrual period, and during that time there is often a migraine which is akin to a menstrual migraine. Tri-cycling the combined contraceptive pill back-to-back can be helpful in treating this[13]. Also shortening that pill free period from seven days to four days can also be helpful[14].
If a woman develops aura for the first time when starting the combined contraceptive pill she could be switched to a progestogen only contraceptive. There was a study from Colhoun[15] looking at switching from oral combined oral contraceptive to the vaginal ring and that did improve aura and this was thought to be because the dose of oestrogen in the vaginal ring is lower.
A German group investigated the levels in women of two key peptides thought to be involved in the pathophysiology of migraine: calcitonin gene-related peptide (CGRP)[16] and pituitary adenylate cyclase-activating polypeptide (PACAP-38)[17]. The design of the study was that there were two groups of women, 60 with migraine and 60 without migraine, and half of them were on contraceptive pill and half of them were not. PACAP levels in women with menstrual migraine were higher during menstruation and in women with migraine compared with controls[18]. Interestingly, looking at the women with migraine who were on the combined contraceptive pill, there was no difference between the levels of PACAP between them and the control patients on the contraceptive pill. The same applied to the CGRP study[19]. These studies demonstrate how being on the combined contraceptive pill can modulate levels of these peptides which are thought to be involved in migraine.
THE EFFECT OF PROGESTOGEN ONLY METHODS ON MIGRAINE
A meta-analysis of the use of desogestrel in migraine included four studies, two of which involved women with migraine with aura[20]. Desogestrel is thought to inhibit ovulation. There was a significant but modest reduction in the number of migraine attacks, reduced analgesic use and improved headache quality of life scores. However, the analysis suggested that the quality of evidence was low for each outcome measure, mainly because the number of patients in the study were low, and these were not randomised placebo-controlled trials. Current evidence is observational and based on small samples of women using only one oral progestin-only formulation. Two of the studies compared the progestone-only pill to the combined oral contraceptive. One of these suggested some benefits of being on the progestin pill rather than being on the combined pill taken with a break after 21 days. About 10% on desogestrel had troublesome bleeding, which is often a problem with progestogen only methods of contraception.
There is even less evidence regarding non-oral formulations of progesterone. One study looked at the effects of amenorrhoea on menstrual migraine and suggested that if a woman develops amenorrhea, then she is more likely to have an improvement in menstrual migraine[21]. In this particular study, 19 out of 31 women were on the levonorgestrel containing intrauterine device.
The depot and the implant versions of progesterone inhibit ovulation. In theory, inhibition of ovulation reduces the fluctuations of oestrogen and progesterone, which is thought to be quite important in triggering migraines. So theoretically there should be some improvement but there is no good trial data.
![Figure 3. Contraceptive methods and their effects in migraine. Adapted from Lohouizen et al [24]](/pdf-to-wp-converter/uploads/images/contraceptive-use-women-migraine-figure-3.png)
What are the safety concerns?
The safety concerns for migraineurs regarding oral contraceptives revolve around vascular concerns. There are two issues; venous thromboembolism and the risk of stroke. The former is an issue regardless of migraine. The combined contraceptive pill does increase the risk of venous thromboembolism, the progestin-only contraceptive pill does not. The intrauterine contraceptive device also does not. The risk of a thromboembolism is also determined by the oestrogen dose of the combined contraceptive pill and by which generation of progestogen is used. Levonorgestrel tends to have a lower risk then gestodene, cyproterone, desogestrel and drospirenone[23].
The main concern specific to migraine and the contraceptive pill is stroke. Over the last two decades there has been evidence suggesting a twofold higher stroke risk among women with migraine with aura[24]. The stroke risk was also found to be higher in those with greater migraine attack frequency[25]. However, there is less clear evidence regarding stroke risk in women who have migraine without aura.
A review of the meta analyses of migraine and risk of stroke demonstrates a lower risk with the more recent studies compared with the older ones (see table 1 where studies are listed in chronological order)[26]. The most recent meta-analysis was from Mahmoud et al[27]. They conducted a meta-analysis of 16 observational cohort studies including over 1.1 million subjects (394 942 migraineurs) and an extended follow-up duration up to 26 years looking at both cardiovascular and cerebrovascular events. Their study reports a relative risk of stroke of 1.56 for migraine with aura and 1.11 in migraine without aura. The studies included dated from 2006–2017.
Table 1. Meta-analysis of migraine and relative risk of ischaemic stroke (Tietjen et al[26])
| Study | Design | Migraine Overall | Migraine Without Aura | Migraine With Aura | Migraine in Women |
|---|---|---|---|---|---|
| Etminan et al[3] | 11 case-control, 3 cohort | 2.16 (1.89–2.48) | 1.56 (1.03–2.36) | 2.88 (1.89–4.39) | <45 y: 2.76 (2.17–3.52), OCP: 8.72 (5.05–15.05) |
| Schürks et al[4] | 13 case-control, 10 cohort, 2 cross-control | 1.73 (1.31–2.29) | 1.23 (0.90–1.69) | 2.16 (1.53–3.03) | 2.08 (1.31–3.84), <45 y: 3.65 (2.21–6.04), OCP: 7.02 (1.51–32.68) |
| Spector et al[5] | 13 case-control, 8 cohort | 2.04 (1.72–2.43) | 1.24 (0.86–1.79) | 2.25 (1.53–3.33) | Not reported |
| Hu et al[6] | 11 cohort (6 with ischemic stroke) | 1.64 (1.22–2.20) | 1.02 (0.68–1.51) | 2.14 (1.33–3.43) | Not reported |
| Mahmoud et al[7] | 16 cohort (7 with ischemic stroke) | 1.29 (1.08–1.54) | 1.11 (0.94–1.31) | 1.56 (1.30–1.87) | Not reported |
In the meta-analysis by Schürks et al the relative risk of stroke in migraine with aura was found to be two-fold. Adding in the combined contraceptive pill has a multiplicative effect with a sevenfold increase in stroke and smoking increases the relative risk to 9[28]. It is important to note that although the Schürks paper was published in 2009 the data that was used to derive these figures was from four studies, three of which were from the 1990s and the fourth study was the McClellan study[29]. Consequently, this reflects the contraceptive pill that was available in the 1990s, which had a much higher oestrogen dose, usually 50mcg. The McClellan study only included 400 women with migraine with aura in the study, so relatively small numbers.
In a more recent study by Champaloux[30], which is a nested case control study, data was collected from 2006 to 2012 and there were a much larger number of cases; 1884 cases of stroke. The study investigated the effect of having migraine and being on a contraceptive on stroke as a combined effect. It also corrected for other variables as well. In this study migraine with aura confers a 2.7 adjusted odds ratio of stroke while being on the combined contraceptive pill with migraine with aura increases this odds ratio to 6.1.
An important fact to consider is that the risk of stroke changes according to the oestrogen composition of the combined contraceptive pill. A meta analysis demonstrates very clearly that as the dose of oestrogen reduces, the odds ratio of having a stroke declines and that with progesterone-only pills there is no increased risk of stroke (see table 2)[31]. Therefore, the evidence is very clear that the oestrogen dose correlates with the risk of stroke. The combined contraceptive pill is not a single entity because it depends on the composition. The progestogen component of the combined pill also has an effect on the risk of stroke but this is more variable and depends on type of progestogen rather than dose[32].
Table 2. Impact of oestrogen dose on estimated risk of ischaemic stroke – adapted from Xu et al[31]
| OCP formulations | No. of studies | OR (95%CI) |
|---|---|---|
| Estrogen dose | ||
| ≥50 ug | 9 | 3.28 (2.49–4.32) |
| <50 ug | 11 | 1.97 (1.61–2.41) |
| 30–40 ug | 5 | 1.75 (1.61–1.89) |
| 20 ug | 3 | 1.56 (1.36–1.79) |
| POPs | 4 | 0.99 (0.71–1.37) |
There is a paucity of data from low dose oestrogen contraceptives with only one very small study with 127 patients in a tertiary referral centre who have confirmed stroke and only 36 had migraine[33]. This paper suggested there is an odds ratio 1.5 of stroke for women with migraine on a combined contraceptive pill greater than 30 micrograms compared to those without migraine. However, the numbers in this study are too small.
The actual risk of stroke for a woman depends on multiple factors such as smoking and probably the most important factor is age. For a 15 to 19 year old woman the actual risk of stroke is 1.4 in 100,000[34].
So the actual risks of starting a combined contraceptive for a 19 year old who has occasional migraine with aura is very different to the risk to a woman in her forties who may be a smoker with multiple episodes of migraine with aura.
What are the guidelines?
The American guidelines, CDC MEC (U.S. Medical Eligibility Criteria for Contraceptive Use, 2024 Contraception | CDC)[35] state that combined contraceptives are not recommended for migraine with aura and can be used in migraine without aura without risk factors. In the UK the guidance is similar from the Faculty of Sexual Health and Reproductive Healthcare (FSRH)[36]. The consensus guideline from European Headache Federation and European society of contraceptive and reproductive health also advises against the use of oestrogen containing contraceptives in migraine with aura and in women with migraine without aura without risk factors an oestrogen dose of less than 35 micrograms is recommended if combined contraceptives are to be used[37].
Conclusion
The current guidance does not take into account individual risk factors for women with migraine with aura. Further research is required to accurately assess the risk of stroke in migraineurs using current formulations of contraceptives with lower doses of oestrogen and to look at migraine with and without aura separately. This becomes an increasingly challenging prospect as, for the aforementioned reasons, there are less women with migraine with aura on combined contraceptives now. Until better data is available, the existing guidelines suggest avoiding oestrogen based contraceptives in women with migraine with aura.
References
1. Jensen R, Stovner LJ. Epidemiology and comorbidity of headache. Lancet Neurol 2008;7(4): 354-61 doi: 10.1016/S1474-4422(08)70062-0 [published Online First: Epub Date]|.
2. Haakenstad A, Angelino O, Irvine CMS, et al. Measuring contraceptive method mix, prevalence, and demand satisfied by age and marital status in 204 countries and territories, 1970-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2022;400(10348):295-327 doi: 10.1016/S0140-6736(22)00936-9[published Online First: Epub Date]|.
3. Sarayani A, Winterstein A, Cristofoletti R, Vozmediano V, Schmidt S, Brown J. Real-world effect of a potential drug-drug interaction between topiramate and oral contraceptives on unintended pregnancy outcomes. Contraception 2023;120:10 9953 doi: 10.1016/j.contraception.2023.109953 [published Online First: Epub Date]|.
4. Borsook D, Erpelding N, Lebel A, et al. Sex and the migraine brain. Neurobiol Dis 2014;68: 200-14 doi: 10.1016/j.nbd.2014.03.008[published Online First: Epub Date]|.
5. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology 2004;63(2):351-3 doi: 10.1212/01.w nl.0000133134.68143.2e[published Online First: Epub Date]|.
6. Somerville BW. The role of estradiol withdrawal in the etiology of menstrual migraine. Neurology 1972;22(4):355-65 doi: 10.1212/wnl.2 2.4.355 [published Online First: Epub Date]|.
7. Ornello R, De Matteis E, Di Felice C, Caponnetto V, Pistoia F, Sacco S. Acute and Preventive Management of Migraine during Menstruation and Menopause. J Clin Med 2021;10 (11) doi: 10.3390/jcm10112263[published Online First: Epub Date]|.
8. Warfvinge K, Krause DN, Maddahi A, Edvinsson JCA, Edvinsson L, Haanes KA. Estrogen receptors alpha, beta and GPER in the CNS and trigeminal system – molecular and functional aspects. J Headache Pain 2020;21(1):131 doi: 10.1186/s101 94-020-01197-0[published Online First: Epub Date]|.
9. MacGregor EA. Oestrogen and attacks of migraine with and without aura. Lancet Neurol 2004;3(6):354-61 doi: 10.1016/S1474-4422(04)007 68-9[published Online First: Epub Date]|.
10. Chauvel V, Multon S, Schoenen J. Estrogen-dependent effects of 5-hydroxytryptophan on cortical spreading depression in rat: Modelling the serotonin-ovarian hormone interaction in migraine aura. Cephalalgia 2018;38(3):427-36 doi: 10.1177/0 333102417690891[published Online First: Epub Date]|.
11. Rustichelli C, Bellei E, Bergamini S, et al. Comparison of pregnenolone sulfate, pregnanolone and estradiol levels between patients with menstrually-related migraine and controls: an exploratory study. J Headache Pain 2021;22(1):13 doi: 10.1186/s1019 4-021-01231-9[published Online First: Epub Date]|.
12. MacGregor EA. Contraception and headache. Headache 2013;53(2):247-76 doi: 10.1111/head.1 2035[published Online First: Epub Date]|.
13. Sulak P, Willis S, Kuehl T, Coffee A, Clark J. Headaches and oral contraceptives: impact of eliminating the standard 7-day placebo interval. Headache 2007;47(1):27-37 doi: 10.1111/j.1526-4610.2007.00650.x[published Online First: Epub Date]|.
14. De Leo V, Scolaro V, Musacchio MC, Di Sabatino A, Morgante G, Cianci A. Combined oral contraceptives in women with menstrual migraine without aura. Fertil Steril 2011;96(4):917-20
doi:10.1016/j.fertnstert.2011.07.1089[published Online First: Epub Date]|.
15. Calhoun A, Ford S, Pruitt A. The impact of extended-cycle vaginal ring contraception on migraine aura: a retrospective case series. Headache 2012;52(8):1246-53 doi: 10.1111/j.1526-4610.201 2.02211.x[published Online First: Epub Date]|.
16. Edvinsson L, Haanes KA, Warfvinge K, Krause DN. CGRP as the target of new migraine therapies – successful translation from bench to clinic. Nat Rev Neurol 2018;14(6):338-50 doi: 10.1038/s41582-018-0003-1[published Online First: Epub Date]|.
17. Edvinsson L, Tajti J, Szalardy L, Vecsei L. PACAP and its role in primary headaches. J Headache Pain 2018;19(1):21 doi: 10.1186/s10194-018-0852-4[published Online First: Epub Date]|.
18. Storch E, Overeem LH, Terhart M, et al. PACAP-38 and sex hormones in women with migraine: exploratory analysis of a cross-sectional, matched cohort study. J Headache Pain 2024;25 (1):98 doi: 10.1186/s10194-024-01804-4[published Online First: Epub Date]|.
19. Raffaelli B, Storch E, Overeem LH, et al. Sex Hormones and Calcitonin Gene-Related Peptide in Women With Migraine: A Cross-sectional, Matched Cohort Study. Neurology 2023;100(17):e1825-e35 doi: 10.1212/WNL.0000000000207114[published Online First: Epub Date]|.
20. Warhurst S, Rofe CJ, Brew BJ, et al. Effectiveness of the progestin-only pill for migraine treatment in women: A systematic review and meta-analysis. Cephalalgia 2018;38(4):754-64
doi:10.1177/0333102417710636[published Online First: Epub Date]|.
21. Vetvik KG, MacGregor EA, Lundqvist C, Russell MB. Contraceptive-induced amenorrhoea leads to reduced migraine frequency in women with menstrual migraine without aura. J Headache Pain 2014;15(1):30 doi: 10.1186/1129-2377-15-30 [published Online First: Epub Date]|.
22. van Lohuizen R, Paungarttner J, Lampl C, MaassenVanDenBrink A, Al-Hassany L. Considerations for hormonal therapy in migraine patients: a critical review of current practice. Expert Rev Neurother 2023;24(1):1-21 doi: 10.1080/1473 7175.2023.2296610[published Online First: Epub Date]|.
23. van Hylckama Vlieg A, Helmerhorst FM, Vandenbroucke JP, Doggen CJ, Rosendaal FR. The venous thrombotic risk of oral contraceptives, effects of oestrogen dose and progestogen type: results of the MEGA case-control study. BMJ 2009;339: b2921 doi: 10.1136/bmj.b2921[published Online First: Epub Date]|.
24. Kurth T, Chabriat H, Bousser MG. Migraine and stroke: a complex association with clinical implications. Lancet Neurol 2012;11(1):92-100 doi:10.1016/S1474-4422(11)70266-6 [published Online First: Epub Date]|.
25. Kurth T, Slomke MA, Kase CS, et al. Migraine, headache, and the risk of stroke in women: a prospective study. Neurology 2005;64(6):1020-6 doi:10.1212/01.WNL.0000154528.21485.3A [published Online First: Epub Date]|.
26. Tietjen GE, Maly EF. Migraine and Ischemic Stroke in Women. A Narrative Review. Headache 2020;60(5):843-63 doi:10.1111/head.13796 [published Online First: Epub Date]|.
27. Mahmoud AN, Mentias A, Elgendy AY, et al. Migraine and the risk of cardiovascular and cerebrovascular events: a meta-analysis of 16 cohort studies including 1 152 407 subjects. BMJ Open 2018;8(3):e020498 doi: 10.1136/bmjopen-2017-020498[published Online First: Epub Date]|.
28. Schurks M, Rist PM, Bigal ME, Buring JE, Lipton RB, Kurth T. Migraine and cardiovascular disease: systematic review and meta-analysis. BMJ 2009;339:b3914 doi:10.1136/bmj.b3914 [published Online First: Epub Date]|.
29. MacClellan LR, Giles W, Cole J, et al. Probable migraine with visual aura and risk of ischemic stroke: the stroke prevention in young women study. Stroke 2007;38(9):2438-45 doi: 10.1161/STROKEAHA.10 7.488395 [published Online First: Epub Date]|.
30. Champaloux SW, Tepper NK, Monsour M, et al. Use of combined hormonal contraceptives among women with migraines and risk of ischemic stroke. Am J Obstet Gynecol 2017;216(5):489 e1-89 e7 doi:10.1016/j.ajog.2016.12.019[published Online First: Epub Date]|.
31. Xu Z, Li Y, Tang S, Huang X, Chen T. Current use of oral contraceptives and the risk of first-ever ischemic stroke: A meta-analysis of observational studies. Thromb Res 2015;136(1):52-60
doi:10.1016/j.thromres.2015.04.021[published Online First: Epub Date]|.
32. Lidegaard O, Lokkegaard E, Jensen A, Skovlund CW, Keiding N. Thrombotic stroke and myocardial infarction with hormonal contraception. N Engl J Med 2012;366(24):2257-66 doi: 10.1056/N
EJMoa1111840 [published Online First: Epub Date]|.
33. Batur P, Yao M, Bucklan J, et al. Use of combined hormonal contraception and stroke: A case-control study of the impact of migraine type and estrogen dose on ischemic stroke risk. Headache 2023;63(6):813-21 doi:10.1111/head.14473 [published Online First: Epub Date]|.
34. Nightingale AL, Farmer RD. Ischemic stroke in young women: a nested case-control study using the UK General Practice Research Database. Stroke 2004;35(7):1574-8 doi:10.1161/01.STR.00001297 89.58837.e4 [published Online First: Epub Date]|.
35. Appendix D: Classifications for Combined Hormonal Contraceptives. U.S. Medical Eligibility Criteria for Contraceptive Use, 2024 2024
36. UK MEDICAL ELIGIBILITY CRITERIA FOR CONTRACEPTIVE USE The faculty of sexual and reprodutive healthcare 2019
37. Sacco S, Merki-Feld GS, KL AE, et al. Hormonal contraceptives and risk of ischemic stroke in women with migraine: a consensus statement from the European Headache Federation (EHF) and the European Society of Contraception and Reproductive Health (ESC). J Headache Pain 2017;18(1):108
doi:10.1186/s10194-017-0815-1 [published Online First: Epub Date]|.

